Abstract
Dengue is a notorious viral infection, which affects a large segment of world populations in absence of vaccines and anti-viral treatment. The current study evaluates role of effective siRNA in dengue virus replication. Eight siRNA were synthesized against five different genes (Capsid, CprM, NS1, NS3 and NS5) of all serotypes of dengue virus. All serotype of DV were transfected with all synthesized siRNA in vitro, using BHK-21 cell lines. Culture fluid from test and control was tested by Real time PCR for CT value comparison in siRNA treated cell line (test) and untreated cell line (controls). Percent knockdown (%KD) was calculated by ∆∆CT methods to know the difference in test and control CT value. It was found that siRNA targeted against capsid gene worked best and showed inhibition of all four DV serotypes. DV-1, DV-2, DV-3 and DV-4 showed 93.8%, 99.3%, 87.5% and 93.8% knock down (%KD) respectively by siRNA targeted against capsid gene. Additionally, Si2 (target CprM gene 60-899) and Si 6 (target NS1 gene 3007-3025) were also showing inhibition of replication. Most serotypes of DV (with few exceptions) were not inhibited by siRNA targeted against NS-1, NS-3, and NS-5 genes. Animal studies using siRNAs are warranted to establish their therapeutic role.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Dengue virus (DV) infection is responsible for significant mortality and morbidity in world. Due to lack of effective strategy, scientists are unable to find specific remedy against it. Dengue virus is a mosquito borne, single, positive-strand RNA virus of the family Flaviviridae, genus Flavivirus [6]. Four known serotypes (DV 1–4) of the virus are circulating in India [8]. The viral genome is about 11,000 bases of single stranded RNA that codes for three structural protein, capsid protein C (114 Amino acids), membrane protein (166 Amino acids), envelope protein (495 amino acids) and seven non-structural proteins, NS1 (352 amino acid), NS2a (218 amino acid), NS2b (130 amino acid), NS3 (619 amino acid), NS4a (127 amino acid), NS4b (249 amino acid) and NS5 (899 amino acid). It also includes short non-coding regions on both the 5′ and 3′ ends.
All over the world multiple tools are being tried to manage dengue virus infection. RNAi (RNA interference) is one such strategy which has shown promising results in inhibiting other virus replication [4, 13]. RNAi is sequence specific, effective gene silencing process that plays important role in defence against viruses [18]. Several studies have been done for prevention of dengue virus replication through RNAi [9, 25, 29]. RNAi works in cells by either suppressing transcription or by activating a sequence specific RNA degradation process. There are two important components of RNAi; miRNA and siRNA. siRNA (small interfering RNA) can be synthesized artificially, hence can be a successful tool in inhibiting viruses in vitro. Therefore, the present study was planned to study the effect of siRNA targeting five genes (Capsid, CprM, NS1, NS3 and NS3) of dengue virus genome on all four serotypes of dengue virus in vitro.
Materials and methods
siRNA design and synthesis
The nucleotide sequence of standard strains of all four DENV serotypes were retrieved from NCBI database (https://www.ncbi.nlm.nih.gov/genome/?term=dengue+virus) with accession numbers; EU081255.1 (DV-1), FJ467493.1 (DV-2), KU216209.1 (DV-3) and KT794007.1 (DV-4). Selection of best possible siRNAs against the chosen target positions for all four DV serotypes were carried out with the help of I-score designer. It includes nine different siRNA designing scores of Ui-Tei, Amarzguioui, Hsieh‚Takasaki, s-Biopredsi‚ I-Score‚ Reynolds‚ Katoh and DSIR (https://www.med.nagoyau.ac.jp/neurogenetics/i_Score/i_score.html) accessed on 20.3.2017. To design best possible siRNA, target sequences were also screened using Gene runner, siRNA scales, I-score designer and Reynolds’s guidelines [21]. Eight best designed siRNA as shown in Table 1 were synthesized and purified by HPLC (synthesized by Ambion, USA). Negative control (Cat#AM4611) siRNA was labeled with FAM and purified for visualization after transfection (synthesized by Ambion, USA).
Cell lines maintenance and virus propagation
BHK-21cells procured from the National Institute of virology, Pune, India were cultured in 25cm2 cell culture flasks (Corning, USA) and routinely maintained at 37 °C in a 5% CO2 incubator (New Brunswick, USA). Freshly low passaged BHK-21 cells were used in the experiment. Cells were passaged in mid-exponential phase of culture at every 3–4 days at a split ratio of 1:4 after treatment with 0.25% trypsin solution and fed with fresh 5 ml complete cell culture medium containing minimum essential media (DMEM) supplemented with 10% FBS, 3.8 g/L sodium bicarbonate, 100 U/ml penicillin, and 100 µg/ml streptomycin at 37 °C in 5% CO2 [20]. Standard strains of Dengue serotypes 1–4 were procured from ATCC (10801 University Boulevard, Manassas, Virginia, USA) in 1 ml Cryovials (NR-86-D4, NR-82-D2, NR-80-D3 and NR-84-D1). All four dengue serotype were propagated in BHK-21 cells. For virus propagation, BHK-21 cells were inoculated with 200 µl viral stock of 50PFU/ µl of standard strains of Dengue serotypes 1–4. Post inoculation flasks were incubated for 2 h at 37 °C in a 5% CO2 incubator to allow virus adsorption. Later, 5 ml of maintenance medium was added and the flask was incubated in 5% CO2 incubator at 37 °C and observed regularly to monitor the cytopathic effect (CPE). Once majority of cells showed CPE, virus titration was done by TCID50 and plaque assay [20]. Culture fluid was harvested and store at −70 °C.
Transfection by FAM labeled siRNA
To check the adaptability of BHK-21 cell lines for transfection, this experiment was done before transfection experiments. BHK21 cells were seeded over 22 mm round cover slips placed in wells of 6 well tissue culture plates (Nunc, USA). Plates were incubated at 37º C in a 5% CO2 incubator to allow adherence and to reach 70–80% confluence of cells at the time of transfection. 1.5 µl LipofectamineTM 3000 (Invitrogen, USA) and 2 µl FAM labeled negative control siRNA was added in a tube and kept at dark for 20 min to allow the siRNA-Lipofectamine complex formation. In the meantime, cells were rinsed thrice with serum free medium. The siRNA- Lipofectamine complex was overlaid on cells in an even manner and was incubated at 37ºC for 2 h in a CO2 incubator. After this incubation transfection mixture was replaced with maintenance medium and plate was further incubated. Plate was taken out of the incubator after the completion of predetermined time period (at 24 h, at 48 h and at 72 h). Cover glass were removed from the culture wells after 24 h, 48 h and 72 h of incubation and examined to determine the minimum time taken for transfection. For examination cover slips were washed thrice with 0.05 M PBS (pH 7.4). Cells were fixed using chilled methanol. The dried cover slips were taken out and mounted onto 1 mm clean flat glass slides using mountant (65% glycerol in PBS) and observed under a fluorescent microscope [34]. The successful transfection means at least 75% cells should show the fluorescence. Successful transfection was not achieved till 72 h of incubation; hence, further experiments were not terminated before 72 h.
Experimental design
Five experimental groups were designed as per the details provided in Table 2. All experiments of transfection were done in 24 well plates with 70–80% confluent BHK-21 cell lines. Each experiment was repeated in triplicate. Before the transfection experiments were setup, siRNA and Lipofectamine 3000 transfection reagent (Invitrogen, Thermo Fisher scientific, USA) were prepared for each experiment. Total 5 nM of lyophilized commercially synthesized siRNA was suspended in 100 µl of RNase free water to make 50 µM Stock. Total 20 µl of 50 µM stock was diluted with 80 µl RNase free water for preparation of working stock. 2 µl of working siRNA, 96.5 µl DMEM and 1.5 µl Lipofectamine 3000 transfection reagents were used for each test and control well. Tubes with mix were lightly vortexed and incubated for 10–15 min. before use.
100 µl mixture solution of siRNA, Lipofectamine and DMEM was dispensed in each test and control wells containing 70–80% of confluent cells as per plan shown in Table 2. Plates were kept at 37 °C in 5% CO2 for 4 h. After 4 h of incubation, the solution was removed and plates were washed by 1X PBS followed by addition of 250 µl of 10% DMEM. Thereafter plates were incubated for 24 h at 37 °C in 5% CO2. The plates were washed with 1× PBS and challenged with 200 µl of each DV serotype of 50PFU/2 µl [34]. Plates were then incubated for 2 h at 37 °C in 5% CO2. After 2 h, all wells were washed with 1× PBS. Fresh 2% DMEM was added in all wells and incubated at 37 °C in 5% CO2. Total 100 µl culture fluid was removed from each well at 72 h for Real time PCR for CT value comparisons in siRNA treated wells (test) and untreated wells (controls).
Quantitative Real Time PCR analysis for viral load
RNA was extracted from culture fluid taken from test and control wells, by Qiagen kit (Valencia, CA, USA). The extracted RNA was subjected to real time RT-PCR. CDC (Centers for Disease Control and Prevention, Atlanta, Georgia, USA) approved primers and probe was used. Amplicons sizes were 112 bp, 78 bp, 74 bp and 89 bp respectively for D1, D2, D3 and D4 [23]. Amplification conditions were: 45 cycle of denaturation at 95 °C for 15 s, annealing at 55 °C for 20 s and extension at 72 °C for 20 s using 7500 ABI real time PCR machine (Applied Biosystems™ 7500 Real-Time PCR Systems). Relative mRNA expression was calculated by using ΔΔCT method [3]. Percent knockdown (%KD) was calculated by subtracting the normalized ∆∆CT expression from 1 (defined by the level of expression for untreated sample) and multiplying by 100 (%KD).
Statistical analysis
All values were presented as mean ± S.D. Statistical significance was evaluated using the two-tailed Mann–Whitney U-test; P < 0.05 was considered significant.
Result and discussion
siRNA transfection
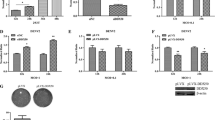
Experiment showed successful transfection of FAM labeled siRNA in the cell line. Fluorescence was observed at 24 h, 48 h and 72 h, with maximum FAM fluorescence was seen at 72 h. Therefore all experiments were set up for maximum period of up to 72 h (Fig. 1).
Inhibitory effect of siRNA on Dengue virus replication
The details of inhibitory effect experiments are shown in Table 3. siRNA1 (target capsid gene 60–401) showed inhibition of all four serotype of DV replication in vitro (87.5–99.3% knockdown). siRNA2 (target CprM gene 60–899) showed inhibition of DV-2 and DV-4 (range 93.6–98% knockdown). There was limited inhibition of DV-1 and DV-3 replication (75% knockdown). siRNA 3 (Target NS1 gene 2385–3440) showed only 50% inhibition with DV-1 and DV-4 and 75% knockdown with DV-2 & DV-3. siRNA 4 (target NS3 gene 4485–6341) showed inhibition of DV-2 and DV-4 (range 96.9–98% knockdown). There was limited inhibition of DV-1 (75% knockdown) and DV-3 (50% knockdown) replication. siRNA 5 (target NS3 gene 4485–6341) showed limited inhibition of DV-4 replication (75% knockdown). There was no significant effect on DV-1, DV-2 and DV-3 replication (50% knockdown). siRNA 6 (target NS1 gene 3007–3025) showed inhibition of DV-1 and DV-3 (range 87–96% knockdown). There was limited inhibition of DV-2 and DV-4 replication (75% knockdown). siRNA 7 (target NS3 gene 5592–5610) showed limited inhibition of DV-1, DV-3 and DV-4 replication (75% knockdown) and no significant effect on DV-2. siRNA 8 (target NS5 gene 7819–7837) showed inhibition of DV-2 and DV-3 (87% knockdown). DV-1 and DV-4 showed only 75% knockdown (Table 3).
RNAi is believed to be an effective endogenous mechanism for host cells to defend against virus attack and has been applied as an exogenous measure to inhibit viral replication, such as HIV [15], influenza A virus [30], HBV [32] and SARS-CoV [26]. DV is one of the first animal viruses that could be efficiently inhibited by RNAi [31]. Some studies done on the siRNA against different target genes of DV in different cell lines/animals are listed in Table 4
In the present study, siRNA targeted against capsid gene (60–401 position) worked best and showed inhibition of replication all four DV serotypes (Table 3). Mature Capsid protein is highly basic protein of 12KDa [12]. N-Terminal region of DV capsid is involved in RNA binding and viral particle formation [22]. One previous study showed more than 98% inhibition of all DV serotypes using siRNA against Capsid gene in comparison to control siRNA. This study found that highly conserved sequence in DV capsid gene, if inhibited, can inhibit all DV serotypes in Huh-7 cell and DV-2 in AG129 mouse model [28].
In the present study siRNA against CprM gene was effective in inhibition only DV-2 and DV-4 serotype replication (Table 3). CprM, also termed as precursor membrane protein, comprises an N terminal domain, that is, predomain (part of membrane of an immature virus) followed by an M domain, stem region, and two transmembrane helices [16].There is a report to demonstrate that PrM gene of DENV-1 was successfully targeted by four synthetic siRNAs. siRNA were transfected in C6/36 cell line with DENV which showed reduced CPE (cytopathogenic effect) and increased cell survival by 2.26 fold. It also estimated reduction in viral RNA by about 97.54% in comparison to virus control group only at 7 dpi (days post infection) [34].
In the present study, siRNA targeting NS1 gene was effective against DV-1 and DV-3 serotype. NS1 protein plays an important role in structural stability and secretion of NS1 in dimeric form [27] and have a role in immune evasiveness, viral replication, viral assembly, viral maturation and release [24]. NS3 protein is responsible for helicase and ATPase/RTPase activities, and should theoretically have role in DV replication [7]. To the best of our knowledge, this is the first report on the role of siRNA targeting NS3 gene, which was found to be effective against DV-2 and DV-4 serotypes only.
Both 5′NTR and 3′NTR have also been proven to be potential targets for siRNA technology. There are two sites (sh-3c and sh-5b) in 5′NTR region which function as promoter for RNA synthesis. These sites with their complementary sequences in 3′NTR contribute interaction between two ends of genomic RNA. With the help of recombinant adenovirus –mediated expression of sh-5b siRNA, DV 1–4 were inhibited in Vero cell line depicting that 5′NTR can be used as target to inhibit all four DV serotypes [14]. NS5 possess guanylyl transferase, guanine N7 methyltransferase and nucleoside 2′O methyltransferase activities required for 5′ RNA capping. It also helps virus in immune evasion [5, 17] and possess RNA dependent RNA polymerase (RdRp) activity for de novo synthesis of viral RNA [10]. In the present study siRNA targeting NS5 gene was effective against DV-1 and DV-4. In past small interfering RNAs (FG-siRNAs) targeting conserved sequences in the NS4B and NS5 regions of the DENV genome are shown to inhibit DENV replication. The FG-siRNAs were effective against DENV-1, -3, and -4, but not DENV-2. siRNA specifically targeting the NS5 region of the DENV-2 genome (SG-siRNA) was designed and tested against two different DENV-2 strains, showing high levels of inhibition in both mammalian and insect cells [33].
The role of siRNA has been studied and proved to be effective for various viruses including West Nile virus (WNV), Japanese Encephalitis Virus (JEV), Influenza Virus, Human Immunodeficiency virus (HIV) etc. Jain et al. [11] identified siRNA that can be used as a universal therapeutic agent against anti-human Influenza virus. It showed 73.6% and 62.1% inhibition of Influenza A/pdm H1N1 and Influenza A/H3N2 respectively at 48 h post infection on MDCK cell line. The siRNAs targeting M gene have demonstrated effective inhibition of Influenza virus replication and provides a basis for the development of siRNA which can be used as universal anti-IAV therapeutic agent [11]. Studies done on the role of siRNA on Hepatitis C virus showed that siRNA targeted against 5′ untranslated region (5’UTR) of HCV genome achieved up to 80% suppression of replication. It also showed efficient delivery of RNAi into hepatocytes using DNA-based vectors, thereby demonstrating feasibility of siRNA based gene therapy in inhibiting HCV replication [35]. In a study on Chikungunya virus, the authors found a combination of siRNAs targeted against E2 and NS1 genes to have an additive effect leading to early and complete inhibition of virus replication in virus infected mice when administered 72 h post infection. They suggested that RNAi may constitute a novel therapeutic strategy for controlling CHIKV infection and transmission [19]. Similarly, siRNA targeting the NS5 gene showed significant antiviral activity against Japanese encephalitis virus infection in both cultured cells and mouse model [1]. siRNA activity was observed in neurons for shorter duration (3 weeks) and RVG9R-siFvEJW-treated animals succumbed to intracerebrally injected WNV a month after treatment. Thus, an effective siRNA reduced systemic exposure can be transformative for treating acute flavivirus neuroencephalitis, particularly in the elderly. This approach also offers better scope for strategic clinical testing in confirmed WNV disease [2].
Self life of siRNA is too short. siRNA molecules are only retained in 8.9% of the cells at the seventh day post transfection. Therefore, maintaining the continuity of RNA interference is crucial for the promotion and application of RNAi technology. There is no effectively vaccine or therapy for DV infection. In this scenario siRNA appears to be an attractive treatment modality, as it is safe chemotherapeutic agent because of its limited side effects and its specificity for viral genomes. However, limitations like lack of effective delivery mechanisms to reach target cells to knock out gene of interest.
In the present study, self-designed siRNA against Capsid gene effectively inhibited all serotypes of DV and appears promising. Additionally, results for siRNA against CprM and NS1 are also acceptable. These results provide a basis for further development of effective siRNA against Dengue virus.
Abbreviations
- RNAi:
-
RNA interference
- BHK-21:
-
Baby hamster kidney cell
- siRNA:
-
Small interfering RNA
- DV:
-
Dengue virus
- CPE:
-
Cytopathic effect
- PBS:
-
Phosphate buffer saline
- DMEM:
-
Dulbecco's Modified Eagle Medium
References
Anantpadma M, Vrati S. siRNA-mediated suppression of Japanese encephalitis virus replication in cultured cells and mice. J Antimicrob Chemother. 2012;67(2):444–51. https://doi.org/10.1093/jac/dkr487.
Beloor J, Maes N, Ullah I, Uchil P, Jackson A, Fikrig E, Lee SK, Kumar P. Small interfering RNA-mediated control of virus replication in the CNS is therapeutic and enables natural immunity to west Nile virus. Cell Host Microb. 2018;23(4):549–5563. https://doi.org/10.1016/j.chom.2018.03.001.
Bustin SA, editor. AZ of quantitative PCR. La Jolla, CA: International University Line; 2004–2006.
Castanotto D, Rossi JJ. The promises and pitfalls of RNA-interference-based therapeutics. Nature. 2009;22:426–33.
Chang DC, Hoang LT, Mohamed Naim AN, et al. Evasion of early innate immune response by 2′Omethylation of dengue genomic RNA. Virology. 2016;499:259–66.
Chen R, Vasilakis N. Dengue–quo tu et quo vadis? Viruses. 2011;9:1562–660.
Clum S, Ebner KE, Padmanabhan R. Cotranslational membrane insertion of the serine proteinase precursor NS2B-NS3 (Pro) of dengue virus type 2 is required for efficient in vitro processing and is mediated through the hydrophobic regions of NS2B. J Biol Chem. 1997;272(49):30715–23.
Gupta N, Srivastava S, Jain A, Chaturvedi UC. Dengue in India. Indian J Med Res. 2012;136:373–90.
Idrees S, Ashfaq UA. RNAi: antiviral therapy against dengue virus. Asian Pac J Trop Biomed. 2013;3:232–6.
Issur M, Geiss BJ, Bougie I, et al. The flavivirus NS5 protein is a true RNA guanylyltransferase that catalyzes a two-step reaction to form the RNA cap structure. RNA. 2009;15(12):2340–50.
Jain B, Jain A, Prakash O, Singh AK, Dangi T, Singh M, Singh KP. In vitro validation of self designed "universal human Influenza A siRNA. Indian J Exp Biol. 2015;53(8):514–21.
Jones CT, Ma L, Burgner JW, Groesch TD, Post CB, Kuhn RJ. Flavivirus capsid is a dimeric alpha-helical protein. J Virol. 2003;77(12):7143–9.
Kakumani PK, Ponia SS, Sood V, Chinnappan M, Banerjea AC, Medigeshi GR, Malhotra P, Mukherjee SK, Bhatnagar RK. Role of RNA interference (RNAi) in dengue virus replication and identification of NS4B as an RNAi suppressor. J Virol. 2013;87(16):8870–83. https://doi.org/10.1128/JVI.02774-12.
Korrapati AB, Swaminathan G, Singh A, Khanna N, Swaminathan S. Adenovirus delivered short hairpin RNA targeting a conserved site in the 5′ non-translated region inhibits all four serotypes of dengue viruses. PLoS Negl Trop Dis. 2012;6:e1735.
Lee SK, Dykxhoorn DM, Kumar P, Ranjbar S, Song E, Maliszewski LE, François-Bongarçon V, Goldfeld A, Swamy NM, Lieberman J, Shankar P. Lentiviral delivery of short hairpin RNAs protects CD4 T cells from multiple clades and primary isolates of HIV. Blood. 2005;106(3):818–26. https://doi.org/10.1182/blood-2004-10-3959.
Li L, Lok SM, Yu IM, et al. The flavivirus precursor membrane envelope protein complex: structure and maturation. Science. 2008;319(5871):1830–4.
Liu L, Dong H, Chen H, et al. Flavivirus RNA cap methyl-transferase: structure, function and inhibition. Front Biol. 2010;5(4):286–303.
Obbard DJ, Gordon KH, Buck AH, Juggins FM. The evolution of RNAi as a defence against viruses and transposable elements. Philos Trans R Soc Lond B Biol Sci. 2009;364:99–115.
Parashar D, Paingankar MS, Kumar S, Gokhale MD, Sudeep AB, Shinde SB, Arankalle VA. Administration of E2 and NS1 siRNAs inhibit Chikungunya virus replication in vitro and protects mice infected with the virus. PLoS Negl Trop Dis. 2013;7(9):e2405. https://doi.org/10.1371/journal.pntd.0002405.
Phanthanawiboon S, A-nuegoonpipat A, Panngarm N, Limkittikul K, Ikuta K, Anantapreecha S, Kurosu T. Isolation and propagation of Dengue virus in Vero and BHK-21 cells expressing human DC-SIGN stably. J Virol Methods. 2014;209:55–61.
Prakash O, Jain B, Jain A. Designing of putative siRNA to inhibit dengue virus replication. Int J Res Dev Pharm Life Sci. 2018;7(5):3115–8.
Samsa MM, Mondotte JA, Caramelo JJ, Gamarnik AV. Uncoupling cis-acting RNA elements from coding sequences revealed a requirement of the N-terminal region of dengue virus capsid protein in virus particle formation. J Virol. 2012;86(2):1046–58.
Santiago GA, Vergne E, Quiles Y, Cosme J, Vazquez J, Medina JF, et al. Analytical and clinical performance of the CDC real time RT-PCR assay for detection and typing of dengue virus. PLoS Negl Trop Dis. 2013;7:7.
Scaturro P, Cortese M, Chatel-Chaix L, Fischl W, Bartenschlager R. Dengue virus non-structural protein 1 modulates infectious particle production via interaction with the structural proteins. PLoS Pathog. 2015;11(11):e1005277.
Schnettler E, Sterken MG, Leung JY, Metz SW, Geertsema C, Goldbach RW, Vlak JM, Kohl A, Khromykh AA, Pijlman GP. Noncoding flavivirus RNA displays RNA interference suppressor activity in insect and Mammalian cells. J Virol. 2012;86(24):13486–500. https://doi.org/10.1128/JVI.01104-12.
Shi Y, Yang DH, Xiong J, Jia J, Huang B, Jin YX. Inhibition of genes expression of SARS coronavirus by synthetic small interfering RNAs. Cell Res. 2005;15(3):193–200. https://doi.org/10.1038/sj.cr.7290286.
Somnuke P, Hauhart RE, Atkinson JP, Diamond MS, Avirutnan P. N-linked glycosylation of dengue virus NS1 protein modulates secretion, cell-surface expression, hexamer stability, and interactions with human complement. Virology. 2011;413(2):253–64.
Stein DA, Perry ST, Buck MD, Oehmen CS, Fischer MA, Poore E, et al. Inhibition of dengue virus infections in cell cultures and in AG129 mice by a small interfering RNA targeting a highly conserved sequence. J Virol. 2011;85:10154–66.
Subramanya S, Kim SS, Abraham S, Yao J, Kumar M, Kumar P, Haridas V, Lee SK, Shultz LD, Greiner DNM, Shankar P. Targeted delivery of small interfering RNA to human dendritic cells to suppress dengue virus infection and associated proinflammatory cytokine production. J Virol. 2010;84(5):2490–501. https://doi.org/10.1128/JVI.02105-08.
Sui HY, Zhao GY, Huang JD, Jin DY, Yuen KY, Zheng BJ. Small interfering RNA targeting m2 gene induces effective and long term inhibition of influenza A virus replication. PLoS One. 2009;4(5):e5671. https://doi.org/10.1371/journal.pone.0005671.
Sánchez-Vargas I, Travanty EA, Keene KM, Franz AW, Beaty BJ, Blair CD, Olson KE. RNA silencing, arthropod-borne virus and mosquitoes. Virus Res. 2004;102:65–74.
Tang KF, Chen M, Xie J, Song GB, Shi YS, Liu Q, Mei ZC, Steinle A, Ren H. Inhibition of hepatitis B virus replication by small interference RNA induces expression of MICA in HepG2.2.15 cells. Med Microbiol Immunol. 2009;198(1):27–32. https://doi.org/10.1007/s00430-008-0101-6.
Villegas PM, Ortega E, Villa-Tanaca L, Barrón BL, Torres-Flores J. Inhibition of dengue virus infection by small interfering RNAs that target highly conserved sequences in the NS4B or NS5 coding regions. Arch Virol. 2018;163(5):1331–5. https://doi.org/10.1007/s00705-018-3757-2.
Wu X, Hong H, Yue J, Wu Y, Li X, Jiang L, Li L, Li Q, Gao G, Yang X. Inhibitory effect of small interfering RNA on dengue virus replication in mosquito cells. Virol J. 2010;7(1):1–8.
Yokota T, Sakamoto N, Enomoto N, Tanabe Y, Miyagishi M, Maekawa S, Yi L, Kurosaki M, Taira K, Watanabe M, Mizusawa H. Inhibition of intracellular hepatitis C virus replication by synthetic and vector-derived small interfering RNAs. EMBO Rep. 2003;4(6):602–8. https://doi.org/10.1038/sj.embor.embor840.
Acknowledgements
Authors acknowledge the Indian Council of Medical Research (ICMR), New Delhi, for providing financial support (Viral Diagnostic Lab Grant No. VIR/70/2013/ECD-1).
Funding
Indian Council of Medical Research (ICMR), New Delhi.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The author(s) declare that they have no competing interests.
Ethics approval
83rd ECM II A/P7 (No.1671/ethics/R.cell.-17) Registration no.ECR/262/inst/UP/3013 (dated 4.2.17).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Prakash, O., Khan, D.N., Singh, T. et al. Effect of siRNA targeting dengue virus genes on replication of dengue virus: an in vitro experimental study. VirusDis. 32, 518–525 (2021). https://doi.org/10.1007/s13337-021-00700-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13337-021-00700-8