Abstract
BACKGROUND:
This study aims to explore the potential mechanism of action of the newly discovered hsa_circ_0128899 (circSPEF2) in diffuse large B-cell lymphoma (DLBCL).
METHODS:
circSPEF2, miR-16-5p and BTB and CNC homologue 2 (BACH2) expression patterns in DLBCL patients and cell lines were studied by RT-qPCR. The biological function of circSPEF2 in vitro and in vivo was investigated by function acquisition experiments. The proliferation activity of lymphoma cells was detected by MTT. Bax, Caspase-3, and Bcl-2 were determined by Western Blot. Apoptosis and the ratio of CD4 to Treg of immune cells in the co-culture system were analyzed by flow cytometry. The mechanism of action of circSPEF2 in DLBCL progression was further investigated by RIP and dual luciferase reporter experiments.
RESULTS:
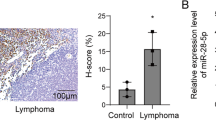
circSPEF2 was a circRNA with abnormally down-regulated expression in DLBCL. Increasing circSPEF2 expression inhibited the proliferative activity and induced apoptosis of lymphoma cells in vitro and in vivo, as well as increased CD4+T cells and decreased Treg cell proportion of immune cells in the tumor microenvironment. Mechanically, circSPEF2 was bound to miR-16-5p expression, while BACH2 was targeted by miR-16-5p. circSPEF2 overexpression-mediated effects on lymphoma progression were reversible by upregulating miR-16-5p or downregulating BACH2.
CONCLUSIONS:
circSPEF2 can influence DLBCL progression by managing cellular proliferation and apoptosis and the proportion of immune cells Treg and CD4 through the miR-16-5p/BACH2 axis.








Similar content being viewed by others
Data availability statement
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
References
Sehn LH, Gascoyne RD. Diffuse large B-cell lymphoma: optimizing outcome in the context of clinical and biologic heterogeneity. Blood. 2015;125:22–32.
Yin X, Xu A, Fan F, Huang Z, Cheng Q, Zhang L, et al. Incidence and mortality trends and risk prediction nomogram for extranodal diffuse large B-cell lymphoma: an analysis of the surveillance, epidemiology, and end results database. Front Oncol. 2019;9:1198.
Jiang X, Wang J, Deng X, Xiong F, Ge J, Xiang B, et al. Role of the tumor microenvironment in PD-L1/PD-1-mediated tumor immune escape. Mol Cancer. 2019;18:10.
Eichmüller SB, Osen W, Mandelboim O, Seliger B. Immune modulatory microRNAs involved in tumor attack and tumor immune escape. J Natl Cancer Inst. 2017. https://doi.org/10.1093/jnci/djx034
Laurent C, Charmpi K, Gravelle P, Tosolini M, Franchet C, Ysebaert L, et al. Several immune escape patterns in non-Hodgkin’s lymphomas. Oncoimmunology. 2015;4:e1026530.
Zou W. Regulatory T cells, tumour immunity and immunotherapy. Nat Rev Immunol. 2006;6:295–307.
Afshar-Sterle S, Zotos D, Bernard NJ, Scherger AK, Rödling L, Alsop AE, et al. Fas ligand-mediated immune surveillance by T cells is essential for the control of spontaneous B cell lymphomas. Nat Med. 2014;20:283–90.
Strasser A, Jost PJ, Nagata S. The many roles of FAS receptor signaling in the immune system. Immunity. 2009;30:180–92.
Kelley TW, Parker CJ. CD4 (+)CD25 (+)Foxp3 (+) regulatory T cells and hematologic malignancies. Front Biosci (Schol Ed). 2010;2:980–92.
Strauss L, Bergmann C, Whiteside TL. Human circulating CD4+CD25highFoxp3+ regulatory T cells kill autologous CD8+ but not CD4+ responder cells by Fas-mediated apoptosis. J Immunol. 2009;182:1469–80.
Mahnke K, Bedke T, Enk AH. Regulatory conversation between antigen presenting cells and regulatory T cells enhance immune suppression. Cell Immunol. 2007;250:1–13.
Kristensen LS, Jakobsen T, Hager H, Kjems J. The emerging roles of circRNAs in cancer and oncology. Nat Rev Clin Oncol. 2022;19:188–206.
Shi Y, Ding D, Qu R, Tang Y, Hao S. Non-coding RNAs in diffuse large B-cell lymphoma. Onco Targets Ther. 2020;13:12097–112.
Deng L, Liu G, Zheng C, Zhang L, Kang Y, Yang F. Circ-LAMP1 promotes T-cell lymphoblastic lymphoma progression via acting as a ceRNA for miR-615-5p to regulate DDR2 expression. Gene. 2019;701:146–51.
Perez de Acha O, Rossi M, Gorospe M. Circular RNAs in blood malignancies. Front Mol Biosci. 2020;7:109.
Lin D, Wang Y, Lei L, Lin C. Circ_0003645 serves as miR-335-5p sponge to promote the biological process of diffuse large B-cell lymphoma by upregulating NFIB. Autoimmunity. 2022;55:127–35.
Zhao CX, Yan ZX, Wen JJ, Fu D, Xu PP, Wang L, et al. CircEAF2 counteracts Epstein-Barr virus-positive diffuse large B-cell lymphoma progression via miR-BART19-3p/APC/beta-catenin axis. Mol Cancer. 2021;20:153.
Cen J, Liang Y, Huang Y, Pan Y, Shu G, Zheng Z, et al. Circular RNA circSDHC serves as a sponge for miR-127-3p to promote the proliferation and metastasis of renal cell carcinoma via the CDKN3/E2F1 axis. Mol Cancer. 2021;20:19.
Wang L, Yang B, Xu Z, Song X, Gong Z, Xue S, et al. NRF1-regulated CircNSUN2 promotes lymphoma progression through activating Wnt signaling pathway via stabilizing HMGA1. Cell Cycle. 2021;20:819–28.
Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116:281–97.
Tagawa H, Ikeda S, Sawada K. Role of microRNA in the pathogenesis of malignant lymphoma. Cancer Sci. 2013;104:801–9.
Yan S, Chen Y, Yang M, Zhang Q, Ma J, Liu B, et al. hsa-MicroRNA-28-5p inhibits diffuse large B-cell lymphoma cell proliferation by downregulating 14-3-3zeta expression. Evid Based Complement Alternat Med. 2022;2022:4605329.
Yu CY, Kuo HC. The emerging roles and functions of circular RNAs and their generation. J Biomed Sci. 2019;26:29.
Hu H, Chen C, Chen F, Sun N. LINC00152 knockdown suppresses tumorigenesis in non-small cell lung cancer via sponging miR-16-5p. J Thorac Dis. 2022;14:614–24.
Lou J, Zhang H, Xu J, Ren T, Huang Y, Tang X, et al. circUSP34 accelerates osteosarcoma malignant progression by sponging miR-16-5p. Cancer Sci. 2022;113:120–31.
Ni C, Fang QQ, Chen WZ, Jiang JX, Jiang Z, Ye J, et al. Breast cancer-derived exosomes transmit lncRNA SNHG16 to induce CD73+gammadelta1 Treg cells. Signal Transduct Target Ther. 2020;5:41.
Zhou Y, Wu H, Zhao M, Chang C, Lu Q. The bach family of transcription factors: A comprehensive review. Clin Rev Allergy Immunol. 2016;50:345–56.
Yang L, Chen S, Zhao Q, Sun Y, Nie H. The critical role of Bach2 in shaping the balance between CD4(+) T cell subsets in immune-mediated diseases. Mediat Inflamm. 2019;2019:2609737.
Roychoudhuri R, Hirahara K, Mousavi K, Clever D, Klebanoff CA, Bonelli M, et al. BACH2 represses effector programs to stabilize T(reg)-mediated immune homeostasis. Nature. 2013;498:506–10.
Kuwahara M, Ise W, Ochi M, Suzuki J, Kometani K, Maruyama S, et al. Bach2-Batf interactions control Th2-type immune response by regulating the IL-4 amplification loop. Nat Commun. 2016;7:12596.
Chen Y, Li M, Cao J, Cai G, Li X, Liu Y, et al. CTLA-4 promotes lymphoma progression through tumor stem cell enrichment and immunosuppression. Open Life Sci. 2021;16:909–19.
Rovira J, Valera A, Colomo L, Setoain X, Rodríguez S, Martínez-Trillos A, et al. Prognosis of patients with diffuse large B cell lymphoma not reaching complete response or relapsing after frontline chemotherapy or immunochemotherapy. Ann Hematol. 2015;94:803–12.
Arnaiz E, Sole C, Manterola L, Iparraguirre L, Otaegui D, Lawrie CH. CircRNAs and cancer: biomarkers and master regulators. Semin Cancer Biol. 2019;58:90–9.
Huang G, Liang M, Liu H, Huang J, Li P, Wang C, et al. CircRNA hsa_circRNA_104348 promotes hepatocellular carcinoma progression through modulating miR-187-3p/RTKN2 axis and activating Wnt/beta-catenin pathway. Cell Death Dis. 2020;11:1065.
Liu Z, Zhou Y, Liang G, Ling Y, Tan W, Tan L, et al. Circular RNA hsa_circ_001783 regulates breast cancer progression via sponging miR-200c-3p. Cell Death Dis. 2019;10:55.
Elkoshi Z. “High Treg” inflammations promote (most) non-hematologic cancers while “Low Treg” inflammations promote lymphoid cancers. J Inflamm Res. 2020;13:209–21.
Mittal S, Marshall NA, Duncan L, Culligan DJ, Barker RN, Vickers MA. Local and systemic induction of CD4+CD25+ regulatory T-cell population by non-Hodgkin lymphoma. Blood. 2008;111:5359–70.
Shang B, Liu Y, Jiang SJ, Liu Y. Prognostic value of tumor-infiltrating FoxP3+ regulatory T cells in cancers: a systematic review and meta-analysis. Sci Rep. 2015;5:15179.
Zhao DM, Thornton AM, DiPaolo RJ, Shevach EM. Activated CD4+CD25+ T cells selectively kill B lymphocytes. Blood. 2006;107:3925–32.
Pandiyan P, Zheng L, Ishihara S, Reed J, Lenardo MJ. CD4+CD25+Foxp3+ regulatory T cells induce cytokine deprivation-mediated apoptosis of effector CD4+ T cells. Nat Immunol. 2007;8:1353–62.
Hu Y, Zhao Y, Shi C, Ren P, Wei B, Guo Y, et al. A circular RNA from APC inhibits the proliferation of diffuse large B-cell lymphoma by inactivating Wnt/beta-catenin signaling via interacting with TET1 and miR-888. Aging (Albany NY). 2019;11:8068–84.
Liu W, Lei L, Liu X, Ye S. CircRNA_OTUD7A upregulates FOXP1 expression to facilitate the progression of diffuse large B-cell lymphoma via acting as a sponge of miR-431-5p. Genes Genom. 2021;43:653–67.
Moloudizargari M, Hekmatirad S, Mofarahe ZS, Asghari MH. et al. Exosomal microRNA panels as biomarkers for hematological malignancies. Curr Probl Cancer. 2021;45:100726.
Peng R, Cao J, Su BB, Bai XS, Jin X, Wang AQ, et al. Down-regulation of circPTTG1IP induces hepatocellular carcinoma development via miR-16-5p/RNF125/JAK1 axis. Cancer Lett. 2022;543:215778.
Saral MA, Tuncer SB, Odemis DA, Erdogan OS, Erciyas SK, Saip P, et al. New biomarkers in peripheral blood of patients with ovarian cancer: high expression levels of miR-16-5p, miR-17-5p, and miR-638. Arch Gynecol Obstet. 2022;305:193–201.
Liu X, Su K, Kuang S, Fu M, Zhang Z. miR-16-5p and miR-145-5p trigger apoptosis in human gingival epithelial cells by down-regulating BACH2. Int J Clin Exp Pathol. 2020;13:901–11.
Ciardullo C, Szoltysek K, Zhou P, Pietrowska M, Marczak L, Willmore E, et al. Low BACH2 expression predicts adverse outcome in chronic lymphocytic leukaemia. Cancers (Basel). 2021;14:23.
Daniels J, Choi J. BACH2 is a putative T-cell lymphoma tumor suppressor that may play a role in product-derived CAR T-cell lymphomas. Blood. 2021;138:2731–3.
Kamio T, Toki T, Kanezaki R, Sasaki S, Tandai S, Terui K, et al. B-cell-specific transcription factor BACH2 modifies the cytotoxic effects of anticancer drugs. Blood. 2003;102:3317–22.
Acknowledgements
Not applicable.
Author information
Authors and Affiliations
Contributions
JZ designed the research study. MX and ZC performed the research. LH and ZW provided help and advice. ZH, LL and JZ analyzed the data. JZ wrote the manuscript. LL reviewed and edited the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethical statement
The present study was approved by the Ethics Committee of Huazhong University of Science and Technology Tongji Medical College Affiliated Union Hospital and written informed consent was provided by all patients prior to the study start (Approval number: C2016TJ1117). All procedures were performed in accordance with the ethical standards of the Institutional Review Board and The Declaration of Helsinki, and its later amendments or comparable ethical standards.
All animal experiments were reviewed and approved by Huazhong University of Science and Technology Tongji Medical College Affiliated Union Hospital Animal Protection and Use Committee (Approval number: C2016TJ1128).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, J., Xu, M., Chen, Z. et al. circ_SPEF2 Regulates the Balance of Treg Cells by Regulating miR-16-5p/BACH2 in Lymphoma and Participates in the Immune Response. Tissue Eng Regen Med 20, 1145–1159 (2023). https://doi.org/10.1007/s13770-023-00585-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13770-023-00585-2