Abstract
Introduction
Trimetazidine (TMZ) is an antianginal agent that acts directly at the myocardial cell level and which is now available in a once-daily (od) formulation.
Methods
ODA, a 3-month, observational, multicenter study in Russia, assessed the effectiveness and tolerability of TMZ 80 mg od in patients with stable angina and persisting symptoms, in real-life settings. The present analysis explored the effects of adding TMZ to background antianginal treatment with respect to the duration of stable angina.
Results
A total of 3032 patients were divided into four groups according to stable angina pectoris duration since diagnosis, ranging from less than 1 year to more than 10 years. A decrease in frequency of angina attacks was observed, including in patients with angina duration < 1 year, in whom the frequency of weekly angina attacks decreased from 3.8 ± 2.9 to 1.4 ± 1.7 at 1 month and 0.6 ± 1.0 at 3 months. Short-acting nitrate consumption and proportion of angina-free patients decreased, and self-reported physical activity and adherence to antianginal therapy improved in all patient groups, including recently diagnosed patients and starting already at month 1.
Conclusions
Addition of TMZ 80 mg od to antianginal treatment was effective in reducing the frequency of angina attacks and the use of short-acting nitrates, improving Canadian Cardiovascular Society (CCS) class, self-reported physical activity, and adherence to antianginal therapy. These beneficial effects were observed in patient groups with different durations of stable angina, suggesting an opportunity for decreasing angina burden even in recently diagnosed patients.
Trial Registration
ISRCTN registry Identifier, ISRCTN97780949.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Trimetazidine (TMZ) is an antianginal treatment whose efficacy has been demonstrated in randomized clinical trials, and which is now available in an 80-mg one-daily (od) dosage. |
In this analysis, the effect of TMZ addition to hemodynamic therapy was assessed in patient groups with different angina durations from diagnosis, from recently diagnosed patients to those with > 10 years of disease duration. |
What was learned from the study? |
Introduction of TMZ 80 mg od to antianginal therapy effectively improved Canadian Cardiovascular Society class, frequency of angina attacks and short-acting nitrate consumption, self-reported patient activity and adherence to antianginal treatment in all patient groups irrespective of angina duration. |
The once-daily formulation of TMZ could provide an opportunity for improving angina symptoms whatever the duration of the disease, including from the very beginning of diagnosis and treatment. |
Introduction
Ischemic heart disease remains today a major cause of mortality for both sexes [1, 2] and the incidence of stable angina pectoris (AP) has been reported to increase with age in both sexes [3]. Angina prevails over myocardial infarction and heart failure as a reason for years lived with disability due to ischemic heart disease [4]. Ischemia, angina, and physical limitation are strongly predictive of major adverse cardiovascular events and poor survival [5,6,7,8], even after percutaneous coronary intervention (PCI) [9]. Thus, alleviating angina symptoms and improving patients’ quality of life remains an important goal in the treatment of angina patients [10, 11]. Noteworthy, the latest ESC CCS Guidelines 2019, for the first time ever, gave a specific temporal definition of the term “recently diagnosed” CCS patients, which is patients within 1 year of the diagnosis, focusing on the importance of a tight follow-up and timely angina alleviation in this specific group of angina patients. Also in the same document, there is a specific part dedicated to the patients with a long-standing diagnosis of chronic coronary syndrome where the authors highlight the importance of “life-long treatment and surveillance” [11]. Yet, importantly, more than 30% of patients continue to experience symptoms despite antianginal treatment [12] and the rate of angina remains high even after successful revascularization in the course of the disease and is among the most common factors affecting rehospitalization [13,14,15]. Moreover, angina patients live longer nowadays and often have comorbidities [16], which increases the number of medications thus impacting adherence. This could be even more pronounced in patients who have had angina for a long time.
Patients with angina can remain symptomatic despite standard hemodynamic treatment [17]. Combining hemodynamic drugs does not necessarily translate into improved antianginal efficacy [18, 19], as in spite of hemodynamic treatment combinations, patients remain symptomatic [20]. Trimetazidine (TMZ) acts directly at the myocardial cell level [21] and its addition to the antianginal therapy offers an opportunity for treatment optimization.
The ODA (anti-anginal effectiveness and tolerability of trimetazidine modified release 80 mg once daily in stable angina patients in real-world practice) study assessed the effectiveness and tolerability of TMZ 80 mg once daily (od) in stable angina patients in a real-life clinical practice setting in Russia and its rationale, design, and main results have been previously published [22]. In the present analysis of the ODA study, we sought to assess the effect of TMZ 80 od addition to hemodynamic therapy with regard to angina duration focusing on its impact on effectiveness and adherence to treatment in the patients corresponding to the new ESC CCS Guidelines definition of “recently diagnosed” CCS patients, as well as on patients with a diagnosis duration longer than 1 year, corresponding to the group of patients with long-standing diagnosis of CCS.
Methods
ODA was a 3-month observational, multicenter, prospective study in 3066 stable angina patients with persistent symptoms despite therapy, conducted in Russia from March 2017 to June 2017 in a real-world clinical setting. The methods and main findings of this study have been published previously [22]. Patients were treated according to current recommendations for coronary artery disease management and their inclusion was exclusively determined by the decision of the physician regarding medical meaningfulness and indication for treatment with TMZ od 80 mg.
For the analyses presented herein, patients with known duration of disease (n = 3032) were divided into four groups according to AP duration: (1) group 1, AP duration less than 1 year; (2) group 2, AP duration 1–5 years; (3) group 3, AP duration 5–10 years; and (4) group 4, AP duration more than 10 years. The temporal segmentation of the groups of patients with a duration of the diagnosis more than 1 year was purely empirical, more related to local real clinical practice, and aiming to provide a balanced number of patients for analysis in the respective groups.
Data were collected at baseline, month 1, and month 3, regarding the number of angina attacks, short-acting nitrate (SAN) consumption in the week prior to the visit, Canadian Cardiovascular Society (CCS), patient self-assessment of physical activity, and adherence to antianginal treatment. For self-assessment of their daily activity, patients rated the impact of angina on their daily activity on a scale of 1–10 and answers were categorized into the following categories: no limitation (0), slight limitation (1–2), moderate limitation (3–4), substantial limitation (5–7), and very marked reduction (8–10). Adherence to treatment was assessed using a six-item questionnaire [23], with the following definitions: good adherence—patient responded “NO” to all questions; moderate adherence—patient responded “YES” to 1–2 questions; non-adherence—patient responded “YES” to three or more questions.
Compliance with Ethics Guidelines
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study. This study was approved by the Interuniversity Ethical Committee, Moscow.
Statistical Analysis
A descriptive statistical analysis was performed with SAS software, version 9.1. Patients were considered if they had valid data from all visits. All parameters were analyzed using descriptive statistics methods. The number of patients, mean value, standard deviation, minimum and maximum value, or proportion by category were specified for each parameter. Differences in the numbers of angina pectoris episodes and in the necessity to use anti-anginal drugs were evaluated by the Wilcoxon’s signed-rank test. p < 0.05 was considered significant. The dynamics of the parameters analyzed from visit to visit were studied using both Wilcoxon signed-rank test and Student’s paired t test.
Results
A total of 3032 patients were divided into four groups according to AP duration since diagnosis: (1) group 1, AP duration less than 1 year (n = 88); (2) group 2, AP duration 1–5 years (n = 1323); (3) group 3, AP duration 5–10 years (n = 854); and (4) group 4, AP duration more than 10 years (n = 767). Demographic and baseline characteristics are presented in Table 1. As AP duration increased, patients were on average older, the proportion of patients aged ≥ 65 years increased and the proportion of patients with some comorbidities (arterial hypertension, diabetes) and with history of myocardial infarction also increased. Unfavorable family history of coronary artery disease (CAD) did not seem to be related to AP duration.
Antianginal treatments at baseline are shown in Table 2. Monotherapy was frequent in all patient groups, even those with long disease duration. The proportion of patients on monotherapy with any hemodynamic antianginal drug was 54.7% in group 1, 43.4% in group 2, 37.6% in group 3, and 30.4% in group 4. Beta-blockers (BB) were the most often prescribed drug, either alone or in combination. In patients with longer duration of AP, the frequency of monotherapy with BB was significantly lower compared with that in patients with short (< 1 year) AP duration. The proportion of patients on monotherapy with CCB or long-acting nitrates was low overall and did not differ between groups with different AP duration. A combination of BB + CCB was received by one in three or four patients, with no significant differences observed between patient groups. The proportion of patients treated with a combination of BB + ivabradine or CCB + long-acting nitrates was low and, in most cases, did not differ between groups with different duration of the disease. The frequency of administration of the triple combination of BB + CCB + long-acting nitrates increased with increasing disease duration and was significantly higher in groups 3 and 4.
At baseline, the distribution of patients according to CCS class did not differ between patients with a disease duration of less than 1 year and those with AP duration of 1–5 years (Table 3). With longer AP duration (groups 3 and 4), a significant increase in the proportion of patients with CCS class III and decrease in the proportion of patients with CCS Class I were observed. When TMZ 80 mg od was added to treatment, in patients with angina duration < 1 year, the proportion of patients with CCS Class I increased by twofold by month 1 (56.8%) and by almost threefold by month 3 (80.7%). Similar improvements were observed in patients with 1–5 years angina duration and were even more pronounced in patients with more than 5 years of angina duration. Significant decreases in proportion of patients with CCS Class III compared to baseline were observed in all patient groups.
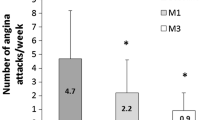
At baseline, the number of angina attacks per week was greater in patients with longer angina duration. Significant decreases in the frequency of angina attacks were observed already at month 1 across all patient groups, with a further decrease observed at month 3 (Fig. 1). In patients with angina duration < 1 year, the frequency of weekly angina attacks decreased by almost threefold, reaching a mean of 1.4 ± 1.7 and by sixfold at month 3, reaching a mean of 0.6 ± 1.0. Similarly, a significant decrease in short-acting nitrate (SAN) consumption was observed starting from month 1 in all patient groups (Fig. 2). The decrease in the frequency of angina attacks in patients with a disease duration of more than 10 years was significantly more pronounced, but the frequency of angina attacks at 3 months remained higher in this patient group than in patients with shorter disease duration. Similar changes were observed with regard to SAN consumption.
The proportion of angina-free patients increased significantly and consistently in all groups, starting already at month 1, with the highest proportion of angina-free patients observed in the group of recently diagnosed patients (70% of patients at month 3) (Fig. 3).
At baseline, physical activity as self-assessed by patients was less limited in patients with AP duration of less than 1 year (Fig. 4). Patients with a disease duration of more than 1 year were more likely to have moderate limitation, substantial limitation, or very marked reduction of physical activity. In patients treated with TMZ 80 mg OD, an improvement in physical activity was observed already at 1 month of therapy, in all patient groups, with further improvement at month 3. The proportion of patients with no or slight limitation of physical activity increased and the proportion of patients with substantial limitation or very marked reduction significantly decreased already after 1 month of therapy.
The share of patients with no or slight physical limitation in group 1 and group 2 was already significantly increased at 1 M (p < 0.01 for no limitation and p < 0.001 for slight limitation increase). The share of patients with no or only a slight physical limitation increased progressively by M3 and in all groups regardless of the duration of the disease (p < 0.001)
At baseline, adherence to antianginal therapy was lower as disease duration increased. Good adherence to antianginal therapy increased significantly in all patient groups at month 1 and continued to increase by month 3 (Fig. 5). The highest proportion of patients with good adherence at month 3 (over 70%) was observed in patients with disease duration of less than 1 year.
Discussion
In the present analysis, we assessed the effect of introducing TMZ 80 mg od to the therapy of stable angina patients with various durations of disease, who were symptomatic despite antianginal therapy, in real clinical practice conditions.
Improvements in CCS Class were observed across all patient groups, with a significant increase in the proportion of CCS Class I patients. Weekly angina attacks and SAN consumption were significantly reduced in all patient groups, regardless of disease duration. The results were significant already at month 1 and continued to improve until month 3. At baseline, there were no angina-free patients in any of the groups. An increase in the proportion of angina-free patients was observed in all groups, particularly in recently diagnosed patients (< 1 year), with half of the patients being asymptomatic at visit 1 and 70% at month 3. Including TMZ in the therapeutic scheme also improved self-reported physical capacity and adherence to antianginal therapy, which might be related to the improvement observed in treatment effectiveness, in all patient groups.
In the newly diagnosed patient group, we observed the highest proportion of patients with CCS Class I, which reached 57% at month 1 in group 1 and 81% at month 3, and the lowest frequency of angina attacks and SAN consumption, although it must be kept in mind that this patient group was in a better state with regard to these parameters at baseline, compared to the other patient groups. Patients with short angina duration (< 1 year) were the most adherent at baseline, which could be related to the fact that they were younger and had fewer comorbidities, and thus were probably taking fewer pills. However, there was still room for adherence to improve, reaching 70% of patients with good adherence at month 3.
Introduction of TMZ led to pronounced improvements in patients with long duration of disease (> 10 years), but the significant improvements observed in patients diagnosed within 1 year highlights the opportunity to improve the clinical status with trimetazidine 80 mg od early on after the diagnosis. This group corresponds to the definition of the new Chronic Coronary Syndrome (CCS) 2019 guidelines of “recently diagnosed patients” (< 1 year from diagnosis) [11].
According to these guidelines, fast symptom relief with timely check-up of the medical treatment result within 2–4 weeks is recommended. Angina is a multifactorial syndrome [11, 24] and symptom alleviation with more than one antianginal drug is described in the guidelines [11]. Very often, the combination of hemodynamic drugs is insufficient to control angina symptoms [18, 20]. There is no evidence from randomized controlled trials of the superiority of the so-called “first-line” antianginal agents coming from a direct comparison with antianginal therapies [25].
The new CCS Guidelines rather recommend a new treatment paradigm of antianginal medical treatment, which should be tailored around the patient profile, co-morbidities, drug tolerance, and potential drug interactions, which is why the guidelines also recommend achieving symptom alleviation with early combination of “first-line” and “second-line” antianginal drugs.
In such a patient-tailored approach, “second-line” drugs like TMZ can be used early alongside “first-line” drugs (beta-blockers, CCBs), for more efficient antianginal relief. TMZ in combination with beta-blockers has been shown to be superior to beta-blockers alone, and to combination of beta-blockers with long-acting nitrates [26, 27]. Therefore, introducing TMZ in combination with a beta-blocker in patients who are still symptomatic despite treatment with the beta-blocker provides an opportunity for more efficient and earlier symptom alleviation. In our study, despite being symptomatic, many patients were on monotherapy at baseline. Even in patients with more than 5 years of angina duration, the proportion of patients treated only with a beta-blocker was 24–30%. Introducing TMZ was particularly beneficial for patients with < 1-year angina duration (recently diagnosed), where more than 50% were taking only a beta-blocker in their therapeutic antianginal scheme. Undertreatment of symptomatic angina patients in clinical practice is not a rare phenomenon [12, 17, 28, 29]. It has been reported that angina is under-treated in clinical practice with 38.5% of patients with daily or weekly angina taking only one antianginal drug [12]; in a large study including more than 12,000 post primary revascularization MI patients, 68% of the patients who reported angina within 6 weeks after the procedure remained treated only with a with beta-blocker [29]. As many patients at diagnosis are already treated with a hemodynamic antianginal drug for reasons other than angina (beta-blockers 50.3%, CCB 17%, LAN 5%) [30], early inclusion of TMZ 80 mg od might be beneficial for these patients, instead of up titrating or adding another hemodynamic agent, which might be related to compromising tolerability due to the enhanced hemodynamic effects of the hemodynamic agent, which might compromise adherence [31] or might have doubtful additional anti-angina efficacy [18].
According to the new guidelines, in the context of antianginal drugs, optimal medical therapy can be defined as the treatment that satisfactorily controls symptoms with effective symptom alleviation, maximum adherence, and minimum side effects [11]. TMZ od is therefore an important part of antianginal therapy, as it effectively improves angina symptoms and shows a good tolerability profile. Tolerability was not assessed in this analysis, but as reported in the main analysis, there were only 0.3% suspected adverse drug reactions [22].
Limitations
Study limitations include limitations inherent to its design (open-label, observational, lack of placebo or other control group) and short follow-up duration. We acknowledge, that due to the chronic and dynamic nature of Chronic Coronary Syndrome, an observational period of at least a year would have brought more rich and accurate data. On the other hand, a short observation period excludes possible changes in the state associated with seasonal, weather changes that can affect the clinical presentation of the disease.
Additionally, the tools used to test physical activity were subjective, and not previously validated, and therefore we could not exclude the possibility of potential bias of the results. The conclusions of the analyses in the subgroup of recently diagnosed patients might be limited due to the low number of patients in this group (n = 88) as compared to the other groups.
Conclusions
Including TMZ 80 mg od to antianginal therapy effectively improved CCS class, frequency of angina attacks and SAN consumption, self-reported patient activity and adherence to antianginal treatment in all patient groups irrespective of angina duration, including patients with recently diagnosed angina, in real clinical practice conditions, thus providing an opportunity to improve symptoms and quality of life whatever the duration of the disease, but most importantly, to provide effective angina relief from the very beginning of diagnosis and treatment.
References
Sanchis-Gomar F, Perez-Quilis C, Leischik R, Lucia A. Epidemiology of coronary heart disease and acute coronary syndrome. Ann Transl Med. 2016;4(13):256.
Timmis A, Townsend N, Gale CP, Torbica A, Lettino M, Petersen SE, et al. European Society of Cardiology: cardiovascular disease statistics 2019. Eur Heart J. 2020;41(1):12–85.
Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, Cushman M, et al. Heart disease and stroke statistics–2015 update: a report from the American Heart Association. Circulation. 2015;131(4):e29–32222.
Moran AE, Forouzanfar MH, Roth GA, Mensah GA, Ezzati M, Flaxman A, et al. The global burden of ischemic heart disease in 1990 and 2010: the Global Burden of Disease 2010 study. Circulation. 2014;129(14):1493–501.
Spertus JA, Jones P, McDonell M, Fan V, Fihn SD. Health status predicts long-term outcome in outpatients with coronary disease. Circulation. 2002;106(1):43–9.
Mozaffarian D, Bryson CL, Spertus JA, McDonell MB, Fihn SD. Anginal symptoms consistently predict total mortality among outpatients with coronary artery disease. Am Heart J. 2003;146(6):1015–22.
Steg PG, Greenlaw N, Tendera M, Tardif J-C, Ferrari R, Al-Zaibag M, et al. Prevalence of anginal symptoms and myocardial ischemia and their effect on clinical outcomes in outpatients with stable coronary artery disease: data from the International Observational CLARIFY Registry. JAMA Intern Med. 2014;174(10):1651–9.
Beatty AL, Spertus JA, Whooley MA. Frequency of angina pectoris and secondary events in patients with stable coronary heart disease (from the Heart and Soul Study). Am J Cardiol. 2014;114(7):997–1002.
Gaglia MA, Torguson R, Lipinski MJ, Gai J, Koifman E, Kiramijyan S, et al. Frequency of angina pectoris after percutaneous coronary intervention and the effect of metallic stent type. Am J Cardiol. 2016;117(4):526–31.
Montalescot G, Sechtem U, Achenbach S, Andreotti F, Arden C, Budaj A, et al. 2013 ESC guidelines on the management of stable coronary artery disease: the task force on the management of stable coronary artery disease of the European Society of Cardiology. Eur Heart J. 2013;34(38):2949–3003.
Knuuti J, Wijns W, Saraste A, Capodanno D, Barbato E, Funck-Brentano C, et al. 2019 ESC guidelines for the diagnosis and management of chronic coronary syndromes. Eur Heart J. 2020;41(3):407–77.
Kureshi F, Shafiq A, Arnold SV, Gosch K, Breeding T, Kumar AS, et al. The prevalence and management of angina among patients with chronic coronary artery disease across US outpatient cardiology practices: insights from the Angina Prevalence and Provider Evaluation of Angina Relief (APPEAR) study. Clin Cardiol. 2017;40(1):6–10.
Niccoli G, Montone RA, Lanza GA, Crea F. Angina after percutaneous coronary intervention: the need for precision medicine. Int J Cardiol. 2017;248:14–9.
Ben-Yehuda O, Kazi DS, Bonafede M, Wade SW, Machacz SF, Stephens LA, et al. Angina and associated healthcare costs following percutaneous coronary intervention: a real-world analysis from a multi-payer database. Catheter Cardiovasc Interv. 2016;88(7):1017–24.
Kwok CS, Shah B, Al-Suwaidi J, Fischman DL, Holmvang L, Alraies C, et al. Timing and causes of unplanned readmissions after percutaneous coronary intervention: insights from the nationwide readmission database. JACC Cardiovasc Interv. 2019;12(8):734–48.
Steg PG, Ferrari R, Ford I, Greenlaw N, Tardif J-C, Tendera M, et al. Heart rate and use of beta-blockers in stable outpatients with coronary artery disease. PLoS ONE. 2012;7(5):e36284.
Qintar M, Spertus JA, Gosch KL, Beltrame J, Kureshi F, Shafiq A, et al. Effect of angina under-recognition on treatment in outpatients with stable ischaemic heart disease. Eur Heart J Qual Care Clin Outcomes. 2016;2(3):208–14.
Fox KM, Mulcahy D, Findlay I, Ford I, Dargie HJ. The Total Ischaemic Burden European Trial (TIBET). Effects of atenolol, nifedipine SR and their combination on the exercise test and the total ischaemic burden in 608 patients with stable angina. The TIBET study group. Eur Heart J. 1996;17(1):96–103.
Pehrsson SK, Ringqvist I, Ekdahl S, Karlson BW, Ulvenstam G, Persson S. Monotherapy with amlodipine or atenolol versus their combination in stable angina pectoris. Clin Cardiol. 2000;23(10):763–70.
Al-Lamee R, Thompson D, Dehbi H-M, Sen S, Tang K, Davies J, et al. Percutaneous coronary intervention in stable angina (ORBITA): a double-blind, randomised controlled trial. Lancet. 2018;391(10115):31–40.
Fragasso G, Perseghin G, de Cobelli F, Esposito A, Palloshi A, Lattuada G, et al. Effects of metabolic modulation by trimetazidine on left ventricular function and phosphocreatine/adenosine triphosphate ratio in patients with heart failure. Eur Heart J. 2006;27(8):942–8.
Glezer MG, Vygodin VA. Anti-anginal effectiveness and tolerability of trimetazidine modified release 80 mg once daily in stable angina patients in real-world practice. Adv Ther. 2018;35(9):1368–77.
Girerd X, Radauceanu A, Achard JM, Fourcade J, Tournier B, Brillet G, et al. Evaluation de l'observance par l'interrogatoire au cours du suivi des hypertendus dans des consultations spécialisées. Arch Mal Coeur Vaiss. 2001;94(8):839–42.
Ferrari R, Camici PG, Crea F, Danchin N, Fox K, Maggioni AP, et al. Expert consensus document: a ‘diamond’ approach to personalized treatment of angina. Nat Rev Cardiol. 2018;15(2):120–32.
Ferrari R, Pavasini R, Camici PG, Crea F, Danchin N, Pinto F, et al. Anti-anginal drugs-beliefs and evidence: systematic review covering 50 years of medical treatment. Eur Heart J. 2019;40(2):190–4.
Michaelides AP, Spiropoulos K, Dimopoulos K, Athanasiades D, Toutouzas P. Antianginal efficacy of the combination of trimetazidine-propranolol compared with isosorbide dinitrate-propranolol in patients with stable angina. Clin Drug Investig. 1997;13(1):8–14.
Szwed H, Sadowski Z, Elikowski W, Koronkiewicz A, Mamcarz A, Orszulak W, et al. Combination treatment in stable effort angina using trimetazidine and metoprolol: results of a randomized, double-blind, multicentre study (TRIMPOL II). TRIMetazidine in POLand. Eur Heart J. 2001;22(24):2267–74.
Shafiq A, Arnold SV, Gosch K, Kureshi F, Breeding T, Jones PG, et al. Patient and physician discordance in reporting symptoms of angina among stable coronary artery disease patients: Insights from the Angina Prevalence and Provider Evaluation of Angina Relief (APPEAR) study. Am Heart J. 2016;175:94–100.
Fanaroff AC, Kaltenbach LA, Peterson ED, Hess CN, Cohen DJ, Fonarow GC, et al. Management of persistent angina after myocardial infarction treated with percutaneous coronary intervention: insights from the TRANSLATE-ACS study. J Am Heart Assoc. 2017;6:e007007. https://doi.org/10.1161/JAHA.117.007007.
Westermann D, Konstantinos Savvatis DW. Prevalence of obstructive coronary artery disease in ambulatory patients with stable angina pectoris. J Clin Exp Cardiolog. 2015;6:387. https://doi.org/10.4172/2155-9880.1000387.
Newby LK, Allen LaPointe NM. Long-term adherence to evidence-based secondary prevention therapies in coronary artery disease. Circulation. 2006;113(2):203–12.
Acknowledgements
The authors would like to thank the participants of the study.
Funding
Sponsorship for this study was provided by Servier, Moscow, Russian Federation. Editorial assistance and the journal’s rapid service fee were funded by Servier, France. All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analysis.
Medical Writing and Editorial Assistance
Writing and editorial assistance was provided by Dr. Diana Toli and Dr. Parvoleta Petrova (Servier, France).
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
List of Investigators
The full list of ODA study investigators is available in the supplementary material.
Disclosures
Maria G. Glezer, scientific coordinator of this study, received honoraria for lectures from Servier, Moscow, Russian Federation. Vladimir A. Vygodin has nothing to disclose.
Compliance with Ethics Guidelines
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study. This study was approved by the Interuniversity Ethical Committee, Moscow.
Data Availability
The datasets during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Consortia
Corresponding author
Additional information
Digital Features
To view digital features for this article go to https://doi.org/10.6084/m9.figshare.12162033.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Glezer, M.G., Vygodin, V.A. & ODA investigators. Effectiveness of Trimetazidine in Patients with Stable Angina Pectoris of Various Durations: Results from ODA. Cardiol Ther 9, 395–408 (2020). https://doi.org/10.1007/s40119-020-00174-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40119-020-00174-7