Abstract
This study aimed to investigate the compostability of treatment sludge and to determine the effects of adding different animal manures (cattle, pig, horse, and chicken) and agricultural waste (sunflower stalks) on the composting process. Five different compost reactors (M1, M2, M3, M4, and M5) were operated. Sunflower stalks were added to all reactors as a bulking agent at the rate of 20%. Treatment sludge (80%) and bulking agent were added to the M1 reactor, where animal manure was not added, while 40% treatment sludge and 40% animal manure were added to the other reactors. In the reactors where animal manure was added, cattle (M2), pig (M3), horse (M4), and chicken (M5) manure were used, respectively. The temperature, pH, electrical conductivity, organic matter (OM), carbon to nitrogen ratio (C/N), total Kjeldahl nitrogen, and total phosphorus were monitored during the 21-day process. The highest OM loss (64.8%) occurred in the M2 reactor, and the maximum temperature (62.7 °C) was achieved. The high temperatures reached in the reactor increased microbial activity, resulting in rapid decomposition of OM and elimination of pathogens. With a maximum reduction in the C/N ratio (40%), carbon and nitrogen balance was achieved, thus obtaining a more stable and higher quality compost for plants in this reactor. In the M1 reactor, where only treatment sludge was composted, lower microbiological activity occurred compared to the other mixtures due to the lack of substrate. It can be said that composting treatment sludge together with animal manures will provide higher quality end products.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
With the increase in population and industry, the amount of biodegradable waste continues to increase rapidly. The generated biomass can be converted into useful bioproducts using various bioconversion methods. Many studies have been conducted on obtaining bioproducts through different bioconversion methods [1,2,3]. Some industrial wastes rich in organic matter and nutrients can be utilized to transform into beneficial bioproducts such as biopesticides, biohydrogen, biofuels, enzymes, and bioplastics through bioconversion methods [4].
Managing biodegradable waste produced in significant amounts, such as treatment sludge, animal manure, and agricultural waste, through bioconversion methods and controlling these wastes is an important issue. Treatment sludge and animal manure cause many environmental problems if they are not disposed of properly. The large amount of domestic treatment sludge from treatment facilities is still being disposed of in landfills in Turkey, causing operational problems. In recent years, the disposal of treatment sludge by incineration has also risen. However, these methods cause different ambients to be adversely affected. If treatment sludge is not properly managed, it can create problems such as odor, toxic gases, vector attraction, and esthetic issues. Also, the leachate formed due to high moisture content contaminates soil and groundwater with high organic matter, pathogens, and heavy metals [5, 6]. Due to their high moisture content, transporting them to disposal sites or burning them in incinerators without pre-treatment also poses a problem. Animal manure is also a biodegradable waste rich in organic substance content. The application of animal manure to the soil positively affects the microorganism population and provides significant macro- and micronutrients to the soil [7]. In our country, animal manures are dried only in rural areas and used in the soil for the purpose of growing vegetables and fruits. In most settlements, they are left to nature in an uncontrolled manner, posing environmental risks. Inappropriate disposal of animal manure causes various problems such as odor, leaching of pollutants like nitrogen and phosphorus into groundwater, and eutrophication in lakes [8, 9]. Additionally, 30–60% of antibiotics used in animal farming are released through animal manure, posing risks to the environment and human health [10]. Considering the waste management hierarchy, waste reduction and recovery are among the priority waste management alternatives. Due to the richness of organic matter and nutrient content, treatment sludge and animal manures can be recovered by using them in agriculture [11]. However, these wastes have harmful properties such as pathogens and synthetic organic pollutants. For this reason, the composting process, which is a clean and sustainable strategy, is widely used to remove the harmful properties of this type of waste [12, 13]. The composting process is a complex biological process in which wastes are decomposed by various microorganisms into stable end products under controlled conditions in aerobic surroundings [14,15,16,17]. Through the composting process, the odor problem caused by treatment sludge and animal manure is eliminated; their moisture content and quantities are reduced; unstable forms of carbon, nitrogen, and phosphorus are converted into stable forms beneficial to the soil; and antibiotics contained in animal manure are reduced. Additionally, hygiene is ensured by eliminating the pathogens they contain [18,19,20,21].
Due to the low porosity properties caused by high moisture content and very small particle size, treatment sludge and animal manure cannot be composted alone and must be mixed with bulking agents [22]. Bulking agents added to the compost mix provide benefits such as moisture, optimization of the C/N ratio, and porosity balancing [23]. Due to the substantial quantities of treatment sludge and animal manures, the appropriate selection and use of bulking agents are crucial for the quality and cost-effectiveness of the resulting compost. Many researchers have worked on the composting of high moisture organic wastes with different bulking agents. In these studies, brush clippings, sawdust, green waste, broken wooden pallets, mushroom waste, acacia shavings, food processing wastes, office paper, newsprint, and tree branches were used as bulking agents [24,25,26,27].
There are many studies in which treatment sludge [22, 28, 29] and animal manures [30,31,32] are composted separately. Although the number of studies in which these wastes are composted together is more limited, they are available in the literature [33,34,35,36,37]. Jalili et al. [34] aimed to achieve the appropriate C/N ratio by creating two different mixture recipes and carried out a 60-day laboratory-scale study. Pistachio shell waste, cattle manure, sewage sludge, and sawdust were used as raw materials. In a study by Arias et al. [33], they created four mixtures by using green forest waste, pig manure, food waste, and sludge from chipboard production. At the end of the study, they concluded that the final product could be used for soil fertilization due to its rich nutrient content and low heavy metal and electrical conductivity (EC) content. The forms and amounts of nutrients contained in different animal manures vary. In a study by Li et al. [38], phosphorus characterization in dairy, swine, and broiler manures was compared, and total phosphorus was determined to be 9, 34, and 29% in these manures, respectively.
In this study, four different animal manures (cattle (CAM), pig (PM), horse (HM), and chicken (CHM)) were composted together with food industry treatment sludge (TS) and sunflower stalk (SS) as bulking agent. The objective of this study was to determine the effect of these four different animal manures on the composting of treatment sludge. In the literature, there is no study comparing the effects of the animal manures used in this study on the composting of treatment sludge. In the study, the physical and chemical changes occurring during the composting of different animal manures with treatment sludge were monitored, and the effects of different manures on the composting of treatment sludge were determined.
2 Materials and methods
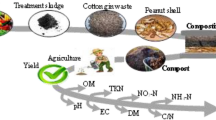
Treatment sludge originating from the food industry was composted alone and together with four different types of animal manure (cattle, pig, horse, and chicken) used as additional substrate materials in five separate aerobic compost reactors (M1, M2, M3, M4, and M5). Sunflower stalks were used as a bulking agent in the composting process. The overall process flow diagram is provided in Fig. 1.
2.1 Raw materials
In this study, treatment sludge was taken from the filter press outlet of a canned food factory in Bursa, and animal manures (cattle, pig, horse, chicken) were taken from the Farm of the Veterinary Faculty of Bursa Uludag University. Sunflower stalks were obtained from the Farm of the Agriculture Faculty of Bursa Uludag University. The characterization of the raw materials (TS, SS, CAM, PM, HM, and CHM) is presented in Table 1. As illustrated in Table 1, the OM content of animal manure is higher than that of treatment sludge, the moisture content of treatment sludge is higher than that of animal manure, and the electrical conductivity of chicken manure is higher than that of the other compost materials used.
2.2 Laboratory-scale composters and method
In the composting experiments, aerobic composting reactors made of stainless-steel material and with a volume of 30 l were used. All reactors were insulated with 50 mm of glass wool and aluminum sheet. A perforated grid was placed 5 cm above the bottom of the reactor to ensure the homogeneous distribution of air within the compost mixture and to allow the leachate formed to drain from the compost mass. Air was supplied to the reactors by means of an aquarium pump, and the airflow rates were measured with a rotameter and recorded online. Aeration was carried out to give an airflow rate of 600–700 ml/min for 15 min every hour. The temperature data in the reactors were measured by means of temperature sensors (thermocouples, TCR-M06-L180-K04.J). The reactors were covered with insulation material in order to maintain the temperature of the waste mass and to prevent heat exchange with the environment.
The composting process was performed as an active composting phase for 21 days in five different reactors. In the compost experiments, the treatment sludge and animal manures were mixed moistly without any pre-treatment. The sunflower stalks used in the experiments were ground to sizes of 0.5–1 cm. The M1 reactor was prepared by mixing only treatment sludge and sunflower stalk without adding animal manure. The other reactors were prepared by adding treatment sludge and sunflower stalk, and respectively, cattle manure to the M2 reactor, pig manure to the M3 reactor, horse manure to the M4 reactor, and chicken manure to the M5 reactor. The mixture ratios of all reactors are given in Table 2. The reactors were mixed manually every other day and on the sampling days for aeration purposes. The temperature and airflow values were recorded with SCADA software for 21 days.
2.3 Laboratory analysis
Samples were taken from the reactors on the 0th, 2nd, 7th, 14th, and 21st days, and experimental studies were carried out. All analytical tests were conducted in triplicate. The dry matter (DM) content of the samples was calculated by measuring the weight loss of samples dried at 105 °C, taking into account the volatile matter content determined based on combustion loss at 550 °C [39]. Moisture content was calculated by subtracting the DM amount from 100. The amount of volatile matter was accepted as organic matter (OM) [40, 41]. Easily oxidizable organic carbon concentrations of the raw materials and the compost mixtures used in the composting were calculated based on the OM content of the samples using a factor of 1.83 (%C = (100 − %ash)/1.83) [42, 43]. The pH and electrical conductivity (EC) values were determined in 1:10 (w/v) distilled water extract [44, 45]. To determine the total Kjeldahl nitrogen (TKN) contents, samples were burned using the Kjeldahl method, and the TKN concentration was determined by water vapor distillation [46]. Free air space (FAS) was determined using the method suggested by Madejón et al. [47]. Total phosphorus concentrations were determined by the ascorbic acid method [39].
3 Results and discussion
3.1 The initial properties of the compost mixtures
The initial properties of the compost mixtures are shown in Table 3. Compost mixtures subjected to the composting process must have a certain amount of moisture, C/N, and FAS in order for the process to run efficiently. For efficient composting processes, it is recommended that the C/N ratio be within the range of 20–40, and the FAS content be between 20–30% [48, 49]. In this study, initial C/N ratios ranged from 18.3 to 37.8 in the reactors. C/N ratios exceeding the optimal range may result in insufficient nitrogen for microbial growth and development, thereby requiring a longer composting period. Conversely, if the C/N ratio remains below the optimal range, nitrogen loss as ammonia gas can occur through evaporation [50]. Wu et al. [51] stated that an initial C/N ratio of 25 in their composting trials with pig manure, rice straw, and corn stalks was suitable for the formation of high-quality compost.
The moisture content of raw materials used for composting provides a suitable transport environment for the nutrients required by microorganisms. In compost mixtures with low moisture content, microorganisms undergo dehydration as they cannot obtain the necessary nutrients for their metabolism. Conversely, in mixtures with high moisture content, water accumulation within compost mass leads to anaerobic conditions during the process [52, 53]. Li et al. [54] determined the optimum moisture content under different moisture levels (45–61%) in their 45-day composting trials using chicken manure. Their research found that a moisture content of 53% achieved the highest composting temperature and longest thermophilic period. In this study, the treatment sludge and 4 different animal manures (CAM, PM, HM, and CHM) to be composted have high moisture content (85.2%, 81.6%, 78.7%, 71.2%, and 77.0%, respectively). Bulking agent was used to increase the moisture value to the required values for composting [55, 56]. For this purpose, sunflower stalk, which has a low moisture (10.3%) value, was added to the compost mixtures. The initial moisture values in the compost reactors ranged between 61.1 and 70.2% (Table 3). The initial pH values in the reactors ranged between 6.7 to 7.6. Ji et al. [57] also suggested that the initial pH values should range from 6.5 to 8.5 in their composting study with animal manures. The initial OM content of the compost reactors increased in consequence of mixing SS, which has a high OM content (91.6%), with animal manures and treatment sludge (56.1%), which have a lower OM content. The organic matter content in all prepared compost mixtures was over 75%.
3.2 Temperature
The temperatures in the compost reactors and ambient temperatures are given in Fig. 2. In general, temperatures increased to the thermophilic phase (> 40 °C) in all reactors. This temperature increase in the compost masses is due to microorganisms breaking down and consuming the easily degradable components and, meanwhile, generating heat during the first days [58]. Maximum temperature was measured as 48 °C in reactor M1, which was prepared only with treatment sludge without adding animal manure, 62.7 °C in reactor M2 with cattle manure added to treatment sludge, 51.5 °C in reactor M3 with pig manure added, 60.2 °C in reactor M4 with horse manure added, and 62.2 °C in reactor M5 with chicken manure added. An increase of temperature indicates the degradation rate of organic matter and can be related to microorganisms present in the environment [59]. Temperature is the main factor used to evaluate the change of pathogenic bacteria in the composting process. High temperature can effectively reduce pathogenic bacteria during the composting process [60]. According to the U.S. Environmental Protection Agency (EPA), for significant pathogen control, the wastes must be kept in the reactor at 40 °C for at least 5 days, during which the temperature must exceed 55 °C for 4 h. Compliance with the EPA’s pathogen reduction criteria is essential for producing safe and effective compost. The impact of effective composting on public health is significant. It provides numerous benefits such as preventing diseases, protecting the environment, improving soil health, and ensuring safe agricultural practices [61]. According to Yin et al. [62], temperatures need to exceed 55 °C to eliminate pathogenic microorganisms. In reactor M1, the temperature remained at 40 °C and above for 3 days, but did not reach 55 °C; in reactor M2, it remained above 40 °C for 8 days and above 55 °C for 2 days; while in reactor M3, it remained above 40 °C for 8 days, but did not reach 55 °C. The temperature in the M4 reactor stayed above 40 °C for 7 days and remained above 55 °C for 1.5 days during this period. The temperature in the M5 reactor, on the other hand, rose above 40 °C for 4 days and remained above 55 °C for 2 days during this period. Considering the EPA criteria in terms of the temperature values, an adequate level of pathogen control could not be achieved in M1 with only sludge, and in reactor M3 with pig manure added. In reactors M2, M4, and M5, however, it is thought that pathogens were significantly reduced and that microbial stabilization was achieved to a large extent. Abouelwafa et al. [63] stated that in a well-functioning composting process, temperatures can reach 50–65 °C with microbial activity. In the study conducted by Zhan et al. [64], the aim was to examine the changes in pathogenic bacteria during the co-composting of sewage sludge, food waste, and corn stalks. At the beginning of the study, they identified 50 pathogenic bacteria in the raw mixture, while after composting, they found 27 pathogenic bacteria. A significant reduction in most bacteria was observed after transitioning to the thermophilic phase. In this study, a significant decrease in the concentration of pathogens was detected at temperatures above 50 °C. Tan et al. [65] stated that during the composting of sewage sludge, the compost piles remained in the thermophilic phase (> 55 °C) for 21 days, with the temperature rising to 78 °C during this period. They indicated that during the thermophilic phase, where high temperatures were reached during composting, temperature-sensitive potential pathogenic bacteria were likely to be affected.
It can be stated that in the conducted composting trials, a more efficient process was achieved in the reactors in which cattle, horse, and chicken manure were added as substrate (M2, M4, and M5, respectively), compared to the composting of treatment sludge alone, and that a significant amount of pathogen removal and hygienization was able to be achieved.
3.3 pH and electrical conductivity
The pH values for the reactors are shown in Fig. 3a. The pH values in the reactors ranged between 6.5 and 8.5 throughout the process. The pH of compost mass can affect microorganisms and therefore the temperature of the compost and evaporation of ammonia [66]. Moreover, pH is affected by many reactions (decomposition, mineralization, etc.) during composting [67]. pH values increased during the first days due to intense microorganism activities in all reactors except M1 and M3. According to Jamroz et al. [68] and Hanc et al. [28], these increases in pH values can be explained by the degradation of organic substances, and the release of ammonium ions or volatile ammonia. From another perspective, Negi et al. [69] stated that throughout the composting, an increase in pH values was observed in parallel with temperature increase, and that the rate of degradation increased as pH increased. In the M1 and M3 reactors, however, a significant change did not occur during the first days. It can be said that, initially, no significant microbial activity or mineralization took place in these two reactors. In the following days, the pH value increased in both these reactors. During the process, active microorganisms need different pH values in order to secrete their enzymes. This environment was provided for microorganisms at pH ranges varying from 6.7 to 8.5 throughout the process. In all reactors, pH values at the end of the process were in the range of 7.8–8.3. During successful and fully developed composting, the pH value usually rises to around 8–9 [70].
The EC value is a parameter that reflects the content of soluble salts found in the mixture. In other words, it can also be defined as the salinity indicator, which shows the salt toxicity levels during composting [71]. The EC values for the reactors are shown in Fig. 3b. Values in all reactors ranged between 3.85 and 6.85. The increase in EC values during the process may be due to the decomposition of organic matter which is not reduced by binding to stable organic complexes [72]. In this study, it was determined that the highest increase in EC was in the M2, M4, and M5 reactors, in which the temperature rose above 60 °C and the decomposition of organic matter was the highest. If the compost formed at the end of the process is to be used for plants, the EC values must be below 4 mS/cm for the plants to tolerate it [73, 74]. It can be said that as a result of the formation of more stable forms in the mature compost obtained at the end of the maturation phase following the active composting phase, the EC values may decrease to more favorable values for plants.
3.4 Organic matter (OM) and C/N ratio
The changes in OM for the reactors are shown in Fig. 4a. OM concentrations decreased in all reactors during the process. Organic matter can be decomposed through the biochemical reactions of various microorganisms, including bacteria, fungi, plants, and algae. These microorganisms have the ability to transform pollutants into stable and harmless substances [75]. According to Li et al. [76], these decreases occur due to the fact that OM is a source of carbon and energy for microorganisms. Similarly, Zhong et al. [77] mentioned that many types of microorganisms are involved in composting and that some specific bacteria are effective in the degradation and transformation of organic materials during the process. When the OM losses occurring in the reactors were determined, they were found to be 46.2%, 64.8%, 51.2%, 58.3%, and 60.3% for reactors M1, M2, M3, M4, and M5, respectively. It was determined that the highest loss of OM was in the M2 reactor in which cattle manure and treatment sludge were used, and in which the temperature was measured as 62.7 °C. In the composting study conducted with dairy manure and sugar cane leaves by Jiang et al. [78], high temperatures (68 °C) were attained, and similar to this study, a highest OM loss rate of 61.8% was obtained. In the M1 reactor, in which there was the lowest OM degradation, the temperature (48 °C) was unable to reach the desired levels, and the microorganisms were unable to achieve adequate mineralization.
The C/N ratio of the organic material to be used for composting is important, since it has an effect on the microbial compost community, the product quality in terms of degree of stabilization, and the ultimately available nutrients [79]. While carbon is used as a primary source of energy and to form microbial cells, nitrogen is required for microbial growth and reproduction via protein synthesis [53, 80]. The changes in the C/N ratios are given in Fig. 4b. In general, the C/N ratios vary according to the raw materials used. For composting, C/N ratios of 25:1 and 30:1 are optimal, but values of 20:1 and 40:1 are also acceptable. Since higher C/N values will cause problems in microbial growth, the composting process will be prolonged [49]. At low values, however, the C/N ratio indicates that there will be a need for a higher N loss through evaporation, and longer maturation periods associated with the short duration of the thermophilic phase [51, 81]. Since the C/N ratio is widely used as a parameter of compost maturity, it is usually evaluated at the beginning and end of the composting process [82]. As can be seen in Fig. 4b, the initial C/N ratios in this study correspond with the values in the literature. The decrease in the C/N ratio due to the simultaneous consumption of carbonaceous and nitrogenous organic substrates indicates an effective composting process [83, 84]. Among the reactors, the highest reduction in the C/N ratio (40%) occurred in the M2 reactor. However, the lowest decrease in the C/N ratio (3.3%) was observed in the M1 reactor, which was set up with the addition of treatment sludge and sunflower stalk, and in which microbial activity remained at the lower limits and the temperature increase (48 °C) was not great. In the M3, M4, and M5 reactors, decreases in the C/N ratio of 26.7%, 35.7%, and 27.4% occurred, respectively.
3.5 Total Kjeldahl nitrogen (TKN) and total phosphorus (TP)
The changes in TKN during the composting process are given in Fig. 5a. The initial TKN values in all reactors ranged between 1.2 and 2.3%. Since nitrogen concentrations, which play an important role in the functioning of the process, directly affect the C/N ratio, they can cause unsuitable conditions if they are found in high values in the mixture. Throughout the process, increases and decreases were observed in TKN values, which are affected by many factors, especially microorganism activities, temperature changes, and dry matter losses. Moreover, increases and decreases in TKN values could be observed throughout the process depending on the amount of waste and the ventilation rate [85]. TKN values increased at the end of the process in all reactors except for the M1 reactor, which was set up with the addition of 80% treatment sludge and 20% sunflower stalk. The reason for this was that since the temperature increase (48 °C) in the M1 reactor was not at the desired level, sufficient mineralization did not occur in the waste mass, and organic materials could not be broken down. The co-composting of various organic wastes results in a more efficient composting process because it influences the biochemical processes through a greater number of microorganisms and minimizes nutrient losses during the composting process [86, 87].
Phosphorus is an essential macronutrient for all living organisms. Animal manure also contains 70–80% of the phosphorus consumed by the animal [88]. In their study in China, Li et al. [38] stated that the TP concentrations of feeds given to poultry and livestock were higher than the specified feed standards, and that, therefore, they encountered high phosphorus concentrations in excreted manure. For this reason, the aerobic composting of animal manure is a long-term and sustainable way to increase phosphorus production for use in the agricultural sector [89]. By composting animal manures, treatment sludge, and agricultural wastes separately or together, a stabilized soil amendment is obtained that can provide higher amounts of available phosphorus compared to inorganic fertilizers [35, 90, 91]. The changes in TP for the reactors are given in Fig. 5b. The initial phosphorus concentrations in all reactors ranged between 1.04 and 1.38%. Except for the M1 reactor, increases in phosphorus concentrations were observed due to the increase in temperatures, decomposition of OM, decline in the C/N ratio, and rise in pH in the reactors. The increase in TP concentration can be termed as the “concentration effect” due to the losses that occur with the outgassing as CO2, H2O, and NH3 [92]. Another reason for the increase in total phosphorus values was the loss of mass occurring in the reactors. In their study, Rehman et al. [35] co-composted sewage sludge and farm manure, and they noted that while sewage sludge contained a significant amount of phosphorus, the addition of farm manure contributed to an increase in TP.
3.6 Free air space (FAS)
Free air spaces allow air to diffuse in the environment and provide the oxygen needed by microorganisms. For the biodegradation of many materials by mixing and for providing adequate aerobic conditions, a FAS ratio of 20–30% is recommended [48]. At very high FAS values, thermophilic temperatures cannot be maintained during the composting process, and the biodegradation rate of volatile solids decreases [93, 94]. The FAS values for the reactors are shown in Fig. 6. In the study, the lowest FAS value (22.8%) was determined in the mixture formed with 80% treatment sludge (M1), while the highest (38.5%) was determined in the mixture formed with the addition of horse manure and treatment sludge (M4). Except for the M4 reactor, the FAS values were between or very close to the values in the literature. Since bulking agents directly affect the FAS values, these values can be found within the recommended ranges provided that the required amounts of bulking agents are used.
4 Conclusions
Co-composting animal manure with treatment sludge can provide a more sustainable solution for managing treatment sludge. In this study, the compostability of treatment sludge with the addition of different types of animal manures (cattle, pig, horse, and chicken) and sunflower stalk was investigated. Adding animal manure to the treatment sludge resulted in greater microbial activity, leading to higher temperatures in the reactors and greater organic matter losses. The maximum temperatures reached in the reactors were 62.7 °C (cattle), 62.2 °C (chicken), 60.2 °C (horse), 51.5 °C (pig), and 48 °C (only treatment sludge). Similarly, the highest organic matter loss occurred in the M2 reactor with cattle manure, while the lowest organic matter loss occurred in the M1 reactor without animal manure. When treatment sludge was composted alone, the composting efficiency was lower due to fewer microbial communities and less substrate. On the other hand, it was determined that better results were achieved in composting experiments with different types of animal manures as an additional substrate and inoculum added to the treatment sludge. The most successful composting process occurred in the reactor with added cattle manure (M2), followed by the reactors with added chicken (M5), horse (M4), and pig (M3) manure, respectively. The addition of animal manure increased the population of microorganisms responsible for carrying out the composting process and the substrate quantity, thereby enhancing composting efficiency. It might be possible to achieve even better results with different bulking agent, different animal manures, and different composting recipes, or after the maturation phase.
References
Tang DYY, Chew KW, Gentili FG et al (2023) Dechlorophyllization of microalgae biomass for the bioconversion into lipid-rich bioproducts. Ind Eng Chem Res 62:14478–14483. https://doi.org/10.1021/acs.iecr.3c00780
Saravanan A, Kumar PS, Mat Aron NS et al (2022) A review on bioconversion processes for hydrogen production from agro-industrial residues. Int J Hydrogen Energy 47:37302–37320. https://doi.org/10.1016/j.ijhydene.2021.08.055
Zhang S, Wang J, Jiang H (2021) Microbial production of value-added bioproducts and enzymes from molasses, a by-product of sugar industry. Food Chem 346:128860. https://doi.org/10.1016/j.foodchem.2020.128860
Chavan S, Yadav B, Atmakuri A et al (2022) Bioconversion of organic wastes into value-added products: a review. Bioresour Technol 344:126398. https://doi.org/10.1016/j.biortech.2021.126398
Aydın Temel F (2023) Evaluation of the influence of rice husk amendment on compost quality in the composting of sewage sludge. Bioresour Technol 373:128748. https://doi.org/10.1016/j.biortech.2023.128748
Liew C-S, Kiatkittipong W, Lim J-W et al (2021) Stabilization of heavy metals loaded sewage sludge: reviewing conventional to state-of-the-art thermal treatments in achieving energy sustainability. Chemosphere 277:130310. https://doi.org/10.1016/j.chemosphere.2021.130310
Havlin J, Tisdale SL, Nelson WL, Beaton JD (2014) Soil fertility and fertilizers: an introduction to nutrient management. Pearson
Şevik F, Tosun İ, Ekinci K (2018) The effect of FAS and C/N ratios on co-composting of sewage sludge, dairy manure and tomato stalks. Waste Manag 80:450–456. https://doi.org/10.1016/j.wasman.2018.07.051
Zhou C, Liu Z, Huang Z-L et al (2015) A new strategy for co-composting dairy manure with rice straw: addition of different inocula at three stages of composting. Waste Manag 40:38–43. https://doi.org/10.1016/j.wasman.2015.03.016
Su Y, Zhou L, Zhuo Q et al (2024) Microbial mechanisms involved in negative effects of amoxicillin and copper on humification during composting of dairy cattle manure. Bioresour Technol 399:130623. https://doi.org/10.1016/j.biortech.2024.130623
Urra J, Alkorta I, Lanzén A et al (2019) The application of fresh and composted horse and chicken manure affects soil quality, microbial composition and antibiotic resistance. Appl Soil Ecol 135:73–84. https://doi.org/10.1016/j.apsoil.2018.11.005
Fu J, Pan F, Song S et al (2013) Biodegradation of phthalic acid esters in sewage sludge by composting with pig manure and rice straw. Environ Earth Sci 68:2289–2299. https://doi.org/10.1007/s12665-012-1915-5
González D, Barrena R, Moral-Vico J et al (2024) Addressing the gaseous and odour emissions gap in decentralised biowaste community composting. Waste Manag 178:231–238. https://doi.org/10.1016/j.wasman.2024.02.042
Partanen P, Hultman J, Paulin L et al (2010) Bacterial diversity at different stages of the composting process. BMC Microbiol 10:94. https://doi.org/10.1186/1471-2180-10-94
Dias BO, Silva CA, Higashikawa FS et al (2010) Use of biochar as bulking agent for the composting of poultry manure: effect on organic matter degradation and humification. Bioresour Technol 101:1239–1246. https://doi.org/10.1016/j.biortech.2009.09.024
Gao X, Tan W, Zhao Y et al (2019) Diversity in the mechanisms of humin formation during composting with different materials. Environ Sci Technol 53:3653–3662. https://doi.org/10.1021/acs.est.8b06401
Gattupalli M, Dashora K, Javed Z, Tripathi GD (2024) Exploring garbage enzymes as novel biocatalyst for enhancing bioprocess performance in composting. Process Saf Environ Prot 188:73–80. https://doi.org/10.1016/j.psep.2024.05.080
Liu C, Li H, Ni J-Q et al (2024) Effect of municipal sludge-based biochar produced at different pyrolysis temperatures on humification and oxytetracycline degradation of pig manure composting. Sci Total Environ 906:167816. https://doi.org/10.1016/j.scitotenv.2023.167816
Shan G, Wei X, Li W et al (2024) Effect of aqueous phase from hydrothermal carbonization of sewage sludge on heavy metals and heavy metal resistance genes during chicken manure composting. J Hazard Mater 471:134398. https://doi.org/10.1016/j.jhazmat.2024.134398
Singh A, Sawant M, Herlekar M et al (2024) Assessing feasibility of sewage sludge composting in rotary drum reactor. Bioresour Technol 394:130219. https://doi.org/10.1016/j.biortech.2023.130219
Benito M, Masaguer A, Moliner A et al (2009) Dynamics of pruning waste and spent horse litter co-composting as determined by chemical parameters. Bioresour Technol 100:497–500. https://doi.org/10.1016/j.biortech.2008.06.005
Zhao XL, Li BQ, Ni JP, Xie DT (2016) Effect of four crop straws on transformation of organic matter during sewage sludge composting. J Integr Agric. https://doi.org/10.1016/S2095-3119(14)60954-0
Ingelmo F, Molina MJ, Soriano MD et al (2012) Influence of organic matter transformations on the bioavailability of heavy metals in a sludge based compost. J Environ Manage 95:S104–S109. https://doi.org/10.1016/j.jenvman.2011.04.015
Banegas V, Moreno JL, Moreno JI et al (2007) Composting anaerobic and aerobic sewage sludges using two proportions of sawdust. Waste Manag 27:1317–1327. https://doi.org/10.1016/j.wasman.2006.09.008
Yañez R, Alonso JL, Díaz MJ (2009) Influence of bulking agent on sewage sludge composting process. Bioresour Technol. https://doi.org/10.1016/j.biortech.2009.05.073
Komilis D, Evangelou A, Voudrias E (2011) Monitoring and optimizing the co-composting of dewatered sludge: a mixture experimental design approach. J Environ Manage 92:2241–2249. https://doi.org/10.1016/j.jenvman.2011.04.012
Meng L, Zhang S, Gong H et al (2018) Improving sewage sludge composting by addition of spent mushroom substrate and sucrose. Bioresour Technol 253:197–203. https://doi.org/10.1016/j.biortech.2018.01.015
Hanc A, Dume B, Hrebeckova T et al (2024) The fate of pharmaceuticals and personal care products during composting of sewage sludge. Sustain Chem Pharm 38:101498. https://doi.org/10.1016/j.scp.2024.101498
Uçaroğlu S, Alkan U (2016) Composting of wastewater treatment sludge with different bulking agents. J Air Waste Manag Assoc 66:288–295. https://doi.org/10.1080/10962247.2015.1131205
Cheng Q, Shen Y, Zhao S et al (2024) Study of the variation law of the airflow resistance and related physical parameters of the pile throughout compost process with cattle manure. Waste Manag 174:263–272. https://doi.org/10.1016/j.wasman.2023.11.032
Wen X, Chen M, Ma B et al (2024) Removal of antibiotic resistance genes during swine manure composting is strongly impaired by high levels of doxycycline residues. Waste Manag 177:76–85. https://doi.org/10.1016/j.wasman.2024.01.037
Zhang H, Ma L, Li Y et al (2024) Control of nitrogen and odor emissions during chicken manure composting with a carbon-based microbial inoculant and a biotrickling filter. J Environ Manage 357:120636. https://doi.org/10.1016/j.jenvman.2024.120636
Arias O, Viña S, Uzal M, Soto M (2017) Composting of pig manure and forest green waste amended with industrial sludge. Sci Total Environ 586:1228–1236. https://doi.org/10.1016/j.scitotenv.2017.02.118
Jalili M, Mokhtari M, Eslami H et al (2019) Toxicity evaluation and management of co-composting pistachio wastes combined with cattle manure and municipal sewage sludge. Ecotoxicol Environ Saf 171:798–804. https://doi.org/10.1016/j.ecoenv.2019.01.056
Rehman RA, Qayyum MF (2020) Co-composts of sewage sludge, farm manure and rock phosphate can substitute phosphorus fertilizers in rice-wheat cropping system. J Environ Manage 259:109700. https://doi.org/10.1016/j.jenvman.2019.109700
Hachicha S, Sellami F, Cegarra J et al (2009) Biological activity during co-composting of sludge issued from the OMW evaporation ponds with poultry manure—physico-chemical characterization of the processed organic matter. J Hazard Mater 162:402–409. https://doi.org/10.1016/j.jhazmat.2008.05.053
Awasthi MK, Pandey AK, Bundela PS et al (2016) Co-composting of gelatin industry sludge combined with organic fraction of municipal solid waste and poultry waste employing zeolite mixed with enriched nitrifying bacterial consortium. Bioresour Technol 213:181–189. https://doi.org/10.1016/j.biortech.2016.02.026
Li G, Li H, Leffelaar PA et al (2014) Characterization of phosphorus in animal manures collected from three (dairy, swine, and broiler) farms in China. PLoS ONE 9:e102698. https://doi.org/10.1371/journal.pone.0102698
APHA AWWA W (1998) Standart methods for the examination of water and wastewater, 20th editi. Copyright by American Public Health Association, Baltimore, USA
Diaz LF, De Bertoldi M, Bidlingmaier W (2007) Compost science and technology. Elsevier
Khalil AI, Hassouna MS, El-Ashqar HMA, Fawzi M (2011) Changes in physical, chemical and microbial parameters during the composting of municipal sewage sludge. World J Microbiol Biotechnol 27:2359–2369. https://doi.org/10.1007/s11274-011-0704-8
Barrington S, Choinière D, Trigui M, Knight W (2002) Effect of carbon source on compost nitrogen and carbon losses. Bioresour Technol 83:189–194. https://doi.org/10.1016/s0960-8524(01)00229-2
Adhikari BK, Barrington S, Martinez J, King S (2009) Effectiveness of three bulking agents for food waste composting. Waste Manag 29:197–203. https://doi.org/10.1016/j.wasman.2008.04.001
Rhoades JD (1982) Soluble salts. Methods Soil Anal: Part 2 Chem Microbiol Properties 10:167–179
McLean EO (1982) Soil pH and lime requirement. Methods Soil Anal: Part 2 Chem Microbiol Properties 12:199–224
Bremner JM, Mulvaney CS (1982) Nitrogen - total. Methods Soil Anal: Part 2 Chem Microbiol Properties 31:595–624
Madejón E, Jesús Díaz M, López R, Cabrera F (2002) New approaches to establish optimum moisture content for compostable materials. Bioresour Technol 85:73–78. https://doi.org/10.1016/S0960-8524(02)00030-5
Haug RT (1993) The practical handbook of compost engineering. Taylor \& Francis
Ezugworie FN, Igbokwe VC, Onwosi CO (2021) Proliferation of antibiotic-resistant microorganisms and associated genes during composting: an overview of the potential impacts on public health, management and future. Sci Total Environ 784:147191. https://doi.org/10.1016/j.scitotenv.2021.147191
Rao JN, Parsai T (2023) A comprehensive review on the decentralized composting systems for household biodegradable waste management. J Environ Manage 345:118824. https://doi.org/10.1016/j.jenvman.2023.118824
Wu S, Shen Z, Yang C et al (2017) Effects of C/N ratio and bulking agent on speciation of Zn and Cu and enzymatic activity during pig manure composting. Int Biodeterior Biodegradation 119:429–436. https://doi.org/10.1016/j.ibiod.2016.09.016
Ge M, Shen Y, Ding J et al (2022) New insight into the impact of moisture content and pH on dissolved organic matter and microbial dynamics during cattle manure composting. Bioresour Technol 344:126236. https://doi.org/10.1016/j.biortech.2021.126236
Ahmad R, Jilani G, Arshad M et al (2007) Bio-conversion of organic wastes for their recycling in agriculture: an overview of perspectives and prospects. Ann Microbiol 57:471–479. https://doi.org/10.1007/BF03175343
Li M-X, He X-S, Tang J et al (2021) Influence of moisture content on chicken manure stabilization during microbial agent-enhanced composting. Chemosphere 264:128549. https://doi.org/10.1016/j.chemosphere.2020.128549
Uçaroǧlu S (2014) Use of sunflower stalks as a bulking agent in sewage sludge composting. Fresenius Environ Bull 23:1302–1308
Nikaeen M, Nafez AH, Bina B et al (2015) Respiration and enzymatic activities as indicators of stabilization of sewage sludge composting. Waste Manag 39:104–110. https://doi.org/10.1016/j.wasman.2015.01.028
Ji Z, Zhang L, Liu Y et al (2023) Evaluation of composting parameters, technologies and maturity indexes for aerobic manure composting: a meta-analysis. Sci Total Environ 886:163929. https://doi.org/10.1016/j.scitotenv.2023.163929
Kulikowska D (2016) Kinetics of organic matter removal and humification progress during sewage sludge composting. Waste Manag 49:196–203. https://doi.org/10.1016/j.wasman.2016.01.005
Putranto A, Chen XD (2017) A new model to predict diffusive self-heating during composting incorporating the reaction engineering approach (REA) framework. Bioresour Technol 232:211–221. https://doi.org/10.1016/j.biortech.2017.01.065
Pandey PK, Cao W, Wang Y et al (2016) Simulating the effects of mesophilic anaerobic and aerobic digestions, lagoon system, and composting on pathogen inactivation. Ecol Eng 97:633–641. https://doi.org/10.1016/j.ecoleng.2016.10.047
USEPA (1993) Standards for the use and disposal of sewage; final rules. Federal Register, Washington
Yin J, Xie M, Yu X et al (2024) A review of the definition, influencing factors, and mechanisms of rapid composting of organic waste. Environ Pollut 342:123125. https://doi.org/10.1016/j.envpol.2023.123125
Abouelwafa R, AitBaddi G, Souabi S et al (2008) Aerobic biodegradation of sludge from the effluent of a vegetable oil processing plant mixed with household waste: physical–chemical, microbiological, and spectroscopic analysis. Bioresour Technol 99:8571–8577. https://doi.org/10.1016/j.biortech.2008.04.007
Zhan J, Han Y, Xu S et al (2022) Succession and change of potential pathogens in the co-composting of rural sewage sludge and food waste. Waste Manag 149:248–258. https://doi.org/10.1016/j.wasman.2022.06.028
Tan Y, Cao X, Chen S et al (2023) Antibiotic and heavy metal resistance genes in sewage sludge survive during aerobic composting. Sci Total Environ 866:161386. https://doi.org/10.1016/j.scitotenv.2023.161386
Spencer JL, Van Heyst BJ (2013) Effect of different intermediate amendments on pH and ammonia emissions of composted poultry mortalities. J Appl Poult Res 22:700–714. https://doi.org/10.3382/japr.2012-00632
Mao H, Zhang H, Fu Q et al (2019) Effects of four additives in pig manure composting on greenhouse gas emission reduction and bacterial community change. Bioresour Technol 292:121896. https://doi.org/10.1016/j.biortech.2019.121896
Jamroz E, Bekier J, Medynska-Juraszek A et al (2020) The contribution of water extractable forms of plant nutrients to evaluate MSW compost maturity: a case study. Sci Rep 10:12842. https://doi.org/10.1038/s41598-020-69860-9
Negi S, Mandpe A, Hussain A, Kumar S (2020) Collegial effect of maggots larvae and garbage enzyme in rapid composting of food waste with wheat straw or biomass waste. J Clean Prod 258:120854. https://doi.org/10.1016/j.jclepro.2020.120854
Sundberg C, Smårs S, Jönsson H (2004) Low pH as an inhibiting factor in the transition from mesophilic to thermophilic phase in composting. Bioresour Technol 95:145–150. https://doi.org/10.1016/j.biortech.2004.01.016
Ravindran B, Awasthi MK, Karmegam N et al (2022) Co-composting of food waste and swine manure augmenting biochar and salts: nutrient dynamics, gaseous emissions and microbial activity. Bioresour Technol 344:126300. https://doi.org/10.1016/j.biortech.2021.126300
Jain MS, Jambhulkar R, Kalamdhad AS (2018) Biochar amendment for batch composting of nitrogen rich organic waste: effect on degradation kinetics, composting physics and nutritional properties. Bioresour Technol 253:204–213. https://doi.org/10.1016/j.biortech.2018.01.038
Lasaridi K, Protopapa I, Kotsou M et al (2006) Quality assessment of composts in the Greek market: the need for standards and quality assurance. J Environ Manage 80:58–65. https://doi.org/10.1016/j.jenvman.2005.08.011
López R, Antelo J, Silva AC et al (2021) Factors that affect physicochemical and acid-base properties of compost and vermicompost and its potential use as a soil amendment. J Environ Manage 300:113702. https://doi.org/10.1016/j.jenvman.2021.113702
Chan SS, Khoo KS, Chew KW et al (2022) Recent advances biodegradation and biosorption of organic compounds from wastewater: microalgae-bacteria consortium - a review. Bioresour Technol 344:126159. https://doi.org/10.1016/j.biortech.2021.126159
Li L, Guo X, Zhao T, Li T (2021) Green waste composting with bean dregs, tea residue, and biochar: effects on organic matter degradation, humification and compost maturity. Environ Technol Innov 24:101887. https://doi.org/10.1016/j.eti.2021.101887
Zhong X-Z, Ma S-C, Wang S-P et al (2018) A comparative study of composting the solid fraction of dairy manure with or without bulking material: Performance and microbial community dynamics. Bioresour Technol 247:443–452. https://doi.org/10.1016/j.biortech.2017.09.116
Jiang Z, Lu Y, Xu J et al (2019) Exploring the characteristics of dissolved organic matter and succession of bacterial community during composting. Bioresour Technol 292:121942. https://doi.org/10.1016/j.biortech.2019.121942
Han W, Clarke W, Pratt S (2014) Composting of waste algae: a review. Waste Manag 34:1148–1155. https://doi.org/10.1016/j.wasman.2014.01.019
Sweeten JM, Auvermann BW (2008) Composting manure and sludge. Agrilife Extension, Texas FARMER Collect 479:6–8. https://oaktrust.library.tamu.edu/bitstream/handle/1969.1/87650/pdf_346.pdf?sequence=1&isAllowed=y
Ren J, Deng L, Li C et al (2021) Effects of added thermally treated penicillin fermentation residues on the quality and safety of composts. J Environ Manage 283:111984. https://doi.org/10.1016/j.jenvman.2021.111984
Nasini L, De Luca G, Ricci A et al (2016) Gas emissions during olive mill waste composting under static pile conditions. Int Biodeterior Biodegradation 107:70–76. https://doi.org/10.1016/j.ibiod.2015.11.001
Wang X, Selvam A, Wong JWC (2016) Influence of lime on struvite formation and nitrogen conservation during food waste composting. Bioresour Technol 217:227–232. https://doi.org/10.1016/j.biortech.2016.02.117
Song B, Manu MK, Li D et al (2021) Food waste digestate composting: feedstock optimization with sawdust and mature compost. Bioresour Technol 341:125759. https://doi.org/10.1016/j.biortech.2021.125759
de Guardia A, Mallard P, Teglia C et al (2010) Comparison of five organic wastes regarding their behaviour during composting: part 2, nitrogen dynamic. Waste Manag 30:415–425. https://doi.org/10.1016/j.wasman.2009.10.018
Sanchez-Monedero MA, Cayuela ML, Roig A et al (2018) Role of biochar as an additive in organic waste composting. Bioresour Technol 247:1155–1164. https://doi.org/10.1016/j.biortech.2017.09.193
Zhang D, Luo W, Li Y et al (2018) Performance of co-composting sewage sludge and organic fraction of municipal solid waste at different proportions. Bioresour Technol 250:853–859. https://doi.org/10.1016/j.biortech.2017.08.136
Karunanithi R, Szogi AA, Bolan N, et al (2015) Chapter three - Phosphorus recovery and reuse from waste streams. In: Sparks DL (ed). Academic Press, pp 173–250
Zhang T, Wu X, Shaheen SM et al (2021) Effects of microorganism-mediated inoculants on humification processes and phosphorus dynamics during the aerobic composting of swine manure. J Hazard Mater 416:125738. https://doi.org/10.1016/j.jhazmat.2021.125738
Rehman MZ, Rizwan M, Ali S et al (2016) Contrasting effects of biochar, compost and farm manure on alleviation of nickel toxicity in maize (Zea mays L.) in relation to plant growth, photosynthesis and metal uptake. Ecotoxicol Environ Saf 133:218–225. https://doi.org/10.1016/j.ecoenv.2016.07.023
Gagnon B, Demers I, Ziadi N et al (2012) Forms of phosphorus in composts and in compost-amended soils following incubation. Can J Soil Sci 92:711–721. https://doi.org/10.4141/cjss2012-032
Wei Y, Zhao Y, Xi B et al (2015) Changes in phosphorus fractions during organic wastes composting from different sources. Bioresour Technol 189:349–356. https://doi.org/10.1016/j.biortech.2015.04.031
Michel FC, Pecchia JA, Rigot J, Keener HM (2004) Mass and nutrient losses during the composting of dairy manure amended with sawdust or straw. Compost Sci Util 12:323–334. https://doi.org/10.1080/1065657X.2004.10702201
Külcü R (2015) Determination of the relationship between FAS values and energy consumption in the composting process. Ecol Eng 81:444–450. https://doi.org/10.1016/j.ecoleng.2015.04.026
Funding
Open access funding provided by the Scientific and Technological Research Council of Türkiye (TÜBİTAK).
Author information
Authors and Affiliations
Contributions
S.U. supervised the study and wrote the paper. B.O. conducted research methodology, experiments, and drafting of the manuscript. All the authors read the manuscript and approved to submit it.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ucaroglu, S., Ozbek, B. Bioconversion of organic wastes: treatment sludges, animal manures, and agricultural wastes. Biomass Conv. Bioref. (2024). https://doi.org/10.1007/s13399-024-05883-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13399-024-05883-w