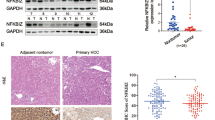
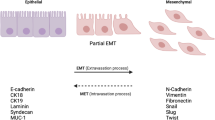
Abstract
The function of the epithelial-to-mesenchymal transition (EMT) during hepatocellular carcinoma (HCC) progression is well established. However, the regulatory mechanisms modulating this phenomenon remain unclear. Homeobox B9 (HOXB9) has been proposed as an oncogene in many cancer developments, but its function and underlying mechanisms in HCC metastasis remain unknown. HOXB9 modulates EMT through the transforming growth factor-β1 (TGF-β1) pathway, which is a recognized regulator of EMT in HCC cells. The knockdown of HOXB9 decreased the migration and invasion of HCC cells. Conversely, the HOXB9 overexpression led to an increase in the above-mentioned phenotypes in HCC cells. Further analysis of HOXB9-regulated cellular functions showed the ability of this transcription factor to induce EMT. Moreover, we demonstrated that the TGF-β1 pathway is important in HOXB9-induced EMT in HCC cells. These findings define a novel cellular mechanism regulated by HOXB9, which controls EMT phenotype in HCC. This study is the first to illustrate the pivotal function of HOXB9 in regulating the metastatic behavior of HCC cells.








Similar content being viewed by others
References
Bruix J, Boix L, Sala M, Llovet JM (2004) Focus on hepatocellular carcinoma. Cancer Cell 5(3):215–219
Zaret KS (2002) Regulatory phases of early liver development: paradigms of organogenesis. Nat Rev Genet 3(7):499–512
Lai E, Prezioso VR, Tao WF, Chen WS, Darnell JE Jr (1991) Hepatocyte nuclear factor 3 alpha belongs to a gene family in mammals that is homologous to the Drosophila homeotic gene fork head. Genes Dev 5(3):416–427
Bossard P, Zaret KS (1998) GATA transcription factors as potentiators of gut endoderm differentiation. Development 125(24):4909–4917
Abate-Shen C (2002) Deregulated homeobox gene expression in cancer: cause or consequence? Nat Rev Cancer 2(10):777–785
Nagel S, Burek C, Venturini L, Scherr M, Quentmeier H, Meyer C, Rosenwald A, Drexler HG, MacLeod RA (2007) Comprehensive analysis of homeobox genes in Hodgkin lymphoma cell lines identifies dysregulated expression of HOXB9 mediated via ERK5 signaling and BMI1. Blood 109(7):3015–3023
Nunes FD, de Almeida FC, Tucci R, de Sousa SC (2003) Homeobox genes: a molecular link between development and cancer. Pesqui Odontol Bras 17(1):94–98
Cantile M, Cindolo L, Napodano G, Altieri V, Cillo C (2003) Hyperexpression of locus C genes in the HOX network is strongly associated in vivo with human bladder transitional cell carcinomas. Oncogene 22(41):6462–6468
Chen F, Capecchi MR (1999) Paralogous mouse Hox genes, Hoxa9, Hoxb9, and Hoxd9, function together to control development of the mammary gland in response to pregnancy. Proc Natl Acad Sci USA 96(2):541–546
Miller GJ, Miller HL, van Bokhoven A, Lambert JR, Werahera PN, Schirripa O, Lucia MS, Nordeen SK (2003) Aberrant HOXC expression accompanies the malignant phenotype in human prostate. Cancer Res 63(18):5879–5888
Nguyen DX, Chiang AC, Zhang XH, Kim JY, Kris MG, Ladanyi M, Gerald WL, Massague J (2009) WNT/TCF signaling through LEF1 and HOXB9 mediates lung adenocarcinoma metastasis. Cell 138(1):51–62
Vider BZ, Zimber A, Hirsch D, Estlein D, Chastre E, Prevot S, Gespach C, Yaniv A, Gazit A (1997) Human colorectal carcinogenesis is associated with deregulation of homeobox gene expression. Biochem Biophys Res Commun 232(3):742–748
Shrestha B, Ansari KI, Bhan A, Kasiri S, Hussain I, Mandal SS (2012) Homeodomain-containing protein HOXB9 regulates expression of growth and angiogenic factors, facilitates tumor growth in vitro and is overexpressed in breast cancer tissue. FEBS J 279(19):3715–3726
Hayashida T, Takahashi F, Chiba N, Brachtel E, Takahashi M, Godin-Heymann N, Gross KW, Vivanco M, Wijendran V, Shioda T, Sgroi D, Donahoe PK, Maheswaran S (2010) HOXB9, a gene overexpressed in breast cancer, promotes tumorigenicity and lung metastasis. Proc Natl Acad Sci USA 107(3):1100–1105
Kalluri R, Weinberg RA (2009) The basics of epithelial-mesenchymal transition. J Clin Investig 119(6):1420–1428
Franco-Chuaire ML, Magda Carolina SC, Chuaire-Noack L (2013) Epithelial-mesenchymal transition (EMT): principles and clinical impact in cancer therapy. Invest Clin 54(2):186–205
Wang Y, Wen M, Kwon Y, Xu Y, Liu Y, Zhang P, He X, Wang Q, Huang Y, Jen KY, Labarge MA, You L, Kogan SC, Gray JW, Mao JH, Wei G (2014) CUL4A induces epithelial-mesenchymal transition and promotes cancer metastasis by regulating ZEB1 expression. Cancer Res 74(2):520–531
Lim J, Thiery JP (2012) Epithelial-mesenchymal transitions: insights from development. Development 139(19):3471–3486
Le Bras GF, Taubenslag KJ, Andl CD (2012) The regulation of cell–cell adhesion during epithelial-mesenchymal transition, motility and tumor progression. Cell Adh Migr 6(4):365–373
Gomes LR, Terra LF, Sogayar MC, Labriola L (2011) Epithelial-mesenchymal transition: implications in cancer progression and metastasis. Curr Pharm Biotechnol 12(11):1881–1890
Katsuno Y, Lamouille S, Derynck R (2013) TGF-beta signaling and epithelial-mesenchymal transition in cancer progression. Curr Opin Oncol 25(1):76–84
Drabsch Y, ten Dijke P (2012) TGF-beta signalling and its role in cancer progression and metastasis. Cancer Metastasis Rev 31(3–4):553–568
Fuxe J, Karlsson MC (2012) TGF-beta-induced epithelial-mesenchymal transition: a link between cancer and inflammation. Semin Cancer Biol 22(5–6):455–461
Xiong M, Jiang L, Zhou Y, Qiu W, Fang L, Tan R, Wen P, Yang J (2012) The miR-200 family regulates TGF-beta1-induced renal tubular epithelial to mesenchymal transition through Smad pathway by targeting ZEB1 and ZEB2 expression. Am J Physiol Renal Physiol 302(3):F369–F379
Naber HP, Drabsch Y, Snaar-Jagalska BE, ten Dijke P, van Laar T (2013) Snail and Slug, key regulators of TGF-beta-induced EMT, are sufficient for the induction of single-cell invasion. Biochem Biophys Res Commun 435(1):58–63
Kang H, Lee M, Jang SW (2013) Celastrol inhibits TGF-beta1-induced epithelial-mesenchymal transition by inhibiting Snail and regulating E-cadherin expression. Biochem Biophys Res Commun 437(4):550–556
Sun VC, Sarna L (2008) Symptom management in hepatocellular carcinoma. Clin J Oncol Nurs 12(5):759–766
Yang JD, Harmsen WS, Slettedahl SW, Chaiteerakij R, Enders FT, Therneau TM, Orsini L, Kim WR, Roberts LR (2011) Factors that affect risk for hepatocellular carcinoma and effects of surveillance. Clin Gastroenterol Hepatol 9(7):617–623.e611
Cabrera R, Nelson DR (2010) Review article: the management of hepatocellular carcinoma. Aliment Pharmacol Ther 31(4):461–476
Chang AC, Fu Y, Garside VC, Niessen K, Chang L, Fuller M, Setiadi A, Smrz J, Kyle A, Minchinton A, Marra M, Hoodless PA, Karsan A (2011) Notch initiates the endothelial-to-mesenchymal transition in the atrioventricular canal through autocrine activation of soluble guanylyl cyclase. Dev Cell 21(2):288–300
Larue L, Bellacosa A (2005) Epithelial-mesenchymal transition in development and cancer: role of phosphatidylinositol 3′ kinase/AKT pathways. Oncogene 24(50):7443–7454
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY, Brooks M, Reinhard F, Zhang CC, Shipitsin M, Campbell LL, Polyak K, Brisken C, Yang J, Weinberg RA (2008) The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 133(4):704–715
Lai W, Liu L, Zeng Y, Wu H, Xu H, Chen S, Chu Z (2013) KCNN4 channels participate in the EMT induced by PRL-3 in colorectal cancer. Med Oncol 30(2):566
Na DC, Lee JE, Yoo JE, Oh BK, Choi GH, Park YN (2011) Invasion and EMT-associated genes are up-regulated in B viral hepatocellular carcinoma with high expression of CD133-human and cell culture study. Exp Mol Pathol 90(1):66–73
Zheng X, Gai X, Wu Z, Liu Q, Yao Y (2013) Metastasin leads to poor prognosis of hepatocellular carcinoma through partly inducing EMT. Oncol Rep 29(5):1811–1818
Kapur RP, Gershon MD, Milla PJ, Pachnis V (2004) The influence of Hox genes and three intercellular signalling pathways on enteric neuromuscular development. Neurogastroenterol Motil 16(Suppl 1):8–13
Acknowledgments
This work was supported by the National Natural Science Foundation of China (NSFC H1617).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Lin Sha and Lei Dong have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sha, L., Dong, L., Lv, L. et al. HOXB9 promotes epithelial-to-mesenchymal transition via transforming growth factor-β1 pathway in hepatocellular carcinoma cells. Clin Exp Med 15, 55–64 (2015). https://doi.org/10.1007/s10238-014-0276-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-014-0276-7