Abstract
Introduction
A survey was conducted by The Harris Poll on behalf of Arcutis Biotherapeutics in the USA to understand perspectives and burden of patients with psoriasis using prescription topical treatments for their disease. This manuscript presents results from the subset of patients with intertriginous psoriasis.
Methods
The survey was conducted online October 21–November 24, 2021, among 507 US adults aged 18+ years with psoriasis diagnosed by a healthcare provider and currently using prescription topical treatment. Participants with intertriginous psoriasis were patients with plaque psoriasis reporting symptoms in the armpit, groin, under breast, stomach fold, or between the buttocks.
Results
Of the 507 respondents, 320 (64%) reported symptoms in intertriginous areas at some point, typically between the buttocks (31%). Most patients with intertriginous psoriasis reported it made them feel embarrassed (80%), anxious (79%), or depressed (69%). In addition, 45% of these patients reported intertriginous psoriasis caused a negative impact on sexual anxiety or distress. Quality of life impact was reported as “very strong negative impact” in 16% of patients with groin involvement vs. 6% in patients with no groin involvement and 15% in women vs. 6% in men. Patients with intertriginous psoriasis reported that itch (61%), scaling (53%), redness (49%), and skin cracking (46%) related to intertriginous psoriasis had the greatest negative impact on quality of life. Most (86%) of these patients said they would be more adherent if a single treatment option could be used to treat all affected areas of their body.
Conclusion
Psoriasis involvement in intertriginous areas over the course of disease is common and has a negative impact on these patients’ quality of life, particularly emotional well-being and sexual health.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
A survey was conducted by The Harris Poll on behalf of Arcutis Biotherapeutics in the USA to understand perspectives and burden of patients with psoriasis using prescription topical treatments for their disease. |
Most patients described their intertriginous psoriasis as moderate and reported that the most commonly affected areas were between buttocks (47%) and armpits (44%). |
Nearly all participants said their intertriginous psoriasis has an effect on their overall quality of life; female participants and participants with groin involvement reported a higher impact on quality of life. |
Patients with intertriginous psoriasis prefer a topical treatment and agreed that they would be more adherent if a single topical treatment could be used on multiple parts of their body. |
Introduction
Psoriasis is a chronic, inflammatory skin condition commonly affecting the scalp, extensor surfaces, and intertriginous areas [1]. Psoriasis affects about 3% of adults [2], with prevalence of intertriginous involvement varying from 3.2% to 36% [3]. The treatment of psoriasis in intertriginous areas is challenging with typical topical corticosteroid therapies because these areas are associated with greater risk for local adverse events such as skin atrophy, striae, and telangiectasia. Therefore, lower potency steroids, calcipotriene, and topical calcineurin inhibitors are recommended for use in intertriginous areas [4, 5]. Additional nonsteroidal topical treatment options have also been recently approved for patients with psoriasis, with roflumilast cream 0.3% including an indication for use in intertriginous areas.
In a global survey of 8338 patients with moderate-to-severe psoriasis, 84% of patients with psoriasis reported they experienced discrimination and/or humiliation due to psoriasis, which can affect their work, intimate relationships, sleep, and mental health [6, 7]. Similarly in a narrative interview of 90 patients with moderate-to-severe psoriasis, increasing severity of psoriasis symptoms was correlated with an increasingly negative effect on quality of life. Itching, flaking, pain, and burning were the most common symptoms negatively impacting quality of life [8]. Negative feelings from psoriasis symptoms affecting quality of life include anger, frustration, helplessness, embarrassment, and self-consciousness [9]. Surveys of patients with psoriasis showed that psoriasis involving intertriginous areas has a disproportionately negative effect on quality of life [1]. Furthermore, intertriginous involvement is associated with genital psoriasis, which particularly affects quality of life and sexual health [10].
A survey of US patients with plaque psoriasis was conducted by The Harris Poll on behalf of Arcutis Biotherapeutics to understand perspectives of patients with psoriasis who use topical treatments to manage their disease. This manuscript reports perspectives from the subset of respondents with psoriasis involving intertriginous areas.
Methods
This was an approximately 15-min survey conducted online October 21, 2021 through November 24, 2021, among 507 adults (aged ≥ 18 years) in the USA previously diagnosed with psoriasis by a healthcare provider (HCP). To be eligible, participants had to be using a topical treatment at the time of the survey and not be using a prescription injectable treatment for their psoriasis. The survey was conducted in accordance with the principles of the Declaration of Helsinki of 1964 and its later amendments and Good Clinical Practice guidelines of the International Council for Harmonisation. No identifying information is included for any participant. Before enrollment of patients, the study protocol and informed consent form were reviewed and approved by an appropriate institutional review board or independent ethics committee.
Data were weighted, where necessary, to align them with actual proportions in the population using a multistep weighting process. The first step was the initial total opt-in online sample of all US adults aged ≥ 18 years weighted to population benchmarks from the March 2020 Current Population Survey. The natural fallout sample of qualified respondents was 247/9584 US adults (the Gen Pop sample). In the second step, the total combined sample of 507 psoriasis patients from two sample sources (Gen Pop sample from step 1 and a second sample targeted to patients with psoriasis) was weighted using the estimated demographic distributions of the initial weighted opt-in online subset sample of patients with psoriasis from step 1.
Demographic variables included were education, age by gender, race/ethnicity, census region, household size, and marital status. A propensity score variable was included to adjust for respondents’ propensity to be online. Ranked weights were estimated using random iterative method weighting. The respondent level weights from step 2 were used as the final weights. Data are reported as percentages; percentages may not add up to 100% because of rounding and the acceptance of multiple responses. Response percentages were adjusted using a propensity score variable to adjust for respondents’ propensity to be online and a model using ranked weights and random iterative method weighting.
Psoriasis in intertriginous areas was defined as occurring in the groin, armpits, stomach folds, under breast, and between the buttocks. Patients with psoriasis in intertriginous areas were those who reported exhibiting symptoms in intertriginous areas when presented with a list of areas of the body or when aided with pictures showing psoriasis in this area.
Results
Of the 507 respondents, 320 (64%) patients with psoriasis reported having symptoms in intertriginous areas during the course of their disease, with the majority of patients with intertriginous psoriasis describing their symptoms as moderate in severity (67%). Approximately two times more patients with intertriginous area involvement reported having psoriasis in intertriginous areas after they saw pictures of psoriasis in these areas than prior to seeing the pictures. The most commonly affected intertriginous areas once patients saw the photos were between buttocks (47%), armpits (44%), and groin (40%; Fig. 1). When patients were asked where they typically exhibited their symptoms of plaque psoriasis, 10% overall reported having genital psoriasis, which was not included in the intertriginous assessment. In patients with intertriginous psoriasis, the respondents’ mean age was 45.1 years, most were male (55%), and 58% were White. The mean number of treatment types ever used by patients with intertriginous psoriasis was 2.6 prescription topicals, 2.3 over the counter, and 0.9 prescription oral.
Patients’ Perspective on Healthcare Provider Visits
Patient visits for participants with intertriginous psoriasis with HCPs varied considerably, with some patients reporting they disrobe for their exam and others indicating they remain fully clothed. Although 70% of patients with intertriginous area involvement showed their psoriasis to their HCP, the most common area of intertriginous involvement (between buttocks) was the least likely area to be shown to an HCP (Fig. 1). Patients with intertriginous psoriasis reported that the reasons for not showing certain areas of their body to their HCP were being too embarrassed (27%), not being asked to disrobe (19%), not being asked about psoriasis in intertriginous areas (18%), and being unsure if the symptoms experienced in certain areas are psoriasis (17%).
Patient-Reported Burden of Intertriginous Psoriasis
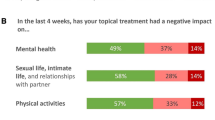
Nearly all patients (97%) said their intertriginous psoriasis has some negative effect on their overall quality of life and most (86%) said this impact is slightly (20%), moderately (46%), or strongly (20%) negative (Fig. 2). The percentages of patients reporting a “very strong negative impact” on their quality of life were directionally higher in patients with groin involvement (16%) versus no groin involvement (6%) and in women (15%) versus men (6%).
Patients with psoriasis in intertriginous areas reported that it makes them feel embarrassed (80%), anxious (79%), or depressed (69%). Of patients with intertriginous involvement, reported rates of emotional impact were directionally higher in women versus men (embarrassed, 85% vs. 76%; anxious, 87% vs. 74%; depressed, 77% vs. 62%), and rates of emotional impact were also higher in patients with groin involvement versus those without groin involvement (embarrassed, 86% vs. 76%; anxious, 84% vs. 76%; depressed, 76% vs. 63%).
Patients reported intertriginous psoriasis had a “very strong or strong negative impact” on sexual distress/anxiety (45%), intimate relationships (39%), and sexual function (37%) (Fig. 3). Patients with groin involvement reported directionally higher rates of impact versus patients with no groin involvement (sexual distress/anxiety, 52% vs. 39%; intimate relationships, 45% vs. 34%; sexual function, 44% vs. 31%; Fig. 3). Symptoms that caused the greatest negative impacts (based on percentage of patients reporting very strong or strong negative impact) on the quality of life among patients with psoriasis in intertriginous areas were itching (61%), scaling (53%), redness (49%), and skin cracking (46%; Fig. 4).
Patterns and Preferences of Patients with Intertriginous Psoriasis
When asked about the forms of treatment they preferred, 70% of patients with intertriginous psoriasis reported they prefer topical treatments, with 26% preferring pills and 4% preferring injections. Fifty-two percent of patients reported using multiple topical treatments on different parts of their body. Among those who use multiple treatments on different body parts, over half (54%) reported that the medications were time-consuming to apply and 40% reported that the medications were expensive; when these patients were asked about their experience using multiple topical therapies, 38% reported the experience was frustrating. Additionally, among these patients who used multiple topical treatments on different parts of their body, 43% reported using treatments on areas of their skin not discussed with their HCP and 31% reported using treatment longer than prescribed. A strong majority (87%) of the patients agreed that they would prefer a single treatment option for their psoriasis to treat different parts of their body. Furthermore, if a single treatment option could be used to treat different parts of their body, 86% of patients agreed that they would be more adherent (i.e., use treatments as they are intended to be used).
Discussion
A gap in the literature has been the lack of studies reporting the specific impact of intertriginous psoriasis, and this study fills that gap by characterizing the negative impact of intertriginous psoriasis on patients’ quality of life. Almost two in three patients with psoriasis reported symptoms in intertriginous areas at some point in the course of their disease. Consistent with other studies, patients with intertriginous involvement reported itching, scaling, redness, and cracking to be the most bothersome signs and symptoms of psoriasis [11]. Some patients with psoriasis in intertriginous areas were not able to identify the lesions as psoriasis until after seeing example photos. This suggests HCPs may need to show patients pictures and/or examine all their intertriginous areas to enable proper treatment of all areas involved.
Over 6 in 10 patients reported that they do not show HCPs all areas of their body with psoriasis involvement. Approximately one-quarter of these patients reported they are too embarrassed to do so and nearly 1 in 5 did not think the lesions in those areas were psoriasis, perhaps mistaking them for an infection or allergic reaction [12]. Intertriginous psoriasis, specifically vulvar involvement, can mimic candidiasis, which can make diagnosis difficult [13]. Therefore, the opportunity to discuss the differences in psoriasis presentation with patients and, if possible, to conduct a full-body exam may help address the proper diagnosis and thus treatment of patients with intertriginous and genital psoriasis. Patients should be educated about the value of full-body skin exams and encouraged to request a full-body skin exam, including examination of the intertriginous area.
Long-term use or intertriginous application of topical high-potency corticosteroids is associated with local side effects [14]. Concerningly in this poll, 43% of patients who used multiple topical treatments on different parts of their body reported using treatments on areas of their skin not discussed with their HCP. Additionally, of these patients who use multiple treatments, 31% reported using treatment longer than prescribed. This is specifically concerning since the areas that patients did not show their HCP are likely the areas where many topical treatments should not be used, and the potential exists for patients to use high-potency topical corticosteroids and other contraindicated topical treatments in intertriginous areas or for longer durations than typically prescribed.
Psoriasis in intertriginous areas has a disproportionately negative impact on patients’ lives, particularly their emotional well-being and sexual health [1]. In this study, almost 8 in 10 patients agreed that intertriginous psoriasis made them feel anxious and 45% of patients disclosed that it had a very strong or strong negative impact on their sexual distress/anxiety. A 2018 survey of 3821 patients with psoriasis along with anxiety and depression experienced a significant increase in the likelihood of itch, and several studies have reported an association between increased severity of itch and significantly lower quality of life [15, 16]. Although the current survey did not specify genital involvement as part of the intertriginous areas, patients with intertriginous and groin involvement reported more impact to sexual health parameters. This is consistent with a study of 20 patients with HCP-diagnosed genital psoriasis in which most patients reported impaired sexual experience during sexual activity (80%), worsening of symptoms after sexual activity (80%), decreased frequency of sexual activity (80%), avoidance of sexual relationships (75%), and reduced sexual desire (55%) [17]. This is also consistent with a survey of 776 patients with self-identified genital psoriasis that reported high rates of itching, burning, and dyspareunia, as well as poor sexual health, especially in women [10].
Most patients with psoriasis in intertriginous areas who use different topical treatments for different parts of their body agreed that the ability to use a single treatment would be beneficial and increase adherence. These results are similar to those of a 2014 survey of 3426 patients with psoriasis, in which 85% thought better psoriasis treatments are needed and 46% of patients reported that psoriasis treatments can be worse than the disease itself [11].
A limitation of this study is patients were not examined by a dermatology HCP to confirm diagnosis of intertriginous psoriasis and may have had other flexural dermatoses. Because this was a patient survey, validated indices to measure the emotional impact of intertriginous psoriasis were unable to be conducted. This could have led to higher levels of patient-reported embarrassment, anxiety and depression in this survey. Another limitation was patients in this survey were using topical treatments, which could have introduced bias toward a preference for topical treatments.
Conclusion
Patients with intertriginous psoriasis report considerable burden of disease that particularly affects their emotional and sexual quality of life. Patients with psoriasis may not associate lesions in their intertriginous areas with their psoriatic disease and may use prescriptions in these areas without discussing with their physician. These data highlight the opportunity to enhance the dialogue with patients to include discussing how psoriasis can present differently depending on the area of the body, the need for more frequent full body exams to identify psoriasis in these areas, and the appropriate treatment options for psoriasis in these body areas.
Data Availability
Data collected for this study will be made available to others. Proposals for data requests will be reviewed and considered for sharing following approval of the indication. Information about when data availability will begin and end will be provided following approval of the indication.
References
Armstrong AW, Read C. Pathophysiology, clinical presentation, and treatment of psoriasis: a review. JAMA. 2020;323:1945–60. https://doi.org/10.1001/jama.2020.4006.
Armstrong AW, Mehta MD, Schupp CW, Gondo GC, Bell SJ, Griffiths CEM. Psoriasis prevalence in adults in the United States. JAMA Dermatol. 2021;157:940–6. https://doi.org/10.1001/jamadermatol.2021.2007.
Dopytalska K, Sobolewski P, Błaszczak A, Szymańska E, Walecka I. Psoriasis in special localizations. Reumatologia. 2018;56:392–8. https://doi.org/10.5114/reum.2018.80718.
Elmets CA, Korman NJ, Prater EF, et al. Joint AAD-NPF Guidelines of care for the management and treatment of psoriasis with topical therapy and alternative medicine modalities for psoriasis severity measures. J Am Acad Dermatol. 2021;84:432–70. https://doi.org/10.1016/j.jaad.2020.07.087.
Kalb RE, Strober B, Weinstein G, Lebwohl M. Methotrexate and psoriasis: 2009 National Psoriasis Foundation Consensus Conference. J Am Acad Dermatol. 2009;60:824–37. https://doi.org/10.1016/j.jaad.2008.11.906.
Armstrong A, Jarvis S, Boehncke WH, et al. Patient perceptions of clear/almost clear skin in moderate-to-severe plaque psoriasis: results of the clear about psoriasis worldwide survey. J Eur Acad Dermatol Venereol. 2018;32:2200–7. https://doi.org/10.1111/jdv.15065.
Dowlatshahi EA, Wakkee M, Arends LR, Nijsten T. The prevalence and odds of depressive symptoms and clinical depression in psoriasis patients: a systematic review and meta-analysis. J Invest Dermatol. 2014;134:1542–51. https://doi.org/10.1038/jid.2013.508.
Pariser D, Schenkel B, Carter C, Farahi K, Brown TM, Ellis CN. A multicenter, non-interventional study to evaluate patient-reported experiences of living with psoriasis. J Dermatolog Treat. 2016;27:19–26. https://doi.org/10.3109/09546634.2015.1044492.
Armstrong AW, Schupp C, Wu J, Bebo B. Quality of life and work productivity impairment among psoriasis patients: findings from the National Psoriasis Foundation survey data 2003–2011. PLoS ONE. 2012;7:e52935. https://doi.org/10.1371/journal.pone.0052935.
Larsabal M, Ly S, Sbidian E, et al. GENIPSO: a French prospective study assessing instantaneous prevalence, clinical features and impact on quality of life of genital psoriasis among patients consulting for psoriasis. Br J Dermatol. 2019;180:647–56. https://doi.org/10.1111/bjd.17147.
Lebwohl MG, Bachelez H, Barker J, et al. Patient perspectives in the management of psoriasis: results from the population-based Multinational Assessment of Psoriasis and Psoriatic Arthritis Survey. J Am Acad Dermatol. 2014;70:871-81.e1-30. https://doi.org/10.1016/j.jaad.2013.12.018.
Czuczwar P, Stępniak A, Goren A, et al. Genital psoriasis: a hidden multidisciplinary problem—a review of literature. Ginekol Pol. 2016;87:717–21. https://doi.org/10.5603/gp.2016.0074.
Mauskar MM, Marathe K, Venkatesan A, Schlosser BJ, Edwards L. Vulvar diseases: conditions in adults and children. J Am Acad Dermatol. 2020;82:1287–98. https://doi.org/10.1016/j.jaad.2019.10.077.
Lebwohl M, Ali S. Treatment of psoriasis. Part 1. Topical therapy and phototherapy. J Am Acad Dermatol. 2001;45:487–502. https://doi.org/10.1067/mjd.2001.117046.
Zhu B, Edson-Heredia E, Guo J, Maeda-Chubachi T, Shen W, Kimball AB. Itching is a significant problem and a mediator between disease severity and quality of life for patients with psoriasis: results from a randomized controlled trial. Br J Dermatol. 2014;171:1215–9. https://doi.org/10.1111/bjd.13065.
Griffiths CEM, Jo S-J, Naldi L, et al. A multidimensional assessment of the burden of psoriasis: results from a multinational dermatologist and patient survey. Br J Dermatol. 2018;179:173–81. https://doi.org/10.1111/bjd.16332.
Cather JC, Ryan C, Meeuwis K, et al. Patients’ perspectives on the impact of genital psoriasis: a qualitative study. Dermatol Ther (Heidelb). 2017;7:447–61. https://doi.org/10.1007/s13555-017-0204-3.
Acknowledgements
The authors thank the patients and their families for participation in this trial. Medical writing support was provided by Lauren Ramsey, PharmD, for Alligent Biopharm Consulting, which was supported by Arcutis Biotherapeutics, Inc.
Medical Writing and Editorial Assistance
Medical writing support was provided by Lauren Ramsey, PharmD, for Alligent Biopharm Consulting, which was supported by Arcutis Biotherapeutics, Inc.
Funding
This work was supported by Arcutis Biotherapeutics, Inc, and Arcutis Biotherapeutics, Inc will be funding the journal’s Rapid Service Fee.
Author information
Authors and Affiliations
Contributions
All authors (Teri Greiling, Melodie Young, Melissa S. Seal, Robert C. Higham, April Armstrong) had full access to all the data in the study and they all take responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: all authors. Acquisition, analysis, or interpretation of data: all authors. Drafting of the manuscript: all authors. Critical revision of the manuscript for important intellectual content: all authors.
Corresponding author
Ethics declarations
Conflict of Interest
Teri Greiling has received investigator fees, speaker fees, clinical research grants, and/or honoraria from Arcutis Biotherapeutics, Inc. Melodie Young has received investigator fees, speaker fees, clinical research grants, and/or honoraria from AbbVie, Arcutis Biotherapeutics, Inc., Celgene Corporation, Eli Lilly, Janssen, Merck/Sun, Novartis, and UCB. April Armstrong has served as a research investigator, scientific adviser, or speaker to AbbVie, Almirall, Arcutis, ASLAN, Beiersdorf, BI, BMS, EPI, Incyte, Leo, UCB, Janssen, Lilly, Mindera, Nimbus, Novartis, Ortho Dermatologics, Sun, Dermavant, Dermira, Sanofi, Regeneron, and Pfizer. Melissa S. Seal and Robert C. Higham are employees of Arcutis Biotherapeutics, Inc.
Ethical Approval
The trial was conducted in accordance with the principles of the Declaration of Helsinki of 1964 and its later amendments and Good Clinical Practice guidelines of the International Council for Harmonisation. Before enrollment of patients, the study protocol and informed consent form were reviewed and approved by an appropriate institutional review board or independent ethics committee. Participants provided consent to participate in the survey and to report data in aggregate, not individual participant data.
Additional information
Prior Presentation: MauiDerm NP/PA Spring, June 22–25, 2022, Colorado Springs, CO.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Greiling, T., Young, M., Seal, M.S. et al. Patient Perspectives on the Prevalence and Burden of Intertriginous Psoriasis: Results from a National Survey of Adults with Psoriasis in the United States. Dermatol Ther (Heidelb) 14, 1839–1847 (2024). https://doi.org/10.1007/s13555-024-01190-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13555-024-01190-4