Abstract
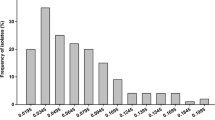
Triflumizole is a broad-spectrum fungicide in the chemical group of imidazoles. In the present study, the biological activity of triflumizole on Botrytis cinerea was determined in vitro, and the protective and curative activity against the fungal pathogen was determined on cucumber true leaves. Moreover, the baseline sensitivity for mycelial growth of B. cinerea to triflumizole was determined in vitro with a set of 79 isolates obtained from different geographical regions in Shandong Province of China. Inhibition of mycelial growth was found to be the most sensitive growth stage affected by triflumizole, whereas spore germination of B. cinerea was the least sensitive growth stage. Triflumizole provided excellent curative activity against B. cinerea at concentrations of 100 and 200 μg mL−1 and also was highly protective against the fungus at 200 μg mL−1. However, the control efficacy of protective activity and curative activity were relatively low when the concentration decreased to 50 μg mL−1. The baseline sensitivities were distributed as a unimodal curve, with a mean EC50 value of 0.58 μg mL−1. The individual EC50 values for triflumizole ranged from 0.15 to 1.49 μg mL−1. Triflumizole had no cross-resistance with other botryticides, including carbendazim, iprodione, diethofencarb, pyrimethanil and boscalid. However, cross-resistance to SPY-Z048, which is also a sterol biosynthesis inhibitor, was observed. In field trials in 2014 and 2015, triflumizole used at a concentration of 150 g a.i. ha−1 provided control efficacy on leaf ranging from 80.76 to 86.32 %, and its control efficacy on fruit could reach 84.93 % ~89.06 %, which were higher than the control efficacy of other treatments. These results suggested that triflumizole will be a good alternative fungicide for control of grey mould.



Similar content being viewed by others
References
Bardas GA, Veloukas T, Koutita O, Karaoglanidis GS (2010) Multiple resistance of Botrytis cinerea from kiwifruit to SDHIs, QoIs and fungicides of other chemical groups. Pest Manag Sci 66:967–973
Duan YB, Ge CY, Liu SM, Wang JX, Zhou MG (2013) A two-component histidine kinase Shk1 controls stress response, sclerotial formation and fungicide resistance in Sclerotinia sclerotiorum. Mol Plant Pathol 14:708–718
Elad Y (1992) Reduced sensitivity of Botrytis cinerea to two sterol biosynthesis-inhibiting fungicides: fenetrazole and fenethanil. Plant Pathol 41:47–54
Elad Y, Shtienberg D (1995) Botrytis cinerea in greenhouse vegetables: chemical, cultural, physiological and biological controls and their integration. Integr Pest Manag Rev 1:15–29
Fernández-Ortuño D, Chen F, Schnabel G (2012) Resistance to pyraclostrobin and boscalid in Botrytis cinerea isolates from strawberry fields in the Carolinas. Plant Dis 96:1198–1203
Hammer PE, Evensen KB, Janisiewicz WJ (1993) Postharvest control of Botrytis cinerea on cut rose flowers with pyrrolnitrin. Plant Dis 77:283–286
Han P, Liu XL, Liu PF, Si NG (2006) Study on the effect of SYP-Z048 on ergosterol biosynthesis of Botrytis cinerea by HPLC. Chin J Anal Chem 10:1467–1470
Hilber UW, Hilber-Bodmer M (1998) Genetic basis and monitoring of resistance of Botryotinia fuckeliana to anilinopyrimidines. Plant Dis 82:496–500
Hsiang T, Yang L, Barton W (1997) Baseline sensitivity and cross-resistance to demethylation-inhibiting fungicides in Ontario isolates of Sclerotinia homoeocarpa. Eur J Plant Pathol 103:409–416
Ishii H, Takeda H, Nagamatsu Y, Nakashima H (1990) Sensitivity of the pear scab fungus (Venturia nashicola) to three ergosterol biosynthesis-inhibiting fungicides. Pestic Sci 30:405–413
Leroux P, Fritz R, Debieu D, Albertini C, Lanen C, Bach J, Chapeland F (2002) Mechanisms of resistance to fungicides in field strains of Botrytis cinerea. Pest Manag Sci 58:876–888
Lin HK, Knoche HW (1974) Origin of sterols in uredospores of Uromyces phaseoli. Phytochemistry 13:1795–1799
Ma ZH, Morgan DP, Felts D, Michailides TJ (2002) Sensitivity of Botryosphaeria dothidea from California pistachio to tebuconazole. Crop Prot 21:829–835
Ma JY, Zhang XF, Wang WQ, Han XY, Ma ZQ, Ding HB (2009) The sensitivity of Botrytis cinerea to SYP-Z048 and cross resistance against diverse fungicides. Acta Phys Sin 36:61–64
Markoglou AN, Ziogas BN (2002) SBI-fungicides: fungicidal effectiveness and resistance in Botrytis cinerea. Phytopathol Mediterr 41:120–130
Mavroeidi VI, Shaw MW (2005) Sensitivity distributions and cross-resistance patterns of Mycosphaerella graminicola to fluquinconazole, prochloraz and azoxystrobin over a period of 9 years. Crop Prot 24:259–266
Moyano C, Gómez V, Melgarejo P (2004) Resistance to pyrimethanil and other fungicides in Botrytis cinerea populations collected on vegetable crops in Spain. J Phytopathol 152:484–490
Myresiotis CK, Karaoglanidis GS, Tzavella-Klonari K (2007) Resistance of Botrytis cinerea isolates from vegetable crops to annilinopyrimidine, phenylpyrrole, hydroxyanilide, benzimidazole, and dicarboximide fungicides. Plant Dis 91:407–413
Pontzen R, Scheinpflug H (1989) Effects of triazole fungicides on sterol biosynthesis during spore germination of Botrytis cinerea, Venturia inaequalis and Puccinia graminis f sp Tritici Netherlands. Journal of Plant Pathology 95:151–160
Robbertse B, van der Rijst M, van Aarde IMR, Lennox C, Crous PW (2001) DMI sensitivity and cross-resistance patterns of Rhynchosporium secalis isolates from South Africa. Crop Prot 20:97–102
Rodríguez A, Acosta A, Rodríguez C (2014) Fungicide resistance of Botrytis cinerea in tomato greenhouses in the Canary Islands and effectiveness of non-chemical treatments against gray mold. World J Microbiol Biotechnol 30:2397–2406
Russell PE (2004) Sensitivity Baselines in Fungicide Resistance Research and Management. Brussels, Belgium: Crop Life International: FRAC Monograph No. 3.
Siegel MR (1981) Sterol-inhibiting fungicides: effects on sterol biosynthesis and sites of action. Plant Dis 65:986–989
Stehmann C, de Waard MA (1996) Factors influencing activity of triazole fungicides towards Botrytis cinerea. Crop Prot 15:39–47
Tateishi H, Chida T (2000) Sensitivity of Fusarium moniliforme isolates to ipconazole. J Gen Plant Pathol 66:353–359
Veloukas T, Karaoglanidis GS (2012) Biological activity of the succinate dehydrogenase inhibitor fluopyram against Botrytis cinerea and fungal baseline sensitivity. Pest Manag Sci 68:858–864
Yuan NN, Chen SN, Zhai LX, Schnabel G, Yin LF, Luo CX (2013) Baseline sensitivity of Monilia yunnanensis to the DMI fungicides tebuconazole and triadimefon. Eur J Plant Pathol 136:651–655
Zhang CQ, Yuan SK, Sun HY, Qi ZQ, Zhou MG, Zhu GN (2007a) Sensitivity of Botrytis cinerea from vegetable greenhouses to boscalid. Plant Pathol 56:646–653
Zhang CQ, Zhu JW, Wei FL, Liu SY, Zhu GN (2007b) Sensitivity of Botrytis cinerea from greenhouse vegetables to DMIs and fenhexamid. Phytoparasitica 35:300–313
Zhang CQ, Hu JL, Wei FL, Zhu GN (2009) Evolution of resistance to different classes of fungicides in Botrytis cinerea from greenhouse vegetables in eastern China. Phytoparasitica 37:351–359
Zhang XK, Wu DX, Duan YB, Ge CY, Wang JX, Zhou MG, Chen CJ (2014) Biological characteristics and resistance analysis of the novel fungicide SYP-1620 against Botrytis cinerea. Pestic Biochem Physiol 114:72–78
Acknowledgments
This study was supported by the Research Award Fund for Excellent Young Scientist of Shandong Province (BS2011NY012).
Author information
Authors and Affiliations
Corresponding author
Additional information
Yingying Song and Datong Xu contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Song, Y., Xu, D., Lu, H. et al. Baseline sensitivity and efficacy of the sterol biosynthesis inhibitor triflumizole against Botrytis cinerea . Australasian Plant Pathol. 45, 65–72 (2016). https://doi.org/10.1007/s13313-015-0384-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13313-015-0384-1