Abstract
Purpose
The present work aims to isolate and identify bacterial community from the rotary drum compost of green waste such as the water hyacinth. Compost quality was also investigated with the physicochemical parameters and the heavy metal analysis.
Methods
For rotary drum composting, a waste mixture of 150 kg with water hyacinth, cow dung and sawdust were prepared in the proportion of 6:3:1, respectively. The physicochemical parameters such as pH, temperature, volatile solids and the electrical conductivity were analyzed to detect the compost quality. Pb, Ni, Zn, and Cd were investigated for total heavy metals, toxicity characteristic leaching procedure (TCLP), diethylenetriaminepentaacetic acid (DTPA) and water solubility tests. Consistent and active bacterial community were isolated from the rotary drum compost of water hyacinth. Culture-dependent and culture-independent techniques were approached for the isolation process.
Results
Twelve bacteria were isolated and identified by 16S rDNA sequencing and phylogenetic analysis; they majorly belonged to the Bacillus and Enterobacter family. The analysis of temperature, pH, EC, VS and heavy metals depicted the good quality of compost. Heavy metals concentration was in Pb > Ni > Zn > Cd; however, for Pb, Cd and Ni water solubility remained non-detectable, DTPA concentration for Pb and Cd was also found to be non-detectable.
Conclusions
The microbes in water hyacinth compost are metabolically active degrading the organic matter, surviving in the heavy metal-loaded compost environment. Thus, green waste (water hyacinth) can be utilized to isolate bacterial species in anticipation of their application in heavy metal removal in liquid and solid waste sources for micro-bioremediation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Water hyacinth (Eichhornia crassipes) is an aquatic weed, originating in South America. It covers around 23.2% wetland area of the northeast part of India, it was introduced in West Bengal as an ornamental plant, but now it has become a nuisance throughout the country (Abbasi 1998; Husain 2003). Since then, water hyacinth growth has caused various problems in the whole area, e.g., reduction of fishes, blockage of shipping routes, and impeding other irrigational activities (Gunnarsson and Petersen 2007). There are many documentations in the literature regarding the utilization of water hyacinth as a substitute for animal fodder (Tag El-Din 1992), fermentation feed and biogas production (Singhal and Rai 2003), pulp and paper industry and the vermicomposting and composting (Goswami and Saikia 1994; Gajalakshmi et al. 2001; Singh and Kalamdhad 2013a, b). Composting of water hyacinth followed by its use in the land application has come out to be the best method for treatment and utilization of this weed (Malik 2007). However, a huge amount of heavy metals has been reported in the compost of water hyacinth (Singh and Kalamdhad 2012). Additionally, a high count of actinomycetes, streptomycetes, bacteria and fungi was also observed throughout the whole period of compost formation of water hyacinth (Vishan et al. 2014). Contaminated regions such as industrial waste, wastewater, soil, municipal waste and sewage sludge have been used as source of the isolation of the most robust microbes (Affan et al. 2009; Bahig et al. 2008; Bautista-Hernández et al. 2012; Colak et al. 2011; Congeevaram et al. 2007; Jiang et al. 2008; Pal and Paul 2004).
Composting is a rigorous process of aerobic degradation of organic waste by the metabolic activities of the microorganisms, therefore it serves for the abundance of metabolically active microbes (Anastasi et al. 2005; Bhatia et al. 2013; Hassen et al. 2001; Pan and Sen 2013; Ryckeboer et al. 2003). The microbes are dependent on the surrounding conditions such as the temperature, availability of organic waste, pH, species diversity and availability of heavy metals. A large variety of mesophilic, thermo-tolerant microorganisms have been reported in composting of different types of waste materials, it includes bacteria, actinomycetes, streptomycetes and fungi (Hassen et al. 2001; Ryckeboer et al. 2003).
For the current study, the water hyacinth compost was utilized as a source of isolation of the most robust and persistent microbes. However, no work has been reported regarding the detection, isolation, and identification of bacteria present during the compost formation of water hyacinth using rotary drum composter. The hypothesis is that the bacterial community which is capable of sustaining the variable conditions of composting from the initial till the final day could be further utilized for the purpose of micro-bioremediation. Hence, there is an immense possibility in exploitation of microbes for bioadsorption, biotransformation, and/or bioaccumulation of the heavy metals in future (del Carmen Vargas-García et al. 2012; Anastasi et al. 2005). Accordingly, in the present study, basic physiochemical parameters and heavy metal content of compost were examined, along with this culture-dependent and culture-independent approaches were adopted to isolate and identify the bacterial strains.
Materials and methods
Rotary drum composting of water hyacinth
-
Rotary drum composter
Figure 1 depicts the diagram of a rotary drum composter, with a capacity of 550 L; it works in batch mode operation. The pilot-scale rotary drum composter can hold around 150 kg of waste. The period of 20 days is sufficient for composting for proper degradation and stabilization of waste material as previously been studied during in-vessel composting (Kalamdhad and Kazmi 2009). The frequency of turning of the drum manually was per day (per 24 h) with a full rotation of the rotary drum ensuring that the waste moves from one portion to another, for higher temperature in order to degrade the waste properly (Kalamdhad and Kazmi 2009). To maintain the aerobic condition, the side doors at the ends of the rotary drum were left open.
-
Feedstock materials
The waste mixture was prepared by mixing of water hyacinth, cow dung, and sawdust. Water hyacinth was obtained from the contaminated water in an industrial area called Amingaon near the campus of IIT Guwahati, Assam, India. Cow dung was brought from a dairy farm. Sawdust was purchased from a saw mill near to the campus. Fine powder of sawdust and cow dung was prepared by mixing them thoroughly. The water hyacinth was chopped down into small pieces of the order of 1 cm for maintaining proper aeration and moisture control. All the three components, i.e., 90 kg of water hyacinth, 45 kg of cow dung and 15 kg of sawdust were mixed thoroughly in the best proportion of 6:3:1 to the fine mixture as per the previous studies (Vishan et al. 2014).
-
Sampling and analysis of physicochemical parameters
Compost sample was grabbed from the end terminals and mid portion with the help of compost sampler in without disturbing the adjacent materials. On every 2nd-day, samples were collected in triplicates after manual turning of the rotary drum, air dried and separated through 0.2 mm of the sieve, followed by storing for physiochemical analysis.
The observation of temperature was done thrice a day using digital thermometer throughout 20 days of the composting period. Thereafter, the samples were dried in an oven at 105 °C for 24 h to calculate the moisture content, other dried samples were used for analysis of pH (1:10 w/v, waste: water extract), volatile solids (loss on ignition at 550 °C for 2 h), total metal analysis was done by digesting 0.2 g of dried sample in 10 mL mixture of sulphuric acid perchloric acid in the ratio of 5:1 was fed in a block digester for 2 h at 300 °C (Pelican equipment Chennai-India). For water-soluble heavy metal analysis, 2.5 g of sample was extracted with distilled water of 50 mL volume, where sample and solution were in the ratio of 1:20 and kept in a shaker at 100 rpm for 2 h at room temperature. For DTPA (diethylenetriaminepentaacetic acid), 4 g of ground sample (sieved through 0.22 mm sieve) was mixed with 0.005 M DTPA (40 mL), 0.01 M CaCl2 and 0.1 M (triethanolamine) (pH 7.3) and mechanically shaken at 100 rpm. The leachability of heavy metals was determined by TCLP (toxicity characteristic leaching procedure), following the EPA method 1311, 5 g of sample having size less than 9.5 mm was mixed with CH3COOH (100 mL) at pH 4.93 ± 0.05. The sample and solution were taken in a ratio of 1:20 in 125 mL of reagent bottle and kept at 30 ± 2 rpm for 18 h at room temperature. The remaining suspensions were filtered by Whatman no. 42 filter paper after being centrifuged at 10,000 rpm for 5 min. The samples were stored in a specimen tube at 4 °C for heavy metal analysis. The heavy metal analysis was accomplished using atomic absorption spectrophotometer (AAS) (Brand Varian Spectra 55B). All the experiments were done in triplicates, and the result is depicted as the means of them.
For the calculation of the extraction efficiency of heavy metals extractable by DTPA method, the following equation was used (Singh and Kalamdhad 2013a):
where CDTPA and CTotal denote the concentration of heavy metals extractable by DTPA, and the total heavy metal concentration, respectively.
Isolation of bacteria
-
Sampling for microbial analysis
Samples were collected from mid-area, and end terminals on every fourth day such as 0, 4, 8, 12, 16, 20th day after manual turning of the rotary drum composter. The count of microbes was performed by compost addition (10 g) into a sterile distilled water (90 mL) having 0.85% (w/v) sterile NaCl solution into 250-mL Erlenmeyer flasks. Mechanical mixing of the solution was done at 150 rpm for 2 h and 25 °C. Finally, the serial dilution of waste suspensions was done for count of microbes on the different media.
-
Media optimization
The first step towards the isolation of bacterial pure culture was by selecting appropriate growth media. The medium used for bacterial growth can have a major impact on the isolation efficiency. As the source of isolation was water hyacinth compost, the appropriate growth medium for bacteria was unknown. As mentioned in Table 1, four different types of media including two growth media and two minimal media were tested for a maximum number of bacterial species and consistency of performance in supporting the growth of targeted microbes. Cycloheximide (0.2 g/L) was added as antibiotic in all media to avoid contamination by fungi.
Different types of media are mentioned in the literature, on the basis of the goal for the isolation and its source. Growth can be affected by components of media, the difference can be observed in the same sample cultured in nutrient enriched and minimal media. Selection of suitable growth medium is a crucial step for the isolation of novel strains from a waste source, which is enriched with heterotrophic unidentified bacteria. However, a comparative study of bacterial growth and isolation in different nutrient media is missing in the literature.
-
Culture-dependent bacterial identification
The sampling was done as described in the sampling procedure for microbial analysis. After the serial dilution of waste suspensions, diluted samples were used for microbial counts on all four media separately. Spread plate technique was performed to enumerate the bacterial colonies in the compost samples. The petri-plates were then incubated in an inverted position for 24–72 h at 25 °C. Plates with a lower number of distinguished bacterial colonies were picked up for purification and separation by quadrant streaking. Initially, streaking was done on every fourth day, and then the time interval was increased to 15 days. Plating was done till the final 20th day of composting with the observance of more number and sustainable bacterial colonies in different media. The serially diluted samples were plated on MacConkey agar for counting Escherichia coli number in plates kept for 24–48 h at 37 °C. Glucose minimal media depicted the maximum growth of bacterial colonies giving 12 distinguished robust bacterial isolates. Morphological testing of these 12 isolates was performed with HiMedia bacterial kit for the biochemical tests. The identifications were confirmed by using the Bergey’s Manual of systematic bacteriology (Aneja 1999).
-
Culture-independent bacterial identification
-
Genomic DNA isolation from bacterial isolates
Genomic DNA was extracted from anexic culture pellets using the GeNei DNA Purification kit as per manufacturer’s instruction. The purity of DNA was then checked by horizontal gel electrophoresis in 0.8% agarose gel.
-
PCR amplification and 16S rDNA analysis
For PCR amplifications 8F (5′-AGAGTTTGATCCTGGCTCAG-3′) (forward) and 1492R (5′-CGGTTACCTTGTTACGACTT-3′) (reverse) primers were used. Reactions were performed in 50-µL volumes using the Ready mix Taq PCR reaction Mix (Sigma-Aldrich Ready Mix Taq PCR mixture) in PCR-peQLab thermocycler and following a protocol with a denaturation step at 94 °C for 5 min, followed by 35 amplification cycles (95 °C, 60 s; 52 °C, 60 s; 68 °C, 90 s) and a final extension step at 68 °C for 10 min. The amplification products were cleaned using the GenElute PCR Clean-Up kit (Sigma-Aldrich, St. Louis, MO, USA), sequenced and compared to the nucleotide database at GenBank by using BLAST (NCBI).
Results and discussion
Physiochemical analysis of compost
Microbial activities are inter-related with the variations of compost temperature; compost degradation takes place by the metabolic interventions of microbes with the organic wastes. The variability in temperature gives an index of the rate of decomposition by the biological process, and thus plays an important role in the succession of microbes throughout the composting period (Hassen et al. 2001). Figure 2 depicts the variability of the temperature throughout the 20 days of the composting period.
A maximum of 56.5 °C temperature was achieved on the fourth day. A proper combination of waste materials is important for the achievement of high temperature as it gives a proper combination of carbon and nitrogen to the microbial population for their growth and activities (Singh and Kalamdhad 2012). The temperature started rising within 24 h of feeding of the rotary drum with the feedstock and reached its maximum on the fourth day of composting, afterwards it became stabilized. As at the initial time of feeding, organic materials are readily available for microbes to degrade and decompose. In a composting process for proper degradation and maintenance of thermophilic activity, a temperature range of 52–60 °C is suitable (Ryckeboer et al. 2003).
Variability in pH causes changes in the availability of hydrogen ions which thereby affects the interaction and transport across the cell membrane of microbes, consequently affecting the composting process (Plette et al. 1995). Therefore, pH is also an important factor which influences the composting process. Bacteria can develop and survive in the optimal range of pH 6.0–7.0. A pH of 4.0 and 6.5 had been reported suitable for composting of food waste and garden waste, respectively. In composting of wheat straw, a pH range of 6.5–8.0 was reported to be favorable for microbial growth (Pan and Sen 2013). In the present study, initial pH characterization of water hyacinth, cattle (cow) manure, and sawdust was observed to be 6.5, 7.2 and 5.9, respectively. While the initial and final pH of the compost sample mixture was in the range of 5–6.6 and 7–7.6, respectively, as reported in Fig. 3. The pH value of the solution influences the precipitation, speciation, biosorption, hydrolysis and complexation process (Wang et al. 2009). These pH values favored the development of optimum conditions for bacterial growth and activities in the compost sample.
Electrical conductivity (EC) gives the chemical indication of soluble salts during composting process (Pan and Sen 2013). Transportation of nutrients and water into the plants can be reduced due to high EC, thereby affecting the growth of plants (Singh and Kalamdhad 2013a). As depicted in Fig. 3, the final EC value of compost was reduced down to 2.9 dS/m, similar values had been reported by Singh and Kalamdhad (2012). The decrease in EC at the later phase of composting can possibly be due to the precipitation of mineral salts and ammonia volatilization (Ganesh et al. 2012). Also, the EC reduction could be attributed to the release of humic substances which can reduce water solubility by interacting with the metal ions.
Volatile solids in the composting process account for the amount of organic matter oxidized due to the temperature rise. Microbes utilize the soluble organic matter as a primary source of nutrition for their growth and development. However, in the present study, the plant material, i.e., water hyacinth is rich in cellulose, hemicelluloses, reducing sugars, lignin, and lignocellulose. Where the lignocelluloses part is difficult to be solubilized (almost non-degraded) and forms humus-like substances, rest of the parts of the plant are easily degraded (Juan et al. 2013; Sarika et al. 2014). The volatile solids analyzed from 0th to 20th day reduced from 67 to 36%, i.e., an amount of 46% VS reduction was reported (Fig. 4). But this was an incomplete reduction which depicts that more time is needed for the degradation of the complex lignocellulosic material.
The total concentration of metals increases with the increase in organic matter degradation with the release of CO2 and water (Singh and Kalamdhad 2013a). The presence of heavy metals in compost makes it too hazardous to be used in the field. Enhancement of heavy metals in soil laden with compost can increase the heavy metal concentration in plants. Table 2 demonstrates the variation in total concentration of metals (Pb, Zn, Ni, and Cd) during 20 days of the composting period. The order of heavy metals in the compost was Pb > Ni > Zn > Cd; whereas, Zn concentration was reported to be highest of about 220–370 mg/kg in the compost of sewage sludge (Chiang et al. 2007). However, all total metals concentration decreased for the compost of municipal solid waste (Castaldi et al. 2006). Total metal concentration gives information about the level of contamination, however, very less information is available on the bioavailability and leachability of heavy metals, whereby the present study focuses on the scenario of the toxicity of heavy metals in the compost and the microbes present in that contaminated environment.
Bioavailability of heavy metals during rotary drum composting process
-
Water-soluble heavy metals
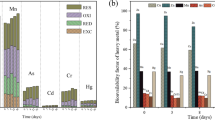
With the decomposition of organic matter, the fraction of heavy metals increases comparatively, but the water-soluble fraction of metals may decrease due to the changes of ionic and oxidizing conditions of surrounding compost environment. The extent of total metal content in compost is not sufficient to predict its potential toxicity, but the bioavailability and leachability of heavy metals are also important (Singh and Kalamdhad 2012). It is essential to determine the water-soluble fraction of metals as they can contaminate the food chain by being present in the compost (Singh and Kalamdhad 2013a). Table 3 shows the changes in water-soluble fractions of Pb, Zn, Ni and Cd during 20 days of the composting period. Water-soluble concentration of Zn was reported to decrease by 41% at the end of the composting period. Water-soluble fraction of Ni, Cd and Pb was not detected throughout the composting period. The increase in water-soluble concentration of Zn was reported by Singh and Kalamdhad (2012) in water hyacinth composting. A decrease of 95 and 60% water-soluble fraction of Pb, Cd and Zn, respectively, was reported in municipal solid waste composting by Castaldi et al. (2006).
-
DTPA-extractable (plant available heavy metals)
DTPA-extractable heavy metal is the potentially available metal for the plant uptake (Chiang et al. 2007). It is a supplementary approach for finding out the potential ecotoxicity of heavy metals (Singh and Kalamdhad 2012). Table 4 shows the variations in DTPA-extractable fractions of Pb, Zn, Ni and Cd during 20 days of the composting period. Pb and Cd DTPA fractions were not detectable throughout the 20 days. The concentration of Ni was, however, reported to increase by 10% from initial till the final 20th day. Hydrolysis of metal ions due to alteration in pH contributes to the variability of metals. The concentration of Zn was observed to increase by 80% from its initial concentration. Similarly, not detectable Pb and Cd along with increased concentration of Ni and Zn were reported in water hyacinth composting (Singh and Kalamdhad 2013a).
-
Leachable heavy metals during composting
TCLP depicts the mobility of the organic and inorganic components present in the solid and liquid waste materials (Singh and Kalamdhad 2013b). A heavy metal found in the leachable state can gradually percolate into the groundwater table causing contamination. For the compost, TCLP should be determined to detect its suitability of being non-hazardous for the land application. Table 5 denotes the leachability changes of Pb, Cd, Zn and Ni throughout the 20 days of the composting period. The concentration of Pb was reduced by 33% from the initial day of composting; however, an increase in Pb concentration was reported in water hyacinth composting by Singh and Kalamdhad (2012), whereas for Cd, the concentration was observed to be reduced by 57%. For Ni, 39% of reduction was reported, whereas 6% of the increase was observed for Zn from their initial till the final day concentration. The influence of pH on the solubility of metal ions is well known, either by solubility equilibria or by complexation pH increase affects the solubility of divalent trace metals (Cambier and Charlatchka 1999).
-
Isolation and identification analysis
Media optimization was done on the basis of their maximum consistency and ability to support the growth of bacterial populations. For this, the different selective media were used for culturing bacterial populations during the whole composting period. From the previous study, it was found that about 104–107 dilutions consistently gave a good colony count in case of mesophilic bacteria. The initial count was as high as 50 for glucose minimal media which decreased to 18 gradually. This was synchronous with nutrient-rich media which yielded 41 colonies initially, reducing to 17 colonies in the end. The total number of colonies obtained from the compost was 656. The glucose minimal media gave the maximum 222 colonies followed by nutrient media with 214 colonies. Sucrose minimal media and tryptic soy agar media yielded a total of 102 and 118 colonies, respectively. The quadrant streaking was continued for 10 cycles which concluded with total 12 isolates from all four media. A total of 12 distinguishable isolates were obtained at the end. All the discrete isolates underwent morphological and biochemical tests as described in Table 5. Gram staining and phase contrast microscopy gave the shape identification of bacterial colonies, which were spotted to be in different forms such as, rod, circular and oval shapes (Bhatia et al. 2013). The colors of the strains were mostly, white, yellow, cream, orange and pink.
-
Molecular identification
The sequence analysis of the 16S rRNA gene is an accurate and faster method to identify the phylogenetic position of bacteria. A full-length (about 1500 bp) 16S rDNA of all 12 bacterial isolates was sequenced and used for construction of a phylogenetic tree. The 16S rDNA gene sequence was used to carry out the BLAST alignment of NCBI GenBank database. On the basis of maximum identity score, the sequences were selected and aligned by using a multiple alignment software program such as Clustal W. Distance matrix was generated using RDP database and the phylogenetic tree was constructed using MEGA 5 (Tamura et al. 2013). Figure 5 shows that the evolutionary history was inferred using the neighbor-joining method (Saitou and Nei 1987). The bootstrap consensus tree inferred from 1000 replicates is taken to represent the evolutionary history of the taxa analyzed (Felsenstein 1985). Branches corresponding to partitions reproduced in less than 50% bootstrap replicates were collapsed. The percentage of replicate trees in which the associated taxa clustered together in the bootstrap test (1000 replicates) is shown next to the branches (Felsenstein 1985). The evolutionary distances were computed using the Kimura 2-parameter method (Kimura 1980) and are in the units of the number of base substitutions per site. The accession nos. received from NCBI were KR780040–KR780050 and KP216715. The bacterial groups were Enterobacter, Bacillus subtilis, Bacillus cereus, Bacillus badius, Bacillus thuringiensis (Fig. 5). Although composting is an aerobic process, members of facultative anaerobic family, i.e., Bacillus are much prevalent which signifies the versatility and survival capacity of these microbes.
Conclusion
Rotary drum composting of water hyacinth is enriched with numerous microorganisms. Bacteria in this environment are metabolically active which leads to degradation of the organic matter, also they are surviving in a heavy metal-loaded compost environment. 12 new robust bacterial strains were isolated as being the most consistent throughout the 20 days of the composting period. Huge degradation of organic matter, temperature, pH and heavy metals was observed. Both the culture-dependent and culture-independent approach yielded new bacterial strains. They survived not only the rigorous variations of composting process, but also the presence of toxic heavy metals in the compost. Reduction in water solubility, leachability and DTPA values of heavy metals indicated the efficient degradation of the organic matter by the microbial activities during the composting process. These bacterial species can be investigated further for their capability of heavy metal removal in liquid and solid waste sources. In this way, these bacteria can contribute to micro-bioremediation.
References
Abbasi SA (1998) Weeds of despair, and hope. Wetl India 3:12–21
Affan QUA, Shoeb E, Badar U, Akhtar J (2009) Isolation and characterization of bacterial isolates having heavy metal tolerance. J Basic Appl Sci 5:55–60
Anastasi A, Varese GC, Marchisio VF (2005) Isolation and identification of fungal communities in compost and vermicompost. Mycologia 97:33–44. doi:10.3852/mycologia.97.1.33
Aneja KR (1999) Experiments in microbiology plant pathology and biotechnology, 4th edn. New Age International Publishers, New Delhi. ISBN 81-224-1494-x
Bahig AE, Aly EA, Khaled AA, Amel KA (2008) Isolation, characterization and application of bacterial population from agricultural soil at Sohag Province, Egypt. Malays J Microbiol 4:42–50. doi:10.21161/mjm.11808
Bautista-Hernández DA, Ramírez-Burgos LI, Duran-Páramo E, Fernández-Linares L (2012) Zinc and lead biosorption by Delftia tsuruhatensis: bacterial strain resistant to metals isolated from mine tailings. J Water Resour Prot 4(4):207–216. doi:10.4236/jwarp.2012.44023
Bhatia A, Madan S, Sahoo J, Ali M, Pathania R, Kazmi AA (2013) Diversity of bacterial isolates during full scale rotary drum composting. Waste Manag 33(7):1595–1601. doi:10.1016/j.wasman.2013.03.019
Cambier P, Charlatchka R (1999) Influence of reducing conditions on the mobility of divalent trace metals in soils. In: Selim HM, Iskandar IK (eds) Fate and transport of heavy metals in the vadose zone. Lewis Publishers, Boca Raton/London/New York
Castaldi P, Santona L, Melis P (2006) Evolution of heavy metals mobility during municipal solid waste composting. Fresen Environ Bull 15(9):1133–1140
Chiang KY, Huang HJ, Chang CN (2007) Enhancement of heavy metal stabilization by different amendments during sewage sludge composting process. J Environ Eng Manag 17(4):249–256
Colak F, Atar N, Yazicioglu D, Olgun A (2011) Biosorption of lead from aqueous solutions by Bacillus strains possessing heavy-metal resistance. Chem Eng J 173:422–428. doi:10.1016/j.cej.2011.07.084
Congeevaram S, Dharani S, Park J, Dexillin M, Thamaraiselvi K (2007) Bio-absorption of chromium and nickel by heavy metal resistant fungal and bacterial isolates. J Hazard Mater 146(1–2):270–277. doi:10.1016/j.jhazmat.2006.12.017
del Carmen Vargas-García M, López MJ, Suárez-Estrella F, Moreno J (2012) Compost as a source of microbial isolates for the bioremediation of heavy metals: in vitro selection. Sci Total Environ 431:62–71. doi:10.1016/j.scitotenv.2012.05.026
El-Din Tag (1992) Utilization of water hyacinth hay in feeding of growing sheep. Indian J Anim Sci 62:89–992
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution. doi:10.2307/2408678
Gajalakshmi S, Ramasamy EV, Abbasi SA (2001) Assessment of sustainable vermiconversion of water hyacinth at different reactor efficiencies employing Eudrilus eugeniae Kinberg. Bioresour Technol 80:131–135. doi:10.1016/S0960-8524(01)00077-3
Ganesh CD, Roshan SW, Khwairakpam M, Kalamdhad AS (2012) Composting of water hyacinth using sawdust/rice straw as a bulking agent. Int J Environ Sci 2(3):1223–1238
Goswami T, Saikia CN (1994) Water hyacinth—a potential source of raw material for greaseproof paper. Bioresour Technol 50:235–238. doi:10.1016/0960-8524(94)90095-7
Gunnarsson CC, Petersen CM (2007) Water hyacinth as a resource in agriculture and energy production: a literature review. Waste Manag 27:117–129. doi:10.1016/j.wasman.2005.12.011
Hassen A, Belguith K, Jedidi N, Cherif M, Boudabous A (2001) Microbial characterization during composting of municipal solid waste. Bioresour Technol 80:217–225. doi:10.1016/S0960-8524(01)00065-7
Husain Z (2003) Environmental issues of Northeast India. Regency Publication, New Delhi
Jiang CY, Sheng XF, Qian M, Wang QY (2008) Isolation and characterization of heavy-metal resistant bacteria Burkholderia sp. from heavy-metal contaminated paddy field soil and its potential in promoting plant growth and heavy metal accumulation in metal-polluted soil. Chemosphere 72(2):157–164. doi:10.1016/j.chemosphere.2008.02.006
Juan ALG, Lopez MJ, Vargas-Garcia MC, Suarez-Estrella F, Jurado M, Moreno J (2013) Tracking organic matter and microbiota dynamics during the stages of lignocellulosic waste composting. Bioresour Technol 146:574–584. doi:10.1016/j.biortech.2013.07.122
Kalamdhad AS, Kazmi AA (2009) Effects of turning frequency on compost stability and some chemical characteristics in a rotary drum composter. Chemosphere 74:1327–1334. doi:10.1016/j.chemosphere.2008.11.058
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16(2):111–120. doi:10.1007/BF01731581
Malik A (2007) Environmental challenge vis a vis opportunity: the case of water hyacinth. Environ Int 33(1):122–138. doi:10.1016/j.envint.2006.08.004
Pal A, Paul AK (2004) Aerobic chromate reduction by chromium-resistant bacteria isolated from serpentine soil. Microbiol Res 159:347–354. doi:10.1016/j.micres.2004.08.001
Pan I, Sen SK (2013) Microbial and Physico-chemical analysis of composting process of wheat straw. Indian J. Biotechol 12:120–128
Parungao MM, Tacata PS, Tanayan CRG, Trinidad LC (2007) Biosorption of copper, cadmium and lead by copper-resistant bacteria isolated from Mogpog River, Marinduque. Philipp J Sci 136(2):155
Plette ACC, Van Riemsdijk WH, Benedetti MF, Van der Wal A (1995) pH dependent charging behavior of isolated cell walls of a gram-positive soil bacterium. J Colloid Int Sci 173:354–363
Ryckeboer J, Mergaert J, Vaes K, Klammer S, De Clercq D, Coosemans J, Insam H, Swings J (2003) A survey of bacteria and fungi occurring during composting and self-heating processes. Ann Microbiol 53:349–410
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4(4):406–425
Sarika D, Singh J, Prasad R, Vishan I, Varma VS, Kalamdhad AS (2014) Study of physico-chemical and biochemical parameters during rotary drum composting of water hyacinth. Int J Recycl Org Waste Agric 3(3):1–10. doi:10.1007/s40093-014-0063-1
Sheng XF, Xia JJ, Jiang CY, He LY, Qian M (2008) Characterization of heavy metal-resistant endophytic bacteria from rape (Brassica napus) roots and their potential in promoting the growth and lead accumulation of rape. Environ Pollut 156(3):1164–1170. doi:10.1016/j.envpol.2008.04.007
Singh J, Kalamdhad AS (2012) Concentration and speciation of heavy metals during water hyacinth composting. Bioresour Technol 124:169–179. doi:10.1016/j.biortech.2012.08.043
Singh J, Kalamdhad AS (2013a) Assessment of bioavailability and leachability of heavy metals during rotary drum composting of green waste (water hyacinth). Ecol Eng 52:59–69. doi:10.1016/j.ecoleng.2012.12.090
Singh J, Kalamdhad AS (2013b) Reduction of bioavailability and leachability of heavy metals during vermicomposting of water hyacinth. Environ Sci Pollut Res 20:8974–8985. doi:10.1007/s11356-013-1848-x
Singhal V, Rai JPN (2003) Biogas production from water hyacinth and channel grass used for phytoremediation of industrial effluents. Bioresour Technol 86:221–225. doi:10.1016/S0960-8524(02)00178-5
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30(12):2725–2729. doi:10.1093/molbev/mst197
Vishan I, Kanekar H, Kalamdhad A (2014) Microbial population, stability and maturity analysis of rotary drum composting of water hyacinth. Biologia 69(10):1303–1313. doi:10.2478/s11756-014-0450-0
Wang SG, Sun XF, Gong WX, Ma Y (2009) Biosorption of metals on to granular sludge. Heavy metals in the environment. Taylor and Francis Group/CRC Press, Boca Raton, pp 201–223
Acknowledgements
The authors gratefully acknowledge the support by Central Instrument facility (CIF) IITG, Centre for the Environment Laboratory, IITG.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Vishan, I., Sivaprakasam, S. & Kalamdhad, A. Isolation and identification of bacteria from rotary drum compost of water hyacinth. Int J Recycl Org Waste Agricult 6, 245–253 (2017). https://doi.org/10.1007/s40093-017-0172-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40093-017-0172-8