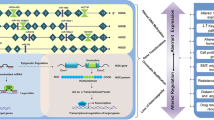
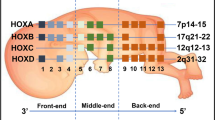
Abstract
The Homeobox (HOX) gene family is essential to regulating cellular processes because it maintains the exact coordination required for tissue homeostasis, cellular differentiation, and embryonic development. The most distinctive feature of this class of genes is the presence of the highly conserved DNA region known as the homeobox, which is essential for controlling their regulatory activities. Important players in the intricate process of genetic regulation are the HOX genes. Many diseases, especially in the area of cancer, are linked to their aberrant functioning. Due to their distinctive functions in biomedical research—particularly in the complex process of tumor advancement—HOXA9 and HOXB9 have drawn particular attention. HOXA9 and HOXB9 are more significant than what is usually connected with HOX genes since they have roles in the intricate field of cancer and beyond embryonic processes. The framework for a focused study of the different effects of HOXA9 and HOXB9 in the context of tumor biology is established in this study.

Similar content being viewed by others
Data availability
Not applicable.
Abbreviations
- HOX:
-
Homeobox
- TALE:
-
Three amino acid loop extension
- CCND1:
-
Cyclin D1
- CDK6:
-
Cyclin-dependent kinase 6
- VEGF:
-
Vascular endothelial growth factor
- CAFs:
-
Cancer-associated fibroblasts
- EMT:
-
Epithelial-mesenchymal transition
- CRC:
-
Colorectal cancer
- SC:
-
Stem cell
- PTEN:
-
Phosphatase and TENsin homolog deleted on chromosome 10
- CIP1:
-
Cyclin-dependent kinase inhibitor p21
- WAF1:
-
Wildtype p53-activated fragment 1
- PBX:
-
Pre-B cell leukemia transcription factor
- MEIS1:
-
Myeloid ecotropic viral integration site 1
- MAPK:
-
Mitogen-activated protein kinase
- ERK:
-
Extracellular signal-regulated kinase
- TCF4:
-
Transcription factor 4
- WNT:
-
Wingless-related integration site
- GalNAc-T14:
-
N-acetyl-galactosaminyl-transferases 14
- bFGF:
-
Fibroblast growth factor
- MDSCs:
-
Myeloid-derived suppressor cells
- TGF-β:
-
Transforming growth factor-β
- PI3K:
-
Phosphatidylinositol 3-kinase
- AKT:
-
Protein kinase B
- mTOR:
-
Mammalian target of rapamycin
- NF-κB:
-
Nuclear factor kappa-light-chain-enhancer of activated B cells
- HNSCC:
-
Head and neck squamous cell carcinoma
- SCC:
-
Squamous cell carcinoma
- AML:
-
Acute myeloid leukemia
- ALL:
-
Aacute lymphoblastic leukemia
- RELA:
-
V-rel avian reticuloendotheliosis viral oncogene homolog A
- ATG:
-
Autophagy-related gene
- GRHL2:
-
Grainyhead-like 2
- ALDH:
-
Aldehyde dehydrogenase
- BRCA1:
-
Breast cancer gene 1
- TET1:
-
Ten‐eleven translocation 1
- HMGA2:
-
High-mobility-group protein AT-hook 2
- ANGPTL2:
-
Angiopoietin‐like protein 2
- E2F3:
-
E2F transcription factor 3
- SMAD:
-
Mothers against decapentaplegic homolog 2
- CXCL1:
-
Chemokine ligand 1
- PFS:
-
Progression-free survival
- TAM:
-
Tumor-associated macrophage
- MHC:
-
Major histocompatibility complex
- DC:
-
Dendritic cell
- TLR:
-
Toll-like receptor
- IFN:
-
Type I interferons
- BM:
-
Bone marrow
- FLT3:
-
FMS‐like tyrosine kinase 3
- FLT3L:
-
FMS-like tyrosine kinase 3 ligand
- NK:
-
Natural killer
References
Abu-Amero KK, Kondkar AA, Salih MA, Alorainy IA, Khan AO, Oystreck DT, Bosley TM (2013) Partial chromosome 7 duplication with a phenotype mimicking the HOXA1 spectrum disorder. Ophthalmic Genet 34(1–2):90–96
Afzal Z, Krumlauf R (2022) Transcriptional regulation and implications for controlling Hox gene expression. J Dev Biol 10(1):4
Agrawal-Singh S, Bagri J, Giotopoulos G, Azazi DMA, Horton SJ, Lopez CK, Anand S, Bach AS, Stedham F, Antrobus R, Houghton JW, Vassiliou GS, Sasca D, Yun H, Whetton AD, Huntly BJP (2023) HOXA9 forms a repressive complex with nuclear matrix-associated protein SAFB to maintain acute myeloid leukemia. Blood 141(14):1737–1754
Aguiar GdM, Ramão A, Plaça JR, Simões SC, Scaraboto NV, Freitas-Castro F, Cardoso C, Sousa JdF, Silva WA Jr (2021) Upregulation of HOX genes promotes cell migration and proliferation in head and neck squamous cell carcinoma. Tumor Biology 43(1):263–278
Aksoz M, Turan RD, Albayrak E, Kocabas F (2018) Emerging roles of Meis1 in cardiac regeneration, stem cells and cancer. Curr Drug Targets 19(2):181–190
Almasmoum HA, Airhihen B, Seedhouse C, Winkler GS (2021) Frequent loss of BTG1 activity and impaired interactions with the Caf1 subunit of the Ccr4–Not deadenylase in non-Hodgkin lymphoma. Leuk Lymphoma 62(2):281–290
Alsayegh K, Cortés-Medina LV, Ramos-Mandujano G, Badraiq H, Li M (2019) Hematopoietic differentiation of human pluripotent stem cells: HOX and GATA transcription factors as master regulators. Curr Genomics 20(6):438–452
Alvarado-Ruiz L, Martinez-Silva MG, Torres-Reyes LA, Pina-Sanchez P, Ortiz-Lazareno P, Bravo-Cuellar A, Aguilar-Lemarroy A, Jave-Suarez LF (2016) HOXA9 is underexpressed in cervical cancer cells and its restoration decreases proliferation, migration and expression of epithelial-to-mesenchymal transition genes. Asian Pac J Cancer Prev 17(3):1037–1047
Aros CJ, Pantoja CJ, Gomperts BN (2021) Wnt signaling in lung development, regeneration, and disease progression. Commun Biol 4(1):601
Aryal S, Zhang Y, Wren S, Li C, Lu R (2023) Molecular regulators of HOXA9 in acute myeloid leukemia. FEBS J 290(2):321–339
Bhatlekar S, Ertel A, Gonye GE, Fields JZ, Boman BM (2019) Gene expression signatures for HOXA4, HOXA9, and HOXD10 reveal alterations in transcriptional regulatory networks in colon cancer. J Cell Physiol 234(8):13042–13056
Bhatlekar S, Fields JZ, Boman BM (2014) HOX genes and their role in the development of human cancers. J Mol Med 92:811–23
Bhatlekar S, Fields JZ, Boman BM (2018a) Role of HOX genes in stem cell differentiation and cancer. Stem Cells Int 2018:3569493
Bhatlekar S, Viswanathan V, Fields JZ, Boman BM (2018b) Overexpression of HOXA4 and HOXA9 genes promotes self-renewal and contributes to colon cancer stem cell overpopulation. J Cell Physiol 233(2):727–735
Blecua P, Martinez-Verbo L, Esteller M (2020) The DNA methylation landscape of hematological malignancies: An update. Mol Oncol 14(8):1616–1639
Bondos SE, Mendes GG, Jons A (2020) Context-dependent HOX transcription factor function in health and disease. Prog Mol Biol Transl Sci 174:225–62
Braekeleer ED, Douet-Guilbert N, Basinko A, Bris M-JL, Morel F, Braekeleer MD (2014) Hox gene dysregulation in acute myeloid leukemia. Future Oncol 10(3):475–495
Brechka H, Bhanvadia RR, VanOpstall C, Vander Griend DJ (2017) HOXB13 mutations and binding partners in prostate development and cancer: Function, clinical significance, and future directions. Genes Dis 4(2):75–87
Brison N, Debeer P, Tylzanowski P (2014) Joining the fingers: a HOXD13 Story. Dev Dyn 243(1):37–48
Brotto DB, Siena ÁDD, de Barros II, SdCeS C, Muys BR, Goedert L, Cardoso C, Plaça JR, Ramão A, Squire JA (2020) Contributions of HOX genes to cancer hallmarks: Enrichment pathway analysis and review. Tumor Biol 42(5):1010428320918050
Buffry AD, McGregor AP (2022) Micromanagement of Drosophila post-embryonic development by Hox genes. J Dev Biol 10(1):13
Calvanese V, Capellera-Garcia S, Ma F, Fares I, Liebscher S, Ng ES, Ekstrand S, Aguadé-Gorgorió J, Vavilina A, Lefaudeux D (2022) Mapping human haematopoietic stem cells from haemogenic endothelium to birth. Nature 604(7906):534–540
Calvanese V, Mikkola HK (2023) The genesis of human hematopoietic stem cells. Blood 142(6):519–532
Carbone C, Piro G, Simionato F, Loupakis F, Cremolini C, Fontanini G, Di Nicolantonio F, Moratti E, Ligorio F, Zanotto M (2016) Homeobox B9 (HOXB9) sustains anti-VEGF treatment resistance in gastrointestinal tumors. Cancer Res 76(14_Supplement):3265
Carbone C, Piro G, Simionato F, Ligorio F, Cremolini C, Loupakis F, Alì G, Rossini D, Merz V, Santoro R (2017) Homeobox B9 mediates resistance to anti-VEGF therapy in colorectal cancer patients. Clin Cancer Res 23(15):4312–4322
Chen F, Capecchi MR (1999) Paralogous mouse Hox genes, Hoxa9, Hoxb9, and Hoxd9, function together to control development of the mammary gland in response to pregnancy. Proc Natl Acad Sci 96(2):541–546
Chen Y, Ren B, Yang J, Wang H, Yang G, Xu R, You L, Zhao Y (2020) The role of histone methylation in the development of digestive cancers: a potential direction for cancer management. Sig Transduct Target Ther 5(1):143
Chiba N, Comaills V, Shiotani B, Takahashi F, Shimada T, Tajima K, Winokur D, Hayashida T, Willers H, Brachtel E (2012) Homeobox B9 induces epithelial-to-mesenchymal transition-associated radioresistance by accelerating DNA damage responses. Proc Natl Acad Sci 109(8):2760–2765
Chiba N, Ozawa Y, Hikita K, Okihara M, Sano T, Tomita K, Takano K, Kawachi S (2017) Increased expression of HOXB9 in hepatocellular carcinoma predicts poor overall survival but a beneficial response to sorafenib. Oncol Rep 37(4):2270–2276
Chiba N, Ochiai S, Gunji T, Kobayashi T, Sano T, Tomita K, Kawachi S (2022) HOXB9 mediates resistance to chemotherapy and patient outcomes through the TGFβ pathway in pancreatic cancer. Oncotarget 13:747
Cieslak A, Charbonnier G, Tesio M, Mathieu E-L, Belhocine M, Touzart A, Smith C, Hypolite G, Andrieu GP, Martens JH (2020) Blueprint of human thymopoiesis reveals molecular mechanisms of stage-specific TCR enhancer activation. J Exp Med 217(9):e20192360
Collins CT, Hess JL (2016a) Deregulation of the HOXA9/MEIS1 axis in acute leukemia. Curr Opin Hematol 23(4):354–361
Collins CT, Hess JL (2016b) Role of HOXA9 in leukemia: dysregulation, cofactors and essential targets. Oncogene 35(9):1090–1098
Collins EM, Thompson A (2018) HOX genes in normal, engineered and malignant hematopoiesis. Int J Dev Biol 62(11–12):847–856
Contarelli S, Fedele V, Melisi D (2020) HOX genes family and cancer: a novel role for homeobox B9 in the resistance to anti-angiogenic therapies. Cancers 12(11):3299
Dai M, Song J, Wang L, Zhou K, Shu L (2021) HOXC13 promotes cervical cancer proliferation, invasion and Warburg effect through β-catenin/c-Myc signaling pathway. J Bioenerg Biomembr 53:597–608
de Bock CE, Cools J (2018) JAK3 mutations and HOXA9 expression are important cooperating events in T-cell acute lymphoblastic leukemia. Mol Cell Oncol 5(3):e1458014
de Bessa Garcia SA, Araújo M, Pereira T, Mouta J (1873) Freitas R (2020) HOX genes function in breast cancer development. Biochim Biophys Acta (BBA)-Rev Cancer 2:188358
Deschamps J, van den Akker E, Forlani S, De Graaff W, Oosterveen T, Roelen B, Roelfsema J (2004) Initiation, establishment and maintenance of Hox gene expression patterns in the mouse. Int J Dev Biol 43(7):635–650
Dickson GJ, Liberante FG, Kettyle LM, O’Hagan KA, Finnegan DP, Bullinger L, Geerts D, McMullin MF, Lappin TR, Mills KI (2013) HOXA/PBX3 knockdown impairs growth and sensitizes cytogenetically normal acute myeloid leukemia cells to chemotherapy. Haematologica 98(8):1216
Di-Poi N, Montoya-Burgos JI, Miller H, Pourquie O, Milinkovitch MC, Duboule D (2010) Changes in Hox genes’ structure and function during the evolution of the squamate body plan. Nature 464(7285):99–103
Drabkin H, Parsy C, Ferguson K, Guilhot F, Lacotte L, Roy L, Zeng C, Baron A, Hunger S, Varella-Garcia M (2002) Quantitative HOX expression in chromosomally defined subsets of acute myelogenous leukemia. Leukemia 16(2):186–195
Duan R, Han L, Wang Q, Wei J, Chen L, Zhang J, Kang C, Wang L (2015) HOXA13 is a potential GBM diagnostic marker and promotes glioma invasion by activating the Wnt and TGF-β pathways. Oncotarget 6(29):27778–93
Duan T, Du Y, Xing C, Wang HY, Wang RF (2022) Toll-like receptor signaling and its role in cell-mediated immunity. Frontiers in Immunol 13:812774
Durston AJ (2019) What are the roles of retinoids, other morphogens, and Hox genes in setting up the vertebrate body axis? genesis 57(7–8):e23296
Ekanayake DL, Małopolska MM, Schwarz T, Tuz R, Bartlewski PM (2022) The roles and expression of HOXA/Hoxa10 gene: A prospective marker of mammalian female fertility? Reprod Biol 22(2):100647
Fang L, Xu Y, Zou L (2014) Overexpressed homeobox B9 regulates oncogenic activities by transforming growth factor-β1 in gliomas. Biochem Biophys Res Commun 446(1):272–279
Fenelon K (2022) Linking Nuclear Mechanotransduction and Epigenetics in Morphogenesis: University of Toronto (Canada) 28961822
Feng Y, Zhang T, Wang Y, Xie M, Ji X, Luo X, Huang W, Xia L (2021a) Homeobox genes in cancers: from carcinogenesis to recent therapeutic intervention. Front Oncol 11:770428
Feng Y, Zhang T, Wang Y, Xie M, Ji X, Luo X, Huang W, Xia L (2021b) Homeobox Genes in Cancers: From Carcinogenesis to Recent Therapeutic Intervention. Front Oncol 11:770428
Fish JL (2019) Evolvability of the vertebrate craniofacial skeleton. Semin Cell Dev Biol Elsevier 91:13–22
Fraineau S, Palii CG, Allan DS, Brand M (2015) Epigenetic regulation of endothelial-cell-mediated vascular repair. FEBS J 282(9):1605–1629
Gharbaran R (2016) Insights into the molecular roles of heparan sulfate proteoglycans (HSPGs—syndecans) in autocrine and paracrine growth factor signaling in the pathogenesis of Hodgkin’s lymphoma. Tumor Biol 37:11573–88
Gilbert PM, Mouw JK, Unger MA, Lakins JN, Gbegnon MK, Clemmer VB, Benezra M, Licht JD, Boudreau NJ, Tsai KK (2010) HOXA9 regulates BRCA1 expression to modulate human breast tumor phenotype. J Clin Investig 120(5):1535–1550
Gİrgİn B, Alpaslan MK, KocabaŞ F (2020) Oncogenic and tumor suppressor function of MEIS and associated factors. Turk J Biol 44(6):328–55
Göllner S, Oellerich T, Agrawal-Singh S, Schenk T, Klein H-U, Rohde C, Pabst C, Sauer T, Lerdrup M, Tavor S (2017) Loss of the histone methyltransferase EZH2 induces resistance to multiple drugs in acute myeloid leukemia. Nat Med 23(1):69–78
Gonçalves CS (2013) Regulation of Wnt signaling by HOXA9 in Glioblastoma: Mechanistic, prognostic and therapeutic insights: Universidade do Minho (Portugal)
Gonçalves CS, Le Boiteux E, Arnaud P, Costa BM (2020) HOX gene cluster (de) regulation in brain: from neurodevelopment to malignant glial tumours. Cell Mol Life Sci 77(19):3797–3821
Gong T, Li Y, Feng L, Fang M, Dai G, Huang X, Yang Y, Liu S (2020) CASC21, a FOXP1 induced long non-coding RNA, promotes colorectal cancer growth by regulating CDK6. Aging (Albany NY) 12(12):12086
Grier D, Thompson A, Kwasniewska A, McGonigle G, Halliday H, Lappin T (2005) The pathophysiology of HOX genes and their role in cancer. J Pathol: J Pathol Soc Great Britain Ireland 205(2):154–171
Gu M, Ren B, Fang Y, Ren J, Liu X, Wang X, Zhou F, Xiao R, Luo X, You L, Zhao Y (2024) Epigenetic regulation in cancer. MedComm 5(2):e495
Guéro S (2018) Developmental biology of the upper limb. Hand Surg Rehabil 37(5):265–274
Gwin KA, Shapiro MB, Dolence JJ, Huang ZL, Medina KL (2013) Hoxa9 and Flt3 signaling synergistically regulate an early checkpoint in lymphopoiesis. J Immunol 191(2):745–754
Halanych KM, Passamaneck Y (2001) A brief review of metazoan phylogeny and future prospects in Hox-research. Am Zool 41(3):629–639
Han S, Li X, Liang X, Zhou L (2019) HOXA9 transcriptionally promotes apoptosis and represses autophagy by targeting NF-κB in cutaneous squamous cell carcinoma. Cells 8(11):1360
Hao Z, Dai J, Shi D, Xu Z, Chen D, Zhao B, Teng H, Jiang Q (2014) Association of a single nucleotide polymorphism in HOXB9 with developmental dysplasia of the hip: A case-control study. J Orthop Res 32(2):179–182
Hayashida T, Takahashi F, Chiba N, Brachtel E, Takahashi M, Godin-Heymann N, Gross KW, Vivanco MdM, Wijendran V, Shioda T (2010) HOXB9, a gene overexpressed in breast cancer, promotes tumorigenicity and lung metastasis. Proc Natl Acad Sci 107(3):1100–1105
Hoshino Y, Hayashida T, Hirata A, Takahashi H, Chiba N, Ohmura M, Wakui M, Jinno H, Hasegawa H, Maheswaran S (2014) Bevacizumab terminates homeobox B9-induced tumor proliferation by silencing microenvironmental communication. Mol Cancer 13(1):1–14
Huang Y, Sitwala K, Bronstein J, Sanders D, Dandekar M, Collins C, Robertson G, MacDonald J, Cezard T, Bilenky M (2012) Identification and characterization of Hoxa9 binding sites in hematopoietic cells. Blood J Am Soc Hematol 119(2):388–398
Huang K, Yuan R, Wang K, Hu J, Huang Z, Yan C, Shen W, Shao J (2014) Overexpression of HOXB9 promotes metastasis and indicates poor prognosis in colon cancer. Chin J Cancer Res 26(1):72
Huang C, Hu Y-W, Zhao J-J, Ma X, Zhang Y, Guo F-X, Kang C-M, Lu J-B, Xiu J-c, Sha Y-H (2016) Long noncoding RNA HOXC-AS1 suppresses Ox-LDL-induced cholesterol accumulation through promoting HOXC6 expression in THP-1 macrophages. DNA Cell Biol 35(11):722–729
Hubert KA, Wellik DM (2023) Hox genes in development and beyond. Development 150(1):dev192476
Ibrahim DM, Hansen P, Rödelsperger C, Stiege AC, Doelken SC, Horn D, Jäger M, Janetzki C, Krawitz P, Leschik G (2013) Distinct global shifts in genomic binding profiles of limb malformation-associated HOXD13 mutations. Genome Res 23(12):2091–2102
Ibrahim DM, Tayebi N, Knaus A, Stiege AC, Sahebzamani A, Hecht J, Mundlos S, Spielmann M (2016) A homozygous HOXD13 missense mutation causes a severe form of synpolydactyly with metacarpal to carpal transformation. Am J Med Genet A 170(3):615–621
Jin Y, Kim HK, Lee J, Soh EY, Kim J-H, Song I, Chung Y-S, Choi YJ (2019) Transcription factor HOXA9 is linked to the calcification and invasion of papillary thyroid carcinoma. Sci Rep 9(1):6773
Jonkers J, Pai P, Sukumar S (2020) Multiple roles of HOX proteins in Metastasis: Let me count the ways. Cancer Metastasis Rev 39:661–79
Ju T, Jin H, Ying R, Xie Q, Zhou C, Gao D (2017) Overexpression of NAC1 confers drug resistance via HOXA9 in colorectal carcinoma cells. Mol Med Rep 16(3):3194–3200
Kato F, Wada N, Hayashida T, Fukuda K, Nakamura R, Takahashi T, Kawakubo H, Takeuchi H, Kitagawa Y (2019) Experimental and clinicopathological analysis of HOXB9 in gastric cancer. Oncol Lett 17(3):3097–3102
Kelly ZL, Michael A, Butler-Manuel S, Pandha HS, Morgan RG (2011) HOX genes in ovarian cancer. J Ovarian Res 4(1):1–6
Kelly Z, Moller-Levet C, McGrath S, Butler-Manuel S, Kavitha Madhuri T, Kierzek AM, Pandha H, Morgan R, Michael A (2016) The prognostic significance of specific HOX gene expression patterns in ovarian cancer. Int J Cancer 139(7):1608–1617
Kim J-H, Kim YH, Han JH, Lee KB, Sheen SS, Lee J, Soh E-Y, Park TJ (2012) Silencing of homeobox B9 is associated with down-regulation of CD56 and extrathyroidal extension of tumor in papillary thyroid carcinoma. Hum Pathol 43(8):1221–1228
Ko SY, Ladanyi A, Lengyel E, Naora H (2014) Expression of the homeobox gene HOXA9 in ovarian cancer induces peritoneal macrophages to acquire an M2 tumor-promoting phenotype. Am J Pathol 184(1):271–281
Kostic D, Capecchi MR (1994) Targeted disruptions of the murine Hoxa-4 and Hoxa-6 genes result in homeotic transformations of components of the vertebral column. Mech Dev 46(3):231–247
Kurban M, Wajid M, Petukhova L, Shimomura Y, Christiano AM (2011) A nonsense mutation in the HOXD13 gene underlies synpolydactyly with incomplete penetrance. J Hum Genet 56(10):701–706
Kwon O-S, Oh E, Park J-R, Lee J-S, Bae G-Y, Koo J-H, Kim H, Choi Y-L, Choi YS, Kim J (2015) GalNAc-T14 promotes metastasis through Wnt dependent HOXB9 expression in lung adenocarcinoma. Oncotarget 6(39):41916
Li D-P, Li Z-Y, Sang W, Cheng H, Pan X-Y, Xu K-L (2013) HOXA9 gene expression in acute myeloid leukemia. Cell Biochem Biophys 67:935–8
Li F, Dong L, Xing R, Wang L, Luan F, Yao C, Ji X, Bai L (2014) Homeobox B9 is overexpressed in hepatocellular carcinomas and promotes tumor cell proliferation both in vitro and in vivo. Biochem Biophys Res Commun 444(2):241–247
Li P-D, Chen P, Peng X, Ma C, Zhang W-J, Dai X-F (2018) HOXC6 predicts invasion and poor survival in hepatocellular carcinoma by driving epithelial-mesenchymal transition. Aging (Albany NY) 10(1):115
Li B, Huang Q, Wei G-H (2019a) The role of HOX transcription factors in cancer predisposition and progression. Cancers 11(4):528
Li X-f, Zhang H-B, Huo Y (2022) High HOXA9 gene expression predicts response to chemotherapy and prognosis of high-grade serous ovarian cancer patients. J Int Med Res 50(11):03000605221135864
Li B, Huang Q, Wei GH (2019b) The Role of HOX Transcription Factors in Cancer Predisposition and Progression. Cancers (Basel) 11(4)
Liu W, Deng L, Song Y, Redell M (2014) DOT1L inhibition sensitizes MLL-rearranged AML to chemotherapy. PLoS One 9(5):e98270
Liu K, Li X, Yang Z, Zhang R (2023) Activation of CTHRC1 by HOXB9 Promotes Angiogenesis through Fatty Acid Metabolism in Lung Adenocarcinoma. Rev Invest Clin 75(2):63–75
Maffuid K, Cao Y (2023) Decoding the complexity of immune-cancer cell interactions: empowering the future of cancer immunotherapy. Cancers (Basel) 15(16):4188
Makki N, Capecchi MR (2011) Identification of novel Hoxa1 downstream targets regulating hindbrain, neural crest and inner ear development. Dev Biol 357(2):295–304
Martinou E, Falgari G, Bagwan I, Angelidi AM (2021) A systematic review on HOX genes as potential biomarkers in colorectal cancer: an emerging role of HOXB9. Int J Mol Sci 22(24):13429
Martinou E, Moller-Levet C, Karamanis D, Bagwan I, Angelidi AM (2022) HOXB9 overexpression promotes colorectal cancer progression and is associated with worse survival in liver resection patients for colorectal liver metastases. Int J Mol Sci 23(4):2281
Mehrazarin S, Chen W, Oh J-E, Liu ZX, Kang KL, Jin KY, Kim RH, Shin K-H, Park N-H, Kang MK (2015) The p63 gene is regulated by grainyhead-like 2 (GRHL2) through reciprocal feedback and determines the epithelial phenotype in human keratinocytes. J Biol Chem 290(32):19999–20008
Miyamoto R, Kanai A, Okuda H, Komata Y, Takahashi S, Matsui H, Inaba T, Yokoyama A (2021) HOXA9 promotes MYC-mediated leukemogenesis by maintaining gene expression for multiple anti-apoptotic pathways. Elife 10:e64148
Mo R-J, Lu J, Wan Y-P, Hua W, Liang Y-X, Zhuo Y-J, Kuang Q-W, Liu Y-L, He H-C, Zhong W-D (2017) Decreased HoxD10 expression promotes a proliferative and aggressive phenotype in prostate cancer. Curr Mol Med 17(1):70–78
Morgan R, Hunter K, Pandha HS (2022) Downstream of the HOX genes: Explaining conflicting tumour suppressor and oncogenic functions in cancer. Int J Cancer 150(12):1919–1932
Mortimer T, Wainwright EN, Patel H, Siow BM, Jaunmuktane Z, Brandner S, Scaffidi P (2019) Redistribution of EZH 2 promotes malignant phenotypes by rewiring developmental programmes. EMBO Rep 20(10):e48155
Mouw JK, Yui Y, Damiano L, Bainer RO, Lakins JN, Acerbi I, Ou G, Wijekoon AC, Levental KR, Gilbert PM (2014) Tissue mechanics modulate microRNA-dependent PTEN expression to regulate malignant progression. Nat Med 20(4):360–367
Mullany LK, Wong K-K, Marciano DC, Katsonis P, King-Crane ER, Ren YA, Lichtarge O, Richards JS (2015) Specific TP53 mutants overrepresented in ovarian cancer impact CNV, TP53 activity, responses to nutlin-3a, and cell survival. Neoplasia 17(10):789–803
Nery MF, Borges B, Dragalzew AC, Kohlsdorf T (2016) Selection on different genes with equivalent functions: the convergence story told by Hox genes along the evolution of aquatic mammalian lineages. BMC Evol Biol 16:1–9
Nguyen DX, Chiang AC, Zhang XH-F, Kim JY, Kris MG, Ladanyi M, Gerald WL, Massagué J (2009) WNT/TCF signaling through LEF1 and HOXB9 mediates lung adenocarcinoma metastasis. Cell 138(1):51–62
Osmond B, Facey CO, Zhang C, Boman BM (2022) HOXA9 overexpression contributes to stem cell overpopulation that drives development and growth of colorectal cancer. Int J Mol Sci 23(12):6799
Paço A, de Bessa A, Garcia S, Leitão Castro J, Costa-Pinto AR, Freitas R (2020) Roles of the HOX proteins in cancer invasion and metastasis. Cancers (Basel) 13(1):10
Pai P, Sukumar S (2020) HOX genes and the NF-κB pathway: a convergence of developmental biology, inflammation and cancer biology. Biochim Biophys Acta (BBA)-Rev Cancer 1874(2):188450
Parker HJ, Bronner ME, Krumlauf R (2016) The vertebrate Hox gene regulatory network for hindbrain segmentation: Evolution and diversification: Coupling of a Hox gene regulatory network to hindbrain segmentation is an ancient trait originating at the base of vertebrates. BioEssays 38(6):526–538
Parker HJ, Pushel I, Krumlauf R (2018) Coupling the roles of Hox genes to regulatory networks patterning cranial neural crest. Dev Biol 444:S67–S78
Paul S, Zhang X, He J-Q (2020) Homeobox gene Meis1 modulates cardiovascular regeneration. Semin Cell Dev Biol Elsevier 100:52–61
Platais C, Hakami F, Darda L, Lambert DW, Morgan R, Hunter KD (2016) The role of HOX genes in head and neck squamous cell carcinoma. J Oral Pathol Med 45(4):239–247
Popovic R, Erfurth F, Zeleznik-Le N (2008) Transcriptional complexity of the HOXA9 locus. Blood Cells Mol Dis 40(2):156–159
Raines AM, Magella B, Adam M, Potter SS (2015) Key pathways regulated by Hox A9, 10, 11/HoxD9, 10, 11 during limb development. BMC Dev Biol 15:1–15
Ramos-Mejía V, Navarro-Montero O, Ayllón V, Bueno C, Romero T, Real PJ, Menendez P (2014) HOXA9 promotes hematopoietic commitment of human embryonic stem cells. Blood 124(20):3065–3075
Rastogi S, Mishra SS, Arora MK, Kaithwas G, Banerjee S, Ravichandiran V, Roy S, Singh L (2023) Lactate acidosis and simultaneous recruitment of TGF-β leads to alter plasticity of hypoxic cancer cells in tumor microenvironment. Pharmacol Ther 250:108519
Sakiyama J-i, Yokouchi Y, Kuroiwa A (2000) Coordinated expression of Hoxb genes and signaling molecules during development of the chick respiratory tract. Dev Biol 227(1):12–27
Santaguida M, Schepers K, King B, Sabnis AJ, Forsberg EC, Attema JL, Braun BS, Passegué E (2009) JunB protects against myeloid malignancies by limiting hematopoietic stem cell proliferation and differentiation without affecting self-renewal. Cancer Cell 15(4):341–352
Seki H, Hayashida T, Jinno H, Hirose S, Sakata M, Takahashi M, Maheswaran S, Mukai M, Kitagawa Y (2012) HOXB9 expression promoting tumor cell proliferation and angiogenesis is associated with clinical outcomes in breast cancer patients. Ann Surg Oncol 19:1831–40
Sha L, Dong L, Lv L, Bai L, Ji X (2015) HOXB9 promotes epithelial-to-mesenchymal transition via transforming growth factor-β1 pathway in hepatocellular carcinoma cells. Clin Exp Med 15:55–64
Sharma P, Nag A (2014) CUL4A ubiquitin ligase: a promising drug target for cancer and other human diseases. Open Biol 4(2):130217
Shenoy US, Adiga D, Alhedyan F, Kabekkodu SP, Radhakrishnan R (2023) HOXA9 transcription factor is a double-edged sword: from development to cancer progression. Cancer Metastasis Rev 1–20
Shenoy US, Adiga D, Kabekkodu SP, Hunter KD, Radhakrishnan R (2022a) Molecular implications of HOX genes targeting multiple signaling pathways in cancer. Cell Biol Toxicol 38(1):1–30
Shenoy US, Morgan R, Hunter K, Kabekkodu SP, Radhakrishnan R (2022b) Integrated computational analysis reveals HOX genes cluster as oncogenic drivers in head and neck squamous cell carcinoma. Sci Rep 12(1):7952
Shrestha B, Ansari KI, Bhan A, Kasiri S, Hussain I, Mandal SS (2012) Homeodomain-containing protein HOXB 9 regulates expression of growth and angiogenic factors, facilitates tumor growth in vitro and is overexpressed in breast cancer tissue. FEBS J 279(19):3715–3726
Song J, Wang T, Xu W, Wang P, Wan J, Wang Y, Zhan J, Zhang H (2018) HOXB9 acetylation at K27 is responsible for its suppression of colon cancer progression. Cancer Lett 426:63–72
Song X, Wei C, Li X (2022) The signaling pathways associated with breast cancer bone metastasis. Front Oncol 12:855609
Sun M, Song C-X, Huang H, Frankenberger CA, Sankarasharma D, Gomes S, Chen P, Chen J, Chada KK, He C (2013) HMGA2/TET1/HOXA9 signaling pathway regulates breast cancer growth and metastasis. Proc Natl Acad Sci 110(24):9920–9925
Sun X, Song J, Zhang J, Zhan J, Fang W, Zhang H (2020) Acetylated HOXB9 at lysine 27 is of differential diagnostic value in patients with pancreatic ductal adenocarcinoma. Front Med 14:91–100
Sun C, Han C, Wang P, Jin Y, Sun Y, Qu L (2017) HOXB9 expression correlates with histological grade and prognosis in LSCC. BioMed Res Int 2017:3680305
Symeonidou V, Ottersbach K (2021) HOXA9/IRX1 expression pattern defines two subgroups of infant MLL-AF4-driven acute lymphoblastic leukemia. Exp Hematol 93:38-43. e5
Taghon T, Thys K, De Smedt M, Weerkamp F, Staal F, Plum J, Leclercq G (2003) Homeobox gene expression profile in human hematopoietic multipotent stem cells and T-cell progenitors: implications for human T-cell development. Leukemia 17(6):1157–1163
Tang L, Cao Y, Song X, Wang X, Li Y, Yu M, Li M, Liu X, Huang F, Chen F (2019) HOXC6 promotes migration, invasion and proliferation of esophageal squamous cell carcinoma cells via modulating expression of genes involved in malignant phenotypes. PeerJ 7:e6607
Tang L, Peng L, Tan C, Liu H, Chen P, Wang H (2022) Role of HOXA9 in solid tumors: mechanistic insights and therapeutic potential. Cancer Cell Int 22(1):349
Torres-Reyes LA, Alvarado-Ruiz L, Piña-Sánchez P, Martínez-Silva MG, Ramos-Solano M, Olimón-Andalón V, Ortiz-Lazareno PC, Hernández-Flores G, Bravo-Cuellar A, Aguilar-Lemarroy A (2014) Expression of transcription factor grainyhead-like 2 is diminished in cervical cancer. Int J Clin Exp Pathol 7(11):7409
Wan J, Xu W, Zhan J, Ma J, Li X, Xie Y, Wang J, Zhu W-g, Luo J, Zhang H (2016) PCAF-mediated acetylation of transcriptional factor HOXB9 suppresses lung adenocarcinoma progression by targeting oncogenic protein JMJD6. Nucleic Acids Res 44(22):10662–10675
Wan J, Liu H, Feng Q, Liu J, Ming L (2018) HOXB9 promotes endometrial cancer progression by targeting E2F3. Cell Death Dis 9(5):509
Wang W-M, Xu Y, Wang Y-H, Sun H-X, Sun Y-F, He Y-F, Zhu Q-F, Hu B, Zhang X, Xia J-L (2017) HOXB7 promotes tumor progression via bFGF-induced activation of MAPK/ERK pathway and indicated poor prognosis in hepatocellular carcinoma. Oncotarget 8(29):47121
Wilson L, Maden M (2005) The mechanisms of dorsoventral patterning in the vertebrate neural tube. Dev Biol 282(1):1–13
Winkler GS (2010) The mammalian anti-proliferative BTG/Tob protein family. J Cell Physiol 222(1):66–72
Wu SY, Rupaimoole R, Shen F, Pradeep S, Pecot CV, Ivan C, Nagaraja AS, Gharpure KM, Pham E, Hatakeyama H (2016) A miR-192-EGR1-HOXB9 regulatory network controls the angiogenic switch in cancer. Nat Commun 7(1):11169
Xiong H, Xiao H, Luo C, Chen L, Liu X, Hu Z, Zou S, Guan J, Yang D, Wang K (2019) GRP78 activates the Wnt/HOXB9 pathway to promote invasion and metastasis of hepatocellular carcinoma by chaperoning LRP6. Exp Cell Res 383(1):111493
Xiong R, Yin T, Gao J-l, Yuan Y-f (2020) HOXD9 activates the TGF-β/Smad signaling pathway to promote gastric cancer. OncoTargets Ther 13:2163–72
Xu B, Wellik DM (2011) Axial Hox9 activity establishes the posterior field in the developing forelimb. Proc Natl Acad Sci 108(12):4888–4891
Xu H, Wu S, Shen X, Wu D, Qin Z, Wang H, Chen X, Sun X (2020) Silencing of HOXB9 suppresses cellular proliferation, angiogenesis, migration and invasion of prostate cancer cells. J Biosci 45:1–12
Xu Q, Zhang Q, Dong M, Yu Y (2021) MicroRNA-638 inhibits the progression of breast cancer through targeting HOXA9 and suppressing Wnt/β-cadherin pathway. World J Surg Oncol 19:1–9
Xue M, Zhu F-Y, Chen L, Wang K (2017) HoxB9 promotes the migration and invasion via TGF-β1/Smad2/Slug signaling pathway in oral squamous cell carcinoma. Am J Transl Res 9(3):1151
Xue W, Zhang J, Tong H, Xie T, Chen X, Zhou B, Wu P, Zhong P, Du X, Guo Y (2020) Effects of BMPER, CXCL10, and HOXA9 on neovascularization during early-growth stage of primary high-grade glioma and their corresponding MRI biomarkers. Front Oncol 10:711
Yao Y, Liu C, Wang B, Guan X, Fang L, Zhan F, Sun H, Li H, Lou C, Yan F (2022) HOXB9 blocks cell cycle progression to inhibit pancreatic cancer cell proliferation through the DNMT1/RBL2/c-Myc axis. Cancer Lett 533:215595
Ye S, Xiong F, He X, Yuan Y, Li D, Ye D, Shi L, Lin Z, Zhao M, Feng S (2023) DNA hypermethylation-induced miR-182 silence targets BCL2 and HOXA9 to facilitate the self-renewal of leukemia stem cell, accelerate acute myeloid leukemia progression, and determine the sensitivity of BCL2 inhibitor venetoclax. Theranostics 13(1):77
Yi C, Wei W, Wan M, Chen Y, Zhang B, Wu W (2023) Expression Patterns of HOX Gene Family Defines Tumor Microenvironment and Immunotherapy in Hepatocellular Carcinoma. Appl Biochem Biotechnol 195(8):5072–5093
Yu M, Zhan J, Zhang H (2020) HOX family transcription factors: Related signaling pathways and post-translational modifications in cancer. Cell Signal 66:109469
Yuniati L, Scheijen B, van der Meer LT, van Leeuwen FN (2019) Tumor suppressors BTG1 and BTG2: Beyond growth control. J Cell Physiol 234(5):5379–5389
Zhan J, Wang P, Niu M, Wang Y, Zhu X, Guo Y, Zhang H (2015) High expression of transcriptional factor HoxB9 predicts poor prognosis in patients with lung adenocarcinoma. Histopathology 66(7):955–965
Zhang L, Wu Q, He C, Liang D, Yi Q, Shi J, Wan B, Yang R, Li L, Sha S (2019) HOXB9 inhibits proliferation in gastric carcinoma cells via suppression of phosphorylated-Akt and NF-κB-dependent Snail expression. Dig Liver Dis 51(1):157–165
Zhao P, Tan L, Ruan J, Wei X-P, Zheng Y, Zheng L-X, Jiang W-Q, Fang W-J (2015) Aberrant expression of HOXA5 and HOXA9 in AML. Asian Pac J Cancer Prev 16(9):3941–3944
Zhou M, Sacirbegovic F, Zhao K, Rosenberger S, Shlomchik WD (2020) T cell exhaustion and a failure in antigen presentation drive resistance to the graft-versus-leukemia effect. Nat Commun 11(1):4227
Zhussupova A, Hayashida T, Takahashi M, Miyao K, Okazaki H, Jinno H, Kitagawa Y (2014) An E2F1-HOXB9 transcriptional circuit is associated with breast cancer progression. PLoS One 9(8):e105285
Acknowledgements
The authors are thankful to Prince Sattam bin Abdulaziz University, Al-Kharj 11942, Saudi Arabia, for supporting this study (Project number: PSAU/2024/R/1445).
Funding
This study is supported via funding from Prince Sattam bin Abdulaziz University, Al-Kharj 11942, Saudi Arabia (Project number: PSAU/2024/R/1445).
Author information
Authors and Affiliations
Contributions
Ahmed Hjazi, Aiman Mohammed Baqir Al-Dhalimy, Pooja Bansal, Harpreet Kaur, Maytham T. Qasim, Israa Hussein Mohammed, Mahamedha Deorari, Mohammed Abed Jawad, Ahmed Hussein Zwamel: Conceptualization, Writing—Original Draft, Writing – review & editing. Saade Abdalkareem Jasim: Conceptualization, Supervision. All authors read and approved the final version of the work to be published.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Communicated by Ewa Ziętkiewicz.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hjazi, A., Jasim, S.A., Al-Dhalimy, A.M.B. et al. HOXA9 versus HOXB9; particular focus on their controversial role in tumor pathogenesis. J Appl Genetics (2024). https://doi.org/10.1007/s13353-024-00868-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13353-024-00868-x