Abstract
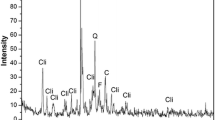
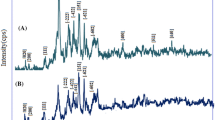
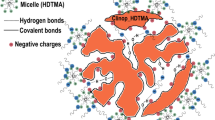
This study presents the results of chromate adsorption upon organically modified clinoptilolite obtained by the treatment of Bigadic clinoptilolite with surfactants in primary amine structure, namely 1-dodecylamine, 1-hexadecylamine and oleylamine. Natural and organo-clinoptilolites were characterized by X-ray diffraction, thermal gravimetry and Fourier transform infrared spectrometry which proved the integration of amine groups followed by the order oleylamine > 1-hexadecylamine > 1-dodecylamine. Organo-clinoptilolites were tested for their efficacy in chromate removal under base case conditions which implied 100 ppm initial concentration, pH 4.0, 10 g l−1 adsorbent dose and at 298 K. Results showed that amine loading induced chromate removal reaching up to 90 % for oleylamine-modified clinoptilolite. Experimentation was continued to determine the effects of pH, adsorbent dosage and initial concentration on chromate adsorption. Accordingly, maximum removal of 94.0 % was attained with 14 g l−1 of oleylamine-modified clinoptilolite, from Cr(VI) solution of 100 ppm initially at pH 3.0. Langmuir isotherm described the adsorption of chromate on oleylamine-modified clinoptilolite with 96.4 % consistency. Maximum theoretical uptake capacity was calculated as 6.72 mg g−1. Kinetic data were consistent with pseudo-second-order model with the controlling steps being film and pore diffusions.








Similar content being viewed by others
References
Anari-Anaraki M, Nezamzadeh-Ejhieh A (2015) Modification of an Iranian clinoptilolite nano-particles by hexadecyltrimethyl ammonium cationic surfactant and dithizone for removal of Pb(II) from aqueous solution. J Colloid Interface Sci 440:272–281. doi:10.1016/j.jcis.2014.11.017
Asgari G, Ramavandi B, Rasuli L, Ahmadi M (2013) Cr(VI) adsorption from aqueous solution using a surfactant-modified Iranian zeolite: characterization, optimization, and kinetic approach. Desalin Water Treat 51:6009–6020. doi:10.1080/19443994.2013
Aydın YA, Deveci Aksoy N (2009) Adsorption of chromium on chitosan: optimization, kinetics and thermodynamics. Chem Eng J 151:188–194. doi:10.1016/j.cej.2009.02.010
Babel S, Kurniawan TA (2004) Cr(VI) removal from synthetic wastewater using coconut shell charcoal and commercial activated carbon modified with oxidizing agents and/or chitosan. Chemosphere 54:951–967. doi:10.1016/j.chemosphere.2003.10.001
Bajda T, Klapyta Z (2013) Adsorption of chromate from aqueous solutions by HDTMA-modified clinoptilolite, glauconite and montmorillonite. Appl Clay Sci 86:169–173. doi:10.1016/j.clay.2013.10.005
Barczyk K, Mozgawa W, Król M (2014) Studies of anions sorption on natural zeolites. Spectrochim Acta A 133:876–882. doi:10.1016/j.saa.2014.06.065
Bowman RS, Haggerty GM, Huddleston RG, Neel D, Flynn M (1995) Sorption of nonpolar organic compounds, inorganic cations and inorganic oxyanions by surfactant modified zeolite. In: Sabatini DA, Knox RC, Harwell JH (eds) Emerging technologies in surfactant enhanced subsurface remediation. ACS Symposium Series, Washington, pp 54–64
Cansever-Erdoğan B, Ulku S (2012) Cr(VI) sorption by using clinoptilolite and bacteria loaded clinoptilolite rich mineral. Microporous Mesoporous Mater 152:253–261. doi:10.1016/j.micromeso.2011.11.002
Chmielewska E (2003) Adsorption of arsenate and chromate from waters on hydrophobized zeolitic media. Turk J Chem 27:639–648
Chutia P, Kato S, Kojima T, Satokawa S (2009) Adsorption of As(V) on surfactant-modified natural zeolites. J Hazard Mater 162:204–211. doi:10.1016/j.jhazmat.2008.05.024
Covarrubias C, Garcia R, Yanez J, Arriagada R (2008) Preparation of CPB-modified FAU zeolite for the removal of tannery wastewater contaminants. J Porous Mater 15:491–498. doi:10.1007/s10934-007-9131-5
Elaiopoulos K, Perraki T, Grigoropoulou E (2008) Mineralogical study and porosimetry measurements of zeolites from Scaloma area, Thrace, Greece. Microporous Mesoporous Mater 112:441–449. doi:10.1016/j.micromeso.2007.10.021
Gottardi G, Galli E (1985) Natural zeolites. Springer, New York
Guertin J, Jacobs JA, Avakian CP (2004) Chromium (VI) handbook. CRC Press, Danvers
Gupta VK, Rastogi A, Nayak A (2010) Adsorption studies on the removal of hexavalent chromium from aqueous solution using a low cost fertilizer industry waste material. J Colloid Interface Sci 342:135–141. doi:10.1016/j.jcis.2009.09.065
Gupta VK, Nayak A, Bhushan B, Agarwal S (2015) A critical analysis on the efficiency of activated carbons from low-cost precursors for heavy metals remediation. Crit Rev Environ Sci Technol 45:613–668. doi:10.1080/10643389.2013.876526
Kurniawan TA, Chan GYS, Lo WH, Babel S (2006) Comparisons of low-cost adsorbents for treating wastewaters laden with heavy metals. Sci Total Environ 366:409–426. doi:10.1016/j.scitotenv.2005.10.001
Leyva-Ramos R, Jacobo-Azuara A, Diaz-Flores PE, Guerrero-Coronado RM, Mendoza-Barron J, Berber-Mendoza MS (2008) Adsorption of chromium(VI) from an aqueous solution on a surfactant-modified zeolite. Colloid Surface A 330:35–41. doi:10.1016/j.colsurfa.2008.07.025
Li Z, Bowman RS (2001) Retention of inorganic oxyanions by organo-kaolinite. Water Res 35:3771–3776. doi:10.1016/S0043-1354(01)00120-8
Li Z, Hong H (2009) Retardation of chromate through packed columns of surfactant-modified zeolite. J Hazard Mater 162:1487–1493. doi:10.1016/j.jhazmat.2008.06.061
Li Z, Alessi D, Allen L (2002) Influence of quaternary ammonium on sorption of selected metal cations onto clinoptilolite zeolite. J Environ Qual 31:1106–1114. doi:10.2134/jeq2002.1106
Lin SH, Juang RS (2009) Adsorption of phenol and its derivatives from water using synthetic resins and low-cost natural adsorbents: a review. J Environ Manage 90:1336–1349. doi:10.1016/j.jenvman.2008.09.003
Ming DW, Bish DL (2001) Natural zeolites: occurence, properties, applications. Mineralogical Society of America, Washington
Misaelides P, Zamboulis D, Pr Sarridis, Warchoł J, Godelitsas A (2008) Chromium (VI) uptake by polyhexamethylene-guanidine-modified natural zeolitic materials. Microporous Mesoporous Mater 108:162–167. doi:10.1016/j.micromeso.2007.03.041
Mohan D, Pittman CU Jr (2006) Activated carbons and low cost adsorbents for remediation of tri- and Cr(VI) from water. J Hazard Mater 137:762–811. doi:10.1016/j.jhazmat.2006.06.060
Mohan D, Singh KP, Singh VK (2005) Removal of hexavalent chromium from aqueous solution using low-cost activated carbons derived from agricultural waste materials and activated carbon fabric cloth. Ind Eng Chem Res 44:1027–1042. doi:10.1021/ie0400898
Mozgawa W, Bajda T (2005) Spectroscopic study of heavy metals sorption on clinoptilolite. Phys Chem Minerals 31:706–713. doi:10.1007/s00269-004-0433-8
Nezamzadeh-Ejhieh A, Shahanshahi M (2013) Modification of clinoptilolite nano-particles with hexadecylpyridynium bromide surfactant as an active component of Cr(VI) selective electrode. J Ind Eng Chem 19:2026–2033. doi:10.1016/j.jiec.2013.03.018
Noroozifar M, Khorasani-Motlagh MH, Naderpour R (2008) Adsorption behavior of Cr(VI) on modified natural zeolite by a new bolaform N, N, N, N′, N′, N′-hexamethyl-1,9-nonanediammonium dibromide reagent. J Hazard Mater 155:566–571. doi:10.1016/j.jhazmat.2007.11.094
Owlad M, Aroua MK, Daud WAW, Baroutian S (2009) Removal of hexavalent chromium contaminated water and wastewater: a review. Water Air Soil Poll 200:59–77. doi:10.1007/s11270-008-9893-7
Perez Cordoves AI, Granda Valdes M, Torres Fernandez JC, Pina Luis G, Garcia-Calzon JA, Diaz Garcia ME (2008) Characterization of the binding site affinity distribution of a surfactant-modified clinoptilolite. Microporous Mesoporous Mater 109:38–48. doi:10.1016/j.micromeso.2007.04.029
Rozic M, Sipusic DI, Sekovanic L, Miljanic S, Curkovic L, Hrenovic J (2009) Sorption phenomena of modification of clinoptilolite tuffs by surfactant cations. J Colloid Interface Sci 331:295–301. doi:10.1016/j.jcis.2008.11.043
Singha S, Sarkar U, Luharuka P (2013) Functionalized granular activated carbon and surface complexation with chromates and bi-chromates in wastewater. Sci Total Environ 447:472–487. doi:10.1016/j.scitotenv.2013.01.006
Vujakovic A, Canovic T, Dakovic A, Dondur VT (2000) The adsorption of sulphate, hydrogenchromate and dihydrogenphosphate anions on surfactant-modified clinoptilolite. Appl Clay Sci 17:265–277. doi:10.1016/S0169-1317(00)00019-3
Vujakovic A, Dakovic A, Lemic J, Mihajlovic A, Canovic T (2003) Adsorption of inorganic anionic contaminants on surfactant modified minerals. J Serb Chem Soc 68:833–841. doi:10.2298/JSC0311833V
Vujakovic AD, Djuricic MA, Tomasevic-Canovic MR (2004) Thermal study of surfactant and anion adsorption on clinoptilolite. J Therm Anal Calorim 63:161–172. doi:10.1023/A:1010196622325
Wang S, Peng Y (2010) Natural zeolites as effective adsorbents in water and wastewater treatment. Chem Eng J 156:11–24. doi:10.1016/j.cej.2009.10.029
Warchoł J, Misaelides P, Petrus R, Zamboulis D (2006) Preparation and application of organo-modified zeolitic material in the removal of chromates and iodides. J Hazard Mater B137:1410–1416. doi:10.1016/j.jhazmat.2006.04.028
Xie Q, Xie J, Wang Z, Wu D, Zhang Z, Kong H (2013) Adsorption of organic pollutants by surfactant modified zeolite as controlled by surfactant chain length. Microporous Mesoporous Mater 179:144–150. doi:10.1016/j.micromeso.2013.05.027
Yang RT (2003) Adsorbents: fundamentals and applications. Wiley, New Jersey
Yariv S, Cross H (2002) Organo-clay complexes and interactions. Marcel Dekker Inc., New York
Zeng Y, Woo H, Lee G, Park J (2010) Removal of chromate from water using surfactant modified Pohang clinoptilolite and Haruna chabazite. Desalination 257:102–109. doi:10.1016/j.desal.2010.02.039
Acknowledgments
This research was supported by Scientific Research Projects Department of Istanbul Technical University (Grant No. 34969).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guzel, P., Aydın, Y.A. & Deveci Aksoy, N. Removal of chromate from wastewater using amine-based-surfactant-modified clinoptilolite. Int. J. Environ. Sci. Technol. 13, 1277–1288 (2016). https://doi.org/10.1007/s13762-016-0954-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-016-0954-y