Abstract
Objectives
The purpose of this paper is to present an easy–to–use and reproducible morphometrical method of determining the density of intraepidermal nerve fibers (IENF) per epidermal area with the corresponding reference range of the IENF–counts.
Methods
Thirty patients and 22 controls were included in this study. The patients were divided into three groups: small–fiber (SFN), diabetic and demyelinating neuropathy. All subjects underwent punch skin biopsy. Specimens were fixed routinely in formalin and thereafter embedded in paraffin. Nerve fibers were revealed using immunoperoxidase staining with panaxonal antibody PGP 9.5. Using light microscopy, immunopositive nerves were counted morphometrically per epidermal area (NPEA) and, for comparison, per epidermal length (NPEL).
Results
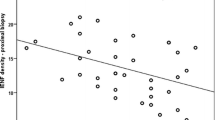
Both the NPEA and NPEL estimates of SFN and diabetic neuropathy group differed significantly from those of control specimen (p < 0.001 and p < 0.001, Mann–Whitney test). Our method of counting, NPEA, shows a good correlation to NPEL (r = 0.945).
Conclusions
IENF–counting by a new morphometric modification is reproducible and diagnostically sensitive and can easily be adopted in any laboratory familiar with the basic immunohistochemical methodology. The method is less dependent on costly technical support systems and seems to be less time consuming when compared with conventional methods for IENF–counting.
Similar content being viewed by others
References
Lacomis D (2002) Small–fiber neuropathy. Muscle Nerve 26:173–188
Dalsgaard C–J, Rydh M, Haegerstrand A (1989) Cutaneous innervation in man visualized with protein gene product 9.5 (PGP 9.5) antibodies. Histochemistry 92:385–389
Levy DM, Terenghi G,Gu XH,Abraham RR, Springall DR, Polak JM (1992) Immunohistochemical measurement of nerves and neuropeptides in diabetic skin: relationship to tests of neurological function. Diabetologia 35:889–897
Herrmann DN, Griffin JW, Hauer P, Cornblath DR, Griffin JW (1999) Epidermal nerve fiber density and sural nerve morphometry in peripheral neuropathies. Neurology 53:1634–1640
Hirai A,Yasuda H, Joko M, Maeda T, Kikkawa R (2000) Evaluation of diabetic neuropathy through the quantitation of cutaneous nerves. J Neurol Sci 172:55–62
Holland NR, Crawford TO, Hauer P, Cornblath DR, Griffin JW, McArthur JC (1998) Small–fiber sensory neuropathies: clinical course and neuropathology of idiopathic cases. Ann Neurol 44:47–59
Scott LJC,Griffin JW, Luciano C, Barton NW, Banerjee T, Crawford T, McArthur JC, Tournay A, Schiffmann R (1999) Quantitative analysis of epidermal innervation in Fabry disease. Neurology 52:1249–1254
Verzé L, Viglietti–Panzica C, Plumari L, Calcagni M, Stella M, Schrama LH, Panzica GC (2000) Cutaneous innervation in hereditary sensory and autonomic neuropathy type IV. Neurology 55:126–128
Lauria G, McArthur JC, Hauer PE, Griffin JW, Cornblath DR (1998) Neuropathological alterations in truncal neuropathy: evaluation by skin biopsy. J Neurol Neurosurg Psychiatry 65:762–766
Holland NR, Stocks MA, Hauer P, Cornblath DR, Griffin JW, McArthur JC (1997) Intraepidermal nerve fiber density in patients with painful sensory neuropathy. Neurology 48:708–711
Kennedy WR,Wendelschafer–Crabb G, Johnson T (1996) Quantitation of epidermal nerves in diabetic neuropathy. Neurology 47:1042–1048
McCarthy BG, Hsieh S–T, Stocks MA, Hauer P, Macko C, Cornblath DR, Griffin JW, McArthur JC (1995) Cutaneous innervation in sensory neuropathies: Evaluation by skin biopsy. Neurology 45:1848–1855
Omdal R, Mellgren SI, Goransson L, Skjesol A, Lindal S, Koldingsnes W, Husby G (2002) Small nerve fiber involvement in systemic lupus erythematosus: a controlled study. Arthritis Rheum 46:1228–1232
Sandroni P, Low PA (2001) Autonomic peripheral neuropathies: clinical presentation, diagnosis, and treatment. J Clin Neuromuscul Dis 2:147–157
McArthur JC, Stocks EA, Hauer P, Cornblath DR, Griffin JW (1998) Epidermal nerve fiber density.Normative reference range and diagnostic efficiency. Arch Neurol 55:1513–1520
Haapasalo H, Pesonen E, Collan Y (1989) Volume corrected mitotic index (M/V–INDEX). The standard of mitotic activity in neoplasms. Path Res Pract 185:551–554
Haapasalo HK, Sallinen PK, Helen PT, Rantala IS, Helin HJ, Isola JJ (1993) Comparison of three quantitation methods for PCNA immunostaining: applicability and relation to survival in 83 astrocytic neoplasms. J Pathol 171:207–214
Periquet MI, Novak V, Collins MP, Nagaraja HN, Erdem S, Nash SM, Freimer ML, Sahenk Z, Kissel JT, Mendell JR (1999) Painful sensory neuropathy. Prospective evaluation using skin biopsy. Neurology 53:1641–1647
Yasuda H, Terada M, Maeda K, Kogawa S, Sanada M, Haneda M, Kashiwagi A, Kikkawa R (2003) Diabetic neuropathy and nerve regeneration. Prog Neurobiol 69:229–285
Chiang MC, Lin YH, Pan CL, Tseng TJ, Lin WM, Hsieh ST (2002) Cutaneous innervation in chronic inflammatory demyelinating polyneuropathy. Neurology 59:1094–1098
Pan CL, Tseng TJ, Lin YH, Chiang MC, Lin WM, Hsieh ST (2003) Cutaneous innervation in Guillain–Barré syndrome: pathology and clinical correlations. Brain 126:386–397
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koskinen, M., Hietaharju, A., Kyläniemi, M. et al. A quantitative method for the assessment of intraepidermal nerve fibers in small–fiber neuropathy. J Neurol 252, 789–794 (2005). https://doi.org/10.1007/s00415-005-0743-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-005-0743-x