Abstract
Introduction
Non-dermatology medical specialties may refer patients for skin biopsies, searching for a particular diagnosis. However, the diagnostic impact of the skin biopsy is not clearly established. This article aims to assess the indications for, and evaluate the clinical relevance of, skin biopsies in non-dermatology medical specialties.
Methods
A questionnaire was sent to 23 non-dermatology specialty departments in a university medical center, requesting a list of indications for skin biopsies, as well as to 10 staff dermatologists to collect the indications of skin biopsies requested by non-dermatology specialties. Once the indications were collected, a literature search was performed to evaluate their clinical value and relevance.
Results
Eleven non-dermatology specialties provided a list of skin biopsy indications, to which staff dermatologists added seven more indications. A literature search revealed evidence-based medicine data for six diseases, that is, amyloidosis, peripheral autonomic neuropathy, Sneddon’s syndrome, intravascular lymphoma, sarcoidosis, and chronic graft-versus-host disease. Results were questionable concerning infectious endocarditis, acute graft-versus-host-disease, and the lupus band test. Skin biopsy were not evidenced as useful for the diagnosis of calciphylaxis, systemic scleroderma, Behçet’s disease, or hypermobile Ehlers–Danlos syndrome. For the diagnosis of Alport’s syndrome, pseudoxanthoma elasticum, and vascular Ehlers–Danlos syndrome, skin biopsy is currently outperformed by genetic analyses. For diagnoses such as Henoch–Schönlein purpura and Sjögren’s syndrome, skin biopsy represents an additional item among other diagnostic criteria.
Conclusion
The usefulness of skin biopsy as requested by non-dermatology specialties is only evidenced for amyloidosis, peripheral autonomic neuropathy, Sneddon’s syndrome, intravascular lymphoma, sarcoidosis, chronic graft-versus-host-disease, Henoch–Schönlein purpura, and Sjögren’s syndrome.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The relevance of skin biopsies for non-dermatology specialties remains unsettled for many indications. |
After collecting the various indications for skin biopsies in non-dermatology specialties, a review was performed for evidence levels. |
The usefulness of a skin biopsy as requested by non-dermatology specialties is evidenced for amyloidosis, peripheral autonomic neuropathy, Sneddon’s syndrome, intravascular lymphoma, sarcoidosis, and chronic graft-versus-host-disease. |
Skin biopsy is one diagnostic criterion among others in calciphylaxis, Sjögren’s syndrome, systemic sclerosis, and Henoch–Schönlein purpura. |
The utility of cutaneous biopsies remains controversial in infectious endocarditis and acute graft-versus-host disease (GVHD) as well as lupus band test for the diagnosis of systemic erythematous lupus. |
Molecular biology has outperformed the sensitivity and specificity of skin biopsies in vascular Ehlers–Danlos, Alport’s syndrome, and pseudoxanthoma elasticum even though skin biopsy remains the fastest and cheaper option. |
Introduction
A skin biopsy performed under local anesthesia is a simple, safe, reproducible, and minimally invasive tool for sampling skin tissue, leaving only a minimal scar. They are of paramount importance to confirm or to help to achieve a precise diagnosis in dermatology. Aside from standard hematoxylin–eosin histochemical staining, a whole array of complementary techniques is available for refining or confirming the histological diagnosis, including a series of special histochemical stains, immunohistochemical and immunofluorescence techniques, in situ hybridization, PCR, and genetic analyses.
Several non-dermatology medical and surgical specialties may also request or perform a skin biopsy of normal or diseased skin in the workup of some specific diagnoses. However, the usefulness and the relevance of these skin biopsies in achieving a specific diagnosis are not always clear. First, there is the textbook phenomenon where standard diagnostic procedures are often accepted as they are and copy/pasted to the next edition. Second, as there is no or little financial interest from pharmaceutical companies, it is difficult to obtain funding to verify the truthfulness of these indications.
The purpose of this work was to collect the indications of skin biopsies as requested by non-dermatology specialties and to analyze the currently available literature concerning the evidence of relevance and usefulness.
Methods
The non-dermatology medical and surgical specialties potentially requesting skin biopsies of their patients were identified by sending a questionnaire to all 23 medical and surgical departments of the university hospital. The questionnaire asked for which specific diseases a skin biopsy is recommended or should be considered. Two reminders were sent.
Another questionnaire was sent to the ten staff dermatologists of the university dermatology department asking for which diseases patients are referred by other medical or surgical specialties for a skin biopsy.
Once the different indications were collected, a literature search was performed to evaluate the clinical value and relevance of skin biopsies according to different levels of evidence. The consulted database was PubMed with English as language, from 1960 to April 2021. The search terms included the names of the specific disease and were subsequently cross-referenced with the following search terms: “diagnosis,” “skin biopsy,” “cutaneous sample,” “diagnostic method,” “histology,” “immunohistochemistry,” “in situ hybridization,” “PCR,” and “genetic analysis.”
The selected articles were then classified according to their level of evidence, ranging from 1 to 3 (1, literature proof available; 2, expert-based recommendation without literature support; 3, no proof retrieved in the literature).
No authorization of the ethical committee was required as this work did not involve direct access to patient data.
Results
Eleven non-dermatology specialties (cardiology, gastroenterology, hematology, nephrology, rheumatology, neurology, general surgery, pneumology, ophthalmology, pediatrics, and physical medicine) reported the following indications for requesting a skin biopsy (Table 1). Gynecology, otorhinolaryngology, emergency medicine, urology, medical oncology, and geriatrics did not reply. Psychiatry, anesthesiology, neurosurgery, stomatology, orthopedic surgery, and esthetic surgery did not report any indication or did not reply.
The returned questionnaires from the dermatologists added seven supplementary indications of requesting a skin biopsy by a non-dermatology specialty (Table 1). The level of evidence is listed as well as whether normal or diseased skin should be biopsied, and how many biopsies should be sampled (Table 1). In addition, when available, alternative diagnostic techniques for skin biopsies are indicated and whether the skin biopsy represents the primary diagnostic tool or is considered an accessory tool for achieving a particular diagnosis (Table 1).
Hereunder we briefly discuss the reported indications.
Discussion
Hereunder we discuss the different indications collected by the two questionnaire rounds.
Vascular Ehlers–Danlos
Ehlers–Danlos syndrome (EDS) is a group of connective tissue disorders comprising 13 subtypes characterized in different proportions by joint hypermobility, skin hyperextensibility, and tissue fragility [1]. The vascular subtype is mainly caused by type III collagen mutations [2, 3]. This collagen type is abundant in the skin, blood vessels, and visceral organs. Skin biopsies seem logically more accessible to sample than vascular samples to explore the disease [3,4,5]. Although some dermatopathologists describe changes on light microscopy and/or electron microscopy examinations, most specialists agree that these modifications are nonspecific [5,6,7,8]. Currently, skin biopsies can be useful to sample for genetic analysis to screen for COL3A1 mutation [1, 9]. In brief, the final diagnosis of the vascular subtype currently relies on molecular genetic testing performed on blood or skin samples with high sensitivity and specificity rather than on light or electron microscopy [1, 10].
Hypermobile Ehlers–Danlos Syndrome
Generalized joint hypermobility as well as milder cutaneous involvement characterizes hypermobile Ehlers–Danlos syndrome (hEDS), a heritable connective tissue disorder [11]. Light microscopy may evidence elastopathy. Electron microscopy can reveal variability in the diameter of the fibrils, irregularities of the interfibril spaces, and flower-shaped fibrils [12]. However, most experts agree that all these modifications are nonspecific [9]. Since the genetic basis of hEDS is still unknown, the diagnosis of this subtype remains mainly clinical [13] and is based on the assessment of joint hypermobility using the Beighton score [14] with, unfortunately, a controversial specificity, sensitivity, and a high inter-examiner variability [1]. The identification of the causal gene(s) would provide a genetic diagnostic tool [13]
Primary and Secondary Amyloidosis
Amyloidosis is characterized by extracellular proteolysis-resistant deposits of fibrils leading to the impairment of organ function [15]. Classification distinguishes primary systemic amyloidosis (AL), often associated with myeloma and lymphoproliferative disorders, from secondary systemic amyloidosis (AA), associated with chronic inflammatory conditions [15]. Skin manifestations present as small, smooth, firm, and waxy papules, macroglossia, periorbital purpura, purpuric lesions, and ecchymoses, which are often found in AL amyloidosis but rarely present in AA forms [15]. The diagnosis relies on the recognition of tissular amyloid deposits [16] and is positive when showing Congo red-positive amyloid deposits, with apple-green birefringence using polarized light [17,18,19,20]. Immunohistochemistry and/or immunofluorescence with anti-light-chain (LC) antibodies are useful for confirmation [19]. Biopsies of clinically involved organs such as liver, heart, and kidney are highly sensitive but also a lot more invasive [16] than skin samples. The histological examination of cutaneous sample in suspected AA amyloidosis is positive in 50–90% of all cases [17, 19], whereas it is shown to be positive in 50% of all cases in AL amyloidosis [15]. Multiple biopsies are recommended to increase the sensitivity for diagnosing amyloidosis. Many studies have shown that a salivary gland biopsy is a highly sensitive and specific method for the diagnosis of both forms and is currently considered the gold standard test to diagnose systemic amyloidosis [18, 20,21,22,23,24,25,26].
Pseudoxanthoma Elasticum
Pseudoxanthoma elasticum (PXE) is an autosomal recessive connective tissue disorder characterized by the accumulation of fragmented and mineralized elastic fibers leading to dermatologic, ophthalmologic, and vascular dysfunction [27, 28]. The disease is characterized by yellowish papules or plaque-like skin lesions often appearing in late childhood. The cervical areas are typically involved [27]. A biopsy of lesional skin reveals calcified elastic fibers, narrowed vessel lumen, calcium deposit in the internal elastic lamina, or fibrous thickening of the endothelium (Fig. 1). Therefore, it can be an interesting diagnostic tool [27], whereas a biopsy of clinically normal skin is not useful [29]. Currently, the underlying genetic defect has been identified in the ABCC6 gene and genetic analysis looking for those mutations are now available with a significantly higher specificity and sensitivity than a skin biopsy of lesional skin [28, 30]. Dermatologic examination and skin biopsy used to be the gold standard, but are nowadays replaced by molecular diagnosis [31].
Infective Endocarditis
Infective endocarditis is a life-threatening disorder that must be rapidly diagnosed and treated to avoid mortality. Osler’s nodes and Janeway lesions represent the cutaneous manifestations but are found in only 5–15% of the infected patients. They are highly suggestive of septicemia [32]. Histological findings of both lesions include septic microemboli in small reticular dermal arterioles with the formation of microabscess in the dermis. Leukocytoclastic vasculitis has also been reported but seems not to be specific [32,33,34]. Microbiological culture may eventually reveal causal microorganisms that can be helpful in the diagnosis. To be more sensitive and specific, skin biopsies have to be performed within 48 h of the onset of the skin lesions [35]. Indeed, the initial skin lesions are later replaced by a nonspecific immunological process. Histology and microbiology can be a prognostic factor, as microabscess formation, visible organisms, and positive bacterial cultures are usually caused by highly virulent organisms [33].
Acute and Chronic Graft-versus-Host Disease
Graft-versus-host disease (GVHD) is a systemic disease due to allogeneic stem cell transplantation divided into acute and chronic forms, respectively defined as within or after 100 days post-transplantation [36, 37]. Skin biopsies are commonly performed to establish the cause of new skin rashes [38]. Histologic diagnosis of GVHD relies on interface dermatitis, vacuolar degeneration of the basal layer, dyskeratosis, and superficial perivascular infiltrate [37]. However, these alterations are sometimes very subtle and may overlap with other skin diseases of the post-transplantation period, such as drug reactions, viral exanthems, and lymphocyte recovery. The usefulness of skin biopsies to confirm an acute GVHD remains controversial in the literature: histological findings are regularly nonspecific, they correlate poorly with the clinical severity of the cutaneous eruption, and they do not allow one to assess the prognosis and progression of a rash [38,39,40,41]. In contrast, when chronic GVHD is suspected, a punch biopsy is a valuable tool [42, 43], although diagnosis requires a clear clinic–pathologic correlation. [36, 44]. However, skin biopsy is superfluous if the diagnosis of chronic GVHD has been established by other clinical, biological, or histological criteria [45]. In a consensus paper on performing skin biopsies, 88% of the participants agreed that a skin biopsy is generally indicated in patients with suspected chronic GVHD, whereas only 62% felt that it was necessary in acute GVHD [46]. Unfortunately, only a very small number of studies attempted to evaluate the usefulness of skin biopsies for acute and chronic GVHD.
Sneddon’s Syndrome
Sneddon’s syndrome is a rare condition, characterized by a combination of episodes of ischemic cerebrovascular events, caused by antiphospholipid antibody deposits and livedo racemosa. The most striking histological aspects is the occlusion of arterioles by subendothelial proliferation [47,48,49]. Ultrastructurally, this thickening corresponds to immigrant medial smooth muscle cells with intermediate filaments colonizing the subendothelial intimal space [47, 50, 51] and shows a positive immunostaining for alpha-smooth muscle actin and tropomyosin [52]. Skin biopsies are often normal, requiring multiple samples [53] to reach a sensitivity up to 80% with three biopsies [47]. The optimal biopsy site is the center of a livedo racemosa ring. In addition, adequate biopsy size and serial sections are required to optimize the detection of a Sneddon’s syndrome [47, 54,55,56].
Intravascular Lymphoma
Intravascular lymphoma (IVL) is a rare lymphoproliferative disorder characterized by the proliferation of neoplastic B cells, NK/T cells, or monocyte/macrophage variants within the lumina of cutaneous blood vessels, or even as primary dermal presentation [57,58,59,60]. Skin lesions observed in about one-third of patients are nonspecific. They mainly appear as erythematous or purpuric nodules and plaques [61, 62]. Diagnosis of IVL involves histopathologic demonstration of neoplastic B or NK/T cells [59]. Since the skin is an easily accessible organ and can show evidence of disease even in apparently healthy skin, cutaneous biopsy must be considered as an important diagnostic tool [59, 63] even without evident skin involvement [61, 62]. However, there are several limitations: studies are rare and have small sample sizes, sometimes with discordant results [63,64,65,66,67,68,69]. Sampling precautions must be respected to increase sensitivity. This includes the sampling of normal skin in at least three sites and preferring lesioned skin and angiomas when present [64, 65, 70, 71].
In sum, skin biopsies should always be performed to rule out lymphoproliferative disorder in patients with “unclear” or “therapy-resistant” skin lesions, or in the event of “unclear” fever. Histopathological analysis, immunohistochemistry, and molecular techniques performed on skin biopsies can be crucial for proper classification of hematologic neoplasia involving the skin and can allow one to stage a lymphoma or a leukemia.
Alport’s Syndrome
Alport’s syndrome is a hereditary disorder combining glomerular nephropathy, hearing impairment, and ophthalmologic alterations [72]. It is related to mutations of the type IV collagen and is caused in 85% of cases by COL4A5 gene mutations on the X chromosome and in 15% by mutations in the COL4A3 or COL4A4 genes located on chromosome 2 [72, 73]. Immunohistochemistry on a skin biopsy can identify X-linked Alport’s syndrome as the α5 chain of collagen IV is absent at the dermal–epidermal junction [73, 74]. Hence, immunohistochemistry with specific anti-α5-chain collagen antibodies does not stain in affected people. The sensitivity exceeds 80%, but its positivity does not exclude the condition and does not permit one to identify the other forms [74,75,76]. Interestingly, in female carriers, immunostaining of the dermal–epidermal membrane presents a fragmented pattern [73, 74]. Currently, most experts recommend genetic testing for the diagnosis of Alport’s syndrome. However, mutation detection is slow, laborious, and expensive whereas the analysis of the skin biopsy is fast, less expensive, and less laborious [77, 78]. In sum, combining clinical data, the family history, and the results of α5-chain collagen immunochemical staining on skin biopsies allows one to identify a large proportion of affected patients, hence saving the patient from a renal biopsy and the cost and wait of genetic testing [72, 73].
Calciphylaxis
The pathogenesis of calciphylaxis, also termed calcific uremic arteriolopathy, an ischemic small-vessel vasculopathy, is multifactorial. Calciphylaxis may occur in patients with renal failure whether dialyzed or not [79, 80] and may present cutaneous ulcerations and/or tissue necrosis [79]. Although the skin lesions are highly characteristic with purplish patches presenting a necrotic center, surrounded by a painful erythematous edge and indurated subcutaneous nodules especially on fat tissue-rich skin regions [79, 81], the clinical diagnosis remains difficult. The gold-standard diagnostic procedure for calciphylaxis used to be cutaneous biopsy revealing medial and perivascular calcification and intimal proliferation of small arteries. Currently, skin biopsies tend to be avoided as they can lead to chronic and difficult-to-heal ulcerations [80, 81] and present a sensitivity varying widely from 18% to 86% owing to various sampling methods and subjective interpretations of the histopathological samples [82,83,84]. Furthermore, false negative results can lead to a potentially lethal delay in the treatment [85]. In brief, skin biopsies are only recommended for patients with an uncertain clinical diagnosis. When performed, benefit–risk balance should be carefully evaluated [86].
Peripheral Autonomic Neuropathy
Peripheral autonomic neuropathy (PAN) comprises a series of disorders characterized by the dysfunction of autonomic nerve fibers [87]. Cutaneous biopsies for the diagnosis of PAN present high sensitivity and specificity, together with the currently available routine autonomic testing [87]. It allows one to characterize and quantify the density of the intraepidermal nerve fibers [88, 89]. Immunohistochemistry using anti-protein-gene-product 9.5 antibodies readily identifies intraepidermal nerve fibers [90,91,92], adequately distinguishing patients with polyneuropathy from controls [91,92,93,94,95] with a moderate-to-good sensitivity and a high specificity [96].
Henoch–Schönlein Purpura
Henoch–Schönlein purpura (HSP) is a systemic vasculopathy of the small vessel mainly observed during childhood. HSP presents a nonthrombocytopenic palpable purpura, arthralgia/arthritis, bowel angina, and hematuria/proteinuria. The skin biopsy was not a prerequisite for diagnosis [97] until a large statistical validation process of selected criteria for HSP evidenced that predominant IgA deposits on skin biopsy are one of the minor criteria, whereas palpable purpura is the major criterion [98, 99]. Biopsy is also a tool to distinguish HSP from other kinds of purpura especially when performed within the first 24–48 h of the onset of lesions[100]. Hence, skin biopsy is a sensitive criterion but not specific, as vascular deposits of IgA can be retrieved in other vasculitic syndromes [101, 102].
Sarcoidosis
Sarcoidosis is a multisystemic, inflammatory disease of unknown etiology that is characterized by noncaseating granulomas. The skin manifestations are divided into specific lesions with histopathologically evident noncaseating granulomas and nonspecific lesions that develop as a result of a reactive process [103]. When specific cutaneous lesions are present, a skin biopsy allows an early diagnosis of sarcoidosis through a nonaggressive procedure. The presence of noncaseating granulomas at one site is usually regarded as sufficient for this diagnosis (Fig. 2) [104, 105].
Behçet’s Disease
Behçet’s disease is a rare multisystemic vasculitis that affects the skin but also the vascular, neurological, ocular, and articular systems [106, 107]. Mucocutaneous manifestations include bipolar (oral and genital) aphthae, which are the most frequent ones, followed by pseudofollicular lesions found in 40–45% of all cases and dermohypodermal nodules present in less than 50% [108]. A skin biopsy is sometimes performed to help establish the diagnosis. However, the histological aspect is nonspecific, revealing polynuclear inflammation with or without arterial obliteration under the necrotic areas. The histological findings are similar in idiopathic aphtous ulcers. Hence, the diagnosis of Behçet’s disease is based on a set of criteria, none of them including a skin biopsy [108, 109].
Systemic Sclerosis
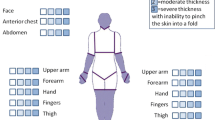
Systemic sclerosis, also known as scleroderma, is a fibrotic process of the skin and various internal organs characterized by three consecutive steps: an inflammatory phase, followed by thickening and finally atrophy of the skin [110]. The histological picture of the skin shows initially microvascular alterations, followed by chronic inflammation evolving toward cutaneous fibrosis and thickening (Fig. 3) [110, 113]. Currently, histological examination does not belong to the diagnostic criteria and is not routinely performed [114]. Skin biopsy is required only in the case of diagnostic doubt with other scleroderma-like disorders or for research purposes [111, 115,116,117,118]. The gold standard method to evaluate skin thickness is the modified Rodnan skin score, which should always be correlated with the clinical grounds and autoantibody profile [110, 111]. Changes in the diagnosis criteria could appear in the coming years as recent studies have shown that multiple histologic parameters correlate with severity [116].
Lupus Band Test in Systemic Lupus Erythematous
Systemic lupus erythematous (SLE) is an autoimmune disease with immunological, genetic, and environmental factors, potentially affecting almost any organ. The lupus band test (LBT) compares deposits of immunoglobulins and complements along the dermal–epidermal junction (DEJ) in photo-exposed and photo-protected skin. It is considered positive when one or more immunoreactants (IgG, IgA, IgM, C3) are detected [119, 120]. The interpretation of a positive LBT implies several variables, including the site of the biopsy (lesional or normal skin), the composition of the fluorescent band, the morphology and brightness of the immunofluorescence in conjunction with clinical findings, and serological and immunopathological testing [103,104,105, 121,122,123]. The predictive value is greater when C4 (100%), properdin (91.3%), and IgA (86.2%) are identified than with IgM (59%). Furthermore, the specificity and predictive value increase with the number of immunoreactants identified [121, 124,125,126]. The LBT is positive in about 70–80% of sun-exposed nonlesional skin and in about 55% of sun-protected nonlesional skin in SLE. The LBT used to be considered a sensitive and specific test in the diagnosis of systemic lupus erythematosus, and some authors described it as a prognostic procedure [121, 127, 128], but currently, it is controversial. Indeed, the intensity of the direct immunofluorescence band fails to show any relationship with the degree of inflammation and does not correlate with the level of inflammation in a clinical lesion [129,130,131].
Sjögren’s Syndrome
Sjögren’s syndrome is an autoimmune disease mainly characterized by xerostomia and xerophthalmia due to disruption of epithelial cells and a lymphoplasmocytic infiltration of exocrine glands [132]. Dermatologic manifestations include xerostomia and tumefactions of salivary glands but also xerosis, vasculitis, angular cheilitis, eyelid dermatitis, and annular erythema [133, 134]. The American–European Consensus Group proposed diagnostic criteria among which histopathology and the presence of autoantibodies are the major ones [135]. A positive biopsy is defined as a lymphocytic sialadenitis (FLS) with a focus score of ≥ 1 defined as at least 50 mononuclear cells per 4 mm2 [136]. Minor salivary gland biopsy is a reliable diagnostic tool with an elevated specificity and a sensitivity, ranging from 63.5% to 93.7% [137]. However, diagnostic value of salivary gland biopsy remains unclear owing to circular reasoning in the literature [136], absence of standardized histopathological interpretation and scoring of samples [138, 139], and the need for trained pathologists to perform the reading [139].
Limitations
It remains difficult to judge why only 11 out of 23 specialties responded to the questionnaire, either because there is no indication for a skin biopsy in their specialty or because they simply did not respond to the questionnaire despite reminders.
A Delphi consensus-based evaluation of the collected indications involving experts from the different specialties could be a next step of research.
Conclusion
This review aimed to clarify the usefulness of the skin biopsy as diagnostic tool in diseases cared for by non-dermatology specialties.
This review concluded that skin biopsy can be a useful diagnostic tool in amyloidosis, peripheral autonomic neuropathy, sarcoidosis, Sneddon’s syndrome, intravascular lymphoma, and chronic GVHD. Although only a small number of studies evaluated the utility of skin biopsies, all the experts seem to agree.
The utility of cutaneous biopsies remains controversial in infectious endocarditis and acute GVHD as well as the LBT for the diagnosis of systemic erythematous lupus. Current literature supports that a skin biopsy is useless in the diagnosis of Behçet disease and hypermobile Ehlers–Danlos. In calciphylaxis, it used to be a useful tool, but since the benefit–risk balance is not favorable except in highly doubtful cases, it should not be routinely performed. In Henoch–Schönlein purpura, systemic scleroderma, and Sjogren’s syndrome, the examination of a skin biopsy represents one additional criterion among many others.
Molecular biology has outperformed the sensitivity and specificity of skin biopsies in multiple indications, including vascular Ehlers–Danlos, Alport’s syndrome, and pseudoxanthoma elasticum, even though skin biopsy remains the fastest and a cheaper diagnostic tool. Once the molecular defect of other diseases is determined, skin biopsy will probably become less useful in these indications.
Currently, a lack of strong evidence persists in many indications. Most studies rely on small samples, and the circular reasoning behind these articles is flawed. The field would benefit from further research and a standardized approach to determine the precise role of biopsy in many indications.
However, even when the evidence base is slim, it should also be kept in mind that a skin biopsy remains a quick, inexpensive tool, and is sometimes the only available procedure on site.
References
Sobey G. Ehlers–Danlos syndrome: how to diagnose and when to perform genetic tests. Arch Dis Child. 2015;100:57–61.
Frank M. Syndrome d’Ehlers–Danlos vasculaire. Rev Prat. 2009;59:459–61.
Bhandari R, Aatre RD, Kanthi Y. Diagnostic approach and management of genetic aortopathies. Vasc Med. 2020;25:63–77.
Shalhub S, Black JH, Cecchi AC, et al. Molecular diagnosis in vascular Ehlers–Danlos syndrome predicts pattern of arterial involvement and outcomes. J Vasc Surg. 2014;60:160–9.
Ong KT, Plauchu H, Peyrol S, et al. Ultrastructural scoring of skin biopsies for diagnosis of vascular Ehlers–Danlos syndrome. Virchows Arch. 2012;460:637–49.
Proske S, Hartschuh W, Enk A, Hausser I. Ehlers–Danlos syndrome: 20 years experience with diagnosis and classification at the university skin clinic of Heidelberg. J Dtsch Dermatol Ges. 2006;4:308–18.
Hausser I, Anton-Lamprecht I. Differential ultrastructural aberrations of collagen fibrils in Ehlers–Danlos syndrome types I–IV as a means of diagnostics and classification. Hum Genet. 1994;93:394–407.
Angwin C, Ghali N, Baker D, et al. Electron microscopy in the diagnosis of Ehlers–Danlos syndromes: correlation with clinical and genetic investigations. Br J Dermatol. 2020;182:698–707.
Germain DP, Herrera-Guzman Y. Vascular Ehlers–Danlos syndrome. Ann Genet. 2004;47:1–9.
Malfait F, Francomano C, Byers P, et al. The 2017 international classification of the Ehlers–Danlos syndromes. Am J Med Genet Part C Semin Med Genet. 2017;175:8–26.
Tinkle B, Castori M, Berglund B, et al. Hypermobile Ehlers–Danlos syndrome (aka Ehlers–Danlos syndrome Type III and Ehlers–Danlos syndrome hypermobility type): clinical description and natural history. Am J Med Genet C Semin Med Genet. 2017;175:48–69.
Hermanns-Lé T, Piérard GE, Piérard-Franchimont C. Syndrome d’Ehlers–Danlos de type hypermobile: une atteinte multi-systémique. Apport de l’ultrastructure cutanée pour une prise en charge personnalisée. Rev Med Liege. 2015;70:325–30.
Colombi M, Dordoni C, Chiarelli N, Ritelli M. Differential diagnosis and diagnostic flow chart of joint hypermobility syndrome/Ehlers–Danlos syndrome hypermobility type compared to other heritable connective tissue disorders. Am J Med Genet C Semin Med Genet. 2015;169:6–22.
Beighton P, Horan F. Orthopaedic aspects of the Ehlers–Danlos syndrome. J Bone Joint Surg Br. 1969;51:444–53.
Ronger-Salvé S. Thomas L. Amyloses cutanées. In : Saurat JH, Grosshans E. Laugier P. Lachapelle JM, editors. Dermatologie et infections sexuellement transmissibles. 4ème édition. Paris; 2004;88–92
Lecadet A, Bachmeyer C, Buob D, Cez A, Georgin-Lavialle S. Minor salivary gland biopsy is more effective than normal appearing skin biopsy for amyloid detection in systemic amyloidosis: a prospective monocentric study. Eur J Intern Med. 2018;57:20–1.
Westermark P. Occurrence of amyloid deposits in the skin in secondary systemic amyloidosis. Acta Pathol Microbiol Scand A 1972.
Rubinow A, Cohen AS. Skin involvement in generalized amyloidosis. A study of clinically involved and uninvolved skin in 50 patients with primary and secondary amyloidosis. Ann Intern Med. 1978;88:781–5.
Lee DD, Huang CY, Wong CK. Dermatopathologic findings in 20 cases of systemic amyloidosis. Am J Dermatopathol. 1998;20:438–42.
Desport E, Bridoux F, Sirac C, et al. Centre national de référence pour l’amylose AL et les autres maladies par dépôts d’immunoglobulines monoclonales. Al Amyloid Orphanet J Rare Dis. 2012;21:7–54.
Real de Asúa D, Costa R, Galván JM, Filigheddu MT, Trujillo D, Cadiñanos J. Systemic AA amyloidosis: epidemiology, diagnosis, and management. Clin Epidemiol. 2014;6:369–77
Hachulla E, Janin A, Flipo RM, et al. Labial salivary gland biopsy is a reliable test for the diagnosis of primary and secondary amyloidosis. A prospective clinical and immunohistologic study in 59 patients. Arthritis Rheum. 1993;36:691–7.
Suzuki T, Kusumoto S, Yamashita T, et al. Labial salivary gland biopsy for diagnosing immunoglobulin light chain amyloidosis: a retrospective analysis. Ann Hematol. 2016;95:279–85.
Mercan R, Bitik B, Tezcan ME, et al. Minimally invasive minor salivary gland biopsy for the diagnosis of amyloidosis in a rheumatology clinic. ISRN Rheumatol. 2014;35:46–8.
Foli A, Palladini G, Caporali R, et al. The role of minor salivary gland biopsy in the diagnosis of systemic amyloidosis: results of a prospective study in 62 patients. Amyloid. 2011;1:80–2.
Jamet MP, Gnemmi V, Hachulla A, et al. Distinctive patterns of transthyretin amyloid in salivary tissue: a clinicopathologic study of 92 patients with amyloid-containing minor salivary gland biopsies. Am J Surg Pathol. 2015;39:1035–44.
Roach ES, Islam MP. Pseudoxanthoma elasticum. Handb Clin Neurol. 2015;132:215–21.
Germain DP. Pseudoxanthoma elasticum. Orphanet J Rare Dis. 2017;12:85.
Brown SJ, Talks SJ, Needham SJ, Taylor AE. Pseudoxanthoma elasticum: biopsy of clinically normal skin in the investigation of patients with angioid streaks. Br J Dermatol. 2007;157:748–51.
Laube S, Moss C. Pseudoxanthoma elasticum. Arch Dis Child. 2005;90:754–6.
Van Loey S, Leys A. Pseudoxanthoma elasticum confirmed by genetic analysis but not by skin biopsy: a case report and review of the literature. Bull Soc Belge Ophtalmol. 2013;322:83–7.
Servy A, Valeyrie-Allanore L, Alla F et al; Association Pour l'Etude et la Prévention de l'Endocardite Infectieuse Study Group. Prognostic value of skin manifestations of infective endocarditis. JAMA Dermatol 2014;150:494–500.
Gunson TH, Oliver GF. Osler’s nodes and Janeway lesions. Australas J Dermatol. 2007;48:251–5.
Alpert JS. Osler’s nodes and Janeway lesions are not the result of small-vessel vasculitis. Am J Med. 2013;126:843–4.
Matsui Y, Okada N, Nishizawa M, et al. An Osler’s node and a Janeway lesion. Intern Med. 2009;48:1487–8.
Kavand S, Lehman JS, Hashmi S, Gibson LE, El-Azhary RA. Cutaneous manifestations of graft-versus-host disease: role of the dermatologist. Int J Dermatol. 2017;56:131–40.
Johnson ML, Farmer ER. Graft-versus-host reactions in dermatology. J Am Acad Dermatol. 1998;38:369–92.
Kohler S, Hendrickson MR, Chao NJ, Smoller BR. Value of skin biopsies in assessing prognosis and progression of acute graft-versus-host disease. Am J Surg Pathol. 1997;21:988–96.
Barksdale SK, Oberlender SA, Barnhill RL. “Rush” skin biopsy specimens in a tertiary medical center: diagnostic yield and clinical utility. J Am Acad Dermatol. 1998;38:548–54.
Paun O, Phillips T, Fu P, Novoa RA, Honda KS, Lu KQ, Lazarus HM. Cutaneous complications in hematopoietic cell transplant recipients: impact of biopsy on patient management. Biol Blood Marrow Transplant. 2013;19:1204–9.
Zhou Y, Barnett MJ, Rivers JK. Clinical significance of skin biopsies in the diagnosis and management of graft-vs-host disease in early postallogeneic bone marrow transplantation. Arch Dermatol. 2000;136:717–21.
Jagasia MH, Greinix HT, Arora M, et al. National institutes of health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. The 2014 diagnosis and staging working group report. Biol Blood Marrow Transplant. 2015;21:389–401.
Filipovich AH, Weisdorf D, Pavletic S, et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and staging working group report. Biol Blood Marrow Transplant. 2005;11:945–56.
Jacobsohn DA, Montross S, Anders V, Vogelsang GB. Clinical importance of confirming or excluding the diagnosis of chronic graft-versus-host disease. Bone Marrow Transplant. 2001;28:1047–51.
Lehman JS, Gibson LE, El-Azhary RA, Chavan RN, Hashmi SK, Lohse CM, Flotte TJ. Acute cutaneous graft-vs-host disease compared to drug hypersensitivity reaction with vacuolar interface changes: a blinded study of microscopic and immunohistochemical features. J Cutan Pathol. 2015;42:39–45.
Hillen U, Hausermann P, Massi D, et al. Consensus on performing skin biopsies, laboratory workup, evaluation of tissue samples and reporting of the results in patients with suspected cutaneous graft-versus-host disease. J Eur Acad Dermatol Venereol. 2015;29:948–54.
Wohlrab J, Fischer M, Wolter M, Marsch WC. Diagnostic impact and sensitivity of skin biopsies in Sneddon’s syndrome. A report of 15 cases. Br J Dermatol. 2001;145:285–8.
Stockhammer G, Felber SR, Zelger B, et al. Sneddon’s syndrome: diagnosis by skin biopsy and MRI in 17 patients. Stroke. 1993;24:685–90.
Wu S, Xu Z, Liang H. Sneddon’s syndrome: a comprehensive review of the literature. Orphanet J Rare Dis. 2014;9:215.
Marsch WC, Muckelmann R. Livedo racemosa generalisata. Livedo racemosa generalisata. Clinical aspects and histopathology of obliterating arteriolopathy with CNS involvement. Z Hautkr. 1985;60:873–4.
Legierse CM, Canninga-Van Dijk MR, Bruijnzeel-Koomen CA, Kuck-Koot VC. Sneddon syndrome and the diagnostic value of skin biopsies-three young patients with intracerebral lesions and livedo racemosa. Eur J Dermatol. 2008;18:322–8.
Tamm E, Jungkunz W, Wolter M, Marsch WC. Immunohistochemical characterization of the “intimal proliferation” phenomenon in Sneddon’s syndrome and essential thrombocythaemia. Br J Dermatol. 1994;131:814–21.
Timoney I, Flynn A, Leonard N, Wynne B. Livedo racemosa: a cutaneous manifestation of Sneddon’s syndrome. BMJ Case Rep. 2019;12:e232670.
Zelger B, Sepp N, Schmid KW, Hintner H, Klein G, Fritsch PO. Life history of cutaneous vascular lesions in Sneddon’s syndrome. Hum Pathol. 1992;23:668–75.
Kong SS, Azarfar A, Bhanusali N. Sneddon syndrome: under diagnosed disease, complex clinical manifestations and challenging diagnosis. A case-based review. Rheumatol Int. 2020;41:987–91.
Bolayir E, Yilmaz A, Kugu N, Erdogan H, Akyol M, Akyuz A. Sneddon’s syndrome: clinical and laboratory analysis of 10 cases. Acta Med Okayama. 2004;58:59–65.
Shimada K, Kinoshita T, Naoe T, Nakamura S. Presentation and management of intravascular large B-cell lymphoma. Lancet Oncol. 2009;10:895–902.
Brunet V, Marouan S, Routy JP, Hashem MA, et al. Retrospective study of intravascular large B-cell lymphoma cases diagnosed in Quebec: a retrospective study of 29 case reports. Med (Baltim). 2017;96(5):e5985.
Asada N, Odawara J, Kimura S, et al. Use of random skin biopsy for diagnosis of intravascular large B-cell lymphoma. Mayo Clin Proc. 2007;82:1525–7.
Wang L, Chen S. Ma H et al Intravascular NK/T-cell lymphoma: a report of five cases with cutaneous manifestation from China. J Cutan Pathol. 2015;42:610–7.
Ferreri AJ, Campo E, Seymour JF, et al. Intravascular lymphoma: clinical presentation, natural history, management and prognostic factors in a series of 38 cases, with special emphasis on the “cutaneous variant”. International Extranodal Lymphoma Study Group (IELSG). Br J Haematol. 2004;127:173–83.
Zuckerman D, Seliem R, Hochberg E. Intravascular lymphoma: the oncologist’s “great imitator.” Oncologist. 2006;11:496–502.
Sitthinamsuwan P, Chinthammitr Y, Pattanaprichakul P, Sukpanichnant S. Random skin biopsy in the diagnosis of intravascular lymphoma. J Cutan Pathol. 2017;44:729–33.
Rozenbaum D, Tung J, Xue Y, Hoang MP, Kroshinsky D. Skin biopsy in the diagnosis of intravascular lymphoma: a retrospective diagnostic accuracy study. J Am Acad Dermatol. 2021;85:665–70.
Cho HG, Sheu SL, Kuo KY, et al. Limited role of random skin biopsy in the diagnosis of intravascular lymphoma in adult patients with hemophagocytic lymphohistiocytosis. Acta Haematol. 2017;138:33–8.
Pongpudpunth M, Rattanakaemakorn P, Fleischer ABJ. Usefulness of random skin biopsy as a diagnostic tool of intravascular lymphoma presenting with fever of unknown origin. Am J Dermatopathol. 2015;37:686–90.
Maekawa T, Komine M, Murata S, Fukushima N, Ohtsuki M. Random skin biopsy of patients with intravascular large B-cell lymphoma associated with thrombocytopenia and coagulation abnormalities: proposal of a modified biopsy method. J Dermatol. 2015;42:318–21.
Higashi Y, Kawai K, Yonekura K, et al. Indication for random skin biopsy for the diagnosis of intravascular large B cell lymphoma. Dermatology. 2012;224:46–50.
Yamada E, Ishikawa E, Watanabe R, et al. Random skin biopsies before brain biopsy for intravascular large B-cell lymphoma. World Neurosurg. 2019;121:364–9.
Arai T, Kato Y, Funaki M, et al. Three cases of intravascular large B-cell lymphoma detected in a biopsy of skin lesions. Dermatology. 2016;232:185–8.
di Fonzo H, Contardo D, Carrozza D, et al. Intravascular large B-cell lymphoma presenting as fever of unknown origin and diagnosed by random skin biopsies: a case report and literature review. Am J Case Rep. 2017;18:482–6.
Bruni V, Petrisano M, Tarsitano F, et al. Alport’s syndrome. J Biol Regul Homeost Agents. 2019;33:19–24.
Delanaye P, Nikkels AF, Martalo O et al. Comment j’explore … le syndrome d'Alport par la biopsie cutanée. Quand la peau parle pour le rein. Rev Med Liege 2002;57:670–1.
van der Loop FT, Monnens LA, Schröder CH, Lemmink HH, Breuning MH, Timmer ED, Smeets HJ. Identification of COL4A5 defects in Alport’s syndrome by immunohistochemistry of skin. Kidney Int. 1999;55:1217–24.
Kashtan CE. Alport syndrome: is diagnosis only skin-deep? Kidney Int. 1999;55:1575–6.
Nakanishi K, Iijima K, Kuroda N, et al. Comparison of alpha5 (IV) collagen chain expression in skin with disease severity in women with X-linked Alport syndrome. J Am Soc Nephrol. 1998;9:1433–40.
Savige J, Ariani F, Mari F, et al. Expert consensus guidelines for the genetic diagnosis of Alport syndrome. Pediatr Nephrol. 2019;34:1175–89.
Savige J, Gregory M, Gross O, Kashtan C, Ding J, Flinter F. Expert guidelines for the management of Alport syndrome and thin basement membrane nephropathy. J Am Soc Nephrol. 2013;24:364–75.
Jeong HS, Dominguez AR. Calciphylaxis: controversies in pathogenesis, diagnosis and treatment. Am J Med Sci. 2016;351:217–27.
Fine A, Zacharias J. Calciphylaxis is usually non-ulcerating: risk factors, outcome and therapy. Kidney Int. 2002;61:2210–7.
Alves C, Ernandez T, Stoermann CC. Calcific uremic arteriolopathy (calciphylaxis): update in 2018. Rev Med Suisse. 2018;14:426–9.
Mochel MC, Arakaki RY, Wang G, Kroshinsky D, Hoang MP. Cutaneous calciphylaxis: a retrospective histopathologic evaluation. Am J Dermatopathol. 2013;35:582–6.
Cassius C, Moguelet P, Monfort JB, et al. Calciphylaxis in haemodialysed patients: diagnostic value of calcifications in cutaneous biopsy. Br J Dermatol. 2018;178:292–3.
Dobry AS, Nguyen ED, Shah R, Mihm MC, Kroshinsky D. The role of skin biopsy in diagnosis and management of calciphylaxis: a retrospective analysis. J Am Acad Dermatol. 2020;85:765–7.
Williams EA, Moy AP, Cipriani NA, Nigwekar SU, Nazarian RM. Factors associated with false-negative pathologic diagnosis of calciphylaxis. J Cutan Pathol. 2019;46:16–25.
Brandenburg VM, Evenepoel P, Floege J et al; ERA-EDTA working group on CKD-MBD and EUCALNET. Lack of evidence does not justify neglect: how can we address unmet medical needs in calciphylaxis? Nephrol Dial Transplant 2016;31:1211–9.
Donadio V, Incensi A, Giannoccaro MP, et al. Peripheral autonomic neuropathy: diagnostic contribution of skin biopsy. J Neuropathol Exp Neurol. 2012;71:1000–8.
Myers MI, Peltier AC. Uses of skin biopsy for sensory and autonomic nerve assessment. Curr Neurol Neurosci Rep. 2013;13:323.
Smith AG, Howard JR, Kroll R, Ramachandran P, Hauer P, Singleton JR, McArthur J. The reliability of skin biopsy with measurement of intraepidermal nerve fiber density. J Neurol Sci. 2005;228:65–9.
Goransson LG, Mellgren SI, Lindal S, Omdal R. The effect of age and gender on epidermal nerve fiber density. Neurology. 2004;62:774–7.
Lauria G, Cornblath DR, Johansson O et al; European Federation of Neurological Societies. EFNS guidelines on the use of skin biopsy in the diagnosis of peripheral neuropathy. Eur J Neurol 2005;12:747–58.
McArthur JC, Stocks EA, Hauer P, Cornblath DR, Griffin JW. Epidermal nerve fiber density: normative reference range and diagnostic efficiency. Arch Neurol. 1998;55:1513–20.
Herrmann DN, McDermott MP, Henderson D, Chen L, Akowuah K, Schifitto G; North East AIDS Dementia (NEAD) Consortium. Epidermal nerve fiber density, axonal swellings and QST as predictors of HIV distal sensory neuropathy. Muscle Nerve 2004;29:420–7.
Koskinen M, Hietaharju A, Kyléniemi M, Peltola J, Rantala I, Udd B, Haapasalo H. A quantitative method for the assessment of intraepidermal nerve fibers in small-fiber neuropathy. J Neurol. 2005;252:789–94.
Li J, Bai Y, Ghandour K, et al. Skin biopsies in myelin-related neuropathies: bringing molecular pathology to the bedside. Brain. 2005;128:1168–77.
England JD, Gronseth GS, Franklin G, et al. Evaluation of distal symmetric polyneuropathy: the role of autonomic testing, nerve biopsy, and skin biopsy (an evidence-based review). Muscle Nerve. 2009;39:106–15.
Ramelli V, Lava SA, Simonetti GD, Bianchetti MG, Ramelli GP, Milani GP. Blistering eruptions in childhood Henoch–Schönlein syndrome: systematic review of the literature. Eur J Pediatr. 2017;76:487–92.
Ruperto N, Ozen S, Pistorio A et al. EULAR/PRINTO/PRES criteria for Henoch-Schönlein purpura, childhood polyarteritis nodosa, childhood Wegener granulomatosis and childhood Takayasu arteritis: Ankara 2008. Part I: overall methodology and clinical characterisation. Ann Rheum Dis. 2010;69:790–7
Yang YH, Yu HH, Chiang BL. The diagnosis and classification of Henoch–Schönlein purpura: an updated review. Autoimmun Rev. 2014;13:355–8.
Chango AJ, Calle Sarmiento PM, Lopetegui Lia N, Alexander SA, Modi V. Leukocytoclastic vasculitis: an early skin biopsy makes a difference. Cureus 2020;12(5):e7912.
Magro CM, Crowson AN. A clinical and histologic study of 37 cases of immunoglobulin A-associated vasculitis. Am J Dermatopathol. 1999;21:234–40.
Carlson JA, Chen KR. Cutaneous vasculitis update: small vessel neutrophilic vasculitis syndromes. Am J Dermatopathol. 2006;28:486–506.
Haimovic A, Sanchez M, Judson MA, Prystowsky S. Sarcoidosis: a comprehensive review and update for the dermatologist: part I. Cutaneous disease. J Am Acad Dermatol. 2012;66:1–18.
Wessendorf TE, Bonella F, Costabel U. Diagnosis of sarcoidosis. Clin Rev Allergy Immunol. 2015;49:54–62.
Mana J, Marcoval J. Skin manifestations of sarcoidosis. Presse Med. 2012;41:355–74.
Davatchi F. Behcet’s disease. Int J Rheum Dis. 2014;17:355–7.
Bessis D. Maladie de Behçet. In :Dermatologie et infections sexuellement transmissibles. 6th ed. 2004;530–2.
Zouboulis CC. Epidemiology of Adamantiades-Behçet’s disease. Ann Med Interne. 1999;150:488–98.
McCarty MA, Garton RA, Jorizzo JL. Complex aphthosis and Behçet’s disease. Dermatol Clin. 2003;21:41–8.
Barsotti S, Orlandi M, Codullo V, et al. One year in review 2019: systemic sclerosis. Clin Exp Rheumatol. 2019;37(Suppl 119):3–14.
Rongioletti F, Ferreli C, Atzori L, Bottoni U, Soda G. Scleroderma with an update about clinico-pathological correlation. G Ital Dermatol Venereol. 2018;153:208–15.
Leask A. Matrix remodeling in systemic sclerosis. Semin Immunopathol. 2015;37:559–63.
Varga J, Denton CP, Wigley FM. Scleroderma: from pathogenesis to comprehensive management. xx ed. Springer, New York; 2012; 689
van den Hoogen F, Khanna D, Fransen J, et al. 2013 Classification criteria for systemic sclerosis: an American College of Rheumatology/European League against Rheumatism collaborative initiative. Arthritis Rheum. 2013;65:2737–47.
Medsger T, Benedek T. History of skin thickness assessment and the Rodnan skin thickness scoring method in systemic sclerosis. J Scleroderma Relat Disord. 2019;4:83–8.
Showalter K, Gordon J. Skin histology in systemic sclerosis: a relevant clinical biomarker. Curr Rheumatol Rep. 2020;26(23):3.
Rodnan GP, Lipinski E, Luksick J. Skin thickness and collagen content in progressive systemic sclerosis and localized scleroderma. Arthritis Rheum. 1979;22:130–40.
Verrecchia F, Laboureau J, Verola O, Roos N, Porcher R, Bruneval P, et al. Skin involvement in scleroderma—where histological and clinical scores meet. Rheumatol (Oxf). 2007;46:833–41.
Alahlafi AM, Wordsworth P, Wojnarowska F. The distribution of IgG subclasses in the lupus band suggests disease-specific alteration in subclass switching rather than polyclonal B-cell activation. Clin Exp Dermatol. 2004;29:288–92.
Mehta V, Sarda A, Balachandran C. Lupus band test. Indian J Dermatol Venereol Leprol. 2010;76:298–300.
Cardinali C, Caproni M, Fabbri P. The utility of the lupus band test on sun-protected non-lesional skin for the diagnosis of systemic lupus erythematosus. Clin Exp Rheumatol. 1999;17:427–32.
Al-Suwaid AR, Venkataram MN, Bhushnurmath SR. Cutaneous lupus erythematosus: comparison of direct immunofluorescence findings with histopathology. Int J Dermatol. 1995;34:480.
Kontos AP, Jirsari M, Jacobsen G, Fivenson DP. Immunoglobulin M predominance in cutaneous lupus erythematosus. J Cutan Pathol. 200;32:352–5
Smith CD, Marino C, Rothfield NF. The clinical utility of the lupus band test. Arthritis Rheum. 1984;27:382–7.
Burnham TK, Neblett TR, Fine G. Immunofluorescent, “band” test for lupus erythematosus. II. Employing skin lesions. Arch Dermatol. 1970;102:42.
Reich A, Marcinow K, Bialynicki-Birula R. The lupus band test in systemic lupus erythematosus patients. Ther Clin Risk Manag. 2011;7:27–32.
Weigand DA. The lupus band test: a re-evaluation. J Am Acad Dermatol. 1984;11:230–4.
Monroe EW. Lupus band test. Arch Dermatol. 1977;113:830–4.
Hung T, Ian Crawford R, Martinka M. Degree of histologic inflammation in lupus erythematosus and direct immunofluorescence results: red and inflamed lesions do not increase the chances of getting a bright band. J Cutan Med Surg. 2013;17:22–6.
Harrist TJ, Mihm MC. Cutaneous immunopathology. The diagnostic use of direct and indirect immunofluorescence techniques in dermatologic disease. Hum Pathol. 1979;10:625–53
Harrist TJ, Mihm MC Jr. The specificity and clinical usefulness of the lupus band test. Arthritis Rheum. 1980;23:479–90.
Roguedas AM, Pers JO, Lemasson G, et al. Memory B-cell aggregates in skin biopsy are diagnostic for primary Sjögren’s syndrome. J Autoimmun. 2010;35:241–7.
Bernacchi E, Amato L, Parodi A, et al. Sjögren’s syndrome: a retrospective review of the cutaneous features of 93 patients by the Italian Group of Immunodermatology. Clin Exp Rheumatol. 2004;22:55–62.
Roguedas AM, Youinou P, Lemasson G, Pennec YL, Misery L. Primary Gougerot–Sjögren syndrome: a dermatological approach. J Eur Acad Dermatol Venereol. 2006;20:243–7.
Vitali C, Bombardieri S, Jonsson R et al.European Study Group on Classification Criteria for Sjögren's Syndrome. Classification criteria for Sjögren's syndrome: a revised version of the European criteria proposed by the American-European Consensus Group. Ann Rheum Dis 2002;61:554–8.
Daniels TE, Cox D, Shiboski CH, et al. Associations between salivary gland histopathologic diagnoses and phenotypic features of Sjögren’s syndrome among 1,726 registry participants. Arthritis Rheum. 2011;63:2021–30.
Guellec D, Cornec D, Jousse-Joulin S, et al. Diagnostic value of labial minor salivary gland biopsy for Sjögren’s syndrome: a systematic review. Autoimmun Rev. 2013;12:416–20.
Fisher BA, Brown RM, Bowman SJ, Barone F. A review of salivary gland histopathology in primary Sjögren’s syndrome with a focus on its potential as a clinical trials biomarker. Ann Rheum Dis. 2015;74:1645–50.
Costa S, Quintin-Roué I, Lesourd A, et al. Reliability of histopathological salivary gland biopsy assessment in Sjögren’s syndrome: a multicentre cohort study. Rheumatol (Oxf). 2015;54:1056–64.
Acknowledgements
Funding
No funding or sponsorship was received for this study or publication of this article.
Authorship
The authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, takes responsibility for the integrity of the work as a whole, and has given approval for this version to be published.
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Sophie Bailleux, Patrick Collins and Arjen F. Nikkels. The first draft of the manuscript was written by Sophie Bailleux and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Disclosures
Sophie Bailleux, Patrick Collins and Arjen F. Nikkels declare that they have no conflicts of interest.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Bailleux, S., Collins, P. & Nikkels, A.F. The Relevance of Skin Biopsies in General Internal Medicine: Facts and Myths. Dermatol Ther (Heidelb) 12, 1103–1119 (2022). https://doi.org/10.1007/s13555-022-00717-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13555-022-00717-x