Abstract
Validation of models for plant disease management is a crucial part in the development of decision support systems in plant protection. Bespoke field trials are usually conducted to determine the performance of a model under practical conditions. However, field trials are very resource-demanding, and the use of already existing field trial data could significantly reduce costs for model validation. In this study, we took this novel approach to verify the performance of models for determining the need of fungicide applications against leaf blotch diseases in wheat by utilising historical weather data and yield data available from fungicide efficacy field trials. Two models based on humidity factors were used in the study. To estimate how specific humidity settings in the two models affect the number of recommended fungicide treatments per season, historical weather data from a 5-year period from weather stations in Denmark, Sweden, Norway, Finland, and Lithuania was used. The model output shows major differences between seasons and regions, typically recommending between one and three treatments per season. To determine the prediction potential of the models, data on yield gains from either one or two fungicide applications in fungicide efficacy trials conducted in wheat over a 5-year period in the five countries was utilised. The yield responses from fungicide treatments in the efficacy trials varied considerably between years and countries, as did the proportion of predictions of profitable treatments. In general, there was a tendency for the models to overestimate the need to apply fungicides (low specificity), but they rarely failed to recommend an application that was needed (high sensitivity). Despite the importance of having specific trials across regions in order to adjust models to local cropping and weather conditions, our study shows that historical weather data and existing field trial data have the potential to be used in model validation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The most important leaf blotch diseases of wheat (Triticum aestivum L.) are septoria tritici blotch (STB) caused by Zymoseptoria tritici (Figure 1a), stagonospora nodorum blotch (SNB) caused by Parastagonospora nodorum (Figure 1b), and tan spot (PTR) caused by Pyrenophora tritici-repentis (Figure 1c).
All these diseases are common in the Nordic-Baltic region (Justesen et al. 2021) and have a negative impact on yield levels (Jalli et al. 2020). Based on estimates from a European survey, STB is considered the most prevalent and yield reducing disease in western Europe, typically resulting in yield reductions within the range of 5–20 dt/ha (Jørgensen et al. 2014). In high disease pressure situations, a yield loss of 50% from STB has been reported in Europe (Fones and Gurr 2015). These significant losses have resulted in an intensive use of fungicides in European wheat production, where the target of 70% of the fungicides used in cereal production is control of leaf blotch diseases, mainly STB (Torriani et al. 2015). This high input is generally unwanted from an economical and environmental perspective. In addition, the intense fungicide use has caused problems with fungicide resistance, especially in populations of Z. tritici (Lucas et al. 2015). It is well-recognised that yield responses to fungicide applications is significantly affected by the disease severity of leaf blotch diseases, which is driven by weather and host susceptibility (Wiik 2009; Jørgensen et al. 2014; Jalli et al. 2020). Rainfall and temperature are specifically known to drive fungal disease epidemics (Shaw and Royle 1993; Te Beest et al. 2009; Wegulo et al. 2011; Wiik and Ewaldz 2009). Temperature influences fungal disease development, but rainfall during the spring and summer season is by far the most important factor for disease outbreaks and progress (Rowlandson et al. 2015; Shaw and Royle 1993). Various risk models, mainly based on precipitation events (Tyldesley and Thompson 1980; Hansen et al. 1994; Wiik and Ewaldz 2009) or incidence of diseased plants (Verreet et al. 2000), have been developed over the years as tools to predict and manage leaf blotch diseases in wheat. Most models have been specifically developed for STB, while a few models cover either only SNB or both SNB and STB together (Tyldesley and Thompson 1980; Hansen et al. 1994; Mehra et al. 2019). The humidity requirements for infection of the fungi causing STB and SNB, respectively, are described to be quite similar. Historically, there has been less focus on models predicting attack of PTR.
Not all models estimating fungicide need have proven to be reliable, and few are actively used by farmers and extension specialists. Based on field trial data from 1994 and 1995, Paveley et al. (1997) suggested that traditional action thresholds (Zadoks 1985) based on disease assessments are not well-aligned with the actual need to apply fungicides. Determining the need of a treatment based on observing disease severities during the season is prone to error. Previous studies have shown that there are no or very poor correlations between yield loss and disease severities at GS 39-51 (flag leaf fully developed, beginning of anthesis) (Zadoks et al. 1974, Jalli et al. 2020). This is supported by Bhathal et al. (2003), who also showed that it is not possible to estimate the effect of early season infections on wheat leaf blotch disease development later in the season. Especially for STB, a poor correlation has been found between early disease severity at GS 31 (early stem elongation) and future damage to the crop (Thomas et al. 1989). These poor correlations are partly linked to the long latency period of some leaf blotch diseases, particularly STB, which makes it difficult to rely on the observed disease intensity from early stem elongation until heading. One example of how models could take infections in the latency phase into account is the use of different “time windows” as predictors for disease risk, as first introduced by Coakley et al. (1985). Later disease assessments that might be better correlated with yield loss are of little value since they are done when it is too late for fungicide treatments, typically during flowering. The weather from stem elongation to anthesis and maturity has proven to be a better predictor to determine the disease progress on the important, upper two leaves (Te Beest et al. 2009; Jørgensen et al. 2020; Hansen et al. 1994; Wiik and Ewaldz 2009). This indicates the potential in using weather-based risk models to estimate future disease development at a time when fungicide applications are still possible.
Several weather-based models, particularly for the control of STB, have been validated for their ability to optimise fungicide input. In Denmark, systems based on recommendations of reduced fungicide rates provided the best economic return (Jørgensen and Hagelskjær 2003), whereas in Ireland, using the simple “septoria timer” gave the highest profitability (Burke and Dunne 2008). Similarly, recent validation of field trials has shown that fungicide input can be reduced, particularly in dry seasons, while maintaining acceptable levels of disease control and profitability by using weather-based models (Jørgensen et al. 2020).
Development and validation of disease prediction models require large amounts of resources, and this is also the case when assessing their use in regions that they were not originally developed for (Jørgensen et al. 2020). The aim of our study was to investigate if historical yield data from field trials with fungicide treatments and historical weather data could be used to evaluate and compare the performance of wheat leaf blotch disease models across different regions in the Nordic-Baltic countries. In order to do this, we conducted a comparative study of two models used to determine the need of fungicide treatment in wheat. The first model uses the number of days with precipitation (Hansen et al. 1994; Henriksen et al. 2000), while the second model is based on continuous hours with high relative humidity (RH), leaf wetness, or rain (Jørgensen et al. 2020, Bligaard et al. 2017). The first aim was to test the temporal and spatial dynamics of the two models. To do this, the models were run with different settings on historical weather data (2012–2016) from weather stations in five Nordic-Baltic countries, and the recommended number of treatments per season and country was used as the response variable. The second aim was to evaluate the usefulness of existing field trial data when validating the performance of the models and model settings by correlating model recommendations to yield responses in fungicide field trials.
2 Method and materials
2.1 Model descriptions
2.1.1 Precipitation model
A well-established model for the management of STB, which is incorporated in Crop Protection Online (CPO) in Denmark, was included in the study. This model uses the number of days with precipitation as an indicator of infection risk (Hansen et al. 1994; Henriksen et al. 2000). For control of STB, the precipitation model recommends treatments after either 4 or 5 days with rain (> 1 mm rain/day) during a 14-day period. In susceptible cultivars, the threshold for recommending a treatment is 4 days with more than 1 mm of rain registered between GS 32 and 71 (stem elongation and early milky ripe stage). In more resistant cultivars, 5 days with more than 1 mm rain is used as threshold in the interval from GS 37 to 71 (flag leaf development to early milky ripe stage). In some of the test runs in this project, we also looked at the impact from 6 days with precipitation, which historically was used as one of the thresholds in CPO (Jørgensen et al. 1999).
2.1.2 Humidity model
The second model to be included in the study was a humidity model, which estimates the need of a fungicide treatment based on a fixed number of consecutive hours with high relative humidity conditions. A humidity hour is counted if pre-set levels of relative humidity, leaf wetness duration, or rainfall have been registered, giving each hour an output of “yes” or “no”. The threshold for leaf wetness is reached when leaves are wet for over 30 min per hour, and the precipitation threshold is over 0.2 mm rain per hour. RH in the range of 85–90% have been used when validating the humidity model under Danish conditions (Bligaard et al. 2017). In many historical weather datasets, measurements of leaf wetness are lacking, and in other cases, only daily rain events were recorded. As a result, hourly values of RH are often the only variable providing humidity input. RH is commonly measured at 2-m height but is expected to reflect the actual humidity in the crop (Bligaard et al. 2017; Djurle et al. 1996).
A threshold of 20 humidity hours has been tested in field trials (Jørgensen et al. 2020). As the optimal model setting is likely to vary, different thresholds were included in this study to see how different settings would fit depending on the regional situation. When validating the models, the start of the season was set to GS 31 (start of stem elongation), but treatments would typically not commence before GS 32. A fungicide treatment was recommended when the specified number of wet hours had been reached.
2.2 Estimated number of treatments based on weather data
To compare weather driven wheat foliar disease models and model settings, datasets with historical weather data were compiled from national weather stations in five countries for the period 2012–2016. The data was collected from four weather stations in Denmark, one in Finland, one in Lithuania, three in Norway, and five in Sweden. A fungicide treatment was recommended when specified numbers of continuous hours (14, 16, 18, 20, 22, and 24) of high RH (85% or 90%), or a specific number of rain days (4 or 5), had been reached. When a given threshold had been exceeded, treatments were assumed to have taken place, and a protection period of 10 days was applied before the model was initiated again. The weather data was read into Microsoft Excel, where all calculations were implemented and run (version Excel for Microsoft 365).
The time range for recommendations was set to the period between the estimated dates of GS 32 and GS 65 (mid flowering). The actual starting and ending dates varied between countries and between regions within countries. All treatment recommendations for individual field trials were compiled as an average of three model runs. The first run represented the local average duration of the time between GS 32 and GS 65. In addition, two runs were done where the seasons were shifted five days earlier respectively five days later compared to the first run to make up for variations in the date when GS 32 was reached.
2.3 Analysis of historical fungicide trial data
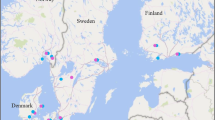
Model performance can be evaluated based on profitability in terms of net yield returns from different numbers of fungicide treatments. A dataset of results from spring and winter wheat field trials carried out during 2012–2016 across the Nordic-Baltic region was collected (N = 263) (see Figure 2). The trials included either one or two fungicide treatments or a combination of both one and two treatments. The dataset has previously been used for assessing the importance of leaf blotch diseases in the region (Jalli et al. 2020).
In order to focus on leaf blotch diseases, data from field trials with more than 5% severity of powdery mildew (Blumeria graminis) and/or yellow rust (Puccinia striiformis) were excluded. Yield levels were determined in fungicide treated and untreated plots, and the difference in yield was used to determine the potential loss caused by leaf blotch diseases in each field trial. Following this, the dataset was used to determine how accurately the models and model settings could recommend the number of fungicide treatments in relation to yield response. The data from Sweden, Denmark, and Lithuania comes from field trials in winter wheat, while the data from Finland and Norway represent spring wheat. The data for field trials with one and two treatments respectively was analysed separately. The fungicide treatments were done with either azoles (prothioconazole, epoxiconazole) alone or mixed with SDHI fungicides (boscalid). Weather data from 5 years (2012–2016) from nearby meteorological stations were used for to predict the risk of yield losses (Figure 2).
Since a fungicide treatment always comes at a cost and the sales price of grain varies, the profitability analysis was done with a requirement expressed as a yield gain of 0, 50, 350, 500, 700, and 1000 kg/ha, respectively, for one and two treatments. The 0 and 50 kg requirements were included to see if there was any yield increase at all, and the other requirements were set as alternatives for generic costs of fungicide, labour, machinery, and wheel track damage for 1 and 2 treatments, respectively. With a fulfilled yield gain requirement, the respective outcomes were classified as 1 (profitable) or 0 (not profitable). The whole range of set profitability requirements was used to compare the profitability profiles of the participating countries (Figure 5). For the comparisons between model prediction and profitability, we used 350 kg/ha and 700 kg/ha as the levels of yield increase that would give an income equal to the cost for one or two fungicide treatments, respectively, with average wheat sale prices for the studied time period.
The model predictions of the number of fungicide treatments averaged between 0 and 4. The predictions from the three model runs (GS 32 to GS 65: 5 days early, normal, 5 days late) averaging between 0 and 0.33 were considered as 0 (= no recommendation), while all averages > 0.33 were set as 1 (= recommendation) regardless of its actual value. For field trials with two treatments, a separate analysis was made in order to compare the yield gains from these trials with model predictions of more than one treatment. In that analysis, only prediction averages greater than 1.0 were classified as 1 (= recommendation).
The number of recommendations to treat or not to treat from all models and model settings for a season or region was compared to the number of treatments that were defined as profitable or not profitable in the same season or region. For each trial, there were four possible outcomes (Table 1). The two correct predictions from models were those that (1) recommended a treatment when there was a yield gain larger than the set requirement (A) and (2) gave no recommendation to treat and no yield gain above the set requirement (D). These are defined as true positive (TP) and true negative (TN) predictions, respectively. Alternative B in Table 1 represents the false positive prediction (FP) where a treatment is recommended without being profitable, and C shows the false negative (FN) predictions, where the model failed to recommend a treatment that would have been profitable.
The true positive proportion of correct predictions for profitable fungicide treatments was calculated as A/(A+C), and the true negative proportion of correct predictions for not profitable treatments was calculated D/(B+D). The calculations were done in SAS/STAT software (SAS version 8) The true positive proportion can be referred to as the sensitivity of a model and the true negative proportion as its specificity (Yuen et al. 1996; Madden 2006). A perfect model would have both sensitivity and specificity equal to 1.0, meaning that all predictions were correct. The total proportions of TP and TN predictions (A+D/(A+C+B+D)) were calculated for models and model settings by country, by year, and by region in a country when applicable. This value represents the total proportion of correct recommendations.
3 Results and discussion
3.1 Recommended number of treatments based on historical weather data
The risk of leaf blotch diseases in wheat varies significantly within the Nordic-Baltic zone, reflecting the different patterns of disease conducive weather. As a result, the number of recommended fungicide treatments differs between sites and seasons (Figure 3 and Figure 4). The need for fungicide applications is further affected by the start and duration of the growing season, which can vary greatly between years and regions across the Nordic-Baltic region. Using weather data from 2012 to 2016, the humidity model recommended between 0 and 3.7 treatments per season for the control of leaf blotch diseases in wheat depending on location and the model setting. The precipitation model, set to require rainfall for 4, 5, or 6 days to recommend a fungicide treatment, gave similar or slightly higher numbers of treatments (Figure 3).
Testing of different humidity and rainy days models. Number of treatments recommended based on historical weather data from 5 countries in the Nordic-Baltic region 2012–2016. Average number of treatments for specific model criteria are conditionally colour-formatted from red (high numbers) to green (low numbers).
Testing of different humidity and rainy days models. Number of treatments recommended based on historical weather data from different locations in 5 countries in the Nordic-Baltic region. Average number of treatments for specific model criteria are conditionally colour formatted from red (high numbers) to green (low numbers).
The number of fungicide treatments showed major variability depending on the tested models, threshold settings, and weather station (Figure 3, Figure 4). Most model settings predicted between one and three treatments per season. The more conservative settings (higher number of humidity hours or days with precipitation) for the models recommended, as expected, fewer treatments. This can particularly be seen for the humidity model settings that required long periods (> 20 h) of continuous wetness at 90% RH. For specific seasons, the setting of the threshold using, e.g. 24 instead of 14 continuous hours with RH at 85%, could change the number of treatments from 0 to 2.7. An increase in the required RH from 85 to 90% resulted in a large reduction in the number of recommended treatments for most countries and years, apart from Finland. Overall, the use of either days with rain in the precipitation model (4 or 5) or RH > 85% for 20 h in the humidity model resulted in similar numbers of recommended treatments. However, for Norway and Finland, there was a tendency that using 4 days with precipitation as a threshold led to recommendations of more treatments compared to 85% RH for 20 h.
3.2 Model performance based on the profitability of fungicide treatments
Treatment profitability (net return) is determined by the relationship between the cost involved with fungicide treatments, the effect on the yield level from the treatment, and the grain sales price. The lowest yield responses to fungicide treatments were observed in the trials conducted in Finland and Sweden, with few profitable treatments when the treatment costs increased above an equivalent of 350 kg/ha. The results from the Danish trials showed a high proportion of profitable treatments with increasing cost, reflecting a generally high yield response from fungicide treatments. This was seen for data from either one or two treatments (Figure 5).
Linking the recommended number of treatments with yield responses is challenging. In our study, the model outputs using local weather data were compared to yield responses using historical data from fungicide efficacy field trials. When evaluating the models’ potential to predict yield losses equivalent to or higher than 350 kg/ha, based on data from one fungicide treatment, the precipitation model and the humidity model using 85% RH resulted in a high percent of correct predictions (88–100%) for Denmark and Lithuania. For Sweden and Norway, the percent of correct predictions from the same model settings was lower (52–71%). Using two fungicide treatments and yield increases corresponding to 700 kg/ha as the cut-off for profitability, the Danish and the Norwegian trials showed the largest percentage of correct predictions (80–90%), while those for Sweden and Lithuania only were correct at 40–60% (Figure 6). Overall, the results from the model validation showed a high degree of correct outputs from most of the models using Danish data. This confirmed previous analyses of Danish fungicide trials, which indicated a positive link between the treatment recommendations from risk models and yield responses due to STB attack (Jørgensen et al. 2018).
Selected model setting tested on data from field trials conducted over 5 years (2012–2016) in winter wheat (Denmark, Lithuania, Sweden) and spring wheat (Norway) using data with either 1 or 2 treatments. For one treatment, 350 kg.ha−1 of yield gain per hectare was used as target for a treatment being profitable, and for two treatments 700 kg/ha. The models are labelled as xx_hh where xx = relative humidity level and hh = the duration in hours of high relative humidity (humidity model), and 4 days, respectively, 5 days for the number of days with precipitation (precipitation model). Correct = percentage of True Negative (TN) recommendations + True Positive (TP) recommendations; Sens. = Sensitivity = Proportion of TP. Spec. = Specificity = Proportion of TN; Spec. = n.a. = all fields profitable to treat.
The variable proportions of correct predictions across the different countries show that there can be challenges when transferring a model from one region to another. A possible explanation for the varying results from the different countries could be linked to the differences in yield levels and yield gains from fungicide treatments. An earlier Nordic-Baltic study showed large differences both in average yield and yield response from fungicide treatments between the countries in the region (Jalli et al. 2020). This is supported by reports of low profitability of fungicide treatments in major parts of Sweden (Wiik and Rosenqvist 2010; Djurle et al. 2018; Willocquet et al. 2021). A study by Cook and Thomas (1990) suggested that higher yielding crops are tolerant to foliar diseases and give the same absolute yield increase at different yield levels, resulting in a reduced relative yield increase at higher potential yield levels. Wiik (2009) reported that absolute yield responses increased with increasing levels of leaf blotch diseases, and when separating data into two yield groups, a significantly higher absolute yield (in kg per hectare) was found at the higher yield level. However, when calculated as percent yield increase, the levels were of a similar size.
To further validate the model performance, values for sensitivity and specificity were calculated (Figure 6 and Figure 7). For all countries, the sensitivity was comparatively high for all model settings, but the precipitation model and the humidity model using the least conservative settings showed mostly low or no specificity. In most cases, the sensitivity value was between 0.7 and 1.0, which means that in the majority of cases, there was a yield increase from the recommendations bases on the set thresholds (350 kg/ha for one treatment and 700 kg/ha for two treatments) and a correct recommendation to treat. At the same time, the models mostly showed low specificity, indicating that the models often overestimated the need for fungicide treatments. However, if a high proportion of the trials shows profitability from fungicide treatments, the proportion of correct recommendations will still be high.
Figure 7 shows the results from different model setting per year that have: (1) previously been used in Denmark (85_20, 85_22, 4 days, and 5 days), and may be considered as the default models, and (2) models that in at least one other country, and for one or several years had a higher rate of correct recommendations compared to the default models (90_18, 90_20). Overall, the results indicate that most models performed well in all five seasons in Denmark, except for the models with a high RH threshold (90% RH). It should be noted that the treatments in all Danish trials, apart from 2015, were profitable (specificity = n.a.). For Lithuania, the prediction to treat or not to treat was correct in two out of three seasons; however, in both these seasons, all treatments gave a yield increase over 350 kg/ha; i.e., they were profitable. The model performance was very variable in Sweden. For example, the number of correct recommendations by year using 20 h with 85% RH varied between 22 and 100%, while the percent correct recommendations using 4 days precipitation varied from 22 to 75%. Similarly, the Norwegian data showed major variability of model performance between years. For Norway, given a treatment threshold of 20 h with 85% RH resulted in 33 and 83% correct recommendations, while the corresponding figures for 4 days precipitation threshold varied from 33 to 100%.
Sensitivity, specificity, and percentage of correct predictions, by year and country, when the yield gain requirement for one treatment was 350 kg/ha. The models are labelled as xx_hh where xx = relative humidity level and hh the duration in hours of high relative humidity (humidity model), and 4 days, respectively, 5 days for the number of days with precipitation (precipitation model). Correct = percentage of True Negative (TN) recommendations + True Positive (TP) recommendations; Sens. = Sensitivity = Proportion of TP; Spec. = Specificity = Proportion of TN; Specificity n.a. = all fields profitable to treat.
The results from Finland are not included in Figures 6 and 7, since the Finnish data consisted of only one field trial per year for 4 years, and in each of these trials, the model prediction was correct. All four trials were profitable to treat, resulting in a sensitivity value of 1, and no value (n.a.) for specificity.
Due to limitations in the number of trials when dividing the data into regions, the calculations of recommended number of treatments, proportion of correct recommendation, and sensitivity-specificity values were done per country. As can be seen in Figure 2, the distances between weather stations used for the model risk calculation and the actual field trial locations vary. Long distances between weather station and the actual trials will have a major influence on the validity of the weather data and, as a result, the performance of the tested models. In the data used for the analyses, the trials in Lithuania were all conducted at the same location with a weather station close by, and most trials carried out in Denmark were done at Flakkebjerg with an on-site weather station. For the other countries, the trial locations were more dispersed from year to year, and distances to the weather stations were in some cases large. This could be one reason for the better model performance in Denmark and Lithuania compared to Sweden and Norway. Precipitation can vary greatly within short distances, while air humidity is usually more stable over larger areas, making it a more reliable variable for predicting the need for fungicide treatments in a situation like the one in our study. Models purely based on rainy days could therefore be more prone to errors in this scenario compared to models based on air humidity.
The models in our study did not include temperature as a prediction parameter, which might have reduced their performance. It is known that fungal infection in general and development of STB in particular are much slower at low temperatures (Shaw 1990). Within the Nordic-Baltic region, STB is found to be less severe in the more northern countries (Norway, Finland), which could be attributed to lower temperatures during the growing season (Jalli et al. 2020). Prolonged periods close to or below freezing during winter and early spring will affect the number of life cycles the pathogen can complete in a season (Shaw and Royle 1993). It has previously been found that the frequency of consecutive days with temperature below 7 °C at the end of tillering and start of stem elongation was negatively correlated with STB severity (Coakley et al. 1985). It should also be noted that the Norwegian and Finnish data came from trials in spring wheat. Winter wheat is often infected in the autumn, which gives the disease a head start in the spring, while spring wheat infections start later and have a shorter timespan to develop. Generally, PTR is more serious in the more northern parts, and SNB is found mainly in Finland and Norway. Since the threshold parameters of the models that were compared in this study were mainly developed to estimate the fungicide input needed to control STB, the geographical distribution of the leaf blotch diseases might have had an influence of the varying performance of the models in the different countries.
The potential yield loss from leaf blotch diseases is highly dependent on the cultivar susceptibility (Jørgensen et al. 2014). The models in CPO using days with precipitation have historically provided simple guidance on when a treatment against STB is expected to be profitable in Denmark. As seen in Figure 4, the number of recommended treatments is considerably reduced if a model with 5 or 6 days with rain is used instead of one with four days. In the current version of Crop Protection Online, a threshold of 4 days of rain is used in susceptible cultivars between GS 32 and 71, and 5 days in the most resistant cultivars between GS 37 and 71. In the latter case, it is rare that more than one treatment provides a positive net return. This separation between susceptible and resistant cultivars is not included in our model evaluation.
Overall, based on our results from historical data, it is not unlikely that differences in climate, yield potential, and wheat type could lead to an overestimation of treatment recommendations in the more northern regions compared to Denmark and Lithuania.
3.3 The use of data from historical fungicide efficacy trials to evaluate model performance
The historical weather data gave good opportunities for testing the dynamics in the number of recommended fungicide treatments from different settings in the models across the Nordic-Baltic region. As could be expected, the historical dataset of fungicide trials did not provide an ideal framework for validating the models. The quality of the analysis depends on how well the design of the field trials fit our research question. Designed for fungicide efficacy testing, rather than IPM strategy testing, the trial yield data left questions unanswered when compared to model recommendations. A proper validation of models to determine the need of fungicide treatments to control leaf blotch diseases in wheat asks for dedicated field trials, designed for this specific purpose to better clarify the potential and possible benefits from using such models. However, determining if treatments recommended by the models were profitable or not by using existing data on the difference in yield from fungicide untreated and treated field trial plots proved to be an important complement to the very resource demanding bespoke validation trials.
4 Conclusion
Decisions related to plant disease management should be based on potential disease development, which is a function of the presence of inoculum or early disease intensity, cultivar resistance, and weather conditions before and after a treatment, as well as farmer’s experiences. The decision-making is also related to risk attitude, and many farmers prefer a certain element of insurance to avoid unexpected yield loss, which can lead to additional fungicide applications or the use of higher doses (Hardwick et al. 2001; te Beest et al. 2013). This points to the need of reducing uncertainties in this decision-making process by the development of better predictive methods, which is a cornerstone in the implementation of IPM. Our study utilised historical weather data in combination with data from fungicide efficiency trials to test models for treatment recommendations. This is a new approach to better understand model performance without the considerable cost of specifically designed field trials. Even though the data showed some inadequacy to fully evaluate the models, the results confirm that major ambiguities still exist when it comes to making farm level decision regarding fungicide treatments.
Data availability
Data sets available at: https://doi.org/10.6084/m9.figshare.19203377.v4
Code availability
The SAS scripts and input files are available at: https://doi.org/10.6084/m9.figshare.19203398.v3
References
Bligaard J, Jørgensen LN, Axelsen J, Hansen JG, Ørum JE, Baby S, Nielsen GC (2017) Udvikling af nye risikomodeller for Septoria (Zymoseptoria tritici) i vinterhvede. Miljø- og Fødevareministeriet, Miljøstyrelsen. Bekæmpelsesmiddelforskning nr. 168. ISBN: 978-87-93529-68-7
Bhathal JS, Loughman R, Speijers J (2003) Yield reduction in wheat in relation to leaf disease from yellow (tan) spot and septoria nodorum blotch. Eur J Plant Pathol 109:435–443
Burke JJ, Dunne B (2008) Field testing of six decision support systems for scheduling fungicide applications to control Mycosphaerella graminicola on winter wheat crops in Ireland. J Agric Sci 146:415–428. https://doi.org/10.1017/S0021859607007642
Cook RJ, Thomas MR (1990) Influence of site factors in responses of winter wheat to fungicide programmes in England and Wales, 1979-1987. Plant Pathol 39:548–557
Coakley SM, McDaniel LR, Shaner G (1985) Model for predicting severity of septoria tritici blotch on winter wheat. Phytopathology 75:1245–1251
Djurle A, Ekbom B, Yuen JE (1996) The relationship of leaf wetness and disease progress of glume blotch, caused by Stagonospora nodorum, in winter wheat to standard weather data. Eur J Plant Pathol 102:9–20
Djurle A, Twengström E, Andersson B (2018) Fungicide treatments in winter wheat: the probability of profitability. Crop Prot 106:182–189. https://doi.org/10.1016/j.cropro.2017.12.018
Fones H, Gurr S (2015) The impact of septoria tritici blotch disease on wheat: an EU perspective. Fungal Genet Biol 79:3–7. https://doi.org/10.1016/j.fgb.2015.04.004
Hansen JG, Secher BJ, Jørgensen LN, Welling B (1994) Threshold for control of Septoria spp. in winter wheat based on precipitation and growth stage. Plant Pathol (43):183–189
Hardwick NV, Jones DR, Slough JE (2001) Factors affecting diseases of winter wheat in England and Wales, 1989-98. Plant Pathol 50:453–462. https://doi.org/10.1046/j.1365-3059.2001.00596.x
Henriksen KE, Jørgensen LN, Nielsen GC (2000) PC-Plant Protection – a tool to reduce fungicide input in winter wheat, winter barley and spring barley in Denmark. Brighton Crop Protection Conference. Pest Diseases:835–840
Jalli M, Kaseva J, Andersson B, Ficke A, Jørgensen LN, Ronis A, Kaukoranta T, Ørum JE, Djurle A (2020) Yield increases due to fungicide control of leaf blotch diseases in wheat and barley as a basis for IPM decision-making in the Nordic-Baltic region. Eur J Plant Pathol 158:315–333. https://doi.org/10.1007/s10658-020-02075-w
Justesen AF, Corsi B, Ficke A, Hartl L, Holdgate S, Jørgensen LN, Lillemo M, Lin M, Mackay IA, Mohler V, Stadlmeier B, Tan K, Turner J, Oliver RP, Cockram J (2021) Hidden in plain sight: a molecular field survey of three wheat leaf blotch fungal diseases in North-Western Europe shows co-infection is widespread. Eur J Plant Pathol 160(2021):949–962. https://doi.org/10.1007/s10658-021-02298-5
Jørgensen LN, Secher BJM, Hossy H (1999) Decision support systems featuring septoria management. In: Septoria on Cereals: A Study of Pathosystems. CABI Publishing. pp 251-262.
Jørgensen LN, Hagelskjær L (2003) Comparative field trials of various decision support systems for cereal disease control. Proceedings of the Crop Protection Conference for the Baltic Sea Region, 28-29 April 2003, Poznan. DIAS report Plant Production (96):114–122
Jørgensen LN, Hovmøller MS, Hansen JG, Lassen P, Clark B, Bayles R, Rodemann B, Jahn M, Flath K, Goral T, Czembor J, Cheyron P, Maumene C, Pope C, Nielsen GC, Berg G (2014) IPM strategies and their dilemmas including an introduction to www.Eurowheat.org. J Integr Agric (13):265–281
Jørgensen LN, Hansen JG, Ørum JE, Jalli M, Ronis A, Djurle A,; Anderson B, Skog TE, Ficke A, Nordskog B (2018) Weather risk models for prediction of septoria tritici blotch. Zymoseptoria tritici community meeting, Sept. 6-7, 2018. Zurich, Switzerland (Poster).
Jørgensen LN, Matzen M, Nielsen GC, Jalli M, Ronis A, Djurle A, Anderson B, Ficke A, Djurle A (2020) Validation of risk models for control of leaf blotch diseases in wheat in the Nordic and Baltic countries. Eur J Plant Pathol 157:599–613
Lucas JA, Hawkins NJ, Fraaije BA (2015) The evolution of fungicide resistance. Adv Appl Microbiol 90:29–92. https://doi.org/10.1016/bs.aambs.2014.09.001
Mehra LK, Adhikari U, Ojiambo PS, Cowger C (2019) Septoria nodorum blotch of wheat. The Plant Health Instructor. https://doi.org/10.1094/PHI-I-2019-0514-01
Madden LV (2006) Botanical epidemiology: some key advances and its continuing role in disease management. Eur J Plant Pathol 115:3–23
Paveley ND, Lockley D, Sylvester-Bradley R, Thomas J (1997) Determinants of fungicide spray decisions for wheat. Pestic Sci 49:379–388
Rowlandson T, Gleason M, Sentelhas P, Gilespie T, Thomas C, Hornbuckle B (2015) Reconsidering leaf wetness duration determination for plant disease management. Plant Dis 99:310–319
Shaw MW (1990) Effects of temperature, leaf wetness and cultivar on the latent period of Mycosphaerella graminicola on winter wheat. Plant Pathol 39:255–268
Shaw MW, Royle DJ (1993) Factors determining the severity of epidemics of Mycosphaerella graminicola (Septoria tritici) on winter wheat in the UK. Plant Pathol 42(1993):882–899
Te Beest DE, Shaw MW, Pietravalle S, Van den Bosch F (2009) A predictive model for early –warning of septoria leaf blotch on winter wheat. Eur J Plant Pathol (124):413–425. https://doi.org/10.1007/s10658-009-9428-0
Te Beest DE, Paveley ND, Shaw MW, van den Bosch F (2013) Accounting for the economic risk caused by variation in disease severity in fungicide dose decisions, exemplified for Mycosphaerella graminicola on winter wheat. Phytopathology 103:666–672. https://doi.org/10.1094/PHYTO-05-12-0119-R
Thomas MR, Cook RJ, King JE (1989) Factors affecting development of Septoria tritici in winter wheat and its effect on yield. Plant Pathol 38:246–257
Torriani SFF, Melichar JPE, Mills C, Pain N, Sierotzki H, Courbot M (2015) Zymoseptoria tritici: a major threat to wheat production, integrated approaches to control. Fungal Genet Biol 79:8–12. https://doi.org/10.1016/j.fgb.2015.04.010
Tyldesley JB, Thompson N (1980) Forecasting Septoria nodorum on winter wheat in England and Wales. Plant Pathol 29:9–20
Wegulo SN, Zwingman MV, Breathnach JA, Baenziger PS (2011) Economic returns from fungicide application to control foliar fungal diseases in winter wheat. Crop Prot (30):685–692. https://doi.org/10.1016/j.cropro.2011.02.002
Verreet JA, Klink H, Hoffmann GM (2000) Regional monitoring for disease prediction and optimization of plant protection measures: the IPM wheat model. Plant Dis 84:816–826. https://doi.org/10.1094/PDIS.2000.84.8.816
Wiik L (2009) Yield and disease control in winter wheat in southern Sweden during 1977–2005. Crop Prot 28:82–89. https://doi.org/10.1016/j.cropro.2008.09.002
Wiik L, Ewaldz T (2009) Impact on temperature and precipitation on yield and plant diseases of winter wheat in southern Sweden 1993-2007. Crop Prot 28:82–89. https://doi.org/10.1016/j.cropro.2009.05.002
Wiik L, Rosenqvist H (2010) The economics of fungicide use in winter wheat in southern Sweden. Crop Prot 29:11–19. https://doi.org/10.1016/j.cropro.2009.09.008
Willocquet L, Meza WR, Dumont B, Klocke B, Feike T, Kersebaum KC, Meriggi P, Rossi V, Ficke A, Djurle A, Savary S (2021) An outlook on wheat health in Europe from a network of field experiments. Crop Prot 139:105335. https://doi.org/10.1016/j.cropro.2020.105335
Yuen J, Twengström E, Sigvald R (1996) Calibration and verification of risk algorithms using logistic regression. Eur J Plant Pathol 102:847–854
Zadoks JC, Chang TT, Konzak CF (1974) A decimal code for the growth stages of cereals. Weed Res 14:415–421
Zadoks JC (1985) On the conceptual basis of crop loss assessment – the threshold theory. Annu Rev Phytopathol 23:455–473
Funding
Open access funding provided by Swedish University of Agricultural Sciences. This study was funded by the Finnish Ministry of Agriculture and Forestry, Danish AgriFish Agency, Lithuanian Research Center for Agriculture and Forestry, Norwegian Research Council, and Swedish Research Council for Environment, Agricultural Sciences and Spatial Planning, and the European Commission, and was carried out as a part of the SpotIT (IT solutions for user-friendly IPM-tools in management of leaf spot diseases in cereals) project (www.nibio.no/en/projects/spotit) funded under the C-IPM ERA-NET. The data analysis is also part of the EU Horizon 2020 project IPM Decision (www.ipmdecisons.net).
Author information
Authors and Affiliations
Contributions
Jens-Erik Ørum and Annika Djurle organised and analysed the risk for LBD based on historical weather data. Lise Nistrup Jørgensen, Björn Andersson, Annika Djurle, Andrea Ficke, Antanas Ronis, and Marja Jalli participated in collecting trial data. Annika Djurle and Björn Andersson analysed the field trial data from the different partners and performed the statistical analysis. Björn Andersson, Lise Nistrup Jørgensen, and Annika Djurle wrote the manuscript in consultation with Andrea Ficke, Marja Jalli, Jens-Erik Ørum, and Antanas Ronis.
Corresponding author
Ethics declarations
Ethics approval
Not applicable
Consent for publication
Not applicable
Conflict of interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Andersson, B., Djurle, A., Ørum, J.E. et al. Comparison of models for leaf blotch disease management in wheat based on historical yield and weather data in the Nordic-Baltic region. Agron. Sustain. Dev. 42, 42 (2022). https://doi.org/10.1007/s13593-022-00767-7
Accepted:
Published:
DOI: https://doi.org/10.1007/s13593-022-00767-7