Abstract
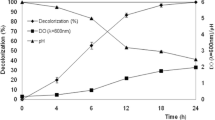
Azo dyes are the largest group of chemically synthesized colorants used in various industries. The hazardous nature of these dyes has led to the development of stringent environmental laws. Thus, mounting pressure exists on the industries for development of cost-effective dye removal systems. The current work focuses on the bioremoval of azo dye Reactive Orange 16 (RO-16) using indigenous bacteria isolated from contaminated sample of a textile industry. The most potent bacterium was capable of decolorizing 89.82% of RO-16 within 24 h under static conditions and was identified as Bacillus flexus VITSP6. When physicochemical parameters were optimized, B. flexus VITSP6 showed efficient decolorization at a temperature of 37 °C, pH 11, glucose as carbon source and peptone as nitrogen source. Interestingly, although B. flexus VITSP6 showed highest decolorization at 37 °C, it was able to maintain its decolorization potency even at elevated temperatures as high as 52 °C. Further the mechanism of dye decolorization was deduced with the help of high-performance liquid chromatography, Fourier transform infrared spectroscopy and liquid chromatography mass spectrometry analyses. These analyses revealed that RO-16 underwent biotransformation to different lower molecular weight aromatic compounds. The toxicity of these biotransformation products were evaluated by conducting phytotoxicity, cytotoxicity and biotoxicity assays. All the toxicity assays indicated that the biotransformed products were non-toxic in nature. The efficient dye decolorization property of B. flexus VITSP6 and its ability to withstand high temperature and alkaliphilic nature make it a highly potential organism to be applied in the dye removal treatments.







Similar content being viewed by others
References
APHA (2005) Standard methods for the examination of water and wastewater, 21st edn. American Public Health Association/American Water Works Association/Water Environment Federation, Washington
Aravindhan R, Rao JR, Nair BU (2007) Removal of basic yellow dye from aqueous solution by sorption of green alga Caulerpa sealpelliformis. J Hazard Mater 142:68–76. https://doi.org/10.1016/j.jhazmat.2006.07.058
Asad A, Amoozegar MA, Pourbabaee AA, Sarbolouki MN, Dastgheib SMM (2007) Decolorization of textile azo dyes by newly isolated halophilic and halotolerant bacteria. Bioresour Technol 98:2082–2088. https://doi.org/10.1016/j.biortech.2006.08.020
Attschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI BLAST: a new generation of protein database search programs. Nucleic Acid Resour 25(17):3389–3402. https://doi.org/10.1093/nar/25.17.3389
Ayed L, Mahdhi A, Cheref A, Bakhrouf A (2011) Decolorization and degradation of azo dye methyl red by an isolated Sphingomonas paucimobilis: biotoxicity and metabolites characterization. Desalination 274:272–277. https://doi.org/10.1016/j.desal.2011.02.024
Bhatt N, Patel KC, Keharia H, Madamwar D (2005) Decolorization of diazo-dye reactive blue 172 by Pseudomonas aeruginosa NBAR12. J Basic Microbiol 45:407–418. https://doi.org/10.1002/jobm.200410504
Bhavsar S, Dudhagara P, Tank S (2018) R software package based statistical optimization of process components to simultaneously enhance the bacterial growth, laccase production and textile dye decolorization with cytotoxicity study. PLoS ONE 13:e0195795. https://doi.org/10.1371/journal.pone.0195795
Carballo JL, Hernández-Inda ZL, Pérez P, García-Grávalos MD (2002) A comparison between two brine shrimp assays to detect in vitro cytotoxicity in marine natural products. BMC Biotechnol 2:17. https://doi.org/10.1186/1472-6750-2-17
Central Pollution Control Board (2010) Pollution control acts, rules and notifications issued thereunder. CPCB, New Delhi
Chavan RB (2011) Environmentally friendly dyes. In: Clark M (ed) Wood head publishing series in textiles, handbook of textile and industrial dyeing. Woodhead Publishing, Sawston, pp 515–561
Chen KC, Wu JY, Liou DJ, Huang SCJ (2003) Decolorization of the textile dyes by newly isolated bacterial strains. J Biotechnol 101:57–68. https://doi.org/10.1016/S0168-1656(02)00303-6
Chen Y, Feng L, Li H, Wang Y, Chen G, Zhang Q (2018) Biodegradation and detoxification of direct black G textile dye by a newly isolated thermophilic microflora. Bioresour Technol 250:650–657. https://doi.org/10.1016/j.biortech.2017.11.092
Chung KT, Fluk GE, Andrews AE (1981) Mutagenicity testing of some commonly used dyes. Appl Environ Microbiol 42(4):641–648
Dave SR, Patel TL, Tipre DR (2015) Bacterial degradation of azo dye containing wastes. In: Singh S (ed) Microbial degradation of synthetic dyes in wastewaters. Environmental science and engineering. Springer, Cham, pp 57–83
Dawkar VV, Jadhav UU, Jadhav SU, Govindwar SP (2008) Biodegradation of disperse textile dye Brown 3REL by newly isolated Bacillus sp. VUS. J Appl Microbiol 105(1):14–24. https://doi.org/10.1111/j.1365-2672.2008.03738.x
EPA (2001) Parameters of water quality: interpretation and standards. Environmental Protection Agency, Wexford
Evangelista-Barreto NS, Albuquerque CD, Vieira RHSF, Campos-Takaki GM (2009) Cometabolic decolorization of the reactive azo dye orange II by Geobacillus stearothermophilus UCP 986. Text Res J 79:1266–1273. https://doi.org/10.1177/0040517508087858
Gahlout M, Gupte S, Gupte A (2013) Optimization of culture condition for enhanced decolorization and degradation of azo dye reactive violet 1 with concomitant production of ligninolytic enzymes by Ganoderma cupreum AG-1. 3 Biotech 3:143–152. https://doi.org/10.1007/s13205-012-0079-z
Guo J, Zhou J, Wang D, Tian C, Wang P, Salah Uddin M (2008) A novel moderately halophilic bacterium for decolorizing azo dye under high salt condition. Biodegradation 19:15. https://doi.org/10.1007/s10532-007-9110-1
Haq I, Kumari V, Kumar S, Raj A, Lohani M, Bhargava R (2016) Evaluation of the phytotoxic and genotoxic potential of pulp and paper mill effluent using Vigna radiata and Allium cepa. Adv Biol 2016:10. https://doi.org/10.1155/2016/8065736
Jain K, Shah V, Chapla D, Madamwar D (2012) Decolorization and degradation of azo dye—reactive violet 5R by an acclimatized indigenous bacterial mixed cultures-SB4 isolated from anthropogenic dye contaminated soil. J Hazard Mater 213–214:378–386. https://doi.org/10.1016/j.jhazmat.2012.02.010
Kapdan IK, Oztekin R (2003) Decolorization of textile dyestuff Reactive Orange 16 in fed-batch reactor under anaerobic condition. Enzyme Microb Technol 33:231–235. https://doi.org/10.1016/S0141-0229(03)00128-5
Khan MZ, Singh S, Sreekrishnan TR, Ahammad SZ (2014) Feasibility study on anerobic biodegradation of azo dye Reactive Orange 16. RSC Adv 4(87):46851–46859. https://doi.org/10.1039/C4RA06716A
Lin YH, Leu JY (2008) Kinetics of reactive azo-dye decolorization by Pseudomonas luteola in a biological activated carbon process. Biochem Eng J 39:457–467. https://doi.org/10.1016/j.bej.2007.10.015
Mielgo I, Moreira MT, Feijoo G, Lema JM (2001) A packed-bed fungal bioreactor for continuous decolourisation of azo-dyes (Orange II). J Biotechnol 89:99–106. https://doi.org/10.1016/S0168-1656(01)00319-4
Misal SA, Lingojwar DP, Shinde RM, Gawai KR (2011) Purification and characterization of azoreductase from alkaliphilic strain Bacillus badius. Process Biochem 46:1264–1269. https://doi.org/10.1016/j.procbio.2011.02.013
OECD (2006) Limits on aromatic amines in textiles coloured with azo dyes. In: Environmental requirements and market access. OECD Publishing, Paris
Pandey AK, Sarada DVL, Kumar A (2016) Microbial decolorization and degradation of reactive red 198 azo dye by a newly isolated alkaligenes species. Proc Natl Acad Sci India Sect B Biol Sci 86:805–815. https://doi.org/10.1007/s40011-015-0497-x
Prasad ASA, Rao KVB (2013) Aerobic biodegradation of Azo dye by Bacillus cohnii MTCC 3616; an obligately alkaliphilic bacterium and toxicity evaluation of metabolites by different bioassay systems. Appl Microbiol Biotechnol 97:7469–7481. https://doi.org/10.1007/s00253-012-4492-3
Prasad ASA, Satyanaryana VSV, Rao KVB (2013) Biotransformation of direct blue 1 by a moderately halophilic bacterium Marinobacter sp. strain HBRA and toxicity assessment of degraded metabolites. J Hazard Mater 262:674–684. https://doi.org/10.1016/j.jhazmat.2013.09.011
Pricelius S, Held C, Murkovic M, Bozic M, Kokol V, Cavaco-Paulo A, Guebitz GM (2007) Enzymatic reduction of azo and indigoid compounds. Appl Microbiol Biotechnol 77(2):321–327. https://doi.org/10.1007/s00253-007-1165-8
Rao KVB, Prasad ASA (2014) Biodecolourisation of azo dye reactive red 22 by Bacillus infantis strain AAA isolated from seawater and toxicity assessment of degraded metabolites. Nat Environ Pollut Technol 13:369–374
Roat C, Kadam A, Patel T, Dave S (2016) Biodegradation of diazo dye, reactive blue 160 by isolate Microbacterium sp. B12 mutant: identification of intermediates by LC-MS. Int J Curr Microbiol Appl Sci 5:534–547. https://doi.org/10.20546/ijcmas.2016.503.063
Rosu CM, Avadanei M, Gherghel D, Mihasan M, Mihai C, Trifan A, Miron A, Vochita G (2018) Biodegradation and detoxification efficiency of azo-dye Reactive Orange 16 by Pichia kudriavzevii CR-Y103. Water Air Soil Pollut 229:15. https://doi.org/10.1007/s11270-017-3668-y
Sahasrabudhe MM, Pathade GR (2011) Biodegradation of sulphonated azo dye C.I. Reactive Orange 16 by Enterococcus faecalis strain YZ 66. Eur J Exp Biol 1:163–173
Sahasrabudhe M, Pathade G (2013) Biodegradation of azo dye C.I. Reactive Orange 16 by an actinobacterium Georgenia sp. CC-NMPT-T3. Int J Adv Res 1:91–99
Saranraj P (2013) Bacterial biodegradation and decolourization of toxic textile azo dyes. Afr J Microbiol Res 7:3885–3890. https://doi.org/10.5897/AJMR2013.5788
Shah MP (2014) Biodegradation of azo dyes by three isolated bacterial strains: an environmental bioremedial approach. J Microbial Biochem Technol S 3:007. https://doi.org/10.4172/1948-5948.S3-007
Shah MP, Patel KA, Nair SS, Darji AM (2013) Optimization of environmental parameters on microbial degradation of reactive black dye. J Bioremed Biodeg 4:183–188. https://doi.org/10.4172/2155-6199.1000183
Shobana S, Thangam EB (2012) Biodegradation and decolorization of Reactive Orange 16 by Nocardiopsis alba soil isolate. J Bioremed Biodeg 3:155–162. https://doi.org/10.4172/2155-6199.1000155
Singh S, Chatterji S, Nandini PT, Prasad ASA, Rao KVB (2015) Biodegradation of azo dye Direct Orange 16 by Micrococcus luteus strain SSN2. Int J Environ Sci Technol 12:2161–2168. https://doi.org/10.1007/s13762-014-0588-x
Solís M, Solís A, Pérez HI, Manjarrez N, Flores M (2012) Microbial decolouration of azo dyes: a review. Process Biochem 47:1723–1748. https://doi.org/10.1016/j.procbio.2012.08.014
Soni RK, Bhatt NS, Modi HA, Acharya PB (2016) Decolorization, degradation and subsequent toxicity assessment of reactive red 35 by Enterococcus gallinarum. Curr Biotechnol 5:325–336. https://doi.org/10.2174/2211550105666151211195703
Spagni A, Grilli S, Casu S, Mattioli D (2010) Treatment of a simulated textile wastewater containing the azo-dye Reactive Orange 16 in an anaerobic-biofilm anoxic-aerobic membrane bioreactor. Int Biodeterior Biodegrad 64(7):676–681. https://doi.org/10.1016/j.ibiod.2010.08.004
Teli MD (2016) Environmental textiles: testing and certification. In: Wang L (ed) Woodhead publishing series in textiles, performance testing of textiles: methods, technology and applications. Woodhead Publishing, Sawston, pp 177–192
Telke A, Kalyani D, Jadhav J, Govindwar S (2008) Kinetics and mechanism of reactive red 141 degradation by a bacterial isolate Rhizobium radiobacter MTCC 8161. Acta Chim Slov 55:320–329
Telke AA, Kalyani DC, Dawkar VV, Govindwar SP (2009) Influence of organic and inorganic compounds on oxidoreductive decolorization of sulfonated azo dye CI Reactive Orange 16. J Hazard Mater 172(1):298–309. https://doi.org/10.1016/j.jhazmat.2009.07.008
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position- specific gap penalties and weight matrix choice. Nucleic Acids Res 22(22):4673–4680
Tony BD, Goyal D, Khanna S (2009) Decolorization of textile azo dyes by aerobic bacterial consortium. Int Biodeterior Biodegrad 63:462–469. https://doi.org/10.1016/j.ibiod.2009.01.003
Ventura-Camargo BC, Maltempi PPP, Marin-Morales MA (2011) The use of the cytogenetic to identify mechanisms of action of an azo dye in Allium cepa meristematic cells. J Environ Anal Toxicol 1:109–120. https://doi.org/10.4172/2161-0525.1000109
Acknowledgments
The authors of this paper wish to thank VIT management for providing a suitable platform to carry out this research work and also CSIR-CLRI for providing with LC–MS service.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors of this paper declare that they have no conflict of interest in the context of this research paper submitted to the journal.
Ethical approval
This article does not contain any studies with human participants. However, invertebrate (Artemia salina) eggs used for lethality assay in this study do not require any ethical committee clearance certificate for usage.
Additional information
Editorial responsibility: Abbaspour.
Rights and permissions
About this article
Cite this article
Saha, P., Rao, K.V.B. Biotransformation of Reactive Orange 16 by alkaliphilic bacterium Bacillus flexus VITSP6 and toxicity assessment of biotransformed metabolites. Int. J. Environ. Sci. Technol. 17, 99–114 (2020). https://doi.org/10.1007/s13762-019-02256-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-019-02256-z