Abstract
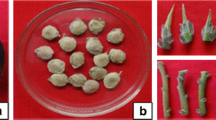
Factors affecting in vitro propagation were evaluated for Ceropegia attenuata Hook., an endemic and endangered plant having ornamental potential but a limited reproductive capacity. Rapid shoot multiplication from nodal explants was established using varying concentrations of cytokinins and auxins either alone or in combinations. The highest frequency of shoot induction was achieved when nodal explants were inoculated on Murashige and Skoog (MS) medium supplemented with 13.31 μM 6-benzylaminopurine with a mean of 12.9 ± 0.5 shoots per explant. High concentrations of TDZ (6.81–11.35 μM) and KN (6.78–11.61 μM) resulted in stunted and vitrified shoots. Factors implicated in the promotion of floral transition of the C. attenuata have been identified which are 4-amino-3, 5, 6-trichloropicolinic acid (picloram), 6-benzylaminopurine, sucrose and photoperiod. The highest frequency of flowering (100%) was obtained when axillary shoot explants were transferred to MS medium supplemented with picloram (4.14 μM) within 4 weeks of culture. Transfer of in vitro regenerated shoots to half strength MS medium with 2.46 μM indole-3-butyric acid (IBA) showed maximum root induction. The in vitro grown plantlets were successfully acclimatized in the glasshouse with 85% of survival and showed normal development. The developed protocol provided a simple, cost-effective approach for the conservation of endangered plant C. attenuata for replenishing its declining populations.



Similar content being viewed by others
Abbreviations
- BAP:
-
6-benzylaminopurine
- IAA:
-
indole-3-acetic acid
- IBA:
-
indole-3-butyric acid
- Kinetin:
-
6-furfurylaminopurine
- TDZ:
-
thidiazuron
- Picloram:
-
4-amino- 3, 5, 6-trichloropicolinic acid
- ½ MS:
-
half-strength MS medium
References
Amoo SO, Finnie JF, Staden JV (2009) In vitro propagation of Huernia hystrix: an endangered medicinal and ornamental succulent. Plant Cell Tiss Org Cult 96:273–278
Anonymous (1992) Ceropegia Linn. (Asclepiadaceae), The wealth of India. Vol. 3. New Delhi, (CSIR), 448
Bapat VA, Yadav SR, Dixit GB (2008) Rescue of endangered plants through biotechnological applications. Nat Acad Sci Lett 31:201–210
Beena MR, Martin KP, Kirti PB, Hariharan M (2003) Rapid in vitro propagation of medicinally important Ceropegia candelabrum. Plant Cell Tiss Org Cult 72:285–289
Bernier G, Perilleux C (2005) A physiological overview of the genetics of flowering time control. Plant Biotech 3:3–16
Britto SJ, Natarajan E, Arockiasamy DI (2003) In vitro flowering and shoot multiplication from nodal explants of Ceropegia bulbosa Roxb var bulbosa. Taiwania 48(2):106–111
Bruyns PV (2003) Three new succulent species of Apocynaceae (Asclepiadoideae) from Southern Africa. Kew Bull 58:427–435
Chandore AN, Nimbalkar MS, Gurav RV, Bapat VA, Yadav SR (2010) An efficient micropropagation protocol for multiplication and restoration of Ceropegia fantastica Sedgw: a critically endangered plant species. Curr Sci 99:1593–1596
Chavan JJ, Nimbalkar MS, Gaikwad NB, Dixit GB, Yadav SR (2011) In vitro propagation of Ceropegia spiralis Wight—an endemic and rare potential ornamental plant of peninsular India. Proc Nat Acad Sci India Sect B 81:120–126
Desai NS, Suprasanna P, Bapat VA (2003) Conservation status and in vitro multiplication of Frerea indica Dalz. an endemic and endangered plant from Western Ghats of Maharashtra, India. Physiol Mol Biol Plants 9(2):265–268
Faisal M, Ahmad N, Anis M (2005) Shoot multiplication in Rauvolfia tetraphylla L. using thidiazuron. Plant Cell Tiss Org Cult 80:187–190
Hagen SR, Letourneau D, Muneta P, Brown J (1990) Initiation and culture of potato tuber callus tissue with picloram. Plant Growth Regul 9:341–345
Hodgkiss RJ (2004) http://www.succulent-plant.com/ceropg.html
Karuppusamy S, Kiranmani C, Aruna V, Pullaiah T (2009) In vitro conservation of Ceropegia intermedia—an endemic plant of south India. Afr J Biotechnol 8:236–239
Kirtikar KR, Basu BD (1935) Indian medicinal plants 3, (M/ s Bishen Singh Mahendrapal Singh, New Delhi), India, 1638
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–497
Nair AK, Naik DD, Pandit SS (2007) High frequency in vitro flowering in six species of Ceropegia. J Plant Biol 50(3):374–377
Nayar MP, Sastry ARK (1987) Red data book of Indian plants, vol 1 & 2. Botanical Survey of India, Kolkata
Patil VM (1998) Micropropagation studies in Ceropegia spp. In Vitro Cell Dev Biol Plant 34:240–243
Patnaik J, Debata BK (1996) Micropropagation of Hemidesmus indicus (L.) R. Br. through axillary bud culture. Plant Cell Rep 15:427–430
Reynolds S (2006) http://www.sagereynolds.com/cero/clist.com
Sheeja TE, Mandal AB (2003) In vitro flowering and fruiting in tomato (Lycopersicon esculentum Mill.). Asia Pac J Mol Biol Biotechnol 11(1):37–42
Sukumar E, Gopal RH, Rao RB, Viswanathan S, Thirugnanasambantham P, Vijayasekaran V (1995) Pharmacological actions of cerpegin, a novel pyridine alkaloid from Ceropegia juncea. Fitoterapia 66(5):403–406
Valverde R, Arias O, Thorpe TA (1987) Picloram-induced somatic embryogenesis in pejibaye palm (Bactris gasipaes H.B.K.). Plant Cell Tiss Org Cult 10:149–156
Zhang T (2007) In vitro flowering of Perilla frutescens. In Vitro Cell Dev Biol Plant 43:91–94
Acknowledgments
Authors are thankful to Department of Biotechnology (DBT), Government of India for providing financial assistance. VAB thankful to Council of Scientific and Industrial Research (CSIR), New Delhi for Emeritus Scientist Fellowship. Authors are also thankful to Head, Department of Botany, Shivaji University, Kolhapur for providing necessary laboratory facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chavan, J.J., Nimbalkar, M.S., Adsul, A.A. et al. Micropropagation and in vitro flowering of endemic and endangered plant Ceropegia attenuata Hook. J. Plant Biochem. Biotechnol. 20, 276–282 (2011). https://doi.org/10.1007/s13562-011-0059-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13562-011-0059-0