Abstract
Kevin Laland and colleagues have put forward a number of arguments motivating an extended evolutionary synthesis. Here I examine Laland et al.'s central concept of reciprocal causation. Reciprocal causation features in many arguments supporting an expanded evolutionary framework, yet few of these arguments are clearly delineated. Here I clarify the concept and make explicit three arguments in which it features. I identify where skeptics can—and are—pushing back against these arguments, and highlight what I see as the empirical, explanatory, and methodological issues at stake.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Over the last 40 years researchers of many stripes have critiqued the structure of evolutionary theory on causal grounds.Footnote 1 This line of criticism has been developed in new ways by Kevin Laland and colleagues. Their arguments have attracted significant attention, especially those arguing for a new evolutionary framework, what they call an extended evolutionary synthesis (EES) (see: Laland et al. 2014, 2015, 2017; Laland 2015). The EES is just one of a number of recent attempts to craft a new evolutionary framework,Footnote 2 yet has quickly assumed a central role in such discussions. There are now a number of publications either building upon (e.g., Mesoudi et al. 2013; Fuentes 2016; Piperno 2017; Zeder 2017; Lu and Bourrat 2018; Uller and Helanterä 2019) or critiquing its proposals (e.g., Dickins and Rahman 2012; Dickins and Barton 2013; Calcott 2013; Wray et al. 2014; Charlesworth et al. 2017; Welch 2017).
Central to the EES critique is whether the consensus practice of current evolutionary theory suffices to explain evolutionary phenomena. Following Kitcher (1993), I use consensus practice to refer to the persisting and shared practices within a scientific domain that enact typical approaches to carrying out research, determine the salience and importance of research questions, and set up the standards for evaluating candidate explanations. Such shared practices are inculcated in researchers through their education, training, and hands-on experience with evolutionary reasoning. Though such inculcation is likely to be highly individualized, it can be approximated by the structure and presentation of evolutionary theory as found in standard textbooks (e.g., Futuyma and Kirkpatrick 2017; see Love 2010).
One important feature of the EES argument concerns the overall integration of evolutionary theory as enacted by consensus practice. Such integration can be more or less centralized or eclectic (Lewens 2015). The difference hinges on the extent to which the consensus practice has a central theoretical, conceptual, or methodological core. The more that consensus practice has a central and well-integrated set of models, theories, assumptions, and standards of explanation the more centralized and “core-like” its organization. By contrast, when there is a loose patchwork of research methods and explanatory criteria, perhaps put in the service of multiple distinct sets of research aims, the more eclectic the organization of consensus practice.
Broadly speaking, EES proponents have argued that consensus practice is centralized, and thus that there is a “core-like” organization of consensus practice. Thus they argue that “core assumptions” (Laland et al. 2015), “central tenets” (Pigliucci and Müller 2010), or “core logic” (Müller 2017) need updating and change.Footnote 3 To put it another way, EES proponents are optimists: they see new tools, models, and concepts as expanding the core of evolutionary theory through methodological and conceptual revision. Opponents of the EES (again, speaking broadly) are skeptics: they hold that consensus practice stands in good stead and that the phenomena motivating optimists’ arguments—at least inclusive inheritance, developmental bias, and phenotypic plasticity—are either non-existent, marginal, already well-understood, currently within reach of standard evolutionary models, or some combination thereof. Such skeptics thus deny that EES research challenges or radically alters the shared core of consensus practice.
To shed some light on these conflicting evaluations of the EES, I analyze one of the optimists’ central concepts: reciprocal causation (Laland et al. 2011, 2013a, b, 2015, 2015). Reciprocal causation is a “defining” and “unifying” theme of the EES, and one taken to challenge causal assumptions embedded in the models and explanations of consensus practice. Yet the positive epistemic merits of this concept are unclear, as a range of competing interpretations seem to show (Calcott 2013; Dickins and Barton 2013; Martínez and Esposito 2014; Watson et al. 2016; Svensson 2018).
In light of these interpretive differences, the central task of this article is identification and clarification: to interpret and carefully distinguish those places where the positive epistemic value of reciprocal causation might be used to argue for changes to consensus practice. Below I find three such arguments. Though supported by quotes and arguments from the EES and its interpreters, these lines of argumentation have not received full articulation by proponents. They are thus best understood as extrapolations or interpretations—possible ways in which the concept of reciprocal causation might be used to argue the case for the EES.
This exegetical and evaluative strategy is important. As the dialectic between EES optimists and skeptics has progressed, there have been increasing claims of misunderstanding on both sides.Footnote 4 It is important to move past the rhetoric and isolate what the potential benefits of the EES are taken to be, whether there are arguments to support these claims, as well as where the concepts and methods of a new synthesis break from the empirical, conceptual, and theoretical understanding of current consensus practice.
Reciprocal Causation
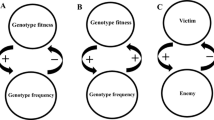
What is reciprocal causation? As I see it, EES researchers use the concept in two ways. The first is in a straightforwardly causal sense. This takes reciprocal causation to be a kind of causal relationship, one where two processes exert a mutual influence on one another (Laland et al. 2011, 2013a; Mesoudi et al. 2013). In many places, this is all that EES optimists have in mind when they use the concept. As Laland et al. (2015, p. 6) write: “The term ‘reciprocal causation’ simply means that process A is a cause of process B, and subsequently, process B is a cause of process A, with this feedback potentially repeated in causal chains.” Though the causal detail of such mutual influence may differ from case to case, the idea is at root a simple one: two causal processes are reciprocally linked insofar as they are coupled processes where the state of one is a function of the other (and vice versa).
On this understanding, reciprocal causal processes contrast with unidirectional ones–causal relationships where mutual influences are negligible or non-existent. So stated, unidirectional causation characterizes many familiar cases of causation: rocks falling to the ground, billiard balls colliding, and solar radiation warming the earth. Speaking more generally, unidirectional causation characterizes situations with asymmetric relationships between causes and effects. To use Woodward’s (2003) terminology, these are situations where there are no significant influences feeding back from the changed effect variable to a subsequent change in the cause variable.
Researchers in the life sciences are familiar with this construal of reciprocal causation. It is the causal relationship enacted in runaway sexual selection, in the positive and negative frequency-dependent selection of population genetics, in gene-network diagrams of evolutionary developmental biology, and the complex interactions of cellular metabolism. In all of these domains and more besides, researchers model coupled processes of mutual influence between elements.
Yet EES proponents also use reciprocal causation in a more substantial manner, often taking it to be central to a revised picture of evolution and evolutionary change (Laland et al. 2014, 2015; Watson et al. 2016; Laland et al. 2017). This construal of reciprocal causation incorporates feedback and interaction across multiple causal levels, with a particular emphasis on organismic behavior as a central cause modulating selection regimes. On this conception of reciprocal causation, organismic activity is locked in relationships of mutual influence with developmental and evolutionary environments through trophic exchange, excretion, and movement. More evocatively, this is a picture where “developing organisms are not solely products, but are also causes, of evolution” (Laland et al. 2015, p. 6). To optimists, the radical theoretical implications of this more substantial construal of reciprocal causation are conspicuous in processes of niche construction, particularly where the activities of a population generate systematic changes in the developmental and selective environments of downstream generations (Odling-Smee et al. 2003).
Again, in the interest of evenhandedness, one should note that consensus-practice researchers are familiar with this more substantial construal of reciprocal causation. Organismic effects on selection regimes are especially prominent in evolutionary ecology as exemplified in the rich empirical and theoretical work on predator–prey dynamics, parental effects, and social evolution. So acknowledging the explanatory value of a more substantial construal of reciprocal causation need not necessitate further methodological and conceptual change. Indeed, skeptics note that niche construction is most often presented—and perhaps is best understood—in a deflationary way. This deflationary understanding takes work in niche construction as an attempt to model a range of novel selection dynamics, rather than a radical challenge to consensus practice (e.g., Wray et al. 2014).
Nonetheless, EES optimists see the substantial view of reciprocal causation as supporting their claims of an expanded and extended consensus practice. Their argument here relies not just on reciprocal causation, but on an idiosyncratic understanding of the history and organization of evolutionary theory. This is visible when EES optimists characterize the modelling assumption of consensus practice they call fractionation (Walsh 2015; Uller and Helanterä 2019).
Fractionation is a strategy for representing and modelling evolutionary dynamics that takes the component processes of evolution to operate autonomously. “Autonomous” here means that causes underpinning evolutionary change—development and survival, replication and inheritance, and the generation of novelty and variation—are distinct realms of causal activity with negligible causal links holding between them. On a fractionated picture of evolution, for instance, the processes involved in generating mutations and innovations (e.g., copying errors, chromosomal recombination) do not directly affect the developmental machinery that generates phenotypes. Instead, mutations merely transform the content-carrying genetic vehicles that are translated by such developmental machinery.
Fractionation is an epistemic strategy, one that represents evolution as occurring in a sequence of noninteracting steps. In this way, fractionation is no different than other modelling assumptions in evolutionary theory, for instance, the assumption of infinite population size present in many population genetic models. Yet when viewed over historical time, representational strategies can become so entrenched that alternatives become difficult to imagine; the strategies begin to seem foundational, perhaps even ontological. Indeed, this claim is central to Walsh’s (2015) historical reconstruction of contemporary evolutionary theory: over the span of the 20th century, the assumptions of fractionation have become so cemented in consensus practice that they are no longer visible as assumptions, but are instead taken to be veridical representations of the evolutionary processes.
Walsh’s position is contentious, and one I have considered elsewhere (Buskell and Currie 2017). Still, even if one were to take Walsh’s argument at face value, one could still be skeptical about its scope: fractionation does not seem to characterize much of evolutionary developmental biology, quantitative genetics, or ecology. To the extent it has purchase, it is on particular modelling strategies in behavioral ecology and population genetics (Welch 2017). It is not unreasonable to wonder, then, what the broader implications of reciprocal causation might be. After all, if reciprocal causation contrasts with fractionation, and this merely characterizes a small subset of methods in evolutionary biology, the broader edifice of the EES seems to be built on shaky ground.
Here EES optimists adopt a distinctive strategy: they suggest that fractionation does in fact characterize much of consensus practice. This is because population genetics forms the core of consensus practice in evolutionary theory. Drawing together a range of historical sources, optimists construct a historical narrative where population genetics is central to the rise of “The Modern Synthesis.” This, they take to be a broad theoretical framework still in place today.
Recall above I suggested that one important dividing line between EES optimists and skeptics is the extent to which they see new research as requiring an overhaul of core consensus practice. Optimists see such changes as either incipient or currently ongoing, while skeptics deny that EES research substantially alters consensus practice. What the optimists’ historical framing aims to achieve is both the identification of a core to consensus practice—the fractionated assumptions of population genetics—as well as a demonstration of plausible, empirically fruitful alternatives.
Yet these arguments are contentious. First, it is unclear to what extent consensus practice is, or was, centralized, around population genetical models, theories, and assumptions. Love’s (2010, 2013), for instance, adopts an erotectic approach and argues that evolutionary theory may be eclectically organized around a range of discipline-specific research questions. Second, the historical narrative is questionable. Historians, philosophers, and evolutionary researchers have consistently criticized narratives that posit a single event, the Modern Synthesis, dominated by formal mathematical models of population genetics.Footnote 5
Still, even if this historical framing is suspect, that EES optimists argue against fractionation does mean arguing against prevailing theoretical and modelling assumptions in high-profile areas of evolutionary research. To the extent that their arguments for theoretical or methodological change are convincing, then, they might still motivate changes to how consensus practice understands, represents, and theorizes about evolution. If organisms are in constant interaction with the world, involved in complex webs of mutual causal influence—perhaps generating environmental regularities in the world that can “flip” latent developmental switches (West-Eberhard 2003)—then the causal assumptions of consensus practice may be called into question.
As all of this makes clear, the positive epistemic value of reciprocal causation is linked to how well it brings complex webs of mutual influence into view. This might involve identifying new kinds of reciprocal causal links, providing new empirical tools, or showing how consensus practice fails to represent crucial causal features in evolutionary change. Yet a key problem for EES proponents is in articulating how exactly reciprocal causation facilitates or renders visible such mutual causal influence. While EES proponents suggest that reciprocal causation is part of a package of ideas that “is more than simply ‘business as usual’ science: it requires conceptual change” (Laland et al. 2015, p. 10), there are countervailing claims. Those most closely aligned with what I identify as the “skeptical” position hold that reciprocal causation is ubiquitous in consensus practice and thus that the concept brings about no radical conceptual or theoretical change (Dickins and Barton 2013; Futuyma 2017; Svensson 2018). Somewhere between the two positions are researchers who argue that while reciprocal causation does bring added conceptual resources to evolutionary theorizing, these merely facilitate the generation of new empirical tools, and that only these further resources can possibly challenge consensus practice (Martínez and Esposito 2014; Watson et al. 2016). What these various interpretations show is that the concept of reciprocal causation is ambiguous, and potentially pressed in the service of multiple aims.
Here I suggest that one can identify three plausible lines of argumentation for the positive empirical value of reciprocal causation. These strategies differ in how reciprocal causation brings added conceptual, theoretical, and empirical resources to bear in understanding complex webs of causal interaction—and as a result, how the concept is supposed to challenge consensus evolutionary practice. These three lines of argumentation are:
Empirical aptness: reciprocal causation is ubiquitous among causes that underpin evolutionary phenomena, and models that employ reciprocal causation are thus more likely to be empirically apt for investigating at least some aspects of these phenomena;
Rigging the system: reciprocal causation partly explains stable selection pressures by highlighting the role of top-down developmental and behavioral constraints;
Keeping an open mind: reciprocal causation provides a causal framework that can correct for insidious practices limiting the power of evolutionary theory.
As suggested above, these arguments involve a certain amount of interpretation and extrapolation. What follows is thus a consideration of several possible ways that reciprocal causation might positively contribute to evolutionary research. In identifying and articulating these lines of arguments, I identify those places where skeptics might push (or are already pushing) back against EES claims.
Empirical Aptness
Reciprocal causation is not new to biology. Causal relationships between key evolutionary processes are familiar from work on sexual selection, parent–offspring conflict, frequency-dependent selection, and density-dependent selection. In each of these, parameter or trait values of a conspecific (for instance, female preference) can both influence, and be influenced by, the parameter or trait value of another conspecific (for instance, male displays). To these conspecific examples, one can add a host of interspecies coevolutionary phenomena including mutualism, crypsis, mimicry, and Red Queen effects. EES optimists, therefore, are in good company when they assert “reciprocal causation to be a typical, perhaps even universal feature of evolving and developing systems” (Laland et al. 2015, p. 7). The ubiquity of reciprocal causation is an assumption shared with consensus evolutionary practice.
On the back of such consensus, then, it seems somewhat unusual to claim that the approach of the life sciences needs to change; that, because of its ubiquity, “reciprocal causation should now be regarded as the norm, rather than the exception” (Laland et al. 2013a, p. 738). Remarks such as these call out for interpretation.
One weak interpretation would take this claim to be an empirical hypothesis about the ubiquity of reciprocal causation in biology, without assuming that theoretical or methodological implications follow. This, for instance, seems to be the position of Svensson (2018) who suggests that reciprocal causation draws our attention to the complexity of evolutionary phenomena and the need for greater collaboration among researchers. While this is one possible interpretation of the role of reciprocal causation, EES optimists seem to think it has broader implications. These come into view if one holds not only that reciprocal causation is ubiquitous, but also that some important causal processes in evolution need to be modelled as such. Embedded within such a stance is a corollary claim—that current consensus practice does not do such modelling or does not have the resources to carry it out. Interpreting reciprocal causation in this way generates an empirical issue: are there kinds of evolutionary phenomena that require, or would be better investigated by, representing evolution in terms of reciprocal causation?
We can construe this as a problem of empirical aptness: does the concept of reciprocal causation help make progress towards the goals of inquiry? Empirical aptness, in other words, is a relationship between a researcher’s resources and the generation of epistemic goods (e.g., explanations, understanding, the articulation of theory). Such goods are produced in the pursuit of what Brigandt and Love (2010, 2012) call “problem” or “explanatory agendas,” research questions that structure and set goals for empirical inquiry. As Brigandt and Love argue, what counts as satisfactory production of goods will be determined and refined as research progresses: empirical investigation reveals “assumptions about what it means to generate an adequate explanatory framework” (Brigandt and Love 2010). Determining the empirical aptness of reciprocal causation means not only evaluating its role in generating empirical methods and tools for everyday research, but also its place in structuring and guiding inquiry across a community of researchers.
Understanding that reciprocal causation may have this dual role—a role in generating empirical tools and also structuring empirical investigations—can help to explain the otherwise puzzling strategy of EES optimists who attempt to argue for change to consensus practice on the back of widespread consensus about empirical matters of fact.
Above I showed that reciprocal causation is widely recognized as an important phenomenon in a number of populational genetic, ecological, and developmental domains. Interestingly, there is also widespread consensus that the reciprocal causal phenomena EES optimists see as central to their framework—at least niche construction, inclusive inheritance, phenotypic plasticity, and developmental bias—are real bona fide phenomena. Nonetheless, whether these phenomena should be central targets for evolutionary research is another matter. Laland et al. (2014) suggest that consensus practice “consistently frames these phenomena in a way that undermines their significance” (p. 164). Yet a response by Wray et al. (2014) argues that, “none of the phenomena championed by Laland et al. are neglected in evolutionary biology… [the] prominence that these four phenomena command in the discourse of contemporary evolutionary theory reflects their proven explanatory power, not a lack of attention” (p. 163). Understanding this debate as about pursuit-worthiness and the broader theoretical and empirical significance—rather than theoretical and empirical validity—helps to clarify what is at stake.
Nonetheless, EES researchers do think that their proposed framework also brings with it new empirical tools and hypotheses. The positive merits of these, I’ve suggested, are tied to their abilities in rendering visible complex webs of mutual influence at multiple levels. Is there evidence that reciprocal causation aids in the generation of such tools? While the EES points to a wide range of sources as being amenable to, and perhaps supporting, a picture of reciprocal causation (reviewed in Laland et al. 2015), much of this work antedates the conceptual and theoretical innovations of the ESS. It is thus unclear whether and to what extent the EES or the concept of reciprocal causation is central to these empirical endeavors. Still, I think there are two examples that support the claim that reciprocal causation does in fact provide a novel conceptual resource that generates new kinds of tools for researchers, and identifies new lines of research to pursue.
The first comes from Uller and Helanterä (2019) who argue that various niche-construction models and concepts are better suited to exploring evolvability. Here, evolvability is understood as the possible trajectories that populations could take through some abstract multidimensional trait space (see also Brown 2014). Uller and Helanterä’s reasoning hinges on the contrastive character of causal explanations; why for example, do orchid mantises look like this orchid rather than that one? As they suggest, given knowledge about the developmental resources, behavioral flexibility, and environmental resources of a particular population, the reciprocal causation concept provides added resources to consider a wider range of explanatory contrasts—especially those where organismic activity alters the circumstances of development.
Their case study considers beach mice (Peromyscus polionotus). Prior work has revealed that a single-nucleotide polymorphism modulates the coat color of these mice and aids in the avoidance of predators by selecting for lighter-coated mice on light, sandy beaches, and darker-coated mice on inland terrain (Hoekstra et al. 2006). Yet Uller and Helanterä argue that this gene-based story is not the only evolutionary trajectory that populations of beach mice could have taken; with the aid of reciprocal causation, a broader range of evolutionary possibilities come into view. Uller and Helanterä focus their energies on characterizing developmental niche construction, where systematic changes in the parent generation can structure the developmental resources of daughter generations. Here they suggest that such systematic changes could have involved changes to burrowing behavior, sensitivities to the signals of aerial predation, movement into new terrains. Importantly, several of these counterfactual trajectories hinge on the role of social learning, where the behavioral strategies of the parent generation influence the daughter generation’s exposure to and affective valence of stimuli.
Uller and Helanterä’s example is speculative, but draws on the rich literatures on parental effects, social transmission, and niche construction. If they are right, then consensus practice does seem to neglect certain kinds of explanatory contrasts—particularly those involving intergenerational effects that shape the plastic capacities of organisms. The concept of reciprocal causation might thus play an important role in drawing attention to, and providing theoretical models for, these underinvestigated evolutionary phenomena.
Yet there are reasons to be skeptical that such explanatory contrasts are, in fact, ignored. There is a large literature within ecology on the plastic responses of populations and individuals within changing environmental circumstance (reviewed in Schlicting and Pigliucci 1998; West-Eberhard 2003; DeWitt and Scheiner 2004). Moreover, this literature covers a wide range of finely demarcated scenarios including “switch-like” change brought about through threshold responses (e.g., Lively 1986), graded change based on continuous variation (e.g., Schoeppner and Relyea 2008), and variable temporal expression such as seasonal polyphenism (e.g., Brakefield and Frankino 2009). Perhaps most important for current purposes, there is now a growing body of empirical and theoretical work investigating the costs of evolving plasticity itself and how this might factor into the understanding of long-term evolutionary trends (Murren et al. 2015). So when it comes to understanding the role of plasticity in bringing about evolutionary change—even change within one or a few generations—it is unclear that the concept of reciprocal causation is central in bringing to light unnoticed or underrepresented explanatory contrasts.
Yet consider a second example drawn from the work of Watson and colleagues (Watson et al. 2014, 2016; Watson and Szathmáry 2016; Kouvaris et al. 2017), who develop an analogy between evolution and learning theory—particularly between gene networks and neural networks (Vohradsky 2001a, b). Building on a rich body of formal theorizing, Watson et al. suggest that the tools and methods from learning theory provide added traction on a number of outstanding theoretical and philosophical issues in evolutionary theory, including major transitions in individuality, ecosystem dynamics, the evolution of genome architecture, and intriguingly, the ability of natural selection to generalize and “anticipate” future selective environments.
Anticipation and generalization are features familiar from the literature on network architectures (Clark 1993). After training on a set of stimuli, networks can generalize to similar stimuli—a feat called “signal” or “prototype extraction.” So, for instance, after being exposed to a training set of unusual dogs—say, obscure breeds like Pulis or Lagotto Romangolos—a connectionist network would be able to recognize and categorize more familiar breeds (Labradors, terriers). Watson et al. suggest that evolution is able to achieve similar feats at multiple causal levels. Evolution can pick up on “deep regularities”—structural clusterings of properties—that facilitate adaptive responses at multiple temporal and spatial scales. In this way, evolution can generalize to novel, though structurally similar, selective environments. As they suggest, “evolution thus acquires information from past selection in the same principled way that simple learning systems acquire information from past experience” (Watson and Szathmáry 2016, p. 148).
Reciprocal causation is central to this story, as it introduces a way of tweaking the parameters of evolutionary change (selection, variation, and heritability) that represent constraints on the mappings between networks and behavior. These reciprocal linkages are seen in the kind of reciprocal causal phenomena highlighted by EES optimists, things like developmental bias, niche construction, and inclusive inheritance, as well as those phenomena highlighted by Watson et al. such as major transitions in individuality (Maynard Smith and Szathmáry 1995) and ecosystem dynamics (Lean 2018). These phenomena are characterized by feedback between different kinds and levels of causation that generate “correlations or covariations between components that were previously independent” (Watson and Szathmáry 2016, p. 153). So, for instance, what they call “EvoDevo” interactions involve causal links between gene-regulatory networks and environmental parameters which together can modify the distribution of phenotypic variation; “EvoEco” how the cumulative interactions among communities of organisms modify selection pressures; and “EvoEgo,” how the interactions among entities at various levels transform and change the mechanisms of inheritance.
To sum up: one way in which optimists seem to argue for the value of reciprocal causation, and for the EES framework more generally, is by emphasizing its empirical aptness. But as I hope to have shown, proponents of the EES argue for such aptness on the back of widespread consensus as to the existence and importance of a range of evolutionary phenomena. So far, little in the debate between optimists and skeptics suggests that reciprocal causation illuminates new explanatory contrasts as of yet undiscovered by evolutionary researchers. Yet the concept may play a central role in new directions of research, where mutually influencing causal relationships at multiple levels are important. I have highlighted the research of Watson et al. here, but I could have equally pointed towards the exciting graph theoretical and dynamical systems models of gene-network effects (e.g., Salazar-Ciudad 2006b; Jaeger et al. 2012). In short, though reciprocal causation may not identify or isolate “new’’ or “neglected” evolutionary phenomena, it may already be playing a role in structuring ongoing research into fundamental evolutionary questions.
Rigging the System
One interesting empirical hypothesis floated by EES optimists concerns how the complex web of feedforward and feedback causal interactions that characterize biological phenomena might ultimately underpin the models of consensus practice. As Laland et al. (2013a) suggest, the empirical success of standard evolutionary models may occur in virtue of underlying reciprocal causation. That is, reciprocal causation explains why the fractionated, unidirectional causal models work:
The external world is likely to be capricious but the selective environment is what matters to evolving organisms and if the selective environment retains some constancy across generations (or is changed in predictable ways), inductive gambles are more likely to pay off. This means that we would expect evolutionarily successful organisms to transform their selective environment in predictable ways … effectively, to “load the dice” as well as predicting the outcome of the roll. (Laland et al. 2013a, pp. 739–740)
Here, organisms that “transform their selective environment” are those engaged in the reciprocal process.
I take it that the argument goes something like this: (1) differences in trait fitnesses reflect the relationship between traits and environments; (2) unless the fitnesses of particular traits are (somewhat) stable over time, directional and cumulative selection cannot occur; (3) key components ensuring the stability of selection pressures over time are the activities of the organisms themselves; and (4) such organismic activities are best characterized in terms of reciprocal causation. Thus, to the extent that the models of consensus practice assume relatively stable selection pressures in their models, they tacitly appeal to the stabilizing effect of organismic activity.
This is a substantial empirical claim. Laland et al. (2013a, p. 739) argue in support of it by pointing to a range of “buffering” activities, notably, the “counteractive” niche-constructing activities of termites, birds, and mammals. Perhaps the clearest example of what they call “counteractive niche construction” comes from the mound-building activity of termites (Turner 2000). Termites in the genus Macrotermes build large complex structures with distinct chambers for food storage, breeding, and the like. What is remarkable about these structures is the way in which they are constantly modified and changed so as to maintain viable living conditions. In the face of wide variation in climatic conditions, these termite colonies are able to modify the structure of the mound in order to regulate the circulation of oxygen and carbon dioxide, as well as to modulate the temperature of the colony.
Similar arguments can be found in evolutionary developmental biology. Kirschner and Gerhart (2005, 2007) have argued that structural features of developing organisms—notably, suites of modularized, exploratory, and weakly regulated processes—facilitate genetic change over evolutionary time (here we should read “facilitating” in terms of making possible). This is because these structural features engender that organisms are built with sufficient robustness, redundancy, and plasticity to accommodate variation among a number of elements. Such variation is constantly tested for viability, in part through interactions with the environment. On this account, variation is generated, accumulated, and expressed in a manner sensitive to organismic functioning. Selection on variation thus only comes about in virtue of the stabilizing characteristics of organisms.
Like other EES arguments considered above, there are two ways of interpreting the claims being made here. A weak interpretation has it that structural and behavioral features help to keep organisms viable. This is the idea that organisms act so as “to increase the chance that [they] and their descendants will remain within their tolerance spaces” (Laland et al. 2013a, p. 739). So stated this weak interpretation is obvious. After all, organisms engage in a wide range of activities—acquiring food, excreting waste, fighting pathogens—in order to increase their viability and opportunities for reproduction.
A stronger interpretation has it that structural features of organisms and some aspects of behavior provide constraints on the variation available for selection. Constraint, here, should be read like “facilitated” above: constraints structure the variations visible to selection by rendering some more likely, and others less likely. On this stronger interpretation, counteractive niche construction and facilitated variation are instances of constraints generated by reciprocal causal processes. These causal interactions—among organisms, their parts, and their environments—together “rig the system” so as to underpin both the stability of selection, and make some selective outcomes more likely than others.
Constraints can be part of evolvability explanations—why certain populations are more likely to evolve certain outcomes than another population—as we saw with the work of Watson et al. above. Selection can only act upon viable variation, and constraints determine what kind of viable variants are made available (Lewens 2004). Understanding selective trajectories should indeed include considerations about the typical range of variants that populations are likely to produce in addition to considering how different environments might affect the distribution of such variation (cf. Brown 2014). But one should not oversell ideas around stabilization and constraint. It may be true that the way that organisms are put together and interact with their environment constrains both possible and viable variation. But unless deterministic, constraints do not determine outcomes, only modify the likelihood of certain variations occurring. So even acknowledging constraints on variation leaves open the possibility that selection might play a powerful creative and directional role. Indeed, as Lewens (2004) argues, both the nature of, and relationship between, constraints and selection are complex and sensitive to the empirical concerns at hand. There will not be a one-size-fits-all approach that determines how constraints should be weighed against selection.
In this way, the EES represents the latest iteration of the long debate about the relationship between constraint and selection (Amundson 1994; Salazar-Ciudad 2006a). As the above makes clear, EES optimists take constraints to represent an underappreciated area of research. Yet skeptics continue to—reasonably, one might add—push back on the idea that developmental or behavioral constraints play an outsized role in shaping the variation visible to selection (e.g., Hoekstra and Coyne 2007)—perhaps by assuming that such constraints are equally present or invariant across multiple phyla or selection events (cf. Lewens 2004). These are questions that are increasingly under investigation, for instance, in empirical studies exploring the constraints on brain size (Logan et al. 2018) and limb length (Young et al. 2010), as well as sophisticated simulation studies that test optimization hypotheses (e.g., Salazar-Ciudad and Marín-Riera 2013). The prevalence and importance of developmental and behavioral constraint is a source of genuine disagreement between EES optimists and skeptics. While both camps put forward competing bets about the causal relevance of constraints on evolutionary change—these are bets that cannot be settled without substantial empirical investigation.
Keeping an Open Mind
EES optimists hold that reciprocal causation both motivates theoretical and methodological pluralism and that such pluralism is increasingly needed in the biological sciences (Laland et al. 2011, 2013a, b, 2014, 2015). Underpinning the urgency of their concerns is a diagnosis that current work in evolution is in some way blinkered. “Blinkered” is used here as a term of art that highlights the way that theoretical frameworks render evidence, concepts, and methodologies invisible or inaccessible to consensus practice.
This blinkered perspective is manifest in the lack of recognition afforded to the empirical and conceptual posits of the EES. This includes the phenomena described above, at least niche construction, organismic constraints, and evolvability. EES optimists blame this parochialism on assumptions that stabilize consensus practice. Key among these is evolutionary externalism. This position holds that organisms fare better or worse in virtue of the way their organismic form is apt to deal with external circumstances.
Evolutionary externalism is an “outside-in” style of investigation. It holds that understanding the evolution of organisms, and why they have the forms that they do, requires knowledge about the environments that have shaped the organism. Two alternatives to externalism are internalism and constructivism (Godfrey-Smith 1996). Internalism contrasts with externalism in being an “inside-out” style of investigation. Here, the evolution of organismic form is related to constraints, limitations, and directionality imposed by the organism itself. Both contrast with evolutionary constructivism, where the organism structures or co-constitutes its environment leading to evolutionary changes in form (Lewens 2004; Walsh 2015).
So when EES proponents argue that consensus practice in evolution is blinkered, what they mean is that consensus practice implicitly endorses evolutionary externalism. Yet how do EES optimists see this favoring taking place? Unsurprisingly, they see the problem as having to do with causation: the “manner in which biologists think about causality has acted like a meta-theoretical conceptual framework to stabilize the dominant scientific paradigm” (Laland et al. 2013a, p. 740). Because of this metatheoretical framework, EES optimists argue, alternative internalist investigatory practices (EvoDevo, DevoEvo) or constructivist ones (niche construction, gene-culture coevolution, EcoEvoDevo) have been unfairly marginalized. So if this is the problem, what is the solution?
The methodological revision that EES optimists push for is one where “potential causal influence should not be ruled out a priori” (Laland et al. 2013a, p. 738). For this to occur, proponents argue, evolutionary theory needs a framework “that allows for feedback encompassed in dynamic cycles of cultural evolution, gene-culture coevolution and organism-environment coevolution” (Laland et al. 2013a, pp. 737–738). Reciprocal causation, we are told, can provide the foundations of such a framework.
What this new framework entails is not, as far as I can tell, the wholescale replacement of externalist investigative strategies with internalist or constructivist ones.Footnote 6 Instead, externalist investigations merely need to be reframed. Such investigations should be understood to be occurring in unique circumstances; they are a “special case of reciprocal causation where feedback is negligible” (Laland et al. 2013a, p. 738).
There is a substantive worry motivating these arguments. Yet it is hard to see. To make these worries more visible, I want to develop an analogy to Elisabeth Lloyd’s (2005, 2015) well-known arguments around the role of adaptationist thinking in evolutionary thinking.
Broadly, adaptationism is a position holding that the adaptiveness or seemingly well-designed character of organisms is important, if not central, to the consensus practice of evolutionary theory. Precisely what it means to be “important” here is more contentious, and commentators have identified and evaluated a range of adaptationist positions (Godfrey-Smith 2001; Lewens 2009). Here what we are concerned with is methodological adaptationism.
Methodological adaptationism can exist in stronger or weaker forms. Keeping with current concerns, consider Lloyd’s picture of insidious methodological adaptationism.Footnote 7 This insidious adaptationism plays a role in setting research objectives and structuring investigations by determining the legitimacy of hypotheses, setting explanatory contrast classes, and regulating standards of evidence. Operating under insidious methodological adaptationism, investigations target the adaptive character of traits and are not complete until the most plausible adaptationist hypothesis has been settled upon. Only when no adaptationist hypothesis is well supported can alternate frameworks be considered. As Lloyd argues, this adaptationist-first strategy marginalizes other explanations; for instance, that traits are the result of exaptation, drift, or developmental constraint. This marginalization is where a “nonselective hypothesis is often treated as the failure to find an explanation” (Lloyd 2015, p. 356) rather than an alternative hypothesis worthy of investigation.
Lloyd’s (2005) key example of such insidious methodological adaptationism centers on the evolution of the female orgasm. She champions the view that the female orgasm is a byproduct resulting from selection on male orgasms: the tissue and nervous connections that support female orgasms result from developmental structures common to all humans, yet the reason why these structures and pathways exist at all is because of prior selection for such structures in males deep in the mammalian clade. On this account, even though the female orgasm has no adaptive function—being a byproduct of selection on male orgasms—it is nonetheless real.
As Lloyd convincingly shows (2015, pp. 351–359), despite empirical evidence for the byproduct account of female orgasm, insidious methodological adaptationism marginalizes such evidence and downplays the byproduct account. Though a plausible evolutionary hypothesis with evidence adduced in its favor, this account “is not on their list of possible answers, which only includes answers like: ‘The function of the female orgasm is to preferentially mate with high-quality males,’ or ‘the function of female orgasm is to aid the pair bond,’ etc.” (Lloyd 2015, p. 358; emphasis in original).
Lloyd’s discussion as to how methodological adaptation dismisses nonselective hypotheses is subtle, and I do not have the space to get into its details here. Suffice to say that Lloyd blames false explanatory dichotomies, lack of “stopping rules,” an inability to recognize alternative sources of evidence, and a marginalization of nonselective hypotheses as uninteresting “nulls.” These standards perpetuate insidious methodological adaptationism and lead consensus practice to ignore other important sources of explanation.
With this in hand, let us shift back to consider reciprocal causation. As I see it, EES optimists are running an analogous argument. That is, they argue that unidirectional causation is part of an explanation for insidious methodological externalism: a blinkered approach to understanding evolutionary phenomena that marginalizes internalist or constructivist alternatives. Here is how the argument works. There is a default assumption in evolutionary biology that organismic traits persist in populations because they solve environmental problems. Researchers thus explain why organisms have evolved the way that they have by understanding the selective environments of the past. But this investigative strategy ignores other kinds of explanatory contrasts that frontload the role of constraints, genomic architectures, niche construction, and the like. Moreover, this investigative strategy is in fact insidious; it leads to the neglect and marginalization of internalist or constructionist alternatives.
This reconstruction of EES optimists’ argument helps to make sense of a number of their claims; for instance, the one cited above that causal assumptions “stabilize” consensus practice. Causal assumptions can play such a role insofar as they lead researchers to neglect or discount alternative causal models. So too can this interpretation make sense of Laland et al. (2013a) claims that, “whether a process is characterized as proximate or ultimate depends critically on the conceptual framework of the researcher” (p. 720) with such a conceptual framework acting to “constrain the set of hypotheses that are deemed to be competing” (p. 729).
Take an instance of reciprocal causation, say, the dam-constructing behavior of beavers. Such a behavior exists because of previous instances of selection: there was either selection for proto-dam-constructing behavior or selection for plastic mechanisms that could be later co-opted into processes of dam-constructing. In either instance, populations of beavers reacted to environmental circumstances, and those that survived had a genetic predisposition to produce dams or to acquire behavior to produce dams. Note that according to this story, reciprocal causation has a limited explanatory role in evolution, characterizing the causal relationships of proximate mechanisms whose dynamics are the expression of latent genetic variation resulting from past selective episodes.
Such a narrative vindicates evolutionary externalism: proximate mechanisms are merely the outcome of previous instances of externalist selection. But this strategy achieves success by ignoring or downplaying internalist or constructivist alternatives. It ignores the possibility that plastic mechanisms may have both been central to the origin and maintenance of dam-constructing behavior in virtue of their role in creating a suitable dam-constructing environment. This, EES optimists might argue, is an instance of how evolutionary investigations can be blinkered. The narrative denies that reciprocal causal processes, like niche construction, play an important evolutionary role insofar as it reinterprets niche construction merely in terms of previous rounds of selection. Just as there is insidious methodological adaptationism, so too may there be insidious evolutionary externalism.
This is an important line of argument. Yet its success requires that optimists secure a number of contentious claims. These are points where skeptics might reasonably push back.
First, skeptics might reasonably doubt that insidious externalism is as central or widespread as insidious adaptationism. There are, to be sure, radical externalists. Williams (1992), for instance, is barefaced in espousing this position, stating that, “Adaptation is always symmetrical; organisms adapt to their environments, never vice versa” (p. 484). Yet to what extent are these views of Williams representative of consensus practice? Work in evolutionary ecology, evolutionary developmental biology, and ecological evolutionary biology seems to show that there is room in contemporary evolutionary science for internalist and constructivist explanations. The increasing prevalence of work examining the architecture of gene-regulatory networks in development—the homeobox-regulated development of crustacean limbs, to take just one example (Martin et al. 2016)—suggests that insidious externalism may not be embedded in the practice of all evolutionary researchers.
These concerns can be amplified. Let me return to considering the image of consensus practice projected by EES optimists. Above I noted that such optimists tend to put forward a historical narrative that sees contemporary consensus practice as organized around a central core constituted by the models and assumptions of population genetics. Yet above I also noted that many historians, evolutionary researchers, and philosophers voice skepticism about such a narrative: both its identification of a single event that could be called the Modern Synthesis, and that there was a coalescing around the modelling assumptions of population genetics. Along the same lines, I pointed to work from Love (2010, 2013) that suggests that work in the life sciences may be more eclectically organized.
Taken together, these considerations put pressure on the claims of a blinkered evolutionary science. Though some areas of consensus practice may be inimical to internalist or constructivist claims, others will be more welcoming. And unsurprisingly, work in evolutionary developmental biology and on gene-regulatory networks is precisely where one would expect internalist assumptions to hold sway.
Yet just because few individuals are bold enough to articulate radically externalist views should not be taken as evidence that such views are rare. And just because some domains of evolutionary research are open to internalist and constructivist positions does not mean that all are. But what these skeptical lines of engagement do show is that to secure a claim of insidiousness, EES optimists will require a great deal of evidence and analysis.
Unfortunately, I do not have the time nor space to try and marshal such evidence here—though I note that Uller and Helanterä (2019) do provide a clear articulation of the logic that might underpin such an insidious position. And such a task is demanding. Consider that Lloyd (2005) dedicated an entire manuscript—surveying all available accounts at the time—in order to document the methodological and empirical flaws at work in empirical research around the female orgasm. In addition, she then showed how such flaws could plausibly be attributed to background assumptions of adaptationism, human uniqueness, and androcentrism. Lloyd not only demonstrates the existence of problems, but also devotes considerable effort to showing how such problems derive from insidious assumptions. A similar evidentiary bar would be required by EES optimists to secure their claims of insidious externalism.
So while EES optimists have some reasons for their claims, their claims here are at best promissory. At the moment, they amount to showing that the assumptions of fractionation and externalism highlight certain kinds of explanatory contrasts over others. What they also show is how such assumptions might plausibly lead to a kind of insidious logic. This is not nothing. But what they have not yet done is secured a claim of insidiousness.
Conclusion
Concepts find a range of different uses in scientific practice. Often, they are directly engaged with empirical practice, picking out specific categories, mechanisms, or processes in the world relevant to researchers. Yet even while deeply tied to empirical use, concepts overlap with a range of other functions. They can facilitate exchange and collaboration, mark out and distinguish communities of like-minded researchers, structure investigations, highlight avenues of pursuit, and draw our attention to unfamiliar or less-than-attended-to phenomena. Here I have tried to highlight how reciprocal causation plays a wide range of roles well beyond the simple identification of mutual influence between processes. In highlighting these roles, I hope to have disentangled some distinct lines of argumentation knotted around the EES. In so doing, I have had as my aim a more productive and helpful dialogue around contemporary evolutionary research, as well as a clearer understanding of the outstanding issues that these lines of argumentation support.
I do not pretend to have come to definite conclusions about either the nature of the reciprocal causation concept or the EES. As things currently stand, the optimists’ alternate vision of evolutionary change, as well as their theoretical and conceptual tools, are still inchoate and incomplete. Yet even so, optimists’ arguments engage consensus practice on many fronts. Moreover, these engagements touch upon fundamental issues in evolutionary theory. Even if consensus practice remains largely unchanged from these confrontations, the interaction may be a productive one—and if so, the positive epistemic value of the EES more generally may lay beyond the somewhat overhyped claims to modify, update, and extend evolutionary theory. Instead, its value may lie in bringing to light and interrogating longstanding assumptions of consensus practice. Here, in disaggregating the arguments around the reciprocal causation concept, I hope to have contributed to such a productive dialectical engagement.
Notes
Perhaps the most congenial narrative to the optimists’ historical framing comes from Provine (1971), though Provine himself denied the centrality of population genetics to evolutionary theory, for instance, as seen in his (1980) work. Other historians have also challenged the idea of a single event that could be called the Modern Synthesis, such as Smocovitis (1996), Cain (2009), and Milam (2010).
Though I call such methodological adaptationism insidious, this is not meant to suggest a vast conspiracy. Historians and sociologists of science have long noted how regimes of teaching (Kuhn 1996), technologies (Kohler 1994), as well as social structures and power relations (Shapin and Schaffer 1985) can lead to the perpetuation of some research objectives, investigative strategies, tools, and explanations at the expense of others. It is this mundane sense—that the practice of science is influenced by material, sociological, and political causes—that I have in mind here.
References
Amundson R (1994) Two concepts of constraint. Philos Sci 61(4):556–578
Brakefield PM, Frankino WA (2009) Polyphenisms in Lepidoptera: multidisciplinary approaches to studies of evolution and development. In: Whitman DW, Ananthakrishnan TN (eds) Phenotypic plasticity in insects: mechanisms and consequences. CRC Press, Enfield, pp 337–368
Brigandt I, Love AC (2010) Evolutionary novelty and the evo-devo synthesis: field notes. Evol Biol 37(2–3):93–99
Brown RL (2014) What evolvability really is. Br J Philos Sci 65(3):549–572
Buskell A, Currie A (2017) Forces, friction, and fractionation: Denis Walsh’s Organisms, agency, and evolution. Biol Philos 32(6):1341–1353
Cain J (2009) Rethinking the synthesis period in evolutionary studies. J Hist Biol 42(4):621–648
Calcott B (2013) Why how and why aren’t enough: more problems with Mayr’s proximate-ultimate distinction. Biol Philos 28:767–780
Carroll SB (2005) Endless forms most beautiful: the new science of evo devo. Norton, New York
Carroll SB (2008) Evo-devo and an expanding evolutionary synthesis: a genetic theory of morphological evolution. Cell 134(1):25–36
Charlesworth D, Barton NH, Charlesworth B (2017) The sources of adaptive variation. Proc R Soc B 284:20162864
Clark A (1993) Associative engines. MIT Press, Cambridge
DeWitt TJ, Scheiner SM (2004) Phenotypic plasticity: functional and conceptual approaches. Oxford University Press, Oxford
Dickins TE, Barton RA (2013) Reciprocal causation and the proximate-ultimate distinction. Biol Philos 28(5):747–756
Dickins TE, Rahman Q (2012) The extended evolutionary synthesis and the role of soft inheritance in evolution. Proc R Soc B 279(1740):2913–2921
Feldman MW, Odling-Smee J, Laland KN (2017) Why Gupta et al.’s critique of niche construction theory is off target. J Genet 96(3):505–508
Fuentes A (2016) The extended evolutionary synthesis, ethnography, and the human niche: toward an integrated anthropology. Curr Anthropol 57(S13):S13–S26
Futuyma DJ (2017) Evolutionary biology today and the call for an extended synthesis. Interface Focus. https://doi.org/10.1098/rsfs.2016.0145
Futuyma DJ, Kirkpatrick M (2017) Evolution, 4th edn. Oxford University Press, Oxford
Gerhart J, Kirschner M (2007) The theory of facilitated variation. Proc Natl Acad Sci USA 104(S1):8582–8589
Godfrey-Smith P (1996) Complexity and the function of mind in nature. Cambridge University Press, Cambridge
Godfrey-Smith P (2001) On the status and explanatory structure of developmental systems theory. In: Oyama S, Griffiths PE, Gray RD (eds) Cycles of contingency. MIT Press, Cambridge, pp 283–298
Gould SJ (2002) The structure of evolutionary theory. Harvard University Press, Cambridge
Griffiths PE, Gray RD (1994) Developmental systems and evolutionary explanation. J Philos 91(6):277–304
Gupta M, Prasad NG, Dey S, Joshi A, Vidya TNC (2017a) Niche construction in evolutionary theory: the construction of an academic niche? J Genet 96(3):491–504
Gupta M, Prasad NG, Dey S, Joshi A, Vidya TNC (2017b) Feldman et al. do protest too much, we think. J Genet 96(3):509–511
Hoekstra HE, Coyne JA (2007) The locus of evolution: evo devo and the genetics of adaptation. Evolution 61(5):995–1016
Hoekstra HE, Hirschmann RJ, Bundley RA, Insel PA, Crossland JP (2006) A single amino acid mutation contributes to adaptive beach mice color pattern. Science 313(5783):101–104
Jaeger J, Irons D, Monk N (2012) The inheritance of process: a dynamical systems approach. J Exp Zool B 318(8):591–612
Kirschner M, Gerhart J (2005) The plausibility of life: resolving Darwin’s dilemma. Yale University Press, New Haven
Kitcher P (1993) The advancement of science: science without legend, objectivity without illusions. Oxford University Press, Oxford
Kohler RE (1994) Lords of the fly: Drosophila genetics and the experimental life. University of Chicago Press, Chicago
Kouvaris K, Clune J, Kounios L, Brede M, Watson RA (2017) How evolution learns to generalise: using the principles of learning theory to understand the evolution of developmental organisation. PLoS Comput Biol. https://doi.org/10.1371/journal.pcbi.1005358
Kuhn TS (1996) The structure of scientific revolutions, 3rd edn. University of Chicago Press, Chicago
Laland KN (2015) On evolutionary causes and evolutionary processes. Behav Proc 117:97–104
Laland KN, Sterelny K, Odling-Smee J, Uller T (2011) Cause and effect in biology revisited: is Mayr’s proximate-ultimate dichotomy still useful? Science 334(6062):1512–1516
Laland KN, Odling-Smee J, Hoppitt W, Uller T (2013a) More on how and why: cause and effect in biology revisited. Biol Philos 28:719–745
Laland KN, Odling-Smee J, Hoppitt W, Uller T (2013b) More on how and why: a response to commentaries. Biol Philos 28(5):793–810
Laland KN, Uller T, Feldman MW, Sterelny K, Müller GB et al (2014) Does evolutionary theory need a rethink? Yes, urgently. Nature 514:161–164
Laland KN, Uller T, Feldman MW, Sterelny K, Müller GB et al (2015) The extended evolutionary synthesis: its structure, assumptions and predictions. Proc R Soc B 282(1813):20151019
Laland K, Odling-Smee J, Endler J (2017) Niche construction, sources of selection and trait coevolution. Interface Focus 7(5):20160147
Lean CH (2018) Indexically structured ecological communities. Philos Sci 85:501–522
Levins R, Lewontin R (1985) The dialectic biologist. Harvard University Press, Cambridge
Lewens T (2004) Organisms and artifacts: design in nature and elsewhere. MIT Press, Cambridge
Lewens T (2009) Seven types of adaptationism. Biol Philos 24(2):161–182
Lewens T (2015) Cultural evolution: conceptual challenges. Oxford University Press, Oxford
Lively CM (1986) Predator-induced shell dimorphism in the Acord Barnacle Chthamalus anisopoma. Evolution 40(2):232–242
Lloyd EA (2005) The case of the female orgasm: bias in the science of evolution. Harvard University Press, Cambridge
Lloyd EA (2015) Adaptationism and the logic of research questions: how to think clearly about evolutionary causes. Biol Theor 10:343–362
Logan CJ, Avin S, Boogert N, Buskell A, Cross FR, Currie A, Jelbert S, Lukas D, Mares R, Navarrete AF, Shigeno S (2018) Beyond brain size: uncovering the neural correlates of behavioral and cognitive specialization. Comp Cognit Behav Rev 13:55–90
Love AC (2010) Rethinking the structure of evolutionary theory for an extended synthesis. In: Pigliucci M, Müller GB (eds) Evolution: the extended synthesis. MIT Press, Cambridge, pp 403–442
Love AC (2013) Theory is as theory does: scientific practice and theory structure in biology. Biol Theor 7(4):325–337
Love AC (2017) Evo-devo and the structure of evolutionary theory. In: Huneman P, Walsh D (eds) Challenging the modern synthesis: adaptation, development, and inheritance. Oxford University Press, Oxford, pp 159–187
Lu Q, Bourrat P (2018) The evolutionary gene and the extended evolutionary synthesis. Br J Philos Sci 69:775–800
Martin A, Serano JM, Jarvis E, Bruce HS, Wang J et al (2016) CRISPR/Cas9 mutagenesis reveals versatile roles of Hox genes in crustacean limb specification and evolution. Curr Biol 26:14–26
Martínez M, Esposito M (2014) Multilevel causation and the extended synthesis. Biol Theor 9(2):209–220
Maynard Smith J (1982) Evolution and the theory of games. Cambridge University Press, Cambridge
Maynard Smith J, Szathmáry E (1995) The major transitions in evolution. Oxford University Press, Oxford
Mesoudi A, Blanchet S, Charmantier A, Danchin E, Fogarty L et al (2013) Is non-genetic inheritance just a proximate mechanism? A corroboration of the extended evolutionary synthesis. Biol Theor 7(3):189–195
Milam EL (2010) The equally wonderful field: Ernst Mayr and organismic biology. Hist Stud Nat Sci 40(3):279–317
Müller GB (2007) Evo–devo: extending the evolutionary synthesis. Nat Rev Genet 8(12):943–949
Müller GB (2017) Why an extended evolutionary synthesis is necessary. Interface Focus. https://doi.org/10.1098/rsfs.2017.0015
Müller GB, Pigliucci M (2010) Extended synthesis: theory expansion or alternative? Biol Theor 5(3):275–276
Murren CJ, Auld JR, Callahan H, Ghalambor CK, Handelsman CA et al (2015) Constraints on the evolution of phenotypic plasticity: limits and costs of phenotype and plasticity. Heredity 115(4):293–301
Odling-Smee J, Laland KN, Feldman MW (2003) Niche construction: the neglected process in evolution. Princeton University Press, Princeton
Oyama S (2000) The ontogeny of information. Duke University Press, Durham
Pigliucci M (2007) Do we need an extended evolutionary synthesis? Evolution 61(12):2743–2749
Pigliucci M (2009) An extended synthesis for evolutionary biology. Ann NY Acad Sci 1168:218–228
Pigliucci M, Müller GB (2010) Elements of an extended evolutionary synthesis. In: Pigliucci M, Müller GB (eds) Evolution: the extended synthesis. MIT Press, Cambridge, pp 3–17
Piperno DR (2017) Assessing elements of an extended evolutionary synthesis for plant domestication and agricultural origin research. Proc Natl Acad Sci USA 114(25):6429–6437
Provine WB (1971) The origins of theoretical population genetics. University of Chicago Press, Chicago
Provine WB (1980) Epilogue. In: Mayr E, Provine WB (ed) The evolutionary synthesis. Harvard University Press, Cambridge, pp 399–411
Raff R (1996) The shape of life: genes, development, and the evolution of animal form. University of Chicago Press, Chicago
Robert JS (2004) Embryology, epigenesis, and evolution: taking development seriously. Cambridge University Press, Cambridge
Salazar-Ciudad I (2006a) Developmental constraints vs. variational properties: how pattern formation can help to understand evolution and development. J Exp Zool 306B(2):107–125
Salazar-Ciudad I (2006b) On the origins of morphological disparity and its diverse developmental bases. BioEssays 28(11):1112–1122
Salazar-Ciudad I, Marín-Riera M (2013) Adaptive dynamics under development-based genotype-phenotype maps. Nature 497:361–364
Schlicting CD, Pigliucci M (1998) Phenotypic evolution: a reaction norm perspective. Sinauer Associates, Sunderland
Schoeppner NM, Relyea RA (2008) Detecting small environmental differences: risk-response curves for predator-induced behavior and morphology. Oecologia 154(4):743–754
Shapin S, Schaffer S (1985) Leviathan and the air-pump. Princeton University Press, Princeton
Smocovitis VB (1996) Unifying biology: the evolutionary synthesis and evolutionary biology. Princeton University Press, Princeton
Svensson EI (2018) On reciprocal causation in the evolutionary process. Evol Biol 45(1):1–14
Turner JS (2000) The extended organism. Harvard University Press, Cambridge
Uller T, Helanterä H (2019) “Niche construction and conceptual change in evolutionary biology. Br J Philos Sci 70:351–375
Vohradsky Jirí (2001a) Neural model of the genetic network. J Biol Chem 276(39):36168–36173
Vohradsky J (2001b) Neural network model of gene expression. Fed Am Soc Exp Biol J 15:846–854
Walsh DM (2015) Organisms, agency, and evolution. Cambridge University Press, Cambridge
Watson RA, Szathmáry E (2016) How can evolution learn? Trends Ecol Evol 31(2):147–157
Watson RA, Wagner GP, Pavlicev M, Weinreich DM, Mills R (2014) The evolution of phenotypic correlations and ‘developmental memory’. Evolution 68(4):1124–1138
Watson RA, Mills R, Buckley CL, Kouvaris K, Jackson A et al (2016) Evolutionary connectionism: algorithmic principles underlying the evolution of biological organisation in evo-devo, evo-eco and evolutionary transitions. Evol Biol 43(4):553–581
Welch JJ (2017) What’s wrong with evolutionary biology? Biol Philos 32:263–279
West-Eberhard MJ (2003) Developmental plasticity and evolution. Oxford University Press, Oxford
Williams GC (1992) Gaia, nature worship and biocentric fallacies. Quart Rev Biol 67(4):479–486
Woodward J (2003) Making things happen: a theory of causal explanation. Oxford University Press, Oxford
Wray GA, Hoekstra HE, Futuyma DJ, Lenski RE, Mackay TF et al (2014) Does evolutionary theory need a rethink? No, all is well. Nature 514:161–164
Young NM, Wagner GP, Hallgrímsson B (2010) Development and the evolvability of human limbs. Proc Natl Acad Sci USA 107(8):3400–3405
Zeder MA (2017) Domestication as a model system for the extended evolutionary synthesis. Interface Focus. https://doi.org/10.1098/rsfs.2016.0133
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Buskell, A. Reciprocal Causation and the Extended Evolutionary Synthesis. Biol Theory 14, 267–279 (2019). https://doi.org/10.1007/s13752-019-00325-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13752-019-00325-7