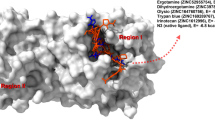
Abstract
Hepatitis B virus (HBV) is an etiological agent of viral hepatitis, which may lead to cirrhosis, and hepatocellular carcinoma. Current treatment strategies have not shown promising effect to date but various complications such as, drug toxicity-resistance have been reported. Study on newly discovered compounds, with minimal side effects, as specific HBV inhibitors is a fundamental subject introducing new biologic drugs. Here, we aimed to, by prediction, estimate interactions of HBF-0259 as a non-toxic anti-HBV compound on inhibiting the HBV through either interaction with the viral entry or HBsAg secreting factors using In Silico procedure. Molecular docking was performed by Hex 8.0.0 software to predict the interaction energy (Etot) between HBF-0259 and known cellular factors involved in HBV entry and HBsAg secreting factors. Hex 8.0.0 also employed to create protein–protein complexes. These interactions were then used to analyze the binding site of HBF-0259 within the assumed receptors by MGLTools software. Finally, the amino acid sequences involved in this interaction were aligned for any conservancy. Here, we showed that HBF-0259 Etot with CypA (−545.41 kcal/mol) and SCCA1 (499.68 kcal/mol), involved in HBsAg secretion and HBV integration, respectively, was higher than other interactions. Furthermore, HBF-0259 predicted interaction energy was even higher than those of CypA inhibitors. In addition, we claim that preS1 and/or preS2 regions within HBsAg are not suitable targets for HBF-0259. HBF-0259 has higher interaction energy with CypA and SCCA1, even more than other known receptors, co-receptors, viral ligands, and secretory factors. HBF-0259 could be introduced as potent anti-viral compound in which CypA and or SCCA1, as previously shown, are involved.


Similar content being viewed by others
References
Berman HM, Westbrook J, Feng Z, Gilliland G, Bhat TN, Weissig H, et al. The protein data bank. Nucleic Acids Res. 2000;28(1):235–42.
Bertoletti A, Gehring AJ. The immune response during hepatitis B virus infection. J Gen Virol. 2006;87(Pt 6):1439–49. doi:10.1099/vir.0.81920-0.
Budkowska A, Bedossa P, Groh F, Louise A, Pillot J. Fibronectin of human liver sinusoids binds hepatitis B virus: identification by an anti-idiotypic antibody bearing the internal image of the pre-S2 domain. J Virol. 1995;69(2):840–8.
Dougherty AM, Guo H, Westby G, Liu Y, Simsek E, Guo JT, et al. A substituted tetrahydro-tetrazolo-pyrimidine is a specific and novel inhibitor of hepatitis B virus surface antigen secretion. Antimicrob Agents Chemother. 2007;51(12):4427–37. doi:10.1128/aac.00541-07.
Fischer G, Bang H, Mech C. Determination of enzymatic catalysis for the cis-trans-isomerization of peptide binding in proline-containing peptides. Biomed Biochim Acta. 1984;43(10):1101–11.
Fletcher SP, Delaney WE. New therapeutic targets and drugs for the treatment of chronic hepatitis B. Semin Liver Dis. 2013;33(2):130–7. doi:10.1055/s-0033-1345713.
Furnham N, Holliday GL, de Beer TA, Jacobsen JO, Pearson WR, Thornton JM. The Catalytic Site Atlas 2.0: cataloging catalytic sites and residues identified in enzymes. Nucleic Acids Res. 2014;42(Database issue):D485–9. doi:10.1093/nar/gkt1243.
Hao Z, Zheng L, Kluwe L, Huang W. Ferritin light chain and squamous cell carcinoma antigen 1 are coreceptors for cellular attachment and entry of hepatitis B virus. Int J Nanomed. 2012;7:827–34. doi:10.2147/IJN.S27803.
Huang Z, Mou L, Shen Q, Lu S, Li C, Liu X, et al. ASD v2.0: updated content and novel features focusing on allosteric regulation. Nucleic Acids Research. 2014;42(Database issue):D510–6. doi:10.1093/nar/gkt1247.
Inoue J, Krueger EW, Chen J, Cao H, Ninomiya M, McNiven MA. HBV secretion is regulated through the activation of endocytic and autophagic compartments mediated by Rab7 stimulation. J Cell Sci. 2015;128(9):1696–706. doi:10.1242/jcs.158097.
Kallen J, Mikol V, Taylor P, Walkinshaw MD. X-ray structures and analysis of 11 cyclosporin derivatives complexed with cyclophilin A. J Mol Biol. 1998;283(2):435–49.
Konig A, Doring B, Mohr C, Geipel A, Geyer J, Glebe D. Kinetics of the bile acid transporter and hepatitis B virus receptor Na+/taurocholate cotransporting polypeptide (NTCP) in hepatocytes. J Hepatol. 2014;61(4):867–75. doi:10.1016/j.jhep.2014.05.018.
Liu Y, Gao P. Modulation of hepatitis B surface antigen secretion by annexin II expressed in hepatitis B virusproducing hepatoma cells. Mol Med Rep. 2014;10(6):3113–7. doi:10.3892/mmr.2014.2602.
Lovell SC, Davis IW, Arendall WB 3rd, de Bakker PI, Word JM, Prisant MG, et al. Structure validation by Calpha geometry: phi, psi and Cbeta deviation. Proteins. 2003;50(3):437–50. doi:10.1002/prot.10286.
Ma S, Boerner JE, TiongYip C, Weidmann B, Ryder NS, Cooreman MP, et al. NIM811, a cyclophilin inhibitor, exhibits potent in vitro activity against hepatitis C virus alone or in combination with alpha interferon. Antimicrob Agents Chemother. 2006;50(9):2976–82. doi:10.1128/AAC.00310-06.
Maupetit J, Derreumaux P, Tuffery P. PEP-FOLD: an online resource for de novo peptide structure prediction. Nucleic Acids Res. 2009;37(Web Server issue):W498–503. doi:10.1093/nar/gkp323.
Meier M, Bider MD, Malashkevich VN, Spiess M, Burkhard P. Crystal structure of the carbohydrate recognition domain of the H1 subunit of the asialoglycoprotein receptor. J Mol Biol. 2000;300(4):857–65. doi:10.1006/jmbi.2000.3853.
Moore PL, Ong S, Harrison TJ. Squamous cell carcinoma antigen 1-mediated binding of hepatitis B virus to hepatocytes does not involve the hepatic serpin clearance system. J Biol Chem. 2003;278(47):46709–17. doi:10.1074/jbc.M302842200.
Neurath AR, Kent SB, Strick N, Parker K. Identification and chemical synthesis of a host cell receptor binding site on hepatitis B virus. Cell. 1986;46(3):429–36.
Ni Y, Lempp FA, Mehrle S, Nkongolo S, Kaufman C, Falth M, et al. Hepatitis B and D viruses exploit sodium taurocholate co-transporting polypeptide for species-specific entry into hepatocytes. Gastroenterology. 2014;146(4):1070–83. doi:10.1053/j.gastro.2013.12.024.
Nigro P, Pompilio G, Capogrossi MC. Cyclophilin A: a key player for human disease. Cell Death Dis. 2013;4:e888. doi:10.1038/cddis.2013.410.
Obchoei S, Wongkhan S, Wongkham C, Li M, Yao Q, Chen C. Cyclophilin A: potential functions and therapeutic target for human cancer. Med Sci Monit Int Med J Exp Clin Res. 2009;15(11):RA221–32.
Pence HE, Williams A. ChemSpider: an online chemical information resource. J Chem Educ. 2010;87(11):1123–4.
Peng J, Xu J. RaptorX: exploiting structure information for protein alignment by statistical inference. Proteins. 2011;79(Suppl 10):161–71. doi:10.1002/prot.23175.
Pettersen EF, Goddard TD, Huang CC, Couch GS, Greenblatt DM, Meng EC, et al. UCSF Chimera—a visualization system for exploratory research and analysis. J Comput Chem. 2004;25(13):1605–12. doi:10.1002/jcc.20084.
Phillips S, Chokshi S, Chatterji U, Riva A, Bobardt M, Williams R, et al. Alisporivir inhibition of hepatocyte cyclophilins reduces HBV replication and hepatitis B surface antigen production. Gastroenterology. 2015;148(2):403–14. doi:10.1053/j.gastro.2014.10.004.
Quarato G, D’Aprile A, Gavillet B, Vuagniaux G, Moradpour D, Capitanio N, et al. The cyclophilin inhibitor alisporivir prevents hepatitis C virus-mediated mitochondrial dysfunction. Hepatology. 2012;55(5):1333–43. doi:10.1002/hep.25514.
Schadler S, Hildt E. HBV life cycle: entry and morphogenesis. Viruses. 2009;1(2):185–209. doi:10.3390/v1020185.
Teissier E, Penin F, Pecheur EI. Targeting cell entry of enveloped viruses as an antiviral strategy. Molecules (Basel, Switzerland). 2011;16(1):221–50. doi:10.3390/molecules16010221.
Tian X, Zhao C, Zhu H, She W, Zhang J, Liu J, et al. Hepatitis B virus (HBV) surface antigen interacts with and promotes cyclophilin a secretion: possible link to pathogenesis of HBV infection. J Virol. 2010;84(7):3373–81. doi:10.1128/JVI.02555-09.
Treichel U, zum Buschenfelde KHM, Stockert RJ, Poralla T, Gerken G. The asialoglycoprotein receptor mediates hepatic binding and uptake of natural hepatitis B virus particles derived from viraemic carriers. J Gen Virol. 1994;75(Pt 11):3021–9. doi:10.1099/0022-1317-75-11-3021.
Trepo C, Chan HL, Lok A. Hepatitis B virus infection. Lancet. 2014;384(9959):2053–63. doi:10.1016/S0140-6736(14)60220-8.
UniProt C. The Universal Protein Resource (UniProt) 2009. Nucleic acids research. 2009;37(Database issue):D169–74. doi:10.1093/nar/gkn664.
Vakonakis I, Staunton D, Rooney LM, Campbell ID. Interdomain association in fibronectin: insight into cryptic sites and fibrillogenesis. EMBO J. 2007;26(10):2575–83. doi:10.1038/sj.emboj.7601694.
Wang Z, Li C, Ellenburg M, Soistman E, Ruble J, Wright B, et al. Structure of human ferritin L chain. Acta Crystallogr D Biol Crystallogr. 2006;62(Pt 7):800–6. doi:10.1107/S0907444906018294.
Wu M, Wang T, Loh E, Hong W, Song H. Structural basis for recruitment of RILP by small GTPase Rab7. EMBO J. 2005;24(8):1491–501. doi:10.1038/sj.emboj.7600643.
Xia HB, Chen ZY, Chen XG. Overexpression of hepatitis B virus-binding protein, squamous cell carcinoma antigen 1, extends retention of hepatitis B virus in mouse liver. Acta Biochim Biophys Sin. 2006;38(7):484–91.
Xu YB, Yang L, Wang GF, Tong XK, Wang YJ, Yu Y, et al. Benzimidazole derivative, BM601, a novel inhibitor of hepatitis B virus and HBsAg secretion. Antivir Res. 2014;107:6–15. doi:10.1016/j.antiviral.2014.04.002.
Xun YH, Zhang YJ, Pan QC, Mao RC, Qin YL, Liu HY, et al. Metformin inhibits hepatitis B virus protein production and replication in human hepatoma cells. J Viral Hepat. 2014;21(8):597–603. doi:10.1111/jvh.12187.
Yan H, Zhong G, Xu G, He W, Jing Z, Gao Z, et al. Sodium taurocholate cotransporting polypeptide is a functional receptor for human hepatitis B and D virus. eLife. 2012;1:e00049. doi:10.7554/eLife.00049.
Yang J, Wang F, Tian L, Su J, Zhu X, Lin L, et al. Fibronectin and asialoglyprotein receptor mediate hepatitis B surface antigen binding to the cell surface. Arch Virol. 2010;155(6):881–8. doi:10.1007/s00705-010-0657-5.
Yu W, Goddard C, Clearfield E, Mills C, Xiao T, Guo H, et al. Design, synthesis, and biological evaluation of triazolo-pyrimidine derivatives as novel inhibitors of hepatitis B virus surface antigen (HBsAg) secretion. J Med Chem. 2011;54(16):5660–70. doi:10.1021/jm200696v.
Zheng B, Matoba Y, Kumagai T, Katagiri C, Hibino T, Sugiyama M. Crystal structure of SCCA1 and insight about the interaction with JNK1. Biochem Biophys Res Commun. 2009;380(1):143–7. doi:10.1016/j.bbrc.2009.01.057.
Acknowledgments
This work has supported by the School of Medicine, Golestan University of Medical Sciences, Gorgan, Iran.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mohebbi, A., Mohammadi, S. & Memarian, A. Prediction of HBF-0259 interactions with hepatitis B Virus receptors and surface antigen secretory factors. VirusDis. 27, 234–241 (2016). https://doi.org/10.1007/s13337-016-0333-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13337-016-0333-9