Abstract
Introduction
PENTAXIM™ (Sanofi), DTaP-IPV//Hib, a pentavalent combination vaccine for protection against diphtheria, tetanus, pertussis, poliomyelitis, and invasive infections caused by Haemophilus influenzae type b, has been licensed in South Korea by the Ministry of Food and Drug Safety (MFDS) on May 9, 2016, and is currently used in routine vaccination. The aim of this phase IV study, conducted as a post-licensure commitment in South Korea, was to evaluate the safety of the DTaP-IPV//Hib vaccine when administered in infants at 2, 4, and 6 months of age in the real-world clinical practice.
Methods
This multicenter, observational, post-marketing surveillance (PMS) study was conducted in real-world practice in South Korea. Infants aged 2 months or older were enrolled across seven centers from July 31, 2018 to February 11, 2020. The study outcomes included occurrence, time to onset, duration, intensity, and causality assessment (for unsolicited adverse events [AEs] only) for several pre-listed solicited injection-site and systemic reactions, unsolicited AEs, and serious adverse events (SAEs).
Results
Data from 619 participants were included in the safety analysis. Overall, 618 AEs were reported by 273 (44.1%) participants consisting of 121 solicited injection-site reactions (15.4%), 344 solicited systemic reactions (24.6%), and 153 unsolicited AEs (15.7%) of which, 124 were unexpected AEs (12.9%) (regardless of intensity). None of the unsolicited AEs were reported to have a causal relationship with the study vaccine. One SAE of pyrexia (solicited reaction) was reported. Most AEs were of mild intensity, and all participants recovered.
Conclusion
This PMS study of the DTaP-IPV//Hib vaccine confirmed its safety profile in a real-life setting in South Korea and justified that the vaccine is well tolerated when used in infants aged 2 months or older for the primary series.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Post-marketing surveillance study of the combined diphtheria, tetanus, acellular pertussis, inactivated poliovirus, and Haemophilus influenzae type b conjugate vaccine, DTaP-IPV//Hib (PENTAXIMTM), allows one to assess its safety profile in a real-life setting in South Korea. |
What was learned from the study? |
No safety concerns related to the DTaP-IPV//Hib vaccine in real-world use were detected in infants aged 2 months or older for the primary series in the Korean population. |
The benefit–risk balance of DTaP-IPV//Hib vaccine was favorable, and it can be continued for immunization against diphtheria, tetanus, pertussis, poliomyelitis, and invasive infections due to Haemophilus influenzae type b, as a three-dose infant primary series given at 2, 4, and 6 months of age. |
Introduction
Diphtheria, tetanus, pertussis, and invasive diseases caused by Haemophilus influenzae type b (Hib) are serious bacterial infections. Causative agents of diphtheria, poliomyelitis, Hib, and pertussis spread from person to person, while for tetanus, they enter the body through cuts or wounds [1, 2]. The most important measure for the control and prevention of these diseases is complete and optimal immunization. The combination of diphtheria, tetanus, and pertussis antigens into a single vaccine (DTaP) has been fundamental to the protection of the pediatric population over the past 50 years. The addition of inactivated poliovirus (IPV) and Hib vaccines into the combination has facilitated the introduction of these vaccines into recommended immunization schedules [3].
Since 2013, the vaccination schedule recommended by the South Korean National Immunization Program (NIP) includes administration of primary series of DTaP, IPV, and Hib at 2, 4, and 6 months of age. A booster dose of DTaP is recommended at 15–18 months of age and in children 4–6 years old. A booster dose of Hib is recommended at 12–15 months of age, and that of IPV at 4–6 years of age [4, 5]. Based on a surveillance study conducted in 2020, the vaccination coverage rates in South Korea at 6 months for the first, second, and third doses of DTaP and IPV were 98.9%, 98.3%, and 94.1%, respectively. For Hib vaccines, they were 98.9%, 98.2%, and 93.9%, respectively [6].
PENTAXIM™ (produced by Sanofi), DTaP-IPV//Hib, is a pentavalent combination vaccine for protection against diphtheria, tetanus, pertussis, poliomyelitis, and invasive infections caused by Hib. It is based on the components of the separate DTaP-IPV (TETRAXIM™) and Hib (Act-Hib™) vaccines [7] that have been used separately or in combination vaccines previously [8, 9]. PENTAXIM has been evaluated in various clinical trials and studies globally, demonstrating its acceptable safety profile and high immunogenicity [8, 10,11,12,13,14,15,16,17,18,19,20], including in South Korea [7]. It has been licensed in Europe, South Korea, and other countries as a three-dose primary vaccination series during the first year of life and/or a booster vaccination during the second year of life [8].
Such combination vaccines improve compliance and vaccination schedule timeliness, increase vaccination coverage rate, decrease the number of vaccination visits and time spent for vaccination by reducing the number of injections, and reduce reactogenicity [3, 21].
In South Korea, DTaP-IPV//Hib was approved by the Ministry of Food and Drug Safety (MFDS) on May 9, 2016. It is recommended as a three-dose primary series in infants to be administered at 2, 4, and 6 months of age. DTaP-IPV//Hib vaccine has demonstrated non-inferior immunogenicity to separately given DTaP-IPV and Hib vaccines, with similar safety profile in a randomized clinical trial conducted in infants from South Korea [7].
Post-marketing surveillance (PMS) studies help in confirming the safety profile of a vaccine in real-life use and to detect adverse events (AEs) in the general population that may not have been detected during clinical development phases. The aim of this phase IV PMS study, conducted as a post-licensure commitment in South Korea, was to evaluate the safety of the DTaP-IPV//Hib vaccine when administered in infants at 2, 4, and 6 months of age in the real-world clinical practice.
Methods
Study Design
This was a multicenter, non-interventional, observational, PMS study, conducted in the real-world after DTaP-IPV//Hib license approval in South Korea in accordance with Pharmaceutical Affairs Law with Enforcement Rule on the Safety Management of Medicinal Products Law. Participants were enrolled across seven centers from July 31, 2018 to February 11, 2020. The trial was approved by The Catholic University of Korea Seoul St Mary’s Hospital (Central institutional review board [IRB]; approval number KC18MODP0049). Per South Korea’s pharmaceutical affairs act, only one central IRB approved the study on behalf of all other study sites. Other study sites were notified of the approval by the central IRB. The study was conducted in accordance with the Declaration of Helsinki and the MFDS standards for re-examination of new drugs. Informed consent was obtained from all the parents/legal representative(s) before enrollment.
Study Participants
Infants aged 2 months or older were enrolled in the study if they were entitled to receive one of the three infant primary series doses of the DTaP-IPV//Hib vaccine according to the locally approved package insert during a routine healthcare visit. A total of 600 evaluable participants were planned to be included in the study. Participants were excluded if they had contraindications to vaccination, had foreign guardians, previously participated in this study, or participated or planned to participate in other studies within 4 weeks from date of enrollment. Participants were followed for one vaccination only, irrespective of their age. Hence, every participant only contributed to one vaccination.
Study Vaccine
DTaP-IPV//Hib vaccine (PENTAXIM) is available as a sterile suspension in a single-dose prefilled syringe containing the DTaP-IPV constituents to be used to rehydrate the content of one single-dose lyophilized Hib vaccine. Each 0.5-mL dose of the final vaccine contains antigens against the five target pathogens, at least 20 international units (IU) of diphtheria toxoids, at least 40 IU of tetanus toxoids, two purified antigens of Bordetella pertussis (25 μg of pertussis toxoid [PT] and 25 μg of filamentous hemagglutinin [FHA], each adsorbed onto aluminum salt), three distinct poliovirus antigens (40 D-antigen units of type 1 poliovirus, 8 D-antigen units of type 2 poliovirus, and 32 D-antigen units of type 3 poliovirus; each produced on VERO cells), and Hib polysaccharide (PRP) (10 μg) conjugated to the tetanus protein (18–30 μg) (PRP~T).
Data Collection
Demographic and other baseline data were collected on a case report form (CRF). Participant’s parents/legally acceptable representative(s) recorded daily body temperature, occurrence of all pre-listed solicited injection-site and systemic reactions, their intensity, and action taken like medications, if any, until day 7 post-vaccination using a diary card. The highest temperature was then recorded by the site in the CRF. Unsolicited AEs were reported up to 28 (+ 7) days post-vaccination in a diary card. Serious AEs (SAEs) were recorded throughout the study, from inclusion to last follow-up with the participant.
Study Outcomes
The study outcomes included occurrence, time to onset, duration, intensity, and causality assessment (for unsolicited AEs only) for pre-listed solicited injection-site and systemic reactions, unsolicited AEs, and SAEs. By definition, all the solicited injection-site and systemic events were considered to have a causal relationship with DTaP-IPV//Hib vaccine, and hence termed as reactions. Unsolicited AEs were defined as AEs not pre-listed in the CRF. Unexpected AEs were defined as AEs not listed in the product label. SAEs were defined as any event that resulted in death, was life-threatening, required hospitalization or the prolongation of hospitalization, resulted in persistent or significant disabilities/incapacity, congenital anomalies, or birth defect, or other medically significant events that jeopardized the participant or required medical or surgical intervention to prevent one of the outcomes. Adverse drug reactions (ADRs) were defined as the AEs that have causal relationship with the study vaccine.
The intensity of the solicited and unsolicited AEs was classified as grade 1 (mild), grade 2 (moderate), or grade 3 (severe).
Actions taken for solicited or unsolicited AEs were classified as 0 (no action), 1 (self-medication with an existing prescription or an over-the-counter medication), 2 (contact with healthcare professional [HCP], but no new medication prescribed), 3 (new medication prescribed by HCP), and 4 ([for solicited reactions only] hospitalization).
The causality of each AE was assessed by the investigator as certain (valid time relationship to vaccine administration and cannot be explained by other concomitant drugs or other disorders), probable/likely (pertinent time relationship to vaccine administration and was not likely to be caused by other concomitant drugs or disorders), possible (pertinent time relationship to vaccine administration, but could be caused by other concomitant drugs or disorders), unlikely (not likely to have causal relationship with vaccine administration; temporary cases), conditional/unclassified (require more data for proper assessment), or unassessable/unclassifiable (insufficient information exist which cannot be supplemented).
Statistical Analysis
Primary analyses were performed on the safety analysis set and were descriptive in nature. The safety analysis set was defined as the participants who received one dose of the study vaccine regardless of the dose number in its infant primary series. AEs were summarized using the International Council on Harmonization Medical Dictionary for Regulatory Activities (MedDRA) preferred term version 20.0. The 95% confidence intervals (CIs) of percentages for participants experiencing the respective AEs were calculated using the exact binomial distribution according to Clopper–Pearson’s method for proportions. All the analyses were carried out on a single population, i.e., the safety analysis set. As required by the MFDS in South Korea, in order to determine the factors that may affect the incidence of AEs, univariate analysis (chi-square test or Fisher’s exact test) was conducted on the demographic and medical factors of participants where at least one event was reported. Gender distribution, vaccination history, concomitant disease, and vaccination dose were assessed using the chi-square test, and concomitant medication/vaccination, allergic history, and concomitant allergic history at the time of vaccination using Fisher’s exact test.
To confirm the statistical significance of the factors that may affect safety of the study vaccine, two-sided test with 5% significance level was used.
Results
Demographic and Baseline Characteristics
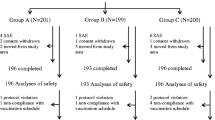
Among the 657 participants enrolled in the study, data from 619 participants were included to the safety analysis (Fig. 1). Data from 38 participants was excluded from the safety analysis set because of off-label vaccine usage (n = 36) and foreign guardian (n = 2). Among 619 participants, 46.5% (n = 288), 30.5% (n = 189), and 22.9% (n = 142) had received their dose 1, dose 2, or dose 3 of the primary series, respectively. Their demographic and baseline characteristics are presented in Table 1. Overall, 50.4% (n = 312) of the participants were female. None of the participants had relevant medical history prior to study vaccine administration, except for allergic manifestations history (n = 1). A total of 5.7% (n = 35) participants had concomitant disease at the time of vaccination. The most frequent concomitant diseases were cough (n = 13), diarrhea (n = 11), allergic manifestations (n = 7) (with one of them having an history of allergic manifestations), and diaper dermatitis (n = 4). Overall, 98.2% (n = 608) of participants were taking concomitant medications (defined as any medication being taken at the time of vaccination with the study vaccine and during the study (up to day 28 [+ 7]) or received one or more concomitant vaccine(s). The most frequent concomitant medications were expectorant combinations with cough suppressants (n = 33) and nasal decongestants (n = 55). The most frequent concomitant vaccinations were bacterial vaccines (n = 558) followed by viral vaccines (n = 536). A total of 7.6% (n = 47) participants had history of vaccination with other vaccines in the 4 weeks prior to the date of study vaccine administration, with hepatitis (n = 41) and influenza (n = 18) vaccines being the most common.
Safety Analysis
Among 619 participants, 288 received dose 1, 189 participants received dose 2, and 142 received dose 3 of the study vaccine. Among 288 participants who received dose 1, 40.6% (n = 117) reported solicited reactions and 11.8% (n = 34) reported unsolicited AEs. Among 189 participants who received dose 2, 33.3% (n = 63) reported solicited reactions and 16.4% (n = 31) reported unsolicited AEs. Among 142 participants who received dose 3, 23.9% (n = 34) reported solicited reactions and 22.5% (n = 32) reported unsolicited AEs.
Overall, 44.1% (n = 273) of participants reported 618 AEs (Table 2): 15.3% (n = 95) of participants reported solicited injection-site reactions and 24.6% (n = 152) of participants reported solicited systemic reactions. A total of 15.7% (n = 97) participants reported 153 unsolicited AEs, of which 124 were unexpected AEs.
The most frequently reported AEs were solicited systemic reactions: irritability (11.0% [n = 68]), followed by somnolence (10.7% [n = 66]), crying (10.2% [n = 63]), decreased appetite (8.7% [n = 54]), and vomiting (8.2% [n = 51]).
Solicited Reactions
Overall, 34.6% (n = 214) of participants reported 465 solicited reactions (Table 3). The most frequent solicited injection site and systemic reactions were injection site swelling (7.4% [n = 46]) and irritability (11.0% [n = 68]), respectively.
Among 214 participants who experienced solicited reactions, the reactions occurred within 3 days of vaccine administration in most (81.3% [n = 174]) of them, were grade 1 (77.6% [n = 166]), grade 2 (29.9% [n = 64]), or grade 3 (5.1% [n = 11]) in intensity and lasted for 1–3 days in 79.9% (n = 171) of them. The most frequently reported grade 3 reaction was somnolence (1.1%).
Among the participants with solicited reactions, 1.9% (n = 4) of participants received medication, 1.4% (n = 3) visited an HCP, 1.9% (n = 4) were prescribed a new medication by the HCP, and 0.5% (n = 1) of participants was hospitalized, all for pyrexia, and all patients recovered. All the solicited reactions were reported to have a causal relationship with DTaP-IPV//Hib vaccine, by definition.
Overall, solicited reactions were reported by 45.8% (n = 132), 46.0% (n = 87), and 38.0% (n = 54) of the participants who received dose 1, dose 2, and dose 3 of the study vaccine, respectively. The difference in the incidences of solicited reactions by injection dose was not statistically significant (p > 0.05) (Table 4).
Unsolicited Adverse Events
Overall, 15.7% (n = 97) of participants reported 153 unsolicited AEs (Table 5). The most frequently reported unsolicited AE was cough (5.0% [n = 31]) followed by diarrhea (3.2% [n = 20]) and bronchitis (2.6% [n = 16]).
Among 97 participants who experienced unsolicited AEs, the AEs occurred post day 8 in most (74.2% [n = 72]) of them and lasted for 1–3 days in 59.8% (n = 58) of them. Among the 153 unsolicited AEs, 95.4% AEs (146 events) were of grade 1 and 4.6% AEs (7 events) were of grade 2 intensity. No unsolicited AEs of grade 3 intensity were reported.
Among the participants with unsolicited AEs, a new medication was prescribed by the HCP for 82.5% (n = 80) of participants, and 16.5% (n = 16) of participants were given an existing or over-the-counter (OTC) medicine. The most frequent AE that required prescription of a new medication was cough (26.8% [n = 26]) followed by bronchitis (16.5% [n = 16]). Nasopharyngitis (6.2% [n = 6]) was the most frequent AE for participants who were given an existing or OTC medication. Overall, 99.0% (n = 96) participants recovered (data was missing for the remaining). None of the unsolicited AEs were reported to have causal relationship with DTaP-IPV//Hib vaccine.
Unexpected Adverse Events
A total of 12.9% (n = 80) of participants reported 124 unexpected AEs. The most frequently reported unexpected AE was cough (5.0% [n = 31]) followed by bronchitis (2.6% [n = 16]) and lower respiratory tract infection (2.4% [n = 15]). None of the unexpected AEs were reported to have causal relationship with DTaP-IPV//Hib vaccine.
Serious Adverse Events
One SAE of pyrexia (solicited systemic reaction) was reported in one (0.2%) participant 2 days after vaccination with the study vaccine. The participant required inpatient hospitalization and recovered. According to the investigator, this SAE was not considered to be related to the study vaccine. No further information was provided.
Occurrence of Adverse Events by Demographic or Medical Characteristics
Demographic and medical factors, including gender, age, body weight, vaccination history, medical history, concomitant diseases, concomitant medication/vaccination, vaccine administration dose, allergic history, concomitant allergic history, and body temperature at the time of vaccination, were assessed for their effect on the incidence of AEs.
The incidence of solicited reactions was lower with increasing age of participants (odds ratio [OR] 0.83; p = 0.0006) and increasing body weight (OR 0.81; p = 0.0018), vaccination dose (p = 0.0026), and was higher in participants with no vaccination history than in those with vaccination history (p = 0.0207). For the other factors the difference was not significant. Further analysis using multiple logistic regression demonstrated that none of the factors had a statistically significant (p > 0.05) effect on the incidence of AEs.
The incidence of unsolicited AEs was higher with increasing age of participants (OR 1.21; p = 0.0042), increasing body weight (OR 1.26; p = 0.0067), and vaccination dose (p = 0.015). For the other factors the difference was not significant. Further analysis using multiple logistic regression demonstrated that none of the factors had a statistically significant effect on the incidence of AEs (p > 0.05).
Discussion
This PMS study evaluated the safety of one DTaP-IPV//Hib vaccine, PENTAXIM (Sanofi), in South Korea, after its approval on May 9, 2016, by the MFDS. It was conducted in participants aged 2 months or older who were eligible to receive one of the three primary infant series doses given at 2, 4, and 6 months of age. All participants were followed only for one vaccination, irrespective of the age/dose number. Results demonstrated an acceptable safety profile. There were no new safety findings of concern identified with this DTaP-IPV//Hib vaccine. All the solicited and unsolicited AEs were mild or moderate in intensity, most of which had a duration of 1–3 days, and all participants recovered. None of the unexpected AEs were reported to have a causal relationship with DTaP-IPV//Hib vaccine. The participant with pyrexia (SAE leading to hospitalization) recovered.
The safety findings from this study were consistent with the safety data generated from two randomized clinical trials conducted in South Korea which assessed the same DTaP-IPV//Hib vaccine [7, 22]. They were also consistent with the information listed on the local product label and are in line with the good safety profile of this DTaP-IPV//Hib vaccine reported globally [8].
The overall incidence of solicited injection-site (15.4%) and systemic (24.6%) reactions in this study was lower than those reported in the randomized clinical trial of the study vaccine in South Korea (solicited injection-site, 75.6%; systemic, 80.9%) [7], and other studies from South Korea (solicited injection-site, 73.5%; systemic, 69.0%) [22], booster study from Mexico (solicited reactions, 40.8%) [23], and a study from Turkey in which the study vaccine was co-administered with hepatitis B vaccine in the comparator arm (solicited injection-site, 38.9%; systemic, 76.30%) [24]. The incidence of solicited reactions was lower in this study, possibly because the safety was only assessed for one of the three doses of the primary series in this observational study, compared with randomized clinical trial conducted in accordance with Good Clinical Practices, in which all study participants had received three doses of the primary series. Some unsolicited and unexpected AEs were reported in this study, none of which were related to the study vaccine. None of the demographic and baseline characteristics had significant association with incidence of AEs in the logistic regression. Overall, as most of the participants may have received concomitant vaccines reflecting the real-life practice, the safety data collected was assessed conservatively, acknowledging that for systemic events it would not be possible to distinguish the role played by the study vaccine or the concomitant vaccines.
Combination multivalent vaccines provide advantages over separate vaccines targeting the same diseases: it helps to achieve better compliance to vaccination schedules, timeliness, and reduction in vaccine preparation time [21, 25, 26]. Head-to-head comparison of combination vaccines with separate vaccines in terms of immunogenicity and safety is thereby necessary. DTaP-IPV//Hib vaccine has demonstrated non-inferiority to licensed DTaP-IPV and Hib (PRP~T) vaccines, administered separately in a randomized clinical trial conducted in 418 healthy infants from South Korea [7]. Both vaccine groups had similar safety profiles, with lower injection-site reactions reported in the DTaP-IPV//Hib group [7]. The advantage of fewer injections and injection-site reactions supports the licensure and incorporation of DTaP-IPV//Hib into the Korean national vaccination schedule.
The booster dose of the study vaccine has been evaluated in multiple countries. The booster dose was well tolerated in children aged 18–19 months from India [27], was highly immunogenic and well tolerated in children aged 18–19 months from South Africa who were previously primed with the same vaccine [28], demonstrated a strong response to each antigen in the second year of life in children aged 18–20 months from China [29], was well tolerated and highly immunogenic following primary vaccination with a hexavalent vaccine in children aged 18–24 months from Thailand [20] and children aged 18 months from Argentina [30], elicited good antibody titers in the Philippines with the 0, 6, and 14-week schedule [10], and demonstrated good antibody persistence in 5–6-year-olds from France who were given the booster at 12–16 months of age [31]. These data support the use of DTaP-IPV//Hib as a toddler booster vaccine following various types and schedules of the primary series of vaccination. It also indicates the need to assess the booster dose in children from South Korea to ensure its safety and immunogenicity in clinical practice. More studies evaluating the toddler booster dose of the study vaccine can be considered in South Korea.
Real-world post-licensure safety studies not only complement the existing safety information but also help in assessing the effect of concomitant medications in healthy participants and participants with concomitant diseases. Monitoring of the safety of DTaP-IPV//Hib vaccine will be continued through voluntary reporting of post-vaccination AEs and collection of safety information.
Study Limitations
This study had some limitations. As the sample size was based on the regulatory requirement in South Korea, it was limited (approximately 600 participants), which would not necessarily allow the detection of rare AEs. Furthermore, the AEs were self-reported by parents, and this might have led to reporting bias. Additionally, as the data for each vaccination dose was collected randomly in the real-world setting, serial follow-up of the dosing data could not be evaluated. However, the vaccine safety management system in South Korea includes a rapid response system to ensure the documentation of rare or serious AEs post-vaccination [32].
Conclusion
This post-marketing surveillance study of the DTaP-IPV//Hib vaccine provided its safety profile in a real-life setting in South Korea and confirmed that the vaccine is well tolerated when used in infants aged 2 months or older for the primary series.
References
Centers for Disease Control and Prevention (CDC). Diphtheria, tetanus, and pertussis vaccines. https://www.cdc.gov/vaccines/hcp/vis/vis-statements/dtap.html#:~:text=DTaP%20vaccine%20can%20prevent%20diphtheria,failure%2C%20paralysis%2C%20or%20death, https://www.cdc.gov/polio/what-is-polio/index.htm#:~:text=Polio%2C%20or%20poliomyelitis%2C%20is%20a,move%20parts%20of%20the%20body. Assessed July 2022.
Centers for Disease Control and Prevention (CDC). Haemophilus influenzae Disease (Including Hib). https://www.cdc.gov/hi-disease/about/causes-transmission.html. Assessed July 2022.
Skibinski DA, Baudner BC, Singh M, O’Hagan DT. Combination vaccines. J Glob Infect Dis. 2011;3(1):63–72.
Jo DS, Kim JH, Choi EH, et al. Recommended immunization schedule for children and adolescents: the Korean Pediatric Society, 2013. Korean J Pediatr. 2013;56(6):231–4.
National Immunization Program for children. Korea Disease Control and Prevention Agency. https://www.kdca.go.kr/contents.es?mid=a30333000000. Assessed July 2022.
Yu JH, Jeong HJ, Kim SJ, et al. Sustained vaccination coverage during the coronavirus disease 2019 epidemic in the Republic of Korea. Vaccines. 2020;9(1):2.
Kang JH, Lee HJ, Kim KH, et al. The immunogenicity and safety of a combined DTaP-IPV//Hib vaccine compared with individual DTaP-IPV and Hib (PRP~T) vaccines: a randomized clinical trial in South Korean infants. J Korean Med Sci. 2016;31(9):1383–91.
Plotkin SA, Liese J, Madhi SA, Ortiz E. A DTaP-IPV//PRP∼T vaccine (Pentaxim): a review of 16 years’ clinical experience. Expert Rev Vaccines. 2011;10(7):981–1005.
Vidor E, Plotkin SA. Immunogenicity of a two-component (PT & FHA) acellular pertussis vaccine in various combinations. Hum Vaccines. 2008;4(5):328–40.
Capeding MR, Cadorna-Carlos J, Book-Montellano M, Ortiz E. Immunogenicity and safety of a DTaP-IPV//PRP approximately T combination vaccine given with hepatitis B vaccine: a randomized open-label trial. Bull World Health Organ. 2008;86(6):443–51.
Dutta AK, Verghese VP, Pemde HK, Mathew LG, Ortiz E. Immunogenicity and safety of a pentavalent diphtheria, tetanus, acellular pertussis, inactivated poliovirus, Haemophilus influenzae type B conjugate combination vaccine (Pentaxim) with hepatitis B vaccine. Indian Pediatr. 2009;46(11):975–82.
Kanra G, Silier T, Yurdakök K, et al. Immunogenicity study of a combined diphtheria, tetanus, acellular pertussis, inactivated poliomyelitis vaccine used to reconstitute a freeze-dried Haemophilus influenzae type b vaccine (DTaP-IPV//PRP-T) administered simultaneously with a hepatitis B vaccine at two, three and four months of life. Vaccine. 1999;18(9–10):947–54.
Li RC, Li FX, Li YP, et al. Immunogenicity and safety of a pentavalent acellular pertussis combined vaccine including diphtheria, tetanus, inactivated poliovirus and conjugated Haemophilus influenzae type b polysaccharide for primary vaccination at 2, 3, 4 or 3, 4, 5 months of age in infants in China. Vaccine. 2011;29(10):1913–20.
Lagos R, Kotloff K, Hoffenbach A, et al. Clinical acceptability and immunogenicity of a pentavalent parenteral combination vaccine containing diphtheria, tetanus, acellular pertussis, inactivated poliomyelitis and Haemophilus influenzae type b conjugate antigens in two-, four- and six-month-old Chilean infants. Pediatr Infect Dis J. 1998;17(4):294–304.
Carlsson RM, Claesson BA, Selstam U, et al. Safety and immunogenicity of a combined diphtheria-tetanus-acellular pertussis-inactivated polio vaccine-Haemophilus influenzae type b vaccine administered at 2-4-6-13 or 3-5-12 months of age. Pediatr Infect Dis J. 1998;17(11):1026–33.
Mallet E, Fabre P, Pines E, et al. Immunogenicity and safety of a new liquid hexavalent combined vaccine compared with separate administration of reference licensed vaccines in infants. Pediatr Infect Dis J. 2000;19(12):1119–27.
Carlsson RM, Claesson BA, Fagerlund E, Knutsson N, Lundin C. Antibody persistence in five-year-old children who received a pentavalent combination vaccine in infancy. Pediatr Infect Dis J. 2002;21(6):535–41.
Madhi SA, Cutland C, Jones S, Groome M, Ortiz E. Immunogenicity and safety of an acellular pertussis, diphtheria, tetanus, inactivated poliovirus, Hib-conjugate combined vaccine (Pentaxim) and monovalent hepatitis B vaccine at 6, 10 and 14 weeks of age in infants in South Africa. S Afr Med J. 2011;101(2):126–31.
Thisyakorn U, Chotpitayasunondh T, Pancharoen C, Chuenkitmongkol S, Ortiz E. Evaluation of an acellular pertussis, diphtheria, tetanus, inactivated poliovirus, Hib-conjugate combined vaccine (Pentaxim) at 2, 4, and 6 months of age plus hepatitis B vaccine at birth, 2, and 6 months of age in infants in Thailand. Southeast Asian J Trop Med Public Health. 2010;41(2):450–62.
Thisyakorn U, Pancharoen C, Chuenkitmongkol S, Ortiz E. Immunogenicity and safety of a DTaP-IPV//PRP-T vaccine (Pentaxim) booster during the second year of life in Thai children primed with an acellular pertussis combined vaccine. Southeast Asian J Trop Med Public Health. 2009;40(2):282–94.
Maman K, Zöllner Y, Greco D, Duru G, Sendyona S, Remy V. The value of childhood combination vaccines: from beliefs to evidence. Hum Vaccines Immunother. 2015;11(9):2132–41.
Kim YK, Vidor E, Kim HM, et al. Immunogenicity and safety of a fully liquid DTaP-IPV-HB-PRP∼T hexavalent vaccine compared with the standard of care in infants in the Republic of Korea. Vaccine. 2017;35(32):4022–8.
Melo FIR, Morales JJR, De Los Santos AHM, Rivas E, Vigne C, Noriega F. Immunogenicity and safety of a booster injection of DTap-IPV//Hib (Pentaxim) administered concomitantly with tetravalent dengue vaccine in healthy toddlers 15–18 months of age in Mexico: a randomized trial. Pediatr Infect Dis J. 2017;36(6):602–8.
Ceyhan M, Yıldırım İ, Tezer H, Devrim İ, Feroldi E. A fully liquid DTaP-IPV-HB-PRP-T hexavalent vaccine for primary and booster vaccination of healthy Turkish infants and toddlers. Turk J Med Sci. 2017;47(4):1247–56.
Tafreshi SH. Efficacy, safety, and formulation issues of the combined vaccines. Expert Rev Vaccines. 2020;19(10):949–58.
Obando-Pacheco P, Rivero-Calle I, Gómez-Rial J, Rodríguez-Tenreiro Sánchez C, Martinón-Torres F. New perspectives for hexavalent vaccines. Vaccine. 2018;36(36):5485–94.
Dutta AK, Verghese VP, Pemde H, Mathew LG, Ortiz E. Immunogenicity and safety of a DTaP-IPV//PRP~T vaccine (Pentaxim) booster dose during the second year of life in Indian children primed with the same vaccine. Indian Pediatr. 2012;49(10):793–8.
Madhi SA, Cutland C, Jones S, Groome M, Ortiz E. One-year post-primary antibody persistence and booster immune response to a DTaP-IPV//PRP~T vaccine (Pentaxim) given at 18–19 months of age in South African children primed at 6, 10 and 14 weeks of age with the same vaccine. S Afr Med J. 2011;101(12):879–83.
Li RC, Li FX, Li YP, et al. Antibody persistence at 18–20 months of age and safety and immunogenicity of a booster dose of a combined DTaP-IPV//PRP∼T vaccine compared to separate vaccines (DTaP, PRP∼T and IPV) following primary vaccination of healthy infants in the People’s Republic of China. Vaccine. 2011;29(50):9337–44.
Tregnaghi M, Zambrano B, Santos-Lima E. Antibody persistence after a primary series of a new DTaP-IPV-Hep B-PRP-T combined vaccine or separate DTaP-IPV//PRP-T and hepatitis B vaccines at 2, 4, and 6 months of age and the effect of a subsequent DTaP-IPV//PRP-T booster vaccination at 18 months of age in healthy Argentinean infants. Pediatr Infect Dis J. 2012;31(1):e24-30.
Mallet E, Matisse N, Mathieu N, Langue J, Boisnard F, Soubeyrand B. Antibody persistence against diphtheria, tetanus, pertussis, poliomyelitis and Haemophilus influenzae type b (Hib) in 5-6-year-old children after primary vaccination and first booster with a pentavalent combined acellular pertussis vaccine: immunogenicity and tolerance of a tetravalent combined acellular pertussis vaccine given as a second booster. Vaccine. 2004;22(11–12):1415–22.
Choe YJ, Bae G-R. Management of vaccine safety in Korea. Clin Exp Vaccine Res. 2013;2(1):40–5.
Acknowledgements
The authors would like to thank all participants who volunteered to take part in the study, the primary investigators, and their site staff.
Funding
This study and the journal’s Rapid Service Fee were both funded by Sanofi.
Medical Writing Assistance and Other Assistance
Medical writing support for this manuscript was provided by Saili Dharadhar (Sanofi), and manuscript coordination support was provided by Roopsha Brahma, PhD (Sanofi).
Authorship
All authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work, and have given their approval for this version to be published.
Authors’ Contributions
JK, KY, and EO contributed to conceptualization of the study, data acquisition, and data analysis and/or interpretation. CH and HK contributed to data analysis and/or interpretation. All authors read and approved the final manuscript and are accountable for accuracy and integrity of the data presented therein.
Disclosures
HK, EO, and CH are employees of Sanofi and may hold stocks in the company. JK received grants from Korea Disease Control and Prevention Agency, consulting fees from HK Innoen, and honoraria from Pfizer. The current affiliation of KY is Gangneung Asan Hospital, Gangneung, South Korea. KY was an employee of Sanofi at the time of the study.
Compliance with Ethics Guidelines
The trial was approved by The Catholic University of Korea Seoul St Mary’s Hospital (Central institutional review board [IRB]; approval number KC18MODP0049). Per South Korea’s pharmaceutical affairs act, only one central IRB approved the study on behalf of all other study sites. Other study sites were notified of the approval by the central IRB. The study was conducted in accordance with the guidelines established by the Declaration of Helsinki and MFDS Notification (Standards for Re-examination of New Drugs). Informed consent was obtained from all participants or their parents/legal representative before enrollment.
Data Availability
The datasets generated during and/or analyzed during the current study are not publicly available but are available from the corresponding author on reasonable request. Qualified researchers may request access to patient-level data and related documents. Patient-level data will be anonymized, and study documents will be redacted to protect the privacy of trial participants. Further details on Sanofi’s data sharing criteria, eligible studies, and process for requesting access can be found at https://www.vivli.org/.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Yang, K., Kim, H., Ortiz, E. et al. Post-Marketing Safety Surveillance of a Childhood Pentavalent Diphtheria–Tetanus–Acellular Pertussis–Polio and Haemophilus influenzae Type B (DTaP-IPV//Hib) Vaccine in South Korea. Infect Dis Ther 12, 499–511 (2023). https://doi.org/10.1007/s40121-022-00724-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40121-022-00724-7