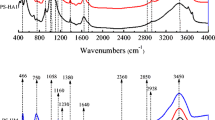
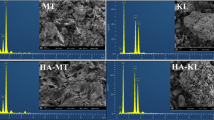
Abstract
Hydrophobic organic compounds (HOCs) exhibit strong hydrophobicity and low water solubility and are readily adsorbed into soil, which is a complex organic compound. In this study, the adsorption effect and mechanism of 10 soil organic matter components on HOCs (represented by aniline) are studied. Furthermore, a chemical extraction method is used to extract different soil organic components to study their properties and functional groups. The adsorption kinetic experiment is conducted to determine the equilibrium time of aniline. The isothermal adsorption experiment is conducted to determine the isothermal adsorption model of aniline and explore its characteristics. Results show that different components have various physical and chemical properties. The pseudo-quadratic kinetic model fitted well with experimental data, indicating that the adsorption of aniline reached equilibrium within 24 h. The Freundlich model fits the isotherm adsorption data of aniline, revealing that the adsorption capacity is significantly positively correlated with the organic carbon content and specific surface area. This study shows that humic acid 2 has the best adsorption capacity for aniline. This study enables the prediction of environmental risks posed by HOCs such as aniline.








Similar content being viewed by others
Data availability
The data that support this study are available in the article and accompanying online supplementary material.
References
Bai YC, Wu FC, Xing BS, Meng W, Shi G, Ma YL, Giesy JP (2015) Isolation and characterization of Chinese standard fulvic acid sub-fractions separated from forest soil by stepwise elution with pyrophosphate buffer. Sci Rep 5(1):1103–1109. https://doi.org/10.1038/srep08723
Begum ZA, Rahman IMM, Tate Y, Ichijo T, Hasegawa H (2018) Binding of proton and iron to lignite humic acid size-fractions in aqueous matrix. J Mol Liq 254:241–247. https://doi.org/10.1016/j.molliq.2018.01.104
Bellotti D, Tocchio C, Guerrini R, Rowińska-Żyrek M, Remelli M (2019) Thermodynamic and spectroscopic study of Cu(ii) and Zn(ii) complexes with the peptide (148–156) fragment of C4YJH2, a putative metal transporter of Candida albicans. Metallomics 11(12):1988–1998. https://doi.org/10.1039/C9MT00251K
Caupos E, Touffet A, Mazellier P, Croue JP (2015) Partitioning of the pesticide trifluralin between dissolved organic matter and water using automated SPME-GC/MS. Environ Sci Pollut Res Int. https://doi.org/10.1007/s11356-014-3614-0
Chen FR, Chefetz B, Thompson ML (2021) Comparison of adsorption behaviors of selected endocrine-disrupting compounds in soil. J Environ Qual 50(3):756–767. https://doi.org/10.1002/jeq2.20221
Christensen BT (2001) Physical fractionation of soil and structural and functional complexity in organic matter turnover: turnover of soil organic matter. Eur J Soil Sci 52(3):345–353. https://doi.org/10.1046/j.1365-2389.2001.00417.x
Cong X, Bi R, Sun SK (2020) Adsorption thermodynamics and kinetics of PCB138 on peat soil and its different soil organic matter FRACTIONS. J Ecol Environ 29:394–401. https://doi.org/10.16258/j.cnki.1674-5906.2020.02.022
Dai SX, Zhao L, Teng Y, Wang XM, Ren WJ (2018) Effect of composition and microstructure of humic acid on 3,3′,4,4′-tetrachlorobiphenyl sorption. Environ Sci Pollut Res 25(15):14656–14665. https://doi.org/10.1007/s11356-018-1536-y
Ezzati R (2020) Derivation of Pseudo-first-order, Pseudo-second-order and modified Pseudo-first-order rate equations from Langmuir and Freundlich isotherms for adsorption. Chem Eng J. https://doi.org/10.1016/j.cej.2019.123705
Han LF, Sun K, Kang MJ, Wu FC, Xing BS (2014) Influence of functional groups and pore characteristics of organic matter on the sorption of hydrophobic organic pollutants. Environ Chem 33(11):1811–1820. https://doi.org/10.7524/j.issn.0254-6108.2014.11.009
He L, Qiu ZF, Lü SG, Lu ZC, Wang ZL, Sui Q, Lin KF, Liu YD (2012) Characteristics and influencing factors of trichloroethylene adsorption in different soil types. Environ Sci 33(11):3976–3982. https://doi.org/10.13227/j.hjkx.2012.11.044
Hua Q, Heng Li, Cong Z et al (2014) Optimization of hum-acid method in soil. Environ Prot Sci 40(06):83–87. https://doi.org/10.16803/j.cnki.issn.1004-6216.2014.06.019
Junhe W, Qiyan S, Jianguo T, Ying L (2021) Analysis on Influencing Factors of soil air exchange process of persistent organic pollutants. Resour Environ Yangtze River Basin 30:879–888
Kaur P, Bansal P, Sud D (2021) Adsorption behavior of quinalphos and its leaching potential in different agricultural soils. Soil Sediment Contam 30(6):730–742. https://doi.org/10.1080/15320383.2021.1890691
Lambert SM (1968) Omega (Omega.), a useful index of soil sorption equilibria. J Agric Food Chem 16(2):340–343. https://doi.org/10.1021/jf60156a038
Leal RMP, de Souza AJ, Reia MY, Alleoni LRF, Tornisielo VL, Regitano JB (2022) Soil factors affecting sulfadiazine sorption in Brazilian soils. Water Air Soil Pollut 233(4):2027–2034. https://doi.org/10.1007/s11270-022-05588-8
Li JR, Wang YX, Wang X, Yuan B, Fu ML (2015) Intercalation and adsorption of ciprofloxacin by layered chalcogenides and kinetics study. J Colloid Interface Sci 453:69–78. https://doi.org/10.1016/j.jcis.2015.03.067
Li FF, Pan B, Liang N, Chang ZF, Zhou YW, Wang L, Li H, Xing BS (2017) Reactive mineral removal relative to soil organic matter heterogeneity and implications for organic contaminant sorption. Environ Pollut 227:49–56. https://doi.org/10.1016/j.envpol.2017.04.047
Liu X, Zhang LF (2015) Removal of phosphate anions using the modified chitosan beads: adsorption kinetic, isotherm and mechanism studies. Powder Technol 277:112–119. https://doi.org/10.1016/j.powtec.2015.02.055
Liu L, Rao L, Hu JH, Zhou WW, Li BT, Tang LM (2021) Effects of different factors on the adsorption–desorption behavior of Glyamifop and its migration characteristics in agricultural soils across China. Microchem J 170:106646. https://doi.org/10.1016/j.microc.2021.106646
Lu J, Wang JH, Li JZ, Xin J, Li M, Liu X (2012) Adsorption desorption characteristics of ethinylestradiol and nonylphenol in soil. J Environ Sci 33:3885–3892. https://doi.org/10.13227/j.hjkx.2012.11.038
Lu MM, Zhang YB, Zhou YL, Su ZJ, Liu BB, Li GH, Jiang T (2018) Adsorption-desorption characteristics and mechanisms of Pb(II) on natural vanadium, titanium-bearing magnetite-humic acid magnetic adsorbent. Powder Technol 344:947–958. https://doi.org/10.1016/j.powtec.2018.12.081
Lu MM, Zhang YB, Su YB, Jiang TB (2020) The NMR and spectral study on the structure of molecular size-fractionated lignite humic acid. Resour Environ Sustain 2:100004. https://doi.org/10.1016/j.resenv.2020.100004
Mudhoo A, Garg VK, Wang S (2012) Removal of heavy metals by biosorption. Environ Chem Lett 10:109–117. https://doi.org/10.1007/s10311-011-0342-2
Mutavdžić PD, Glavač A, Gluhak M, Runje M (2018) Sorption of albendazole in sediments and soils: isotherms and kinetics. Chemosphere 193:635–644. https://doi.org/10.1016/j.chemosphere.2017.11.025
Ofomaja AE, Naidoo EB, Modise SJ (2010) Kinetic and pseudo-second-order modeling of lead biosorption onto pine cone powder. Ind Eng Chem Res 49(6):2562–2572. https://doi.org/10.1021/ie901150x
Patiha HE, Hidayat Y, Firdaus M (2016) The langmuir isotherm adsorption equation: the monolayer approach. IOP Conf Ser Mater Sci Eng 107(1):9. https://doi.org/10.1088/1757-899X/107/1/012067
Peruchi LM, Fostier AH, Rath S (2015) Sorption of norfloxacin in soils: analytical method, kinetics and Freundlich isotherms. Chemosphere 119:310–317. https://doi.org/10.1016/j.chemosphere.2014.06.008
Petridis L, Ambaye H, Jagadamma S, Kilbey SM, Lokitz BS, Lauter V, Mayes MA (2014) Spatial arrangement of organic compounds on a model mineral surface: implications for soil organic matter stabilization. Environ Sci Technol 48(1):79–84. https://doi.org/10.1021/es403430k
Pino-Herrera DO, Pechaud Y, Huguenot D, Esposito G, van Hullebusch ED, Oturan MA (2017) Removal mechanisms in aerobic slurry bioreactors for remediation of soils and sediments polluted with hydrophobic organic compounds: an overview. J Hazard Mater 339:427–449. https://doi.org/10.1016/j.jhazmat.2017.06.013
Rana AK, Mishra YK, Gupta VK, Thakur VK (2021a) Sustainable materials in the removal of pesticides from contaminated water: perspective on macro to nanoscale cellulose. Sci Total Environ 797:149129
Rana AK, Gupta VK, Saini AK, Voicu SI, Thakur AMH, Kumar V (2021b) Water desalination using nanocelluloses/cellulose derivatives based membranes for sustainable future. Desalination. https://doi.org/10.1016/J.DESAL.2021.115359
Rozanov LN (2021) Kinetic equations of non-localized physical adsorption in vacuum for Freundlich adsorption isotherm. Vacuum. https://doi.org/10.1016/J.VACUUM.2021.110267
Sarkar D, De DK, Das R, Mandal B (2014) Removal of organic matter and oxides of iron and manganese from soil influences boron adsorption on soil. Geoderma 214–215:213–216. https://doi.org/10.1016/j.geoderma.2013.09.009
Shalika R, Kumar S, Rana A, Padwad Y, Shashi B (2021) Biological activity of phenolics enriched extracts from industrial apple pomace. Indus Crops Prod. https://doi.org/10.1016/J.INDCROP.2020.113158
Sleep BE, McClure PD (2001) The effect of temperature on adsorption of organic compounds to soils. Can Geotech J 38(1):46–52
Sun JC, Zhou DD, Wang W, Qian KP, Liu Y, Zhang J (2021) Modification of biochar and its adsorption and degradation of pollutants. Environ Chem 40:1503–1513
Tang S, Lin L, Wang X, Sun X, Yu A (2021) Adsorption of fulvic acid onto polyamide 6 microplastics: influencing factors, kinetics modeling, site energy distribution and interaction mechanisms. Chemosphere. https://doi.org/10.1016/J.CHEMOSPHERE.2021.129638
Ukalska-Jaruga A, Klimkowicz-Pawlas A, Smreczak B (2019) Characterization of organic matter fractions in the top layer of soils under different land uses in Central-Eastern Europe. Soil Use Manag 35(4):595–606. https://doi.org/10.1111/sum.12514
Ulantoya (2012) Adsorption behavior and mechanism of water soluble organic matter from landfill leachate in soil. Guangdong Agric Sci 39:43–45. https://doi.org/10.16768/j.issn.1004-874x.2012.20.011
Wang XL, Guo XY, Yang Y, Tao S, Xing BS (2011) Sorption mechanisms of phenanthrene, lindane, and atrazine with various humic acid fractions from a single soil sample. Environ Sci Technol 45(6):2124–2130. https://doi.org/10.1021/es102468z
Wang F, He JT, He BNQ, Zhu XJ, Qiao XC, Peng LY (2018) Formation process and mechanism of humic acid-kaolin complex determined by carbamazepine sorption experiments and various characterization methods. J Environ Sci 69:251–260. https://doi.org/10.1016/j.jes.2017.10.020
Wu QQ, Yang Q, Zhou WJ, Zhu LZ (2015) Sorption characteristics and contribution of organic matter fractions for atrazine in soil. J Soils Sediments 15(11):2210–2219. https://doi.org/10.1007/s11368-015-1162-y
Xu ML, Wei XY, Yu XY, Liu FJ, Wu QC, Li S, Wang SK, Liu GH, Liu ZQ, Guo XH, Zhang YY, Zong ZM (2019) Insight into molecular compositions of soluble species from sequential thermal dissolution of Liuhuanggou bituminous coal and its extraction residue. Fuel 253:762–771. https://doi.org/10.1016/j.fuel.2019.05.045
Xue N, Li FS, Ding Q (2015) Risk management for remediation of organic contaminated sites. Chemical Industry Press
Xue G, Zheng M, Qian Y, Li Q, Gao P, Liu Z, Chen H, Li X (2020) Comparison of aniline removal by UV/CaO2 and UV/H2O2: degradation kinetics and mechanism. Chemosphere. https://doi.org/10.1016/j.chemosphere.2020.126983
Zanin L, Tomasl N, Cesco S, Varanini Z, Pinton R (2019) Humic substances contribute to plant iron nutrition acting as chelators and biostimulants. Front Plant Sci 10:1–10
Zhang D, Hou L, Zhu D, Chen W (2014a) Synergistic role of different soil components in slow sorption kinetics of polar organic contaminants. Environ Pollut 184:123–130. https://doi.org/10.1016/j.envpol.2013.08.022
Zhang T, Xu BB, Wu F (2014b) Study on adsorption kinetics and thermodynamics of benzotriazole by humic acid. Environ Sci Res 27:540–546. https://doi.org/10.13198/j.issn.1001-6929.2014.05.13
Zhang X, Sarmah AK, Bolan NS, He L, Lin X, Che L, Tang C, Wang H (2016) Effect of aging process on adsorption of diethyl phthalate in soils amended with bamboo biochar. Chemosphere 142:28–34. https://doi.org/10.1016/j.chemosphere.2015.05.037
Zhao LX, Zhang YF, Fang SH, Zhu LY, Liu ZT (2014) Comparative sorption and desorption behaviors of PFHxS and PFOS on sequentially extracted humic substances. J Environ Sci 26(12):2517–2525. https://doi.org/10.1016/j.jes.2014.04.009
Zhou YL, Zhang YB, Li GH, Jiang T (2016) Effects of metal cations on the fulvic acid (Fa) adsorption onto natural iron oxide in iron ore pelletizing process. Powder Technol 302:90–99. https://doi.org/10.1016/j.powtec.2016.08.042
Zhou B, Zhang Z, Wang S, Wu Y, Hu S, Sun R (2020) Batch adsorption and column leaching studies of aniline in chinese loess under different hydrochemical conditions. Bull Environ Contam Toxicol 104:511–519. https://doi.org/10.1007/s00128-020-02830-z
Acknowledgements
The authors are grateful to Dr. Yingchen Bai and Dr. Fujun Ma for their valuable suggestions. Y. Ma thanks the China Scholarship Council (CSC) for supporting her one-year study at UMass Amherst.
Funding
This research was financially supported by the National Natural Science Funds of China [Grant number 41701353], the Yue Qi Young Scholar Project at China University of Mining & Technology in Beijing [Grant number 2019QN09], and the Fundamental Research Funds for the Central Universities [Grant number 2020SKHH01]. The funders had no role in the study design; in the collection, analysis, and interpretation of data; in the writing of the report; or in the decision to submit the paper for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Editorial responsibility: Jing Chen.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yan, M., Lu, C., Shengji, A. et al. Influence of soil organic components on the aniline adsorption mechanism. Int. J. Environ. Sci. Technol. 20, 12117–12130 (2023). https://doi.org/10.1007/s13762-023-04773-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-023-04773-4