Abstract
Introduction
Polypharmacy is a growing problem in the United States. The use of multiple medications increases the likelihood that a patient will experience potential drug interactions and adverse drug reactions (ADRs). Those individuals with dementia or Alzheimer’s disease (AD) are at greater risk, due to age, comorbidities, and an increased likelihood of being on a greater number of neuroactive medications.
Methods
uMETHOD Health (uMH) has developed a precision medicine platform to address dementia and mild AD through the creation of personalized, multidomain treatment plans. Many interactions and ADRs may be observed, such as drug–drug interactions (DDIs), drug–gene interactions (DGIs), anticholinergic cognitive burden (ACB), and depression-inducing drugs (DIDs). uMH’s algorithms can parse these interactions, rate them based on input from open-source databases, and then record all these interactions in a generated treatment plan. A total of 295 individuals aged 65 and older were included in this analysis.
Results
Of 295 individuals, 97.59% were on at least one medication, with an overall mean of 11.5 medications per person; 83.66% were on five or more medications. A total of 102 DGIs, 3642 DDIs, and one high-priority DDI were found in this population. There was a significant increase in the number of DDIs as medications per person increased (P value < 0.0001). Of the population, 65.86% were on one or more anticholinergic drugs. There was a significant difference in the ACB score between individuals with cognitive decline and those without. In total, 60.98% of the overall population were on DIDs, with a mean of 1.19 medications per person.
Conclusions
The results of this work show that older populations have a high medication burden. With the growing elderly and AD populations, medication management for polypharmacy is a need that grows direr every year. uMH’s platform was able to identify a multitude of polypharmacy problems that individuals are currently facing.
Funding
uMETHOD Health.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Polypharmacy, defined as the use of five or more medications [1], is a growing problem in the United States [2]. Medication regimes comprised of multiple medications increase the likelihood that a patient will experience potential drug interactions and adverse drug reactions (ADRs) [3]. ADRs place many patients in the hospital every year and can have life-threatening consequences. They are among the top ten leading causes of death [2, 4].
These risks are even higher for the elderly, due to age-related changes of pharmacokinetics (PK) and pharmacodynamics (PD) of many drugs. Changes in PK and PD are related to an overall decrease in total body water which decreases the spread of hydrophilic drugs, increasing body fat that in turn increases the distribution of lipophilic drugs, and a breakdown in the blood–brain barrier that results in a higher distribution of drugs that affect the central nervous system [3]. Moreover, age-related diseases can further change patients’ PK and PD by increasing sensitivity to drug effects and thus increasing their susceptibility to ADRs.
Among those taking any prescription medication, half are exposed to two or more drugs with 5% exposed to eight or more [2]. This high rate of multiple medications is driven in large part by people who have multiple chronic conditions. Approximately half of the US population has at least one chronic condition, and one in four have multiple [5]. Those individuals with dementia or Alzheimer’s disease (AD) are also at greater risk due to age, an increased number of comorbidities, and an increased likelihood of being on a greater number of neuroactive medications. In one study, only 8.7% of AD patients did not have a physical condition, compared to 15.9% of controls [6].
AD is a progressive neurodegenerative condition in which individuals exhibit memory loss, dementia, and impaired metabolism. It is commonly a late-onset disease, with symptoms developing around the age of 65. AD is one of the most common forms of dementia, accounting for 50–80% of all dementia cases [7] and is a growing economic and social burden [8, 9]. Early symptoms include difficulty in recalling recent events, personality changes, trouble with problem-solving, and confusion. As the disease progresses, symptoms include mood swings, irritability, aggression, trouble with language, and long-term memory loss. In the late stages of AD, bodily functions are lost, leading to death. Life expectancy after diagnosis is 7 years [7]. Comorbidity and polypharmacy are both associated with worsening cognition, functional ability, and survival for patients with dementia [6].
AD patients may be at greater risk for ADRs when it comes to neuroactive drugs compared to a normal elderly population, due to the neurological damage that is characteristic of the disease. According to Dr. Pasqualetti at the University of Pisa, “the impaired neurotransmission in AD patients represents the major pharmacological target (especially cholinergic system) for disease treatment, it might also represent a point of weakness for AD patients” [3].
Both chronic diseases and medication numbers increase when considering a population that is 65 and older. Polypharmacy has been shown to be a statistically significant predictor of hospitalization, nursing home placement, death, hypoglycemia, fractures, impaired mobility, pneumonia, and malnutrition for older populations [10]. In the United States, 30% of adults over 65 take 5 or more medications [2].
Each drug has the potential to cause side effects and adverse events, but drugs also interact. These interactions can be so minor as to be recommended to ignore, or they can be very serious, with the potential for death with concurrent use [11]. Interactions and adverse events can arise from multiple factors, including drug–drug interaction (DDI), drug–genome interaction (DGI), anticholinergic burden (ACB), and depression-inducing drugs (DIDs), creating new risks for the patient with each additional medication. Indeed, the inability of patients and providers alike to distinguish drug-induced symptoms from new disease symptoms often results in the addition of more medications to treat drug-induced symptoms, further increasing the risk of DDIs and ADRs [3].
DDIs occur when individuals are exposed to medications that interact to produce an undesirable result. They fall into two categories: PK and PD interactions [12]. Exposure to potential DDIs does not always result in a direct adverse drug event or measurable clinical effect, but they can have an additive effect [12, 13]. As Katie Quinn of Stanford University states in “A dataset quantifying polypharmacy in the United States,” “exposure to multiple drugs puts patients at additive risk of each single drug’s potential adverse outcomes” [2].
Genetic factors also affect the PK and PD of drugs, altering patients’ response [14,15,16]. In recent years, databases have been created to track and aid in the study of DGIs. This has become known as the “druggable genome,” and is defined as “genes or gene products that are known or predicted to interact with drugs, ideally with a therapeutic benefit to patients” [14].
Anticholinergic drugs block the neurotransmitter acetylcholine in the central and peripheral nervous systems. They are indicated for use in many different disease states, including depression, gastrointestinal disorders, Parkinson’s disease, urinary incontinence, epilepsy, and seasonal allergies [17]. In fact, many commonly used drugs have anticholinergic properties, including over-the-counter-medications. These drugs are widely available and include antihistamines, drugs to reduce frequency of urination, and medications for sleep disturbances. The availability and number of non-prescription drugs with anticholinergic properties is increasing in recent years [18]. The cumulative effect of taking multiple medications with anticholinergic properties is referred to as ACB [19].
Many studies have shown that anticholinergic drugs can adversely impact cognition, physical function, cause dizziness, delirium, confusion, falls, and increase the risk of mortality in elderly populations [19, 20]. ACB has also been shown to be a risk factor for developing mild cognitive impairment (MCI) and dementia [17, 21,22,23]. Each definite anticholinergic drug has the potential to increase the risk of cognitive impairment by 46% over 6 years [21]. In addition, the odds ratio for a diagnosis of MCI was 2.73 for adults exposed to at least 3 anticholinergics for a minimum of 90 days. The odds ratio for dementia was 0.43 [22].
Many cognitively impaired and AD patients are simultaneously taking cholinesterase inhibitors along with anticholinergic drugs. Cholinesterase inhibitors are one of the most prescribed drugs types for dementia and AD patients [24]. One study showed that 35.4% of patients received these two drug types in combination [25]. The use of these medications in combination is likely to decrease their pharmacological benefit as they counteract each other [26], making their simultaneous use seldomly appropriate due to the drugs’ invalidating pharmacological effect [25].
Guidelines such as Beers and the Screening Tool of Older Persons’ potentially inappropriate Prescriptions (STOPP) both state that the use of anticholinergic drugs should be avoided in older populations [17, 23]. Even with these accepted guidelines, physicians often prescribe these medications for their anticipated therapeutic outcomes, overlooking the possible risks [19].
Anticholinergic drugs are also one of the most commonly prescribed DIDs. Numerous medications and medication classes have been classified as depression-inducing. Other common DIDs include sedative-hypnotics and analgesics [27, 28]. Drug-induced depression is more likely to occur in individuals who have a higher risk factor for depressive disorder [29], as may be the case for individuals with the APOE ε4 allele and AD patients.
An increase in drug-induced depression is another result of the increasing occurrence of polypharmacy [30]. Many studies that identify potential DIDs are observational in nature, and results have been contradictory. Nevertheless, DIDs should be considered as a potential cause when new symptoms of depression occur [31].
The elderly are less likely to recognize ADRs for what they are and can even be more likely to have their concerns dismissed by medical providers. Elderly people often have lower expectations for what their health should be, and are therefore less likely to let their physician know when they are experiencing new issues. Those with cognitive impairment may also be unable to recognize new symptoms or may have difficulty communicating them. In some cases, ADRs are mistaken for the consequences of old age [18].
The aging population along with the number of people diagnosed with AD are quickly growing in the United States. It is estimated that by 2030, more than 20% of all US residents will be 65 or older, compared with just 13% in 2010 [32].
With the quick growth of these populations and the ever-increasing number of drugs available on the market, medication management is necessary, but too complex to be handled by a single physician. We propose that clinical decision support software (CDSS) with machine-learning algorithms should be employed in this complicated space to increase physician effectiveness and patient safety.
CDSS is software created to provide treatment decision support to medical professionals. CDSS can process large amounts of information in a quick and effective manner, creating actionable recommendations to improve clinical decision-making [33, 34]. Because of this ability, the guidance provided is patient-specific instead of generalized [34]. The output of CDSS contains guidance on the diagnosis, treatment, prevention, cure, or mitigation of disease [35].
Machine learning enables the CDSS platforms to make successful predictions based on past experiences [36, 37]. There may be dozens of issues identified for one patient, so decision-theory techniques can be used to assign weights, priorities, and strategies to the issues. Next, interventions are selected. These interventions have a wide range of costs, including financial costs, pain, and effort by the person being treated which should be taken into account by the CDSS platform. Many interactions may be observed, such as DDIs, DGIs, and drug-to-diet interactions. Algorithms determine an appropriate path forward, given the many potential conflicts.
This method would enable physicians to have access to an automated, repeatable, and reliable analysis that can be applied to every patient, is based on the latest clinical guidelines, and is personalized to the individual. It can process copious amounts of information quickly, making it useful in a clinical setting.
Methods
Population Overview
A total of 295 individuals were included in this analysis. All of the data presented here was previously collected for the purposes of creating treatment plans for patients with a family history of AD, with mild cognitive decline, or mild AD.
Following the guidelines put forth in the 21st Century Cures Act [35], CDSS is not regulated by the US Food and Drug Administration (FDA), and uMETHOD Health’s (uMH) treatment protocol does not include any investigational drugs. Because of this, uMH’s precision medicine platform was taken straight to market and did not need an approval by an ethics committee or board.
When participants were enrolled for the treatment plan, uMH obtained permission from every individual to use their data for future research purposes, in compliance with ethics guidelines. uMH’s Terms and Conditions for the commercially available treatment plan outlined that data would be saved and used only in a de-identified manner.
Each participant receiving a treatment plan accessed an online consent form or was given a printed version if access was an issue, outlining uMH’s Terms and Conditions for the commercially available treatment plan. Individuals who presented with cognitive decline were encouraged by uMH and their physician to have a caretaker or family member review all documents and consent forms before signing.
Before initial enrollment, all individuals were first evaluated by a physician to ensure they were a good fit for the program. Cognitive testing was done at baseline and these results were used to classify the individuals as either cognitively normal or demented. Cut-off scores for dementia classification followed the University of Ohio’s validity and normality data for the Self-administered Gerocognitive Examination (SAGE) with a score of 17–22 considered normal cognition [38, 39].
uMH’s algorithm platform automatically assigns every participant a random eight-character ID when they are first entered into the system to de-identify their information. All data was collected under their randomly assigned ID, and the investigators here only had access to this de-identified information.
Following the guidelines set out by “Coded Private Information or Specimens Use in Research” by the Office for Human Research Protection [40], since the data presented here was not collected for the currently proposed polypharmacy research project and no investigator could ascertain the identity of the individuals, the data presented here is not classified as involving human subjects.
Institutional review board approval was not required as data are recorded in such a manner that subjects cannot be identified, directly or through identifiers linked to the subjects. Meeting these conditions makes this research exempt from the requirements of 45 CFR 46 under the Department of Health and Human Services (HHS): Research, involving the collection or study of existing data, documents, records, pathological specimens, if these sources are publicly available or if the information is recorded by the investigator in such a manner that subjects cannot be identified, directly or through identifiers linked to the subjects.
For the purposes of this analysis, only individuals who were aged 65 and older were included. Our population was 55.25% female (n = 163). See Tables 1 and 2 for a full analysis of the research cohort.
Bioinformatics Platform
uMH has developed a precision medicine platform to address dementia and mild AD through the creation of personalized, multidomain treatment plans [41]. Large data sets are collected and analyzed for each patient to generate a treatment plan. Each treatment plan is reviewed by a trained physician before being delivered to the patient. uMH’s platform identifies and addresses active issues, and creates repeatable and practical treatment plans for use in doctors’ practice.
Many interactions and ADRs may be observed, such as DDIs, DGIs, ACB, as well as DIDs. uMH’s algorithms can parse these interactions, rate them based on input from open-source databases, and then record all these interactions in the person’s generated treatment plan. This allows a physician to easily review and amend a patient’s medication regime.
The internal software of the bioinformatics platform is written in the Python language. It interfaces to an external portal, used by the medical, care, and coaching teams to gather input and return reports written in PHP and Java. External medical databases support the internal rules-processing algorithms. These are sourced from bodies such as the National Institutes of Health (NIH), FDA, pharmaceutical trade groups, and consortia focused on topics such as genetics or allergies.
The initial data input for a person is often about one million data points. This count can vary (generally upwards), depending on several input categories: (1) the completeness of the genetic exome data, (2) the resolution and number of images and image files, (3) the number of historical biospecimen lab results and cognitive assessments, (4) the granularity and history of wearable data samples, and (5) any attached photos, scans, and faxes. Natural language processing (NLP) techniques are used throughout the input steps, particularly for precise identification of lab tests, medications, drug indications, and comorbidities. A range of NLP techniques are employed to normalize input data.
The algorithms that implement this information platform go through a consistent set of steps each time they load a person’s input to generate a new set of reports. These steps are rules-based, so the logic and evidence sources can be tracked (and evaluated). When ADRs are determined, the issues are described in detail, and the physician and care team are alerted.
Every word in every report is generated by the bioinformatics algorithms. Natural language generation (NLG) techniques assure all text, tables, and images are human-readable, in high-quality natural language (such as American English). Multiple versions of the reports are generated suited to the presumed education (e.g., physician), reading ability (e.g., those under treatment and their caregivers), and vocabulary (e.g., dietitian, coach, physical therapist) of the readers.
Input
Data was collected using either paper forms or uMH’s online portal depending on the individual’s computer skills and access.
Personal medical histories were gathered from forms completed by the individual or their caregiver. This information includes current medications, nutraceuticals, over-the-counter drugs, comorbidities, past procedures and surgeries, allergies, imaging such as MRI, EEG, or PET scans, immunization history, and family history of dementia or cardiovascular conditions.
Lifestyle data, information on sleep, diet, stress, educational attainment, physical activity, quality of life, activities of daily vitals, and biometrics were also supplied by the individual, their caregiver, and/or the physician.
Measures
All medications, including prescriptions, vitamins, and over-the-counter drugs, reported were counted toward each person’s total medication number. Three medication categories were chosen for statistical analysis due to their large prevalence, and in some cases accepted guidelines for avoidance, in older and AD populations: neuroactive medications, anticholinergics, and DIDs.
Drug–Drug Interactions
Information on each person’s current medication regimes was collected through uMH’s patient questionnaires or supplied by physicians.
uMH’s CDSS platform utilized existing national databases to determine interactions. DDIs were determined using the NIH RxNorm database, including the “ONC High” subset (ONC is the Office of the National Coordinator for Health Information Technology). The RxNorm database is a normalized naming system for generic and brand-name drugs produced by the National Library of Medicine [42]. The RxNorm system is used by hospitals, pharmacies, and other organizations nationwide to record and process drug information through computer systems. The goal of RxNorm “is to allow computer systems to communicate drug-related information efficiently and unambiguously.” The RxNorm database covers clinical drugs and drug packs. Each drug in the RxNorm database receives a RxNorm concept unique identifier (RxCUI). The RxCUI is normalized to a numeric code that computers are able to parse [42].
DDIs are determined pairwise, using the RxNorm RxCUI code for each of the two drugs. The RxCUIs identify the specific brand of medication (if supplied by the participant), or their generic form (if no brand name was supplied). uMH’s bioinformatics platform has a Representational State Transfer (REST) open application-programming interface (API) to the RxNorm database. Results are cached to speed processing.
DDI priority was derived from the Lexicomp rating system [11]. Lexicomp’s DDI risk-rating scale stratifies risk to provide medical professionals with relevant information about the potential urgency associated with each interaction. The informatics platform examines all DDIs. Those that have a rating of C or above are identified to physicians in uMH’s reports as clinically significant. Those with a rating of D or X are considered high-priority DDIs, as these drug combinations are contraindicated.
Drug–Genome Interactions
uMH’s platform mines publicly available databases to check for DGIs in each individual. Individuals' raw data files of genomic information were collected from consumer-focused companies such as 23andMe or Ancestry.com. Alternatively, a full-exome VCF file of genomic data can be read. The platform analyzes more than 2000 single nucleotide polymorphisms (SNPs) per individual, some of which pertain to DGIs, and others are used to quantify the genetic risk of AD as well as other genetic diseases.
DGI information was drawn from three sources of pharmacogenomic information: the Clinical Pharmacogenetics Implementation Consortium (CPIC) effort [43], DrugBank [13, 44], and SNPedia’s SNP and genoset compilations [45, 46]. DGIs are looked at in two categories: genetics that influence how enzymes metabolize each drug, and relationships between specific genes and drugs. Each medication a person is currently taking is compared against a table of genes that affect it, and these genes are, in turn, compared to those in the person’s genome. CPIC rates each DGI on a scale from A to D, where prescribing action is recommended for levels A or B, but not necessarily for levels C or D. No DGI priorities are assigned in the DrugBank or SNPedia databases.
Anticholinergic Burden
ACB is measured using the ACB scale. This scale was created by expert consensus to rank each medication with known anticholinergic activity from high to low. Each drug that has anticholinergic properties is assigned a score of 1–3 depending on the severity. The list is currently comprised of 195 different medications [19, 21].
Drugs with possible ACB are assigned a value of 1, and those with definite anticholinergics are given a value of 2 or 3. To get the total ACB score for a patient, the value attributed to each medication is summed. Each point a patient has on the ACB scale has been correlated to a decline in mini-mental state examination (MMSE) score of 0.33 over 2 years [21].
The use of the ACB scale in clinical practice is burdensome and challenging, which limits its use in physicians' offices. Although some electronic health systems do flag drugs with ACB, most physicians would have to look up the drugs and their ratings. Hospitals and care homes are most likely to use the scale. The scale is often used for research purposes as well [47,48,49,50,51].
uMH’s platform examined each medication being taken by a person, and determined the ACB score for each. A score (0 through 3) is assigned to each current medication. The cumulative score was calculated, and an estimate was made of how much these drugs might be lowering someone’s MMSE score. Definite ACBs (those with a score of 2 or 3) are tabulated, as those can directly contribute to cognitive decline. A meta-review of ACB medications, which are implemented in this informatics system, can be found in “Anticholinergic burden quantified by anticholinergic risk scales and adverse outcomes in older people: a systematic review” [19].
Depression-Inducing Drugs
DIDs were identified using “Prevalence of Prescription Medications with Depression as a Potential Adverse Effect Among Adults in the United States” [30]. The DIDs listed here were coded into uMH’s informatics platform so that each person’s medication list could be checked against this published list by the algorithms.
Output
Each person’s information was analyzed by the informatics platform and compared to standardized databases, peer-reviewed publications, and reference tables to generate treatment recommendations. Databases and reference tables used were sourced from the Centers for Disease Control, FDA, NIH, DrugBank, and the Online Mendelian Inheritance in Man (OMIM) catalogue. The information in these databases and tables relates to SNPs, DDIs, DGIs, drug indications, and diagnostics.
Along with treatment recommendations, the informatics platform also looks for any and all drug interactions or potential ADRs (see Fig. 1).
Separate care plan reports generated by the software are optimized for the physician, the person under treatment, and their coaching team—with the goal of suggesting what can best be achieved in a clinical setting in the long-term, taken in three-month segments.
Statistical Analysis
All statistical analyses were performed using SAS University Edition. Significance was determined using a 95% confidence interval.
Power calculations for independent, two-sided t tests were completed to compare population means. Either pooled or Satterthwaite P values were used based on equality of variance testing.
Means, standard deviations, and frequencies were established using SAS. One-way analysis of variance (ANOVA) testing was completed to compare mean values between independent groups with categorical and continuous analysis variables. Linear and multiple linear regression models were completed for continuous response variables and singular or multiple continuous explanatory variables.
Results
Of the 295 people analyzed in this group, 97.59% of them were on at least one medication, with an overall mean of 11.5 medications per person; 83.66% were on 5 or more medications (see Tables 3, 4). No significant differences were found between the total number of medications for people with normal cognitive status versus those with cognitive decline.
A higher percentage of people with cognitive decline were taking medications in all the drug categories reviewed: neuroactive medications, anticholinergic drugs, and DIDs. Those with cognitive decline also took more medications for these drug classes compared to those with no cognitive decline (see Tables 5, 6, 7).
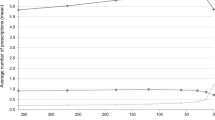
A total of 102 DGIs, 3642 DDIs, and one high-priority DDI were found in this population. Only ten people on medications did not have any observed DDIs or DGIs. There was a significant increase in the number of DDIs as the number of medications per person increased (P value < 0.0001; see Fig. 2).
Neuroactive Medications
A total of 97.23% of people were on at least one neuroactive medication, with a mean of 2.82 medications per person. Females were on a significantly higher amount of neuroactive medications than males (3.03 vs. 2.55, P value 0.02; see Table 5). Those with cognitive decline were also on more neuroactive medications than individuals with normal cognition (3.00 vs. 2.16, P value 0.0004; see Table 5).
Anticholinergics
Of the population, 65.86% were on one or more anticholinergic drug. There was a significant difference in the ACB score between individuals with cognitive decline than those without (2.39 vs. 1.28, P value 0.01). Females were also placed on more anticholinergic drugs than males (1.67 vs. 1.13, P value 0.002), and had a higher ACB score (2.50 vs. 1.71, P value 0.006; see Table 5).
Depression-Inducing Drugs
Females were on a significantly higher number of DIDs than males (1.34 vs. 0.99, P value 0.01). In total, 60.98% of the overall population were on DIDs, with a mean of 1.19 medications per person (see Table 5).
Discussion
The results of this work show that older populations have a high medication burden. Patients had an average of ten DDIs alone. The mean duration of a physician visit in the United States is 21.07 min. [52], most of which is spent discussing a person’s current health and not their medication regime. The numbers presented here as well as in other publications show that physicians and patients need more support to ensure medication regimes are safe.
Those with cognitive impairment face an even higher burden than the average elderly patient, taking more medications that can adversely impair their cognitive abilities and mood.
Females in this population were placed on a higher number of neuroactive and mood-altering drugs. Females are already at a higher risk of developing AD [53,54,55], nearly two thirds of AD patients are women [56]. While our research cannot say if a higher risk of cognitive decline results in more medications or a more complex medication regime, it is apparent that females are at greater risk of drug interactions and ADRs.
With the growing elderly and AD populations, medication management for polypharmacy is a need that grows direr every year. Studies have shown that adding a medical professional with the sole job of medication review to reduce the use of ineffective medications increases patient safety [1], but this remains unrealistic in the real world due to cost and personnel constraints. CDSS does not face the same concerns though.
uMH’s CDSS was able to identify a multitude of polypharmacy problems that individuals are currently facing. With the added knowledge that this type of algorithmic analysis can provide, physicians will have the tools and information needed to amend a person’s medications to avoid interactions and ADRs, and to more closely monitor patients for expected reactions.
While the use of electronic medical records (EMRs) is becoming more prevalent in large hospitals and even private practices, it does not mean that physicians are taking more actions to prevent drug interactions in their patients. Studies have shown that between 33% and 96% of medication-related EMR alerts are overridden and ignored [57]. This is referred to as “alert fatigue.” Even clinically significant alerts are being ignored [57, 58].
The reports generated by uMH’s informatics platform do not allow physicians to dismiss alerts, as it is provided in an un-editable PDF format as opposed to a dismissible alert in an EMR. uMH’s report format allows for review of the recommendations and findings before a patient visit, and presents alerts and recommendations in a consistent format, allowing physicians to focus on whatever piece of the review is relevant at the time and return to others later.
The population presented here was compared to the National Alzheimer’s Coordinating Center’s (NACC) database to check how closely they resemble a more generalized population. Only those aged 65 and older in the NACC database were used to match the age range for the population presented here. We found that our population was comparable in both education, BMI, and age at visit. uMH’s population was on more total medications than those in the NACC's database (see Table 8).
Conclusions
This informatics platform has immense potential for use in clinical practice and helps fulfill the need of increasing patient medication regime safety. It could be further improved by the addition of actionable recommendations for how physicians can safely reduce the medications causing interactions and ADRs. These recommendations could include stopping medications (with information on how to safely wean patients off specific drugs), dosage changes based on precision medicine, and formulary alternatives that would provide the same pharmaceutical benefit without the potential for interactions.
References
Maher RL, Hanlon J, Hajjar ER. Clinical consequences of polypharmacy in elderly. Expert Opin Drug Saf. 2014;13(1):57–65.
Quinn KJ, Shah NH. A dataset quantifying polypharmacy in the United States. Sci Data. 2017;31(4):170167.
Pasqualetti G, Tognini S, Calsolaro V, Polini A, Monzani F. Potential drug–drug interactions in Alzheimer patients with behavioral symptoms. Clin Interv Aging. 2015;8(10):1457–66.
Lazarou J, Pomeranz BH, Corey PN. Incidence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies. JAMA. 1998;279(15):1200–5.
Ward BW, Schiller JS, Goodman RA. Multiple chronic conditions among US adults: a 2012 update. Prev Chronic Dis. 2014;17(11):E62.
Clague F, Mercer SW, McLean G, Reynish E, Guthrie B. Comorbidity and polypharmacy in people with dementia: insights from a large, population-based cross-sectional analysis of primary care data. Age Ageing. 2017;46(1):33–9.
What is Alzheimer’s|Alzheimer’s Association (Internet). https://www.alz.org/alzheimers-dementia/what-is-alzheimers. Accessed 22 Oct 2018.
World Health Organization. First WHO ministerial conference on global action against dementia. Geneva: World Health Organization; 2015.
Winblad B, Amouyel P, Andrieu S, Ballard C, Brayne C, Brodaty H, et al. Defeating Alzheimer’s disease and other dementias: a priority for European science and society. Lancet Neurol. 2016;15(5):455–532.
Frazier SC. Health outcomes and polypharmacy in elderly individuals: an integrated literature review. J Gerontol Nurs. 2005;31(9):4–11.
Interactions|Clinical Drug Information (Internet). https://www.wolterskluwercdi.com/lexicomp-online/user-guide/tools-interactions/. Accessed 26 Oct 2018.
Wiese K, Ellingrod V. Avoiding common drug–drug interactions. Curr Psychiatr. 2015;14(7):21–3.
Ayvaz S, Horn J, Hassanzadeh O, Zhu Q, Stan J, Tatonetti NP, et al. Toward a complete dataset of drug–drug interaction information from publicly available sources. J Biomed Inform. 2015;55:206–17.
Griffith M, Griffith OL, Coffman AC, Weible JV, McMichael JF, Spies NC, et al. DGIdb: mining the druggable genome. Nat Methods. 2013;10(12):1209–10.
Cotto KC, Wagner AH, Feng Y-Y, Kiwala S, Coffman AC, Spies G, et al. DGIdb 3.0: a redesign and expansion of the drug-gene interaction database. Nucleic Acids Res. 2018;46(D1):D1068–D1073.
Pirmohamed M. Personalized pharmacogenomics: predicting efficacy and adverse drug reactions. Annu Rev Genomics Hum Genet. 2014;29(15):349–70.
Richardson K, Fox C, Maidment I, Steel N, Loke YK, Arthur A, et al. Anticholinergic drugs and risk of dementia: case–control study. BMJ. 2018;25(361):k1315.
Mintzer J, Burns A. Anticholinergic side-effects of drugs in elderly people. J R Soc Med. 2000;93:457–62.
Salahudeen MS, Duffull SB, Nishtala PS. Anticholinergic burden quantified by anticholinergic risk scales and adverse outcomes in older people: a systematic review. BMC Geriatr. 2015;25(15):31.
Gorup E, Rifel J, Petek Šter M. Anticholinergic burden and most common anticholinergic-acting medicines in older general practice patients. Zdr Varst. 2018;57(3):140–7.
Anticholinergic cognitive burden (ACB) scale 2012 update. Indianapolis: Regenstrief Institute, Inc.; 2012.
Cai X, Campbell N, Khan B, Callahan C, Boustani M. Long-term anticholinergic use and the aging brain. Alzheimers Dement. 2013;9(4):377–85.
Pfistermeister B, Tümena T, Gaßmann K-G, Maas R, Fromm MF. Anticholinergic burden and cognitive function in a large German cohort of hospitalized geriatric patients. PLoS ONE. 2017;12(2):e0171353.
Orhan IE, Orhan G, Gurkas E. An overview on natural cholinesterase inhibitors—a multi-targeted drug class—and their mass production. Mini Rev Med Chem. 2011;11(10):836–42.
Carnahan RM, Lund BC, Perry PJ, Chrischilles EA. The concurrent use of anticholinergics and cholinesterase inhibitors: rare event or common practice? J Am Geriatr Soc. 2004;52(12):2082–7.
Schultz BR, Takeshita J, Goebert D, Takeshita S, Lu BY, Guilloux A, et al. Simultaneous usage of dementia medications and anticholinergics among Asians and Pacific Islanders. Psychogeriatrics. 2017;17(6):423–9.
Alagiakrishnan K, Wiens CA. An approach to drug induced delirium in the elderly. Postgrad Med J. 2004;80(945):388–93.
Rogers D, Pies R. General medical drugs associated with depression. Psychiatry (Edgmont). 2008;5(12):28–41.
Substance-induced mood disorder: overview, substances linked to depression or mania, etiology (Internet). https://emedicine.medscape.com/article/286885-overview. Accessed 7 Nov 2018.
Qato DM, Ozenberger K, Olfson M. Prevalence of prescription medications with depression as a potential adverse effect among adults in the United States. JAMA. 2018;319(22):2289–98.
Kotlyar M, Dysken M, Adson DE. Update on drug-induced depression in the elderly. Am J Geriatr Pharmacother. 2005;3(4):288–300.
Population estimates and projections. An aging nation: the older population in the United States.
Hunt DL, Haynes RB, Hanna SE, Smith K. Effects of computer-based clinical decision support systems on physician performance and patient outcomes: a systematic review. JAMA. 1998;280(15):1339–46.
Kawamoto K, Houlihan CA, Balas EA, Lobach DF. Improving clinical practice using clinical decision support systems: a systematic review of trials to identify features critical to success. BMJ. 2005;330(7494):765.
H.R.34—114th congress (2015–2016): 21st century cures act |congress.gov| library of congress (Internet). https://www.congress.gov/bill/114th-congress/house-bill/34. Accessed 28 Nov 2018.
Deo RC. Machine learning in medicine. Circulation. 2015;132(20):1920–30.
Baştanlar Y, Ozuysal M. Introduction to machine learning. Methods Mol Biol. 2014;1107:105–28.
Scharre DW, Chang S-I, Murden RA, Lamb J, Beversdorf DQ, Kataki M, et al. Self-administered gerocognitive examination (SAGE): a brief cognitive assessment instrument for mild cognitive impairment (MCI) and early dementia. Alzheimer Dis Assoc Disord. 2010;24(1):64–71.
SAGE—memory disorders|Ohio State Medical Center (Internet). https://wexnermedical.osu.edu/brain-spine-neuro/memory-disorders/sage. Accessed 5 Oct 2018.
Coded private information or specimens use in research, guidance (2008)|HHS.gov (Internet). https://www.hhs.gov/ohrp/regulations-and-policy/guidance/research-involving-coded-private-information/index.html. Accessed 20 Jan 2019.
Keine D, Walker JQ, Kennedy BK, Sabbagh MN. Development, application, and results from a precision-medicine platform that personalizes multi-modal treatment plans for mild Alzheimer’s disease and at-risk individuals. Curr Aging Sci. 2018;18(11):1–1.
RxNorm overview (Internet). https://www.nlm.nih.gov/research/umls/rxnorm/overview.html. Accessed 14 Nov 2018.
Prioritization—CPIC (Internet). https://cpicpgx.org/prioritization/#flowchart. Accessed 14 Nov 2018.
DrugBank (Internet). Pharmaco-transcriptomics. https://www.drugbank.ca/pharmaco/transcriptomics. Accessed 14 Nov 2018.
Pharmacogenomics—SNPedia (Internet). https://www.snpedia.com/index.php/Pharmacogenomics. Accessed 14 Nov 2018.
Genoset—SNPedia (Internet). https://www.snpedia.com/index.php/Genoset. Accessed 14 Nov 2018.
Welsh TJ, van der Wardt V, Ojo G, Gordon AL, Gladman JRF. Anticholinergic drug burden tools/scales and adverse outcomes in different clinical settings: a systematic review of reviews. Drugs Aging. 2018;35(6):523–38.
Campbell NL, Perkins AJ, Bradt P, Perk S, Wielage RC, Boustani MA, et al. Association of anticholinergic burden with cognitive impairment and health care utilization among a diverse ambulatory older adult population. Pharmacotherapy. 2016;36(11):1123–31.
Campbell N, Boustani M, Limbil T, Ott C, Fox C, Maidment I, et al. The cognitive impact of anticholinergics: a clinical review. Clin Interv Aging. 2009;9(4):225–33.
Campbell NL, Boustani MA, Lane KA, Gao S, Hendrie H, Khan BA, et al. Use of anticholinergics and the risk of cognitive impairment in an African American population. Neurology. 2010;75(2):152–9.
Myint PK, Fox C, Kwok CS, Luben RN, Wareham NJ, Khaw K-T. Total anticholinergic burden and risk of mortality and cardiovascular disease over 10 years in 21,636 middle-aged and older men and women of EPIC-Norfolk prospective population study. Age Ageing. 2015;44(2):219–25.
Irving G, Neves AL, Dambha-Miller H, Oishi A, Tagashira H, Verho A, et al. International variations in primary care physician consultation time: a systematic review of 67 countries. BMJ Open. 2017;7(10):e017902.
Podcasy JL, Epperson CN. Considering sex and gender in Alzheimer disease and other dementias. Dialogues Clin Neurosci. 2016;18(4):437–46.
Altmann A, Tian L, Henderson VW, Greicius MD, Alzheimer’s Disease Neuroimaging Initiative Investigators. Sex modifies the APOE-related risk of developing Alzheimer disease. Ann Neurol. 2014;75(4):563–73.
Zhao L, Mao Z, Woody SK, Brinton RD. Sex differences in metabolic aging of the brain: insights into female susceptibility to Alzheimer’s disease. Neurobiol Aging. 2016;18(42):69–79.
Snyder HM, Asthana S, Bain L, Brinton R, Craft S, Dubal DB, et al. Sex biology contributions to vulnerability to Alzheimer’s disease: a think tank convened by the Women’s Alzheimer’s Research Initiative. Alzheimers Dement. 2016;12(11):1186–96.
Phansalkar S, Desai AA, Bell D, Yoshida E, Doole J, Czochanski M, et al. High-priority drug-drug interactions for use in electronic health records. J Am Med Inform Assoc. 2012;19(5):735–43.
Paterno MD, Maviglia SM, Gorman PN, Seger DL, Yoshida E, Seger AC, et al. Tiering drug–drug interaction alerts by severity increases compliance rates. J Am Med Inform Assoc. 2009;16(1):40–6.
Acknowledgments
The authors thank the participants in this study. The NACC database is funded by NIA/NIH grant U01 AG016976. NACC data are contributed by the NIA-funded ADCs: P30 AG019610 (PI Eric Reiman, MD), P30 AG013846 (PI Neil Kowall, MD), P50 AG008702 (PI Scott Small, MD), P50 AG025688 (PI Allan Levey, MD, PhD), P50 AG047266 (PI Todd Golde, MD, PhD), P30 AG010133 (PI Andrew Saykin, PsyD), P50 AG005146 (PI Marilyn Albert, PhD), P50 AG005134 (PI Bradley Hyman, MD, PhD), P50 AG016574 (PI Ronald Petersen, MD, PhD), P50 AG005138 (PI Mary Sano, PhD), P30 AG008051 (PI Thomas Wisniewski, MD), P30 AG013854 (PI M. Marsel Mesulam, MD), P30 AG008017 (PI Jeffrey Kaye, MD), P30 AG010161 (PI David Bennett, MD), P50 AG047366 (PI Victor Henderson, MD, MS), P30 AG010129 (PI Charles DeCarli, MD), P50 AG016573 (PI Frank LaFerla, PhD), P50 AG005131 (PI James Brewer, MD, PhD), P50 AG023501 (PI Bruce Miller, MD), P30 AG035982 (PI Russell Swerdlow, MD), P30 AG028383 (PI Linda Van Eldik, PhD), P30 AG053760 (PI Henry Paulson, MD, PhD), P30 AG010124 (PI John Trojanowski, MD, PhD), P50 AG005133 (PI Oscar Lopez, MD), P50 AG005142 (PI Helena Chui, MD), P30 AG012300 (PI Roger Rosenberg, MD), P30 AG049638 (PI Suzanne Craft, PhD), P50 AG005136 (PI Thomas Grabowski, MD), P50 AG033514 (PI Sanjay Asthana, MD, FRCP), P50 AG005681 (PI John Morris, MD), P50 AG047270 (PI Stephen Strittmatter, MD, PhD).
Funding
All funding for this study was provided by uMETHOD Health. No funding or sponsorship was received for the publication of this article. All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analysis.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
Dorothy Keine is employed by, had a previous financial relationship with, and/or holds stock in uMETHOD Health. Mark Zelek is employed by, had a previous financial relationship with, and/or holds stock in uMETHOD Health. John Q. Walker is employed by, had a previous financial relationship with, and/or holds stock in uMETHOD Health. Marwan Sabbagh is employed by, had a previous financial relationship with, and/or holds stock in uMETHOD Health; he is also Editor in Chief of the journal.
Compliance with Ethics Guidelines
Following the guidelines set out by “Coded Private Information or Specimens Use in Research” by the Office for Human Research Protection, the work presented here does not qualify as human research. The investigators utilized existing, de-identified data from patients who gave previous informed consent to have their data used in future research, but did not involve the patients in any therapy or intervention. Institutional review board approval was not required as data are recorded in such a manner that subjects cannot be identified, directly or through identifiers linked to the subjects. Meeting these conditions makes this research exempt from the requirements of 45 CFR 46 under the Department of Health and Human Services (HHS): Research, involving the collection or study of existing data, documents, records, pathological specimens, if these sources are publicly available or if the information is recorded by the investigator in such a manner that subjects cannot be identified, directly or through identifiers linked to the subjects.
Data Availability
The data that support the findings of this study are available from uMETHOD Health, but restrictions apply to the availability of these data, which are proprietary company information, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of uMETHOD Health.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced digital features
To view enhanced digital features for this article, go to https://doi.org/10.6084/m9.figshare.7739627.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Keine, D., Zelek, M., Walker, J.Q. et al. Polypharmacy in an Elderly Population: Enhancing Medication Management Through the Use of Clinical Decision Support Software Platforms. Neurol Ther 8, 79–94 (2019). https://doi.org/10.1007/s40120-019-0131-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40120-019-0131-6