Abstract
Oriental persimmon, Diospyros kaki Thunb. (Ericales: Ebenaceae), is a generally diclinous tree. Because the representative cultivar, ‘Fuyu’, sets nearly exclusively female flowers and has low parthenocarpic ability, it requires pollen from other cultivars for pollination. Here, we identified the most important pollinator of D. kaki in hilly orchards surrounded by secondary forest in Hiroshima, southwestern Japan. To estimate the contribution of each flower visitor to pollination, we used an index of potential pollen transport, namely the rate of visitation per female flower multiplied by the estimated number of pollen grains on the body surface. Among nine functional groups, the value of this index was greatest for bumblebees followed by small bees, whereas that for honeybees was low because of the relatively few Diospyros pollen grains on the body surface. Bombus ardens ardens Smith (Hymenoptera: Apidae) was the predominant bumblebee species in terms of both monitored frequency of visits by insects and number collected for identification. We also counted the fruit set and seed number of ‘Fuyu’ fruit in various pollination treatments using exclusion mesh to assess the effectiveness of insects of different sizes. Bombus ardens ardens was the most important pollinator and small bees were the second most important.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Oriental (or Japanese) persimmon, Diospyros kaki Thunb. (Ericales: Ebenaceae) is a familiar fruit in Japan. This species is native to China and has been cultivated for over 2500 years in East Asia (Zhang 2008). It is also grown commercially in countries such as Spain, Brazil, and Israel. In 2017, Japan produced 225,300 t of D. kaki fruit—3.9% of the world’s production—making it the fourth-largest producer after China, Spain, and Korea (FAOSTAT 2017). The total domestic area of orchards in Japan was 20,300 ha in 2017, the third largest after citrus and apple (Japanese Government Statistics 2017). Oriental persimmon is produced throughout Japan, except in Hokkaido, the northern end of Honshu, the southern end of Kyushu, and the Nansei Islands.
In D. kaki fruit production, the importance of pollination depends on the cultivar. Trees of D. kaki are generally diclinous, but the representative cultivar, ‘Fuyu’, sets nearly exclusively female flowers (Yonemori et al. 1992) and requires pollen from other cultivars for pollination owing to its low parthenocarpic ability. Pollinizer cultivars, such as ‘Zenjimaru’, are interplanted with the main cultivars or grafted onto their stems. In addition, hives of the European honeybee Apis mellifera L. (Hymenoptera: Apidae) have been placed as necessary in D. kaki orchards because of this insect’s expected role as a pollinator (Yamamura et al. 1989). The degree of parthenocarpy depends on the cultivar; for example, ‘Fuyu’ sets seedless fruit when no pollen is available, but its ability to set these unpollinated fruit is highly variable (Yamada et al. 1987). Pollination of ‘Fuyu’ decreases physiological fruit drop and stabilizes fruit set (Kajiura 1941; Yamada et al. 1987). Fruit drop in the early development stage, which is caused by many factors, including genetic, environmental, and physiological ones (George et al. 1997), could be a primary cause of poor crop yield, especially in young orchards of D. kaki. In addition, the seeded fruit of ‘Maekawa-Jiro’ is slightly bigger, and of better quality, than seedless fruit of this cultivar (Hasegawa and Nakajima 1990a, b). Fruit growers, therefore, optimize the ratio of pollinizer to recipient, to produce fruit containing an adequate number of seeds for the best fruit set and fruit size, when they cultivate D. kaki cultivars with moderate or low parthenocarpy.
Careful investigations are needed to identify the insect species pollinators of D. kaki among the various flower-visiting insects, because frequent visits by a species do not prove that the species makes a large contribution to pollination (Klein et al. 2003). Wild insects visiting the flowers of D. kaki have been observed in Japan; examples are Bombus ignitus Smith (Hymenoptera: Apidae) and Halictus subopacus Smith (Hymenoptera: Halictidae) in Tokyo (Asami and Chow 1936); the Japanese honeybee Apis cerana japonica Radoszkowski (Hymenoptera: Apidae) in Shizuoka (Yokozawa 1951); and Andrena sabopacus Smith (Hymenoptera: Andrenidae) in Kyoto (Tokunaga et al. 1959). However, because few studies have investigated the degree of each species’ contribution to the pollination of D. kaki, the relative importance of each pollinator remains unclear. Therefore, even though we depend on these pollinators for the production of D. kaki, we are uncertain which insect species are responsible for pollination. Enhancing pollination should be effective in decreasing fruit drop in these cultivars. Identifying the contributing pollinators would also give us opportunities to better conserve the landscapes that are their nesting habitats.
We surmised that there were no major differences among the cultivars of D. kaki in terms of attractiveness to flower visitors. Although the timing of blooming differs slightly among cultivars, there are no noticeable differences in the appearance of the flowers. In addition, because the structure of the campanulate female flowers is relatively simple, with four widely opened petals (McGregor 1976), the flowers’ nectaries seem to be accessible to a variety of flower visitors, regardless of proboscis length. However, the details of differences among cultivars in other morphological traits that attract insects, such as nectar guide or flower fragrance, are unknown and need to be studied elsewhere.
Our main objective here was to identify the most important pollinator of D. kaki. We performed field experiments in hilly orchards where A. mellifera had not been introduced. First, we assessed the visitation frequency and species assemblage of insects on D. kaki flowers in the field. Second, to estimate each insect species’ contribution to pollination, we calculated an index of potential pollen transport (PPT), which was a product of the rate of visitation to each female flower multiplied by the number of pollen grains on the body surface of an individual. Third, we compared the fruit-set ratio and number of seeds among pollination treatments using mesh bags to evaluate the pollination effectiveness of insects with different body sizes.
Materials and methods
Study site
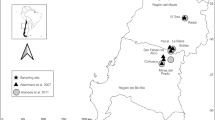
Field experiments were conducted in two orchards of the Institute of Fruit Tree and Tea Science, NARO, in Hiroshima, Japan (34°19.5′N, 132°49.3′E, 130 m above sea level [asl]). The 10-year mean monthly temperature ranged from 5.4 °C in January to 26.7 °C in August (17.4 °C in May), and the mean annual precipitation was 1304 mm, recorded at a nearby automated meteorological data acquisition point of the Japan Meteorological Agency (34°19.8′N, 132°58.9′E, 5 m asl) from 2009 to 2018. The institute’s orchards, in which trees of Oriental persimmon, grape, and kiwifruit have been planted, are located on a hill enclosed by secondary forest surrounded by rice paddy fields and semi-natural grasslands (Fig. S1). In Orchard A, of approximately 1700 m2, 41 D. kaki trees (‘Fuyu’, 16; ‘Soshu’, 15; ‘Taishuu’, 6; ‘Yubeni’, 2; and ‘Hiratanenashi’, 2) have been planted. The male-flower-rich cultivar ‘Zenjimaru’ was grafted as a pollenizer onto five ‘Fuyu’ trees and three ‘Soshu’ trees. In Orchard B, of approximately 1800 m2, 51 trees (‘Taishuu’, 14; ‘Fuyu’, 13; ‘Hiratanenashi’, 8; ‘Shinsyuu’, 7; ‘Soshu’, 4; ‘Taiten’, 3; and ‘Zenjimaru’, 2) have been planted. No domesticated pollinators, such as A. mellifera, have been introduced. The peak blooming dates (when 80% of flowers had bloomed) of ‘Fuyu’ were 25 May in 2017 and 21 May in 2018. Flower-visiting insects were monitored in 2018 and captured in both 2017 and 2018.
Monitoring of flower-visiting insects
Visits by insects to ‘Fuyu’, ‘Taishuu’, ‘Soshu’, and grafted ‘Zenjimaru’ were recorded in Orchards A and B on sunny and cloudy days from 16 to 24 May 2018. Each monitoring plot measured approximately 2 m × 2 m; this size was chosen because it was considered to be the maximum that a researcher could see at a glance. Tree height was 3–4 m. We set each monitoring plot at eye level, because it was difficult to monitor the flowers on the upper branches. After the numbers of male and female flowers in a plot had been recorded, visits by insects to the flowers in the plot were monitored for 15–30 min. Every flower-visiting individual was observed and classified visually into one of the nine functional groups: bumblebees, honeybees, carpenter bees, medium-sized bees, small bees, wasps (including wasp-like bees), syrphid flies, other dipterans, and beetles. We also recorded individuals at the species level if they were identifiable by appearance: Bombus ardens ardens Smith and Bombus hypocrita hypocrita Pérez (Hymenoptera: Apidae) as bumblebees, A. cerana japonica as honeybees, Xylocopa appendiculata circumvolans Smith (Hymenoptera: Apidae) as carpenter bees, and Gametis jucunda Faldermann (Coleoptera: Scarabaeidae) as beetles. Consecutive flower visitations to different flowers by a single individual in a monitoring plot were counted independently. The sex of visited flowers was also recorded. Ants on flowers were not counted; they were found only in female flowers, sucking nectar (data not shown).
Insect species assemblage
In 30-min periods every 2 h between 07:00 and 17:00 h on 26, 29, and 30 May 2017, we used an insect net to collect a total of 102 insect individuals staying on, or flying near, the flowers of D. kaki ‘Fuyu’ and ‘Zenjimaru’ in Orchards A and B. On 17, 21, and 24 May 2018, we collected a total of 371 individuals using another collection method in which a researcher moved the aperture of a 5-mL vial toward an insect that had plunged its head into the bottom of a flower of ‘Fuyu’, ‘Taishuu’, ‘Zenjimaru’, or ‘Hiratanenashi’. We selected the latter method as a way of capturing regular flower visitors—not accidental visitors and passers-by across the orchards. To capture carpenter bees, we used a 50-mL tube. After 3–5 mL of ethanol had been added to each tube, which was given an identification number, the insect samples were stored at room temperature until identification. Flower sex was also recorded during the 2018 insect collections.
To count Diospyros pollen grains on insect body surfaces, at the same time as the monitoring described above was being performed, 53 individuals visiting D. kaki flowers on trees in Orchards A and B were captured individually into 5-mL vials. Each tube containing a captured insect was immediately cooled on ice in a cooler bag; this was effective in preventing decomposition of the pollen loads on the hind legs, because the insect soon ceased to move on the ice. The insects in the tubes were stored at − 30 °C until pollen-counting analysis in the laboratory (see below).
Identification of flower-visiting insects
We morphologically identified all insects captured (526 individuals in total). To check the morphological identification, DNA barcoding was employed as necessary (Table S1). First, we confirmed the accuracy of our morphological discrimination between B. ardens ardens and B. ignitus using DNA barcoding of three and one samples, respectively. Then, in addition to these Bombus species, A. cerana japonica, Eucera nipponensis (Pérez) (Hymenoptera: Apidae), G. jucunda, and X. appendiculata circumvolans were conclusively identified on the basis of their morphologies. For the other species, morphological identification of all individuals was checked by molecular identification using DNA barcoding. If the former was supported by the latter, we adopted the former as conclusive identification (62 individuals). If not, the morphological identification was redone (Scarabaeidae, two individuals; Zygaenidae, one individual), or the morphological identification was kindly verified by Prof. Ryoichi Miyanaga of Shimane University or Dr. Ryuki Murao of Regional Environmental Planning Co., Ltd. (Andrenidae and Halictidae, nine individuals).
DNA from one of the middle legs was extracted using a DNeasy Blood and Tissue Kit (Qiagen, Valencia, CA, USA) in accordance with the manufacturer’s protocol. DNA barcoding analysis was conducted using a segment of mitochondrial cytochrome c oxidase 1 (CO1). The primer combination used for PCR amplification and sequencing of CO1 in all except the Andrenidae and Halictidae was LCO1490 (forward): 5′-GGTCAACAAATCATAAAGATATTGG-3′ and HCO2198 (reverse): 5′-TAAACTTCAGGGTGACCAAAAAATCA-3′ (Folmer et al. 1994). The amplification reaction contained 0.08 μL of Ex Taq polymerase (TaKaRa Bio, Shiga, Japan; 5 U/μL), 1.6 μL of Ex Taq buffer (Mg2+ free), 1.3 μL of MgCl2 (25 mM), 1.3 μL of dNTP mixture (2.5 mM each), 0.5 μL of DNA template, 0.5 μL each of forward and reverse primers (10 μM), and purified water up to 16.6 μL. PCR cycles consisted of an initial denaturation step for 3 min at 94 °C; 30 cycles of denaturation (30 s at 94 °C), annealing (30 s at 45 °C), and extension (1 min 50 s at 72 °C); and a final extension at 72 °C for 7 min.
For the Andrenidae and Halictidae, another primer combination was used for PCR amplification and sequencing of CO1: BarbeeF (forward): 5′-CAACAAATCATAAAAATATTGG-3′ (Françoso and Arias 2013) and MtD9 (reverse): 5′-CCCGGTAAAATTAAAATATAAACTTC-3′ (Simon et al. 1994). The amplification reaction was constructed in the same manner as described above. PCR cycles consisted of an initial denaturation step for 5 min at 94 °C; 35 cycles of denaturation (1 min at 94 °C), annealing (1 min 20 s at 40 °C), and extension (2 min at 64 °C); and a final extension at 64 °C for 10 min.
The PCR products obtained were checked by electrophoresis on 2% (w/v) agarose gel, treated with ExoSAP-IT (USB Corp., Cleveland, OH, USA), and directly sequenced in both directions in an ABI 3130XL genetic analyzer (Applied Biosystems, Foster City, CA, USA) with a Big Dye Terminator Cycle Sequencing Ready Reaction Kit (Applied Biosystems) using the same primers as were used for PCR. Searches for similar sequences were performed using an identification engine searching for “All Barcode Records on BOLD” (http://www.boldsystems.org/index.php/IDS_OpenIdEngine) in BOLD (the Barcode of Life Data Systems) or the Basic Local Alignment Search Tool (BLAST; https://blast.ncbi.nlm.nih.gov/Blast.cgi) in GenBank (NCBI, National Centre for Biotechnology Information). Identifications at the species level were made on the basis of highly similar best matches (> 99% identity). The sequences were deposited in the DNA Data Bank of Japan (DDBJ accession numbers LC477224 to LC477292 and LC490353 to LC490355).
DNA barcoding of pollen loads
To identify flower sources at the species level, we used DNA barcoding to analyze the pollen loads isolated from the insects. This was necessary because, at the study site, species closely related to D. kaki, including D. lotus L., D. oleifera Cheng, D. rhombifolia Hemsl., and D. virginiana L. (Ericales: Ebenaceae), had been planted as genetic resources. It was, thus, possible that the insects we captured would have touched the anthers of these species. Because the Diospyros pollen grains were unidentifiable at the species level by our morphological identification, pollen grains of these Diospyros species could have been present in the samples analyzed.
Twenty-two individuals of the 53 that were captured for counting of pollen grains on the body surface had pollen loads on their hind legs. The pollen load was removed from one of the hind legs of each individual and DNA was extracted using a MightyPrep reagent for DNA (TaKaRa Bio, Shiga, Japan) in accordance with the manufacturer’s protocol. DNA barcoding analysis was conducted using the trnL intron and trnL–trnF intergenic spacer of chloroplast DNA (trnL–trnF). The primer combination used for PCR amplification and sequencing of trnL–trnF was trnL-c (forward), 5′-CGAAATCGGTAGACGCTACG-3′, and trnF-f (reverse), 5′-ATTTGAACTGGTGACACGAG-3′ (Taberlet et al. 1991). The amplification reaction contained 0.2 μL of Tks Gflex DNA Polymerase (TaKaRa Bio, 1.25 U/μL), 5.0 μL of 2 × Gflex PCR buffer (TaKaRa Bio; Mg2+, dNTP plus), 1.0 μL of DNA template, 0.2 μL each of forward and reverse primers (10 μM), and purified water up to 10.0 μL. PCR cycles consisted of an initial denaturation step for 1 min at 94 °C; 35 cycles of denaturation (10 s at 98 °C), annealing (15 s at 52 °C), and extension (1 min at 68 °C); and a final extension at 68 °C for 10 min. The PCR products obtained were checked by electrophoresis on 2% (w/v) agarose gel, purified by using Agencourt AMPure XP (Beckman Coulter, Brea, CA, USA) in accordance with the manufacturer’s protocol, and directly sequenced in both directions in an ABI 3130XL genetic analyzer with a Big Dye Terminator Cycle Sequencing Ready Reaction Kit using the same primers as were used for PCR.
Molecular identification of the amplified sequences was performed by reconstructing a phylogenetic tree for the sequences obtained, along with species with high levels of identity that were retrieved from the GenBank/NCBI database using the BLAST search. The amplified sequences and similar sequences collected from the database were aligned using the ClustalW that was included within MEGA7 (Kumar et al. 2016). Phylogenetic relationships were analyzed using maximum likelihood based on the Tamura–Nei model (Tamura and Nei 1993). The maximum likelihood analyses were performed with MEGA7. The sequences were deposited in the DDBJ (accession numbers LC466582 to LC466584 and LC494287 to LC494301).
Counting of pollen grains
We estimated the number of Diospyros pollen grains on the body surfaces of 53 insect individuals captured on flowers. First, we cut away each sample’s hind legs using scissors to exclude pollen loads, which are considered to be useless for pollination. Then, each body sample was put into a vial with 0.4 M sucrose solution (1.0–6.0 mL, depending on the body size). By shaking the vial tube gently, we disengaged the pollen grains from the body surface and collected them in the sucrose solution (Nikkeshi et al. 2016). We confirmed that only a trace of pollen grains remained on the body surface after this washing procedure (data not shown). Each insect sample was transferred to another vial with 3 mL of ethanol and stored until identification. We then removed 10 μL of solution from the vial and used a microscope (Eclipse E200; Nikon, Tokyo, Japan) to count the number of pollen grains suspended in the solution. We performed this procedure five times and then calculated the average number of pollen grains in 10 μL and the total number of pollen grains on the body surface in accordance with the initial solution volume. In performing these calculations, we did not distinguish insect samples captured on male flowers from those captured on female flowers, because consecutive visits from male to female flowers, and vice versa, were observed during the monitoring in the orchards (data not shown). In the “wasps” functional group, this pollen-counting procedure was performed on only two individuals, which were the only ones captured.
Potential pollen transport
We defined PPT as a product of the average frequency of visitation to each female flower per hour by members of a functional group and the total number of Diospyros pollen grains on an individual’s body surface. We surmised that the PPT value would be closely correlated with the number of pollen grains deposited on a stigma by members of a functional group. We, therefore, expected that the number of pollen grains deposited on a stigma would increase as the value increased.
Pollination experiment
We performed a pollination experiment in which female flower buds in Orchard A were covered with bags with different sizes of mesh, or left uncovered. We compared the resulting fruit set and the number of seeds among treatments. There were six treatments, including three controls: (1) bagging with unwoven cloth bag (polypropylene, white), which prevented insect access, resulting in no pollination (NP); (2) bagging with a 2-mm-mesh bag (2-M; polyethylene, white), which allowed access by minute insects; (3) bagging with a 3.5-mm-mesh bag (3.5-M; polyethylene, white), which prevented access by insects of a size equal to, or greater than, that of honeybees (see Table S2); (4) open pollination (OP) as a control, which did not necessarily result in pollination; (5) hand-pollination followed by enveloping of the flower in a paper bag (HPB) as a control, which evaluated the effect of hand-pollination; and (6) hand-pollination followed by open pollination (HPO) as a control to evaluate potential maximum pollination. The flower buds were thinned beforehand to a ratio of one bud to about 13 leaves. We selected five ‘Fuyu’ trees (which had been planted between 1989 and 1996) adjacent to at least one tree onto which ‘Zenjimaru’ had been grafted and set up five replications of each treatment on a single tree. Thus, each treatment had 25 replications and the total number of female flowers used in the experiment was 150. For the HPB and HPO treatments, the flower buds were covered with paper bags until treatment to prevent insect pollination.
For hand-pollination (HPB and HPO treatments), we used pollen collected from the male flower buds of ‘Zenjimaru’. Before treatment, male flower buds from which the sepals and petals had been removed were dried at 25 °C overnight. On 22 May 2018, we attached pollen to the stigma with the tip of an index finger after dipping the fingertip in the pollen sample collected in a Petri dish. After hand-pollination, the flowers were either covered in a paper bag (HPB treatment) or left open (HPO treatment).
All bags were placed on 22 May and removed on 6 June 2018. Before harvest, seed development was pre-checked on 20 July 2018 in fruit that was not used in the pollination experiments (Fig. S2). Fruit set in each pollination treatment was counted on 1 August 2018, when physiological fruit drop in the early development stage had ceased. Two tags in the HPB treatment group had been lost by harvest of the immature fruit on 1 August 2018 (so n = 23 for the HPB treatment and n = 25 for the other treatments). Diospyros kaki forms, at most, eight seeds per fruit. The number of seeds was counted in each immature fruit harvested. The sequence of pollination experiments was, therefore, performed from 22 May to 1 August 2018.
Statistical analysis
We used a generalized linear model (GLM) to examine whether the number of pollen grains on the body surfaces of individuals differed among six of the nine visitor functional groups: because only two individual “wasps” were available for counting pollen grains, this group was excluded from the statistical analysis. In the GLM, each functional group was an explanatory variable, and the number of deposited pollen grains was the response variable. Family was the negative binomial. If a significant difference was detected, Tukey’s post hoc test was applied to test for significant differences in any pairs of functional groups. Similarly, to examine whether the fruit-set ratio and the number of seeds differed among pollination treatments, we used a generalized linear mixed model (GLMM), in which we set fruit set and the number of seeds as response variables (family = binomial), pollination treatment as explanatory variable, and tree ID as a random effect. If a significant difference was detected, Tukey’s post hoc test was applied to any pairs of pollination treatments. All statistical analyses were performed with the free statistical software R ver. 3.5.1 (R Development Core Team 2018). We used the glht function in the multcomp package (Hothorn et al. 2008) for the two Tukey tests. To assess the significance of the coefficients, we used Wald’s test with the GLM and the GLMM function in the glmmADMB package (Skaug et al. 2016).
Results
Monitoring of flower-visiting insects
We observed a total of 1520 visits by insects in nine functional groups (Table 1). The total number of visits to male flowers (1407) was far greater than that to female flowers (113). Four functional groups made more than one visit to flowers of both sexes. The greatest number of visits was made by bumblebees; two bumblebee species, B. ardens ardens and B. hypocrita hypocrita, were identifiable by their appearance. Honeybees identifiable as A. cerana japonica, carpenter bees identifiable as X. appendiculata circumvolans, and small bees were the other three groups found on the flowers of both sexes. Unlike A. cerana japonica, A. mellifera was not observed as a visitor to D. kaki flowers. Visits by medium-sized bees, syrphid flies, other dipterans, and beetles identifiable as G. jucunda were recorded only on male flowers. Wasps (including wasp-like bees) were found only on female flowers.
Insect species assemblage
Insect species were identified morphologically and, if necessary, also by confirmation using CO1 DNA barcoding (Tables 2, S1). Most bumblebees were identified as B. ardens ardens; this was similar to the monitoring result (Table 1). All honeybee individuals were identified as A. cerana japonica. Therefore, no individual of A. mellifera was observed in either the monitoring or the collection of flower visitors (Tables 1, 2). Eucera nipponensis, a medium-sized bee, was caught on both male and female flowers (Table 2); whereas, no medium-sized bees were monitored on female flowers (Table 1). Among the small bees, nine species in three families were identified: Andrena opacifovea Hirashima, A. parathoracica Hirashima (Hymenoptera: Andrenidae); Hylaeus sp. (Hymenoptera: Colletidae); and Lasioglossum hoffmanni (Strand), L. japonicum (Dalla Torre), L. mutilum (Vachal), L. occidens (Smith), L. proximatum (Smith), and L. scitulum (Smith) (Hymenoptera: Halictidae). In the Hymenoptera, almost all of the 320 individuals caught were females, the exception being a male X. appendiculata circumvolans caught in 2018. We captured insects in two consecutive years. In both years, the species that was captured in the largest number was B. ardens ardens (43 individuals, 42.2% of the total in 2017; 162 individuals, 43.7% in 2018), followed by G. jucunda (11.8% in 2017; 36.1% in 2018) and A. cerana japonica (8.8% in 2017; 5.9% in 2018).
DNA barcoding of pollen loads
To identify at the species level the Diospyros pollen on the body surfaces of flower-visiting insects, we analyzed the pollen loads by using DNA barcoding. Of 22 pollen loads removed from insects captured on D. kaki flowers, 15 samples, which were removed from 11 individuals of B. ardens ardens, two of E. nipponensis, and one each of L. proximatum and X. appendiculata circumvolans, had an identical sequence of the DNA barcoding region trnL–trnF. Because the sequence of these 15 samples was clustered in the clade identical to D. kaki upon reconstruction of a phylogenetic tree (Fig. S3a), they were identified as D. kaki. The remaining seven samples yielded sequences identifiable as that of kiwifruit Actinidia deliciosa (A.Chev.) C.F.Liang & A.R.Ferguson (Ericales: Actinidiaceae), Toxicodendron sp. (Sapindales: Anacardiaceae), or Rosaceae sp. (Rosales) (one sample each; Figs. S3b–S3d), no product from PCR (two samples), or overlaid chromatograms of nucleotide sequences, which seemingly consisted of pollen grains of more than two plant species (two samples). Pollen grains of other Diospyros species were not detected in the analysis.
Counting of pollen grains
The average numbers of Diospyros pollen grains on the body surfaces of bumblebees, carpenter bees, medium-sized bees, small bees, and beetles were approximately 10,000 or more (Table 3). In contrast, the average on honeybees was less than 400. The average number of Diospyros pollen grains on the body surfaces of individual insects differed significantly among six functional groups (GLM, type 2; df = 5, χ2 = 27.985, p < 0.001). Subsequent Tukey’s post hoc tests detected significant differences in the average number of pollen grains on the body surface between honeybees and bumblebees (p = 0.00197), between honeybees and carpenter bees (p < 0.001), and between honeybees and beetles (p = 0.00842). No significant differences were detected (p > 0.05) for the remaining 12 pairs.
PPT
The values of the PPT index are shown in Fig. 1. The far greatest PPT value was seen for bumblebees, followed by small bees; whereas, those of honeybees and wasps were small. Because the rate of visitation of medium-sized bees, syrphid flies, other dipterans, and beetles to female flowers was zero (see Table 1), the PPT values for these groups were also zero.
Potential pollen transport, an index value defined as the rate of visitation to each female flower per hour multiplied by the number of Diospyros pollen grains on the body surface of an insect. The rate of visitation by Bombus ardens ardens was used for that of “bumblebees,” because it was predominant compared with those of the other Bombus species monitored
Pollination experiment
The GLMM revealed significant differences in fruit set among the six pollination treatments (GLMM, type 2; df = 5, χ2 = 58.315, p < 0.001). Tukey’s post hoc test detected significant differences (p < 0.05) in the fruit-set ratio between treatments in one group (NP, 2-M, and 3.5-M treatments) and those in another group (OP, HPB, and HPO treatments) (nine pairs in total; Fig. 2a). In contrast, no significant difference was detected (p > 0.05) between any of the pairs within each group (six pairs in total). The GLMM revealed significant differences in seed number among four pollination treatments (GLMM, type 2; df = 3, χ2 = 32.359, p < 0.001). Tukey’s post hoc test detected significant differences in seed number between the 3.5-M and OP treatments (p = 0.00593), between the 3.5-M and HPB treatments (p < 0.001), and between the 3.5-M and HPO treatments (p < 0.001) (Fig. 2b).
Fruit-set ratio (a) and number of seeds (b) for each pollination treatment. Pollination treatments were as follows: (1) bagging of flower buds with unwoven cloth bag (NP); (2) bagging with 2-mm-mesh bag (2-M); (3) bagging with 3.5-mm-mesh bag (3.5-M); (4) open pollination (OP); (5) hand-pollination followed by enveloping of the flower in paper bag (HPB); and (6) hand-pollination followed by open pollination (HPO). The significance of the coefficients was assessed by using Wald’s test with the GLMM. Tukey’s post hoc test was conducted with the glht function. Ratios with different letters in a differ significantly. In b, significant differences between pollination treatments are shown by **p < 0.01 and ***p < 0.001. Sample sizes in the NP and 2-M treatments were small and were therefore excluded from the statistical analysis in b
Discussion
Our findings revealed that the most important pollinator of D. kaki in Hiroshima, southwestern Japan, was B. ardens ardens. This species was the most abundant in our monitoring of flower-visiting insects. Its abundance was unlikely to be associated with year-to-year fluctuations, because we collected this species for identification more than any others in two consecutive years. These findings implied that if B. ardens ardens were found to possess abundant pollen on its body surface, then it would be the most important pollinator at the study site. In fact, we found that the number of Diospyros pollen grains on the body surfaces of bumblebees was the greatest, followed by carpenter bees and beetles; whereas, the number on honeybees was small. The value of the PPT index, which we surmised would be closely correlated with the number of pollen grains deposited on a stigma—taking into account both visitation frequency and pollen grain number—was far greater for bumblebees than for the other functional groups, demonstrating that bumblebees (i.e., B. ardens ardens) were indeed the most important pollinators of D. kaki. Our pollination experiment revealed that both the fruit-set ratio and the number of seeds in the 3.5-M treatment were significantly smaller than those in the OP treatment. This indicated that the major pollinator was equal in size to, or larger than, insects such as honeybees, supporting our finding that B. ardens ardens was the most important pollinator.
The question arises as to whether B. ardens ardens is also a pollinator of D. kaki in other regions. This bumblebee is distributed almost throughout Japan, except Hokkaido, which is inhabited by another subspecies, Bombus ardens sakagamii Tkalců (Hymenoptera: Apidae), and the Okinawa Islands (Tadauchi and Murao 2014; Suzuki-Ohno et al. 2017). Some studies have reported that B. ardens ardens relies on tree flowers rather than herbaceous ones (Osada et al. 2003; Ushimaru et al. 2008). Additionally, it appears from early April until early July (Katayama 1996), a time that covers the flowering period of D. kaki (from early May in southwestern Japan until early June in northeastern Japan). These studies suggest that B. ardens ardens functions as an important pollinator of D. kaki almost everywhere in Japan, provided that nesting habitats are available in the landscape around the orchards. Our study sites were in orchards surrounded by secondary forest; such habitats are favorable for nesting by this species (Ushimaru et al. 2008).
From the results of the pollination experiment, we can extract other information on pollination of D. kaki at the study site. The fruit-set ratio was significantly smaller in the NP treatment than in the HPB treatment, confirming that pollination is effective in increasing the fruit set of ‘Fuyu’ (Yamada et al. 1987). The lack of a significant difference in fruit-set ratio between the HPB and HPO treatments implied that the hand-pollination in our trial was satisfactory and that additional pollination by insects did not further improve fruit set. The fruit-set ratio in the OP treatment was equivalent to those in the HPB and HPO treatments, showing that natural pollination was sufficient to maximize the fruit set of ‘Fuyu’ under our test conditions. The fruit-set ratio was slightly larger in the 3.5-M treatment than in the 2-M treatment, although the difference was not significant (p = 0.62550). With sufficient pollen grains on their body surfaces, small bees can be considered the second-most-important pollinators. Because their head and thorax widths were approximately 2.5 mm (see Table S2), the 3.5-mm mesh would not have prevented their entry. We do not know why the fruit in the NP treatment set seed (see Fig. 2b), because this treatment was designed to prevent insect access. The most likely explanation is that it was a result of the inadvertent bagging of a flower bud at an inappropriate time—that is, slightly later than anthesis.
Distinguishing between mere flower visitors and effective pollinators is a controversial issue. Simple visitation records are insufficient to discern pollinators, because carryover of pollen grains by an insect and their deposition on a stigma are totally ignored. To better understand this issue, the number of pollen grains on an insect has been used as a factor to assess pollinators’ potential (Lopezaraiza-Mikel et al. 2007; Forup et al. 2008; Devoto et al. 2011). Alarcón (2010) and Popic et al. (2013) used the number of pollen grains on an insect in combination with visitation rate, a method that we basically adopted here. There is, however, a critical view on this concept: it has been noted that quantification of the pollen grains deposited on a stigma is the most important factor in assessing pollination (King et al. 2013). To partially compensate for the fact that here we did not measure pollen deposition on stigmas, we instead conducted pollination treatments using unwoven cloth or mesh bags. The significant decrease in seed number in the 3.5-M treatment compared with that in the OP treatment showed that pollen deposition was performed predominantly by insects that were equal in size to, or larger than, honeybees—that is bumblebees, honeybees, medium-sized bees, or carpenter bees. Among these groups, B. ardens ardens was the only species that both carried large numbers of Diospyros pollen grains and frequently visited the female flowers of D. kaki.
We confirmed by DNA barcoding that 15 pollen loads out of the 22 recovered from flower visitors captured on the flowers of D. kaki constituted pollen grains of this species. This result does not necessarily mean that the other seven individuals possessed no pollen grains of D. kaki on their bodies, because we analyzed the pollen loads recovered from the hind legs—not all the pollen grains attached to the body surface—by molecular identification. Three pollen loads were identified as belonging to kiwifruit (which was simultaneously in bloom at the study site), Toxicodendron sp., and Rosaceae sp. In contrast, two samples gave no amplification with PCR using trnL–trnF as the DNA barcoding region. This could have been due to incompatibility of the primer pair with the plant species whose pollen grains constituted the pollen loads, although the primer pair was designed as universal. We did not conduct further investigations, because the aim of this analysis was to distinguish the pollen of D. kaki from that of other Diospyros species. Other universal primer pairs could also be effective for further identification of pollen loads.
Because A. mellifera had not been introduced to our study site, we did not focus on its effectiveness and efficiency as a pollinator. However, colonies of A. mellifera are often introduced to D. kaki orchards to enhance pollination (Yamamura et al. 1989). We are, therefore, preparing another study to identify the important pollinators in orchards to which A. mellifera has been introduced.
Conclusion
We evaluated the contributions of flower visitors to pollination by multiplying the visitation rate by the number of pollen grains on the body surface, and we found that the most important pollinator of D. kaki at our study site in Hiroshima was B. ardens ardens. The decrease in fruit-set ratio and seed number of ‘Fuyu’ when flower buds were covered with 3.5-mm-mesh bags suggests that B. ardens ardens is an important, large-bodied pollinator. This finding was reasonable, because the orchards at the study site were surrounded by secondary forest, which potentially offers nesting habitats for this bumblebee. The wide distribution of B. ardens ardens in Japan and the timing of its appearance across the flowering season of D. kaki suggest that it functions nationwide as an important pollinator. We are now planning to identify pollinators in several areas of D. kaki production in Japan to see whether B. ardens ardens also serves as an important pollinator in other regions.
References
Alarcón R (2010) Congruence between visitation and pollen-transport networks in a California plant–pollinator community. Oikos 119:35–44. https://doi.org/10.1111/j.1600-0706.2009.17694.x
Asami Y, Chow CT (1936) Is the pollen of Japanese persimmons carried by wind? J Hortic Assoc Jpn 7:247–251. https://doi.org/10.2503/jjshs.7.247
Devoto M, Bailey S, Memmott J (2011) The ‘night shift’: nocturnal pollen-transport networks in a boreal pine forest. Ecol Entomol 36:25–35. https://doi.org/10.1111/j.1365-2311.2010.01247.x
FAOSTAT (Food and Agriculture Organization Corporate Statistical Database) (2017) Production quantities of Persimmons by country. http://www.fao.org/faostat/en/#data/QC/visualize Accessed 14 Feb 2019
Folmer O, Black M, Hoeh W, Lutz R, Vrijenhoek R (1994) DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol Mar Biol Biotechnol 3:294–299. https://www.mbari.org/wp-content/uploads/2016/01/Folmer_94MMBB.pdf. Accessed 19 Feb 2019
Forup ML, Henson KSE, Craze PG, Memmott J (2008) The restoration of ecological interactions: plant–pollinator networks on ancient and restored heathlands. J Appl Ecol 45:742–752. https://doi.org/10.1111/j.1365-2664.2007.01390.x
Françoso E, Arias MC (2013) Cytochrome c oxidase I primers for corbiculate bees: DNA barcode and mini-barcode. Mol Ecol Resour 13:844–850. https://doi.org/10.1111/1755-0998.12135
George AP, Mowat AD, Collins RJ, Morley-Bunker M (1997) The pattern and control of reproductive development in non-astringent persimmon (Diospyros kaki L.): a review. Sci Hortic 70:93–122. https://doi.org/10.1016/S0304-4238(97)00043-5
Hasegawa K, Nakajima Y (1990a) Effects of seediness on fruit quality of Japanese persimmon cv. Maekawa-Jiro. J Jpn Soc Hortic Sci 59:255–262. https://doi.org/10.2503/jjshs.59.255 (in Japanese with English abstract)
Hasegawa K, Nakajima Y (1990b) Effects of bloom date, seediness, GA treatment and location of fruits in the foliar canopy on the fruit quality of persimmon. J Jpn Soc Hortic Sci 59:263–270. https://doi.org/10.2503/jjshs.59.263 (in Japanese with English abstract)
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biom J 50:346–363. https://doi.org/10.1002/bimj.200810425
Japanese Government Statistics (2017) The e-Stat (a portal site for Japanese Government Statistics) of the Statistics Bureau, Ministry of Internal Affairs and Communications. Table f006-29-019 (In Japanese). https://www.e-stat.go.jp/stat-search/files?page=1&layout=datalist&toukei=00500215&tstat=000001013427&cycle=7&year=20170&month=0&tclass1=000001032287&tclass2=000001032927&tclass3=000001120615. Accessed 14 Feb 2019
Kajiura M (1941) Studies on physiological dropping of fruits in the Japanese persimmon II. Relationships among dropping of fruits, pollination and parthenocarpy. J Hortic Ass Jpn 12:247–283. https://doi.org/10.2503/jjshs.12.247 (in Japanese)
Katayama E (1996) Survivorship curves and longevity for workers of Bombus ardens Smith and Bombus diversus Smith (Hymenoptera, Apidae). Jpn J Ent 64:111–121
King C, Ballantyne G, Willmer PG (2013) Why flower visitation is a poor proxy for pollination: measuring single-visit pollen deposition, with implications for pollination networks and conservation. Methods Ecol Evol 4:811–818. https://doi.org/10.1111/2041-210X.12074
Klein AM, Steffan-Dewenter I, Tscharntke T (2003) Fruit set of highland coffee increases with the diversity of pollinating bees. Proc R Soc Lond B 270:955–961. https://doi.org/10.1098/rspb.2002.2306
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874. https://doi.org/10.1093/molbev/msw054
Lopezaraiza-Mikel ME, Hayes RB, Whalley MR, Memmott J (2007) The impact of an alien plant on a native plant–pollinator network: an experimental approach. Ecol Lett 10:539–550. https://doi.org/10.1111/j.1461-0248.2007.01055.x
McGregor SE (1976) Insect pollination of cultivated crop plants. USDA Agriculture Handbook No. 496. Washington DC
Nikkeshi A, Hiraiwa MK, Ushimaru A, Hoshizaki K, Makita A, Inoue M (2016) Established method of deposited pollen grains on the surface of pollinated insect body. Jpn J Palynol 62:3–7 (In Japanese with English abstract)
Osada N, Sugiura S, Kawamura K, Cho M, Takeda H (2003) Community-level flowering phenology and fruit set: comparative study of 25 woody species in a secondary forest in Japan. Ecol Res 18:711–723. https://doi.org/10.1111/j.1440-1703.2003.00590.x
Popic TJ, Wardle GM, Davila YC (2013) Flower-visitor networks only partially predict the function of pollen transport by bees. Austral Ecol 38:76–86. https://doi.org/10.1111/j.1442-9993.2012.02377.x
R Development Core Team (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org. Accessed 31 Oct 2018
Simon C, Frati F, Beckenbach A, Crespi B, Liu H, Flook P (1994) Evolution, weighting, and phylogenetic utility of mitochondrial gene sequences and a compilation of conserved polymerase chain reaction primers. Ann Entomol Soc Am 87:651–701. https://doi.org/10.1093/aesa/87.6.651
Skaug H, Fournier D, Magnusson A, Bolker B, Nielsen A (2016) Generalized linear mixed models using AD model builder. R package version 0.8.3.3. http://glmmadmb.r-forge.r-project.org/. Accessed 31 Oct 2018
Suzuki-Ohno Y, Yokoyama J, Nakashizuka T, Kawata M (2017) Utilization of photographs taken by citizens for estimating bumblebee distributions. Sci Rep 7:11215. https://doi.org/10.1038/s41598-017-10581-x
Taberlet P, Gielly L, Pautou G, Bouvet J (1991) Universal primers for amplification of three non-coding regions of chloroplast DNA. Plant Mol Biol 17:1105–1109. https://doi.org/10.1007/BF00037152
Tadauchi O, Murao R (2014) An illustrated guide to Japanese bees. Bun-ichi Shuppan, Tokyo (in Japanese)
Tamura K, Nei M (1993) Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees. Mol Biol Evol 10:512–526. https://doi.org/10.1093/oxfordjournals.molbev.a040023
Tokunaga M, Sasakawa M, Akiyama J (1959) Studies on the insect-visitors on the flowers of fruit trees. Sci Rep Kyoto Pref Univ Agric 11:59–70 (In Japanese with English abstract)
Ushimaru A, Ishida C, Sakai S, Shibata M, Tanaka H, Niiyama K, Nakashizuka T (2008) The effects of human management on spatial distribution of two bumble bee species in a traditional agro-forestry Satoyama landscape. J Apic Res 47:296–303. https://doi.org/10.1080/00218839.2008.11101478
Yamada M, Kurihara A, Sumi T (1987) Varietal differences in fruit bearing in Japanese persimmon (Diospyros kaki Thunb.) and their yearly fluctuations. J Jpn Soc Hortic Sci 56:293–299. https://doi.org/10.2503/jjshs.56.293 (in Japanese with English abstract)
Yamamura H, Matsui K, Matsumoto T (1989) Effects of gibberellins on fruit set and flower-bud formation in unpollinated persimmons (Diospyros kaki). Sci Hortic 38:77–86. https://doi.org/10.1016/0304-4238(89)90022-8
Yokozawa Y (1951) Insect visitors on the flowers of Japanese persimmon. J Jpn Soc Hortic Sci 20:58–64. https://doi.org/10.2503/jjshs.20.58 (in Japanese)
Yonemori K, Kameda K, Sugiura A (1992) Characteristics of sex expression in monoecious persimmons. J Jpn Soc Hortic Sci 61:303–310. https://doi.org/10.2503/jjshs.61.303 (in Japanese with English abstract)
Zhang JZ (2008) Anthracnose of persimmon caused by Colletotrichum gloeosporioides in China. Asian Australas J Plant Sci Biotechnol 2:50–54. http://www.globalsciencebooks.info/Online/GSBOnline/images/0812/AAJPSB_2(1&2)/AAJPSB_2(2)50-54o.pdf. Accessed 14 Feb 2019
Acknowledgements
We appreciate the helpful advice of Dr. Akihiko Sato (Institute of Fruit Tree and Tea Science, NARO) on the cultivation of Oriental persimmon. We are grateful to Dr. Shoko Nakamura (Forest Research and Management Organization) for identifying Bombus spp. and to Prof. Ryoichi Miyanaga (Shimane University) and Dr. Ryuki Murao (Regional Environmental Planning Co., Ltd.) for verifying our identification of Andrena spp. and Lasioglossum spp. We thank Ms. Satomi Kohara for her technical help. This work was supported financially by the Ministry of Agriculture, Forestry, and Fisheries, Japan, through a research project entitled “Monitoring and enhancement of pollinators for crop production.”
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Nikkeshi, A., Inoue, H., Arai, T. et al. The bumblebee Bombus ardens ardens (Hymenoptera: Apidae) is the most important pollinator of Oriental persimmon, Diospyros kaki (Ericales: Ebenaceae), in Hiroshima, Japan. Appl Entomol Zool 54, 409–419 (2019). https://doi.org/10.1007/s13355-019-00637-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13355-019-00637-x