Abstract
Introduction
We sought to determine the effectiveness and safety of hydroxychloroquine–azithromycin (HCQ-AZM) therapy in hospitalized patients with COVID-19.
Methods
This was a retrospective cohort study of 613 patients hospitalized (integrated health system involving three hospitals) for RT-PCR-confirmed COVID-19 infection between March 1, 2020 and April 25, 2020. Intervention was treatment with HCQ-AZM in hospitalized patients with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. Outcomes of interest were in-hospital all-cause mortality, cardiovascular mortality, pulseless electrical activity (PEA) arrest, non-lethal arrhythmias, and length of hospital stay. Secondary measures included in-hospital corrected QT (QTc) interval parameters and serum biomarkers levels.
Results
Propensity-matched groups were composed of 173 patients given HCQ-AZM and 173 matched patients who did not receive treatment. There was no significant difference in in-hospital mortality (odds ratio [OR] 1.52; 95% confidence interval [CI] 0.80–2.89; p = 0.2), PEA arrest (OR 1.68, CI 0.68–4.15; p = 0.27), or incidence of non-lethal arrhythmias (10.4% vs. 6.8%; p = 0.28). Length of hospital stay (10.5 ± 7.4 vs. 5.8 ± 6.1; p < 0.001), peak CRP levels (252 ± 136 vs. 166 ± 124; p < 0.0001), and degree of QTc interval prolongation was higher for the HCQ-AZM group (28 ± 32 vs. 9 ± 32; p < 0.0001), but there was no significant difference in incidence of sustained ventricular arrhythmias (2.8% vs. 1.7%; p = 0.52). HCQ-AZM was stopped in 10 patients because of QT interval prolongation and 1 patient because of drug-related polymorphic ventricular tachycardia.
Conclusion
In this propensity-matched study, there was no difference in in-hospital mortality, life-threatening arrhythmias, or incidence of PEA arrest between the HCQ-AZM and untreated control groups. QTc intervals were longer in patients receiving HCQ-AZM, but only one patient developed drug-related ventricular tachycardia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
The COVID-19 pandemic has led to an unprecedented global health crisis |
This study sought to ascertain whether hydroxychloroquine and azithromycin therapy is safe and effective in hospitalized patients with COVID-19 |
What was learned from this study? |
In this propensity-matched cohort study, there was no statistical difference in the incidence of in-hospital mortality, PEA arrest, or non-lethal arrhythmias between patients treated with hydroxychloroquine–azithromycin therapy versus matched controls |
Higher degree of corrected QT (QTc) prolongation was observed in the patients treated with hydroxychloroquine–azithromycin, but the incidence of drug-related torsades de pointes was low |
Although likely safe to administer with close QTc monitoring, the findings do not support use of hydroxychloroquine–azithromycin therapy for hospitalized patients with COVID-19 |
Digital Features
This article is published with digital features to facilitate understanding of the article. You can access the digital features on the article’s associated Figshare page. To view digital features for this article go to https://doi.org/10.6084/m9.figshare.13019144.
Introduction
Since emerging from Wuhan, China in December 2019, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has spread throughout the globe at a rapid pace [1]. Although the true prevalence of coronavirus disease 2019 (COVID-19) has been difficult to decipher, a health system crisis triggered by high rates of disease-associated morbidity and mortality as well as prolonged hospital stays has become daunting to address [1,2,3,4]. At the time of this manuscript, over 144,000 patients in the USA and 614,000 worldwide have died from COVID-19 [1]. Given this scenario, investigators have actively sought to repurpose existing antimicrobial drugs in an effort to mitigate progression and improve clinical outcomes in patients with COVID-19 [5].
Chloroquine (N4-(7-chloro-4-quinolinyl)-N1,N1-diethyl-1,4-pentanediamine; CQ) has been used for many decades for treatment of malaria and protozoan infection. A derivative of chloroquine, hydroxychloroquine (HCQ), is more frequently used in clinical practice given its more favorable patient tolerance [6]. Following the publication of several small studies, HCQ has been investigated as a treatment for SARS-CoV-2, in particular for patients with suspected cytokine release syndrome [7,8,9,10]. The potential salutary effects of HCQ include inhibition of angiotensin-converting enzyme 2 (ACE2) receptor-mediated entry of the SARS-CoV2 virus through various actions such as elevation of intravesicular pH, inhibition of lysosomal activity, and alteration of antigen processing [6, 8, 9]. In addition to its potential antiviral properties, HCQ may inhibit various innate immune pathways (Toll-like receptors 7, 8 and 9). The modulation of these pathways could inhibit interleukin-6 (IL6), IL-1b, interferons, or tumor necrosis factor, which are associated with cytokine release syndrome and may be relevant in SARS-CoV2 pathology [11].
One of the clinically important cardiac side effects of HCQ is the potential for QT interval prolongation. Although risk of torsades de pointes (TdP) in patients treated for malaria and autoimmune disorders is believed to be low [12], tolerance for HCQ at treatment doses for SARS-CoV-2 and the related incidence TdP in this population are not well defined. Additionally, azithromycin (AZM; another QT-prolonging agent) has shown promise in reducing viral load of SARS-CoV-2 in combination with HCQ [10], and may be concomitantly prescribed, thus leading to a synergistic effect upon QT interval prolongation and further elevation of TdP risk.
In this study we sought to evaluate the clinical efficacy and in-hospital safety of combination hydroxychloroquine and azithromycin (HCQ-AZM) treatment in a cohort of hospitalized patients with COVID-19.
Methods
The study included consecutive patients admitted for treatment of symptomatic COVID-19 infection at Rush University Medical Center, Chicago, Illinois, and two community hospitals of the integrated health care system between March 1, 2020 and April 25, 2020 were included. Patients were screened and included in the study only if active SARS-CoV-2 infection was confirmed by testing using a reverse transcriptase polymerase chain reaction (RT-PCR) assay.
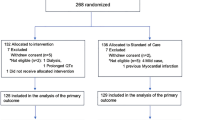
We initially screened 1228 patients who were admitted for suspected COVID-19 infection based upon clinical presentation. Clinical usage criteria for treatment with HCQ-AZM for hospitalized patients with COVID-19 is shown in Fig. 1 of the supplementary material. After exclusion of patients who tested negative for SARS-CoV-2 or who received non-HCQ-AZM experimental therapy for COVID-19 (N = 566), were missing covariables (N = 28), received treatment with HCQ without AZM for treatment of COVID-19 (N = 16), or were already on chronic HCQ therapy for treatment of other illnesses (N = 5), the study cohort comprised 613 patients: 182 patients treated with HCQ and 431 patients who did not receive HCQ during their hospitalizations. After propensity-matched scoring, the final study cohort included 173 patients in the HCQ-AZM treated group and 173 patients who did not receive HCQ-AZM therapy while hospitalized (Fig. 1). The study was undertaken with the approval of the institutional review board of Rush University Medical Center. The study was performed in accordance with the declaration of Helsinki 1964 and its later amendments. Informed consent was not obtained from patients owing to the nature of the study being a retrospective chart review.
Outcomes of interest were in-hospital all-cause mortality, cardiovascular mortality, pulseless electrical activity (PEA) arrest, and incidence of non-lethal cardiac arrhythmias in 173 consecutive patients with COVID-19 treated with a 5-day course of HCQ-AZM combination therapy and an equal number of control patients selected by propensity score matching using the greedy matching algorithm. Secondary outcomes of the study were in-hospital corrected QT (QTc) interval prolongation and peak levels of serum biomarkers. QTc intervals were measured in milliseconds (ms) and heart rate (HR) was measured in beats per minute (BPM). Raw QT intervals were corrected for heart rate and were calculated using the Bazett formula: (QTc = QT/√RR, where QT and RR are measured in seconds). In the matched control group, pre-hospital ECGs up to 12 months prior to the index hospitalization were used for baseline comparison. In the HCQ-AZM treated group, pre-hospital ECGs were only used for baseline QTc interval measurement if an in-hospital ECG prior to treatment was not available.
Given that the investigators did not control assignment of treatment subjects, propensity score matching was used for selection of comparable groups of patients to minimize bias by balancing the distributions of observed (and possibly confounding) covariates (XLSTAT, Anglesey, UK). After estimation of propensity scores for receiving HCQ-AZM therapy for each patient, patients who received HCQ-AZM therapy were matched in a 1:1 ratio to patients who did not receive HCQ-AZM treatment during their index hospitalization using the greedy matching algorithm. Mahalanobis distance along with a caliper size of 0.2 of the standard deviation of propensity scores was used for construction of matched pair samples. These methods have been validated elsewhere [13–15,14,]. Details of the propensity analysis are available in Fig. 2 and Tables 1–6 of the supplementary material.
Continuous data were presented as mean and standard deviation while categorical variables were presented as number and percentages. XLSTAT software was used to compare categorical variables using Pearson χ2 test or chi-square test. Normally distributed variables were compared between the two groups using paired t test. Adjusted odds ratio and confidence intervals for primary outcomes were estimated from the logistical model of covariates and their assigned propensity score [14]. All hypothesis testing was two-tailed, and p values less than 0.05 were considered statistically significant.
Results
Between March 1, 2020 and April 25, 2020, 1298 patients were admitted for suspected active COVID-19 infection. Of these, 662 patients tested positive for SARS-CoV-2/COVID-19 by RT-PCR assay. Baseline characteristics of the final cohort (n = 613) are shown in Table 1. For the overall cohort, in-hospital mortality was 12.1% (74 deaths) and mean length of hospital stay was 7.5 ± 6.8 days. Patient symptoms on presentation for the overall cohort and study groups are shown in Table 7 of the supplementary material.
Among the 182 COVID-19-positive patients that received HCQ-AZM combination therapy of hydroxychloroquine (400 mg twice for 1 day, followed by 200 mg twice daily, the next 4 days) and azithromycin (500 mg day 1, followed by 250 mg per day, the next 4 days), 173 patients were successfully matched on the basis of propensity score to a control patient who did not receive any doses of HCQ or AZM during their index hospitalization. Patient characteristics for HCQ-AZM treated and matched-control groups are shown in Table 2. In the control group, 120 of 173 patients (69%) had a 12-lead ECG obtained in-hospital, which could be compared to a baseline ECG (within 12 months of admission) for comparative QTc interval measurement. In the HCQ-AZM treated group, 139 of 173 patients (80%) had at least one serial ECG available during HCQ-AZ treatment for QTc interval measurement. For the other 34 patients (20%), QTc interval monitoring during HCQ-AZM administration was performed by 3- or 8-lead telemetry.
In the primary analysis, there was no difference in in-hospital all-cause mortality between the HCQ-AZM treated group and the control group (15.0% vs. 10.4%; odds ratio [OR] 1.52; 95% confidence interval [CI] 0.80–2.89; p = 0.2) or for the incidence of in-hospital PEA arrest (7.5% vs. 4.8%; OR 1.68, CI 0.68–4.15; p = 0.27). However, mean length of hospital stay was higher for patients treated with HCQ-AZM in comparison to the propensity-matched control group (10.5 ± 7.4 days vs. 5.8 ± 6.1 days; p < 0.0001) (Table 3). There were no in-hospital arrhythmia-related or cardiovascular deaths in either group of the study cohort.
Patients treated with HCQ-AZM combination therapy had lower QTc intervals than control patients on their baseline 12-lead ECG (436 ± 32 ms vs. 447 ± 33 ms; p = 0.01). Peak QTc intervals obtained in-hospital by serial 12-lead ECG or telemetry measurement and total change (Δ) in QTc intervals were significantly higher in the HCQ-AZM group (28 ± 32 ms vs. 9 ± 22 ms; p < 0.0001). HCQ-AZM therapy was stopped before completion of the 5-day treatment course in 10 of 173 patients (5.7%) because of prolongation of QTc interval (QTc > 500 ms or ΔQTc interval > 60 ms) and was stopped in 1 patient after the development of TdP. Azithromycin was stopped but HCQ treatment was continued in three patients who developed QTc prolongation of greater than 60 ms. Importantly, there was no difference in the incidence of hypokalemia (reference 3.4–5.3 mmol/L) or hypomagnesemia (reference 1.6—2.7 mg/dL) during hospital stays between the HCQ-AZM treated and control groups.
The incidence of in-hospital arrhythmias for the cohort is shown in Table 4. There were no significant differences in the incidence of in-hospital tachyarrhythmias (> 30 s) or bradyarrhythmias between HCQ-AZM patients and propensity-matched controls. Atrial fibrillation was the most commonly occurring arrhythmia in the hospital in both groups. There was no significant difference in the incidence of sustained or hemodynamically unstable VT between groups, although one patient in the HCQ group had premature ventricular contraction (PVC)-triggered polymorphic VT in the setting of drug-induced QTc interval prolongation. None of the patients required implantation of temporary or permanent pacemakers for treatment of sinus bradycardia, pauses, or AV block.
Peak levels of serially drawn inflammatory biomarkers were significantly elevated in both patients groups, consistent with critical illness from SARS-CoV-2 in the cohort (Table 4). Acute kidney injury (AKI) was a frequent complication of SARS-CoV-2 potentially from high incidence of circulatory shock in the cohort. Peak creatinine levels obtained in hospital were significantly higher than baseline creatinine levels for both groups. Peak hospital troponin I levels [reference range 0.01–0.09 ng/mL) were also elevated overall in both groups. However 77% of patients in the cohort did not have troponin levels exceeding normal range (reference 0.00 to 0.09 ng/dL) despite the high prevalence of critical illness in the study population and incidence of severe AKI.
In addition, we performed a subgroup analysis of HCQ-AZM patients by stratifying by significant absolute increase in QTc interval during HCQ-AZM treatment (> vs. ≤ 60 ms). There were no significant differences in comorbidities, baseline QTc interval, or renal function between HCQ-AZM subgroups stratified by ΔQTc interval, although baseline LVEF was higher in patients who had > 60 ms ΔQTc interval. There was no difference in cardiovascular mortality between patients with ΔQTc interval > 60 vs. ≤ 60 ms. However, patients with significant increase in ΔQTc interval (> 60 ms) were significantly more likely to have hypokalemia after starting HCQ-AZM administration (43% vs. 13%; p < 0.001) than the group with ΔQTc interval < 60 ms (Table 5).
Discussion
The major findings of this study were (1) there was no significant difference in in-hospital mortality, incidence of PEA arrest, or non-lethal arrhythmias between HCQ-AZM treated and propensity-matched control groups; (2) mean length of hospital stay and peak C-reactive protein levels were higher for the HCQ-AZM treated group; (3) despite a higher degree of QTc prolongation in the HCQ-AZM group than the control group (6.2% vs. 2.5% increase), drug-related TdP remained rare (0.6%); and (4) patients with greater changes in QTc interval (> 60 ms) were more likely to have electrolyte abnormalities after starting HCQ-AZM therapy.
Despite limited evidence of its effectiveness, HCQ (with or without AZM) remains under investigation for empiric treatment and prophylaxis for treatment of COVID-19 [16, 17]. A potentially clinically important side effect of HCQ administration is the risk for QT interval prolongation and development of lethal ventricular arrhythmias such as TdP. Further, QT interval prolongation can be exacerbated by electrolyte derangements (in particular, depletion of serum potassium or magnesium levels) and co-prescribed medications such as azithromycin which have also been used in the treatment of SARS-CoV-2.
Progression from the early symptomatic phase to acute respiratory distress syndrome and resultant hypoxic respiratory failure may be secondary to uncontrolled release of cytokines. Given HCQ’s clinical use as an anti-inflammatory medication for patients with autoimmune disease [12], HCQ may inhibit cytokine storm by suppressing T cell activation and reduce severity of mortality and morbidity of patients with COVID-19 [6,7,8,9,10]. However, the evidence for the clinical efficacy of HCQ for treatment of SARS-CoV-2 remains limited and there is also debate regarding the optimal therapeutic dosage and duration for treatment. Gautret et al. reported that a combination of HCQ and AZM may be more effective in reducing viral load than HCQ alone [10]. In a recently published study, hospitalized patients with COVID-19 who were treated using a high-dose CQ protocol (600 mg twice daily for 10 days) were more likely to experience QT prolongation and had higher in-hospital mortality than patients treated with low-dose CQ protocol (450 mg twice daily for one day and once daily for 9 more days) (mortality at 13 days 39% vs. 14%). The randomized trial, which was intended to include 440 patients, was stopped prematurely after enrolling only 81 patients because of the high rate of major adverse events [18].
In the current study, we compared in-hospital outcomes between 173 patients treated with a combination of HCQ-AZM for 5 days and 173 propensity-matched controls who did not receive HCQ-AZM during their index hospitalization for COVID-19. We observed no difference in mortality or in-hospital PEA arrest between HCQ-AZM treated and matched control patients (15.0% vs. 10.4% deaths; p = 0.2). Despite similar age, sex, comorbidities, LVEF, and BMI between groups, we found that length of hospital stay was significantly higher for patients treated with HCQ-AZM therapy (10.5 ± 7.4 days vs. 5.8 ± 6.1 days; p < 0.001) than matched control patients. Altogether, the lack of demonstrated clinical effectiveness, requirement for QT monitoring, and longer hospital stay associated with HCQ-AZM may be important considerations for its potential use in patients with COVID-19, especially given strained healthcare resources and limited bed availability needed for critically ill patients in the pandemic.
Although it has been postulated that HCQ may play a beneficial role for treatment of SARS-CoV-2 by inhibiting cytokine release and controlling inflammation, we did not observe any significant reduction in commonly tested, serially drawn inflammatory biomarkers in the HCQ-AZM treatment group versus the matched control group. In fact, peak hospital C-reactive protein levels remained significantly higher in the HCQ-AZM arm of the study despite treatment. CQ is believed to inhibit T cell proliferation by reducing IL-2 production and IL-2 responsiveness [19]. However TH2 cell response may play a role in suppressing inflammation in SARS-CoV-2 infection, and therefore it remains possible that administration of CQ/HCQ could negatively impact the immune response to viral infection [3, 20]. Although patients groups were propensity score matched for baseline covariates in this study, treatment of more severely ill patients in the HCQ-AZM group cannot be completely excluded.
From a cardiovascular standpoint, an interesting finding of the study was the relatively low incidence of elevated serum troponin in the study cohort (77% of patients troponin I level within our hospital’s reference range), despite the high prevalence of critically ill patients and incidence of both acute kidney injury (AKI) and circulatory shock. The incidence of atrial and ventricular tachyarrhythmias was similar between study groups with the most common atrial arrhythmia being atrial fibrillation (Table 2). The incidence of sustained or hemodynamically unstable ventricular arrhythmias was low and statistically similar between groups. One patient in the HCG-AZM treated group developed PVC-induced TdP in the setting of drug-induced QT prolongation. After discontinuation of HCQ, the patient’s QTc interval returned to baseline and the patient did not have further ventricular arrhythmias. The incidence of bradyarrhythmias was 4.2%, although none of the patients in the study required temporary or permanent pacemaker implantation.
Since the start of the COVID-19 pandemic, the widespread usage of CQ and HCQ in the acute hospital setting has been unprecedented. While QT interval prolongation is a known side effect of HCQ administration and has been demonstrated in patients treated with HCQ-AZM combination therapy [12, 18, 21], the degree of QT interval prolongation is variable among patients and may also be modulated by electrolyte abnormalities, myocarditis, and medications exerting QT-prolonging effects on ventricular repolarization. Although HCQ is structurally similar to quinidine and the incidence of TdP has been well established for other potassium channel IKr (hERG/KV11.1) blocking medications [22,23,24], the risk of TdPs in patients taking HCQ at treatment dosages for COVID-19 remains unknown.
The results of the current study confirm that a significant increase in QTc intervals occurs following administration of a moderate HCQ-AZM dosing protocol (HCQ: 400 mg twice day 1, then 200 mg twice daily for 4 days; AZM: 500 mg day 1, then 250 mg per for next 4 days) versus matched controls [18]. In total, HCQ-AZM was stopped prior to completion of the dosing regimen in a minority of patients (6%) because of significant QTc prolongation (QTc interval above 500 ms or total increase in QTc interval > 60 ms) and in one patient because of drug-related polymorphic ventricular arrhythmia. In a subgroup analysis of patients treated with HCQ-AZM, we found that patients with significant increment in QT interval (∆ > 60 ms) were more likely to have hypokalemia (< 3.3 mmol/mL) in hospital. Hypokalemia has been known to potentiate QT prolongation and dispersion of ventricular repolarization, both increasing vulnerability for TdP. Thus, monitoring and correction of electrolyte abnormalities is paramount in patients receiving HCQ or HCQ-AZM combination therapy for treatment of COVID-19 in the hospital.
Limitations
As the study population included only patients hospitalized for COVID-19, the results of the study may not be generalizable to “healthier” symptomatic patients with COVID-19 treated with HCQ-AZM or patients treated with HCQ alone. Although the difference in mortality between groups was not statistically significant, point estimates for all-cause mortality in patients receiving HCQ-AZM were greater than one and our study may have been underpowered. In addition, laboratory and ECG monitoring was not uniformly standardized given the retrospective nature of the study. Although propensity score matching was employed to mitigate differences in confounders between comparator groups, we cannot rule out the possibility of residual confounding with the utilized covariates. Finally the current study utilized an HCQ-AZM treatment course of only 5 days and therefore whether clinical outcomes would differ using a longer course of HCQ-AZM cannot be determined from the current study.
Conclusions
In this study of hospitalized patients with COVID-19 who received HCQ-AZM treatment compared to propensity-matched controls, there was no improvement in overall survival or indices of inflammation. There was no significant difference in the incidence of atrial or ventricular arrhythmias in either group.
References
World Health Organization. Novel coronavirus (2019-nCoV) technical guidance. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/technical-guidance. Accessed 14 Feb 2020.
Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323(11):1061–9.
Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. https://doi.org/10.1016/S0140-6736(20)30183-5(Erratum in: Lancet. 2020 Jan 30).
Centers for Disease Control and Prevention. 2019 Novel coronavirus, Wuhan, China. Information for healthcare professionals. https://www.cdc.gov/coronavirus/2019-nCoV/hcp/index.html. Accessed 14 Feb 2020.
Sanders JM, Monogue ML, Jodlowski TZ, Cutrell JB. Pharmacologic treatments for coronavirus disease 2019 (COVID-19): a review. JAMA. 2020. https://doi.org/10.1001/jama.2020.6019.
Liu J, Cao R, Xu M, et al. Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro. Cell Discov. 2020;18(6):16.
Colson P, Rolain JM, Lagier JC, Brouqui P, Raoult D. Chloroquine and hydroxychloroquine as available weapons to fight COVID-19. Int J Antimicrob Agents. 2020;4:105932.
Yao X, Ye F, Zhang M, et al. In vitro antiviral activity and projection of optimized dosing design of hydroxychloroquine for the treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Clin Infect Dis. 2020;71(15):732–9.
Devaux CA, Rolain JM, Colson P, Raoult D. New insights on the antiviral effects of chloroquine against coronavirus: what to expect for COVID-19? Int J Antimicrob Agents. 2020;11:105938.
Gautret P, Lagier JC, Parola P, Hoang VT, Meddeb L, Mailhe M. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents. 2020;20:105949.
Schrezenmeier E, Dörner T. Mechanisms of action of hydroxychloroquine and chloroquine: implications for rheumatology. Nat Rev Rheumatol. 2020;16(3):155–66.
Haeusler IL, Chan XHS, Guérin PJ, White NJ. The arrhythmogenic cardiotoxicity of the quinoline and structurally related antimalarial drugs: a systematic review. BMC Med. 2018;16(1):200.
Rosenbaum PR. Propensity score. In: Armitage P, Colton T, editors. Encyclopedia of biostatistics. Chichester: Wiley; 1998. p. 3551–3555.
Austin PC. Optimal caliper widths for propensity-score matching when estimating differences in means and differences in proportions in observational studies. Pharm Stat. 2011;10(2):150–61.
Cepeda MS, Boston R, Farrar JT, Strom BL. Comparison of logistic regression versus propensity score when the number of events is low and there are multiple confounders. Am J Epidemiol. 2003;158(3):280–7.
Roden DM, Harrington RA, Poppas A, Russo AM. Considerations for drug interactions on QTc in exploratory COVID-19 treatment. Circulation. 2020;141(24):e906–7.
Kapoor A, Pandurangi U, Arora V, et al. Cardiovascular risks of hydroxychloroquine in treatment and prophylaxis of COVID-19 patients: a scientific statement from the Indian Heart Rhythm Society. Indian Pacing Electrophysiol J. 2020;20(3):117–120.
Borba MGS, Val FFA, Sampaio VS, et al. Effect of high vs low doses of chloroquine diphosphate as adjunctive therapy for patients hospitalized with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection: a randomized clinical trial. JAMA Netw Open. 2020;3(4.23):e208857.
Landewé RB, Miltenburg AM, Verdonk MJ, et al. Chloroquine inhibits T cell proliferation by interfering with IL-2 production and responsiveness. Clin Exp Immunol. 1995;102(1):144–51.
Guastalegname M, Vallone A. Could chloroquine/hydroxychloroquine be harmful in coronavirus disease 2019 (COVID-19) treatment? Clin Infect Dis. 2020;71(15):888–9.
Chorin E, Dai M, Shulman E. The QT interval in patients with COVID-19 treated with hydroxychloroquine and azithromycin. Nat Med. 2020. https://doi.org/10.1038/s41591-020-0888-2.
Wu CI, Postema PG, Arbelo E, et al. SARS-CoV-2, COVID-19, and inherited arrhythmia syndromes. Heart Rhythm. 202017(9):1456–62.
Lin JC, Quasny HA. QT prolongation and development of torsades de pointes with the concomitant administration of oral erythromycin base and quinidine. Pharmacotherapy. 1997;17(3):626–30.
Huang HD, Waks JW, Steinhaus DA, Zimetbaum P. Magnitude of increase in QTc interval after initiation of dofetilide in patients with persistent atrial fibrillation is associated with increased rates of pharmacological cardioversion and long-term freedom from recurrent atrial fibrillation. Heart Rhythm. 2016;13(7):1410–7.
Acknowledgements
Funding
No funding or sponsorship was received for this study or publication of this article.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
Henry Huang, Hani Jneid, Mariam Aziz, Venkatesh Ravi, Parikshit S. Sharma, Timothy Larsen, Neal Chatterjee, Basil Saour, Zaid Aziz, Hemal Nayak, Richard Trohman, and Kousik Krishnan have nothing to disclose.
Compliance with Ethics Guidelines
The study was undertaken with the approval of the institutional review board of Rush University Medical Center. The study was performed in accordance with the declaration of Helsinki 1964 and its later amendments. Informed consent was not obtained from patients owing to the nature of the study being a retrospective chart review.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Huang, H.D., Jneid, H., Aziz, M. et al. Safety and Effectiveness of Hydroxychloroquine and Azithromycin Combination Therapy for Treatment of Hospitalized Patients with COVID-19: A Propensity-Matched Study. Cardiol Ther 9, 523–534 (2020). https://doi.org/10.1007/s40119-020-00201-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40119-020-00201-7