Abstract
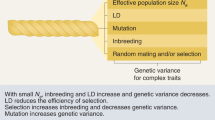
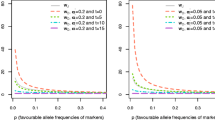
The genetic architecture of a quantitative trait refers to the number of genetic variants, allele frequencies, and effect sizes of variants that affect a trait and their mode of gene action. This study was conducted to investigate the effect of four shapes of allelic frequency distributions (constant, uniform, L-shaped and U-shaped) and different number of trait-affecting loci (50, 100, 200, 500) on allelic frequency changes, long term genetic response, and maintaining genetic variance. To this end, a population of 440 individuals composed of 40 males and 400 females as well as a genome of 200 cM consisting of two chromosomes and with a mutation rate of 2.5 × 10−5 per locus was simulated. Selection of superior animals was done using best linear unbiased prediction (BLUP) with assumption of infinitesimal model. Selection intensity was constant over 30 generations of selection. The highest genetic progress obtained when the allelic frequency had L-shaped distribution and number of trait-affecting loci was high (500). Although quantitative genetic theories predict the extinction of genetic variance due to artificial selection in long time, our results showed that under L- and U-shapped allelic frequency distributions, the additive genetic variance is persistent after 30 generations of selection. Further, presence or absence of selection limit can be an indication of low (<50) or high (>100) number of trait-affecting loci, respectively. It was concluded that the genetic architecture of complex traits is an important subject which should be considered in studies concerning long-term response to selection.





Similar content being viewed by others
References
Barton NH, Keightley PD (2002) Understanding quantitative genetic variation. Nat Rev Genet 3:11–21. doi:10.1038/nrg700
Bost B, de Vienne D, Hospital F, Moreau L, Dillmann C (2001) Genetic and nongenetic bases for the L-shaped distribution of quantitative trait loci effects. Genetics 157:1773–1787
Bulmer M (1971) The effect of selection on genetic variability. Am Nat 201–211
Chamberlain AJ, McPartlan HC, Goddard ME (2007) The number of loci that affect milk production traits in dairy cattle. Genetics 177:1117–1123
Cole JB, VanRaden PM, O’Connell JR, Van Tassell CP, Sonstegard TS, Schnabel RD, Taylor JF, Wiggans GR (2009) Distribution and location of genetic effects for dairy traits. J Dairy Sci 92:2931–2946
Conrad DF, Keebler JE, DePristo MA, Lindsay SJ, Zhang Y, Casals F, Idaghdour Y, Hartl CL, Torroja C, Garimella KV (2011) Variation in genome-wide mutation rates within and between human families. Nature 201:1
Crow JF (2008) Maintaining evolvability. J Genet 87:349–353
Crow JF, Kimura M (1970) An introduction to population genetics theory. Harper & Row, New York
Dempster E (1955) Genetic models in relation to animal breeding. Biometrics 11:535–536
Falconer DS, Mackay TF (1996) Introduction to quantitative genetics (4th edn), 4th edn. Longmans Green, UK
Hayes B, Goddard ME (2001) The distribution of the effects of genes affecting quantitative traits in livestock. Genet Sel Evol 33:209–229. doi:10.1051/gse:2001117
Hayes BJ, Pryce J, Chamberlain AJ, Bowman PJ, Goddard ME (2010) Genetic architecture of complex traits and accuracy of genomic prediction: coat colour, milk-fat percentage, and type in Holstein cattle as contrasting model traits. PLoS Genet 6:e1001139. doi:10.1371/journal.pgen.1001139
Hill WG (1982) Rates of change in quantitative traits from fixation of new mutations. PNAS 79:142–145
Hill WG (1998) Selection with recurrent backcrossing to develop congenic lines for quantitative trait loci analysis. Genetics 148:1341–1352
Hill WG (2005) A Century of corn selection. Science 307:683–684
Hill WG (2010) Understanding and using quantitative genetic variation. Philos Trans R Soc Lond B Biol Sci 365:73–85
Hill WG, Goddard ME, Visscher PM (2008) Data and theory point to mainly additive genetic variance for complex traits. PLoS Genet 4:e1000008
Huang W, Richards S, Carbone MA, Zhu D, Anholt RR, Ayroles JF, Duncan L, Jordan KW, Lawrence F, Magwire MM (2012) Epistasis dominates the genetic architecture of Drosophila quantitative traits. PNAS 109:15553–15559
Keightley PD, Halligan DL (2009) Analysis and implications of mutational variation. Genetica 136:359–369
Laurie CC, Chasalow SD, LeDeaux JR, McCarroll R, Bush D, Hauge B, Lai C, Clark D, Rocheford TR, Dudley JW (2004) The genetic architecture of response to long-term artificial selection for oil concentration in the maize kernel. Genetics 168:2141–2155
Lynch M, Walsh B (1998) Genetics and analysis of quantitative traits. Sinauer, Sunderland, MA
Mackay TFC (2009) A-maize-ing diversity. Science 325:688–689. doi:10.1126/science.1178420
Meuwissen T, Goddard M (2010) Accurate prediction of genetic values for complex traits by whole-genome resequencing. Genetics 185:623–631
Pimentel ECG, Erbe M, König S, Simianer H (2011) Genome partitioning of genetic variation for milk production and composition traits in holstein cattle. Front Genet 2:19. doi:10.3389/fgene.2011.00019
Roberts R (1966) The limits to artificial selection for body weight in the mouse II. The Genetic Nature of the Limits. Genet Res 8:361–375
Robertson FW (1955) Selection response and the properties of genetic variation. Cold spring harbor symposia on quantitative biology. Cold Spring Harbor Laboratory Press, pp 166–177
Robertson A (1960) A theory of limits in artificial selection. Proc R Soc Lond Ser B Biol Sci 153:234–249
Vinkhuyzen AA, Wray NR, Yang J, Goddard ME, Visscher P (2013) Estimation and partitioning of heritability in human populations using whole genome analysis methods. Ann Rev Genet 47:1
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abdollahi-Arpanahi, R., Pakdel, A., Nejati-Javaremi, A. et al. The relation between the genetic architecture of quantitative traits and long-term genetic response. J Appl Genetics 55, 373–381 (2014). https://doi.org/10.1007/s13353-014-0205-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13353-014-0205-1