Abstract
Background:
Wear debris-induced osteolysis leads to periprosthetic loosening and subsequent prosthetic failure. Since excessive osteoclast formation is closely implicated in periprosthetic osteolysis, identification of agents to suppress osteoclast formation and/or function is crucial for the treatment and prevention of wear particle-induced bone destruction. In this study, we examined the potential effect of pentamidine treatment on titanium (Ti) particle-induced osteolysis, and receptor activator of nuclear factor-κB ligand (RANKL)-induced osteoclastogenesis.
Methods:
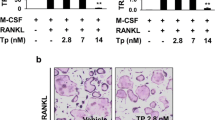
The effect of pentamidine treatment on bone destruction was examined in Ti particle-induced osteolysis mouse model. Ti particles were implanted onto mouse calvaria, and vehicle or pentamidine was administered for 10 days. Then, calvarial bone tissue was analyzed using micro-computed tomography and histology. We performed in vitro osteoclastogenesis assay using bone marrow-derived macrophages (BMMs) to determine the effect of pentamidine on osteoclast formation. BMMs were treated with 20 ng/mL RANKL and 10 ng/mL macrophage colony-stimulating factor in the presence or absence of pentamidine. Osteoclast differentiation was determined by tartrate-resistant acid phosphatase staining, real-time polymerase chain reaction, and immunofluorescence staining.
Results:
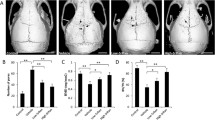
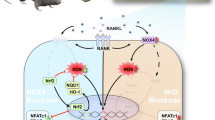
Pentamidine administration decreased Ti particle-induced osteoclast formation significantly and prevented bone destruction compared to the Ti particle group in vivo. Pentamidine also suppressed RANKL-induced osteoclast differentiation and actin ring formation markedly, and inhibited the expression of nuclear factor of activated T cell c1 and osteoclast-specific genes in vitro. Additionally, pentamidine also attenuated RANKL-mediated phosphorylation of IκBα in BMMs.
Conclusion:
These results indicate that pentamidine is effective in inhibiting osteoclast formation and significantly attenuates wear debris-induced bone loss in mice.




Similar content being viewed by others
References
Harris WH. Wear and periprosthetic osteolysis: the problem. Clin Orthop Relat Res. 2001;393:66–70.
Holding CA, Findlay DM, Stamenkov R, Neale SD, Lucas H, Dharmapatni AS, et al. The correlation of RANK, RANKL and TNFalpha expression with bone loss volume and polyethylene wear debris around hip implants. Biomaterials. 2006;27:5212–9.
Kim JA, Ihn HJ, Park JY, Lim J, Hong JM, Kim SH, et al. Inhibitory effects of triptolide on titanium particle-induced osteolysis and receptor activator of nuclear factor-κB ligand-mediated osteoclast differentiation. Int Orthop. 2015;39:173–82.
Zhai Z, Qu X, Li H, Yang K, Wan P, Tan L, et al. The effect of metallic magnesium degradation products on osteoclast-induced osteolysis and attenuation of NF-κB and NFATc1 signaling. Biomaterials. 2014;35:6299–310.
Findlay DM, Haynes DR. Mechanisms of bone loss in rheumatoid arthritis. Mod Rheumatol. 2005;15:232–40.
Feng X, Teitelbaum SL. Osteoclasts: new insights. Bone Res. 2013;1:11–26.
Liu X, Zhu S, Cui J, Shao H, Zhang W, Yang H, et al. Strontium ranelate inhibits titanium-particle-induced osteolysis by restraining inflammatory osteoclastogenesis in vivo. Acta Biomater. 2014;10:4912–8.
Qu S, Bai Y, Liu X, Fu R, Duan K, Weng J. Study on in vitro release and cell response to alendronate sodium-loaded ultrahigh molecular weight polyethylene loaded with alendronate sodium wear particles to treat the particles-induced osteolysis. J Biomed Mater Res A. 2013;101:394–403.
Millett PJ, Allen MJ, Bostrom MP. Effects of alendronate on particle-induced osteolysis in a rat model. J Bone Joint Surg Am. 2002;84A:236–49.
von Knoch M, Wedemeyer C, Pingsmann A, von Knoch F, Hilken G, Sprecher C, et al. The decrease of particle-induced osteolysis after a single dose of bisphosphonate. Biomaterials. 2005;26:1803–8.
Pearson RD, Hewlett EL. Pentamidine for the treatment of Pneumocystis carinii pneumonia and other protozoal diseases. Ann Intern Med. 1985;103:782–6.
Tasaka S. Pneumocystis pneumonia in human immunodeficiency virus-infected adults and adolescents: current concepts and future directions. Clin Med Insights Circ Respir Pulm Med. 2015;9:19–28.
Markowitz J, Chen I, Gitti R, Baldisseri DM, Pan Y, Udan R, et al. Identification and characterization of small molecule inhibitors of the calcium-dependent S100B-p53 tumor suppressor interaction. J Med Chem. 2004;47:5085–93.
Esposito G, Capoccia E, Sarnelli G, Scuderi C, Cirillo C, Cuomo R, et al. The antiprotozoal drug pentamidine ameliorates experimentally induced acute colitis in mice. J Neuroinflammation. 2012;9:277.
Chen YL, Le Vraux V, Giroud JP, Chauvelot-Moachon L. Anti-tumor necrosis factor properties of non-peptide drugs in acute-phase responses. Eur J Pharmacol. 1994;271:319–27.
Rosenthal GJ, Craig WA, Corsini E, Taylor M, Luster MI. Pentamidine blocks the pathophysiologic effects of endotoxemia through inhibition of cytokine release. Toxicol Appl Pharmacol. 1992;112:222–8.
Zhu S, Hu X, Tao Y, Ping Z, Wang L, Shi J, et al. Strontium inhibits titanium particle-induced osteoclast activation and chronic inflammation via suppression of NF-κB pathway. Sci Rep. 2016;6:36251.
Ihn HJ, Lee D, Lee T, Kim SH, Shin HI, Bae YC, et al. Inhibitory effects of KP-A159, a thiazolopyridine derivative, on osteoclast differentiation, function, and inflammatory bone loss via suppression of RANKL-induced MAP kinase signaling pathway. PLoS One. 2015;10:e0142201.
Ihn HJ, Lee T, Kim JA, Lee D, Kim ND, Shin HI, et al. OCLI-023, a novel pyrimidine compound, suppresses osteoclastogenesis in vitro and alveolar bone resorption in vivo. PLoS One. 2017;12:e0170159.
Yoon JY, Baek CW, Kim HJ, Kim EJ, Byeon GJ, Yoon JU. Remifentanil negatively regulates RANKL-induced osteoclast differentiation and bone resorption by inhibiting c-Fos/NFATc1 expression. Tissue Eng Regen Med. 2018;15:333–40.
Ihn HJ, Lee T, Lee D, Kim JA, Kim K, Lim S, et al. A novel benzamide derivative protects ligature-induced alveolar bone erosion by inhibiting NFATc1-mediated osteoclastogenesis. Toxicol Appl Pharmacol. 2018;355:9–17.
Yagi M, Miyamoto T, Sawatani Y, Iwamoto K, Hosogane N, Fujita N, et al. DC-STAMP is essential for cell–cell fusion in osteoclasts and foreign body giant cells. J Exp Med. 2005;202:345–51.
Gallo J, Goodman SB, Konttinen YT, Wimmer MA, Holinka M. Osteolysis around total knee arthroplasty: a review of pathogenetic mechanisms. Acta Biomater. 2013;9:8046–58.
Mediero A, Frenkel SR, Wilder T, He W, Mazumder A, Cronstein BN. Adenosine A2A receptor activation prevents wear particle-induced osteolysis. Sci Transl Med. 2012;4:135ra65.
Boyle WJ, Simonet WS, Lacey DL. Osteoclast differentiation and activation. Nature. 2003;423:337–42.
Franzoso G, Carlson L, Xing L, Poljak L, Shores EW, Brown KD, et al. Requirement for NF-kappaB in osteoclast and B-cell development. Genes Dev. 1997;11:3482–96.
Iotsova V, Caamaño J, Loy J, Yang Y, Lewin A, Bravo R. Osteopetrosis in mice lacking NF-κB1 and NF-κB2. Nat Med. 1997;3:1285–9.
Xu J, Wu HF, Ang ES, Yip K, Woloszyn M, Zheng MH, et al. NF-kappaB modulators in osteolytic bone diseases. Cytokine Growth Factor Rev. 2009;20:7–17.
Takayanagi H, Kim S, Koga T, Nishina H, Isshiki M, Yoshida H, et al. Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts. Dev Cell. 2002;3:889–901.
Acknowledgement
This study was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean Government (MSIT) (NRF-2017R1A2B1005409).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
All animal experiments were approved by the committee on the care and use of animals in research at Kyungpook National University, South Korea, and conducted in accordance with the guidelines for the Care and Use of Laboratory Animals (2015-150).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ihn, H.J., Kim, K., Cho, HS. et al. Pentamidine Inhibits Titanium Particle-Induced Osteolysis In Vivo and Receptor Activator of Nuclear Factor-κB Ligand-Mediated Osteoclast Differentiation In Vitro. Tissue Eng Regen Med 16, 265–273 (2019). https://doi.org/10.1007/s13770-019-00186-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13770-019-00186-y