Abstract
Morbidity and mortality associated with heart failure (HF) has remained high despite advances in therapy. Furthermore, HF-associated risk in patients with type 2 diabetes mellitus (T2D) is even higher than in patients without T2D owing to the strong reciprocal relationship between conditions. However, until recently, no therapy to treat patients with diabetes also reduced cardiovascular risks related to HF. Recent clinical studies (DAPA-HF, EMPEROR-Reduced and EMPEROR-Preserved, SOLOIST-WHF trial) and meta-analysis have demonstrated that sodium–glucose cotransporter-2 inhibitors (SGLT2i) are among the first antidiabetic drugs capable of reducing cardiovascular risks related to HF and improving the prognosis of patients with and without diabetes. Their pleiotropic mechanisms of action place them at the intersection of hemodynamic, metabolic, and neurohumoral pathways, with clear advantages for treating these patients independent of its glucose-lowering effect. Moreover, the benefits of SGLT2i were consistent across the cardiorenal continuum in different populations and clinical settings, which has led to different guidelines introducing SGLT2i as a first-line treatment for HF.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
In heart failure (HF), morbidity and mortality remain high despite advances in therapy, and new therapies are needed to improve the prognosis of these patients, mainly when type 2 diabetes mellitus (T2D) is also present. |
Patients with T2D show a very high risk of developing HF, but those with HF are at higher risk of developing T2D and a worse prognosis when both pathologies are present. |
Recent clinical studies (DAPA-HF, EMPEROR-Reduced and EMPEROR-Preserved, SOLOIST-WHF trial) and meta-analysis have demonstrated that sodium–glucose cotransporter-2 inhibitors (SGLT2i) are among the first antidiabetic drugs capable of reducing cardiovascular risks related to HF and consistently improving the prognosis in different populations and clinical settings. |
On the basis of this clinical evidence, several HF guidelines have introduced SGLT2i as a first-line HF treatment. |
Introduction
The incidence of heart failure (HF) is increasing and is projected to rise by 46% by 2030, according to the American Heart Association's 2017 Heart Disease and Stroke Statistics Update [1]. Its symptoms and multiorgan adverse effects lead to high morbidity, poor survival rates, and significant economic strain on health care systems worldwide [2].
In patients with HF, morbidity and mortality remain high despite advances in therapy. Patients with type 2 diabetes mellitus (T2D) who had five risk-factor variables within target ranges [glycated hemoglobin level, low-density lipoprotein cholesterol level (LDLc), albuminuria, smoking, and blood pressure] appeared to have little or no excess risk of death, myocardial infarction, or stroke compared with the general population. In contrast, the overall hazard ratio for hospitalization for HF among patients with no risk-factor variables outside the target ranges, compared with controls, was 1.45 [95% confidence interval (CI) 1.34–1.57] [3]. Recent estimates indicate that, with the current approach, a third of patients die within 1–2 years after the first admission [4] and 44% after 5 years [5]. Along with this high morbidity and mortality, there is a high rate of hospitalization and readmissions, becoming more likely and severe as the disease progresses. It has been estimated that more than 60% of patients discharged from the hospital with HF are readmitted within 1 year [6].
In this sense, and given the progressive nature of the disease, early identification of possible risk factors would allow the implementation of early intervention strategies to delay the progression or prevent the onset of HF before irreversible remodeling and functional impairment have occurred. In the last decade, several HF risk scores have been published, and most of them have reported a remarkably consistent list of the principal risk factors, including age, sex, coronary artery disease (CAD), myocardial infarction (MI), hypertension, obesity, and diabetes mellitus (DM), particularly T2D [7].
This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
General Mechanisms of Heart Failure (Preserved and Reduced)
Heart failure (HF) has recently been defined by the European Society of Cardiology (ESC) as a clinical syndrome, not a single pathological diagnosis, consisting of cardinal symptoms (e.g., breathlessness, fatigue, and ankle swelling) that may be accompanied by signs (e.g., pulmonary crackles, elevated jugular venous pressure, and peripheral edema). HF is due to a structural and/or functional abnormality of the heart that results in elevated intracardiac pressures and/or inadequate cardiac output at rest and/or during exercise [8].
It is considered a chronic, progressive condition that arises secondary to complex changes in the molecular and cellular composition of the heart that produce an initial decline in its pumping capacity. The molecular and cellular changes may arise from different factors such as sustained hemodynamic overload following a myocardial infarction or exposure to toxic chemotherapies, or may develop secondary to inherited genetic mutations that affect sarcomere function. Following this initial deterioration in heart-pumping capacity, various compensatory mechanisms are activated, including the renin–angiotensin system (RAS), the adrenergic nervous system, and the cytokine system. These systems can restore cardiovascular function to a normal homeostatic range in the short term, resulting in the patient remaining asymptomatic. However, over time, the sustained activation of these systems can lead to secondary end-organ damage within the ventricle, with worsening left ventricle (LV) remodeling and subsequent cardiac decompensation. As a result of worsening LV remodeling and cardiac decompensation, patients transition from asymptomatic to symptomatic HF [9].
HF is categorized into distinct phenotypes based on the left ventricular ejection fraction (LVEF) measurements. HF with reduced ejection fraction (HFrEF) is considered in patients with an LVEF ≤ 40%, with or without signs of clinical HF. Those patients with an LVEF between 41% and 49% have HF with mildly reduced EF (HFmrEF), and those with an LVEF ≥ 50% and symptoms and signs of HF, with evidence of structural and/or functional cardiac abnormalities and/or raised natriuretic peptides (NPs), have HF with preserved ejection fraction (HFpEF). For the diagnosis of HFmrEF, the presence of other evidence of structural heart disease (e.g., increased left atrial size, LV hypertrophy, or echocardiographic measures of impaired LV filling) makes the diagnosis more likely. For the diagnosis of HFpEF, the greater the number of abnormalities present, the higher the likelihood of HFpEF [8].
Although defining HFpEF represents a daunting task, a new entity is being characterized: the “pre-HFpEF stage”. The pre-HFpEF stage may affect an even more significant number of patients than other HF entities and requires the urgent attention of practicing clinicians [10]. Therefore, the definition of pre-HFpEF is taking shape as presenting in an asymptomatic patient (absence of signs or symptoms of HF) with preserved LVEF, structural heart abnormalities (similar to those reported for HFpEF), and elevated biomarker surrogates of cardiac dysfunction (mainly natriuretic peptides, with cut-point values similar to those reported for HFpEF). This stratification may aid in the early and correct identification of patients at increased risk of incident HF and death and may thus be suitable candidates for more stringent preventive strategies [11].
A distinction in the mechanisms implicated in HFrEF and HFpEF development has been described. It has been suggested that, while HFrEF is mainly due to a state of volume overload and systolic dysfunction, HFpEF may primarily result from systemic inflammation. Biomarker profiles specific for HFrEF are related to cellular metabolism and proliferation that promote LV enlargement and cause the LVEF to fall. In contrast, biomarker profiles specific for HFpEF are related to inflammation and extracellular matrix reorganization that limits LV distensibility and dilation, and EF is typically maintained at > 40–50% [12].
Heart Failure in Patients with and without T2DM, Current Understanding and Clinical Practice Consequences
T2D is an independent risk factor for HF in many hospital-based and population-based cardiovascular studies, and in particular, it has been estimated that, compared with patients without diabetes, the risk of HF is fivefold and twofold higher in women and men with diabetes, respectively [13]. Moreover, the presence of diabetes markedly increases the risk of 1-year adverse clinical outcomes in outpatients with HF independent of multiple common risk factors.
Nowadays, it has been demonstrated that diabetes and HF are closely related, and a strong reciprocal relationship between pathologies exists. Patients with T2D have an increased risk of developing HF, but also those with HF are at higher risk for the development of T2D and a worse prognosis when both pathologies are present [14].
Thus, in patients with T2D, HF has been reported to be the most frequent primary cardiovascular disease manifestation, with significant impact in this population since up to two-thirds of patients with T2D develop asymptomatic left ventricular dysfunction in just 5 years after the diagnosis of T2D [15]. Furthermore, the development of HF in these patients produces an increased mortality risk and worse evolution.
For this reason, an early treatment approach is becoming increasingly important in these patients, even before the first symptoms of HF appear, and treatment options are aimed at acting on both conditions simultaneously.
Until recently, no HF therapies were available directed at glucose metabolism, and the antidiabetic therapies for T2D had shown a neutral or harmful effect in HF endpoints [16]. However, recent data on the use of sodium–glucose cotransporter-2 inhibitors (SGLT2i) in patients with HF, with and without diabetes, have changed this paradigm, showing that they target various mechanisms underpinning the HF pathogenesis that go beyond the metabolic control of diabetes.
Description of the Pathological Mechanisms That Link T2D with Heart Failure: Direct and Indirect Effects of SGLT2i on Myocardial Function
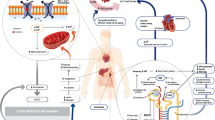
T2D can lead to the development of HFrEF and HFpEF through diverse mechanisms. The available evidence suggests that T2D can promote the development of HFrEF by two main mechanisms: (1) by activating the sodium–hydrogen exchanger and undermining cardiomyocyte survival [17] and (2) by suppressing nutrient deprivation signaling autophagy and thereby promoting cardiomyocyte stress and dysfunction [17]. In parallel, T2D can promote the development of HFpEF by enhancing the expansion of visceral (and especially epicardial) adipose tissue depots, which can promote cardiac inflammation, fibrosis, and microcirculatory dysfunction. Finally, in both HFrEF and HFpEF, insulin action on the renal tubules to stimulate sodium hyperabsorption can increase plasma volume expansion and LV filling pressures [17].
Interestingly, SGLT2i are among the first antidiabetic drugs that have demonstrated the ability to reduce severe HF events and that have significant effects in mitigating each of these mechanisms (Table 1) [18,19,20,21], producing a combination of systemic and direct effects on the myocardium that ultimately leads to the reported cardiovascular benefits (Fig. 1). The pleiotropic mechanisms of action of SGLT2i place them at the intersection of hemodynamic, metabolic, and neurohumoral pathways that impact the heart, the kidney, and the peripheral vasculature, which are essential in the pathogenesis of HF regardless of the LVEF. Nevertheless, all these underlying mechanisms are plausible but not clearly understood.
SGLT2i in Clinical Trials and Meta-Analyses in Heart Failure
Data from several clinical trials and meta-analyses have demonstrated a beneficial effect of SGLT2i on HF cardiac complications that goes beyond its glycemic effects (Table 2).
SGLT2i in Patients with HFrEF, with and without T2D
In 2019, the DAPA-HF study confirmed, for the first time, a significant reduction in the risk of worsening HF or death from cardiovascular causes among patients with HFrEF, in patients with and without T2D treated with this class of drugs [22]. With a population of 4744 patients, this study analyzed whether, compared with placebo, a once-daily dose of dapagliflozin 10 mg improves morbidity, mortality, and quality of life (QoL) in symptomatic patients with HF and a LVEF ≤ 40%. The direct result of cardiovascular death or worsening HF (defined as HF hospitalization or urgent hospital visit for HF treatment) was significantly reduced (HR 0.74, 95% CI 0.65–0.85, P < 0.001) with a sustained statistically significant benefit by 28 days after randomization (HR 0.51, 95% CI 0.28–0.94, P = 0.03) [23].
The number needed to treat (NNT) to prevent one event was 21 over the median follow-up of 18.2 months. A decrease in the risk of other outcomes was also detected, including cardiovascular mortality (HR 0.82, 95% CI 0.69–0.98), all-cause mortality (HR 0.83; 95% CI 0.71–0.97), and HF hospitalization (HR 0.70, 95% CI 0.59–0.83). Furthermore, patients undergoing dapagliflozin treatment were more likely to show a clinically relevant improvement in their QoL after 8 months of treatment, and there was no difference in prespecified adverse severe events between the dapagliflozin and placebo groups. More importantly, there was no evidence of heterogeneity in the efficacy of dapagliflozin in any of the prespecified subgroups, including those with lower LVEF or higher N-terminal-pro B-type natriuretic peptide (NT-proBNP) levels, or in patients with more advanced age or renal insufficiency, which indicates that dapagliflozin may be just as effective in patients with more severe HF [22]. In 2020, a published exploratory analysis of DAPA-HF showed that dapagliflozin’s efficacy was comparable over the whole range of glycosylated hemoglobin values, in both nondiabetic and diabetic populations. These results suggest that the SGLT2 inhibition with dapagliflozin has beneficial effects in HFrEF regardless of T2D status and that the mechanism of action of dapagliflozin in HFrEF extends beyond a simple glucose-lowering effect [24].
Furthermore, another post-hoc analysis of the DAPA-HF trial showed comparable risk reductions in HF hospitalization and mortality with dapagliflozin, regardless of previous HF therapy. These results indicate a complementary value of dapagliflozin in addition to the established combined therapy for HF and further evidence for its use in ambulatory patients with HFrEF to improve clinical outcomes [25]. Finally, an exploratory analysis on the effect of dapagliflozin on incident T2D in the cohort without diabetes enrolled in the trial showed that treatment with dapagliflozin reduced the incidence of new diabetes [26] and therefore could be exerting a protective effect in these patients with HF where the risk of developing diabetes is elevated.
A beneficial independent glucose-lowering effect of SGLT2i on morbidity and mortality in patients with HFrEF, with and without T2D, was confirmed in the EMPEROR-Reduced study. In the EMPEROR-Reduced study, 3730 patients with HFrEF on recommended therapy were randomized to receive a 10 mg once-daily dose of empagliflozin or placebo. The main outcome (cardiovascular death or hospitalization for heart failure) event was significantly reduced in the empagliflozin (19.4%) group compared with the placebo (24.7%) group (HR 0.75, 95% CI 0.65–0.86, P < 0.001; NNT 19), and this reduction was consistent in patients regardless of their diabetes status. In this study, the total number of hospitalizations for HF was also lower in the empagliflozin group than in the placebo group (HR 0.70, 95% CI 0.58–0.85, P < 0.001). It is noteworthy to mention that the annual rate of decline in the estimated glomerular filtration rate (eGFR) was less in the empagliflozin group than in the placebo group (−0.55 versus −2.28 ml/min per 1.73 m2 of body-surface area per year, P < 0.001), and patients treated with empagliflozin had a decreased risk of serious renal outcomes [27]. In patients with prediabetes or normoglycemia, empagliflozin did not lower HbA1c, and there was no increased risk of hypoglycemia, but the effects of the drug over the endpoints analyzed did not differ. When evaluated as a regular variable, baseline HbA1c did not significantly alter the advantages of empagliflozin on the primary outcome (P-interaction = 0.40), suggesting an independent glucose-lowering effect [28].
A meta-analysis using data from both previous studies confirms earlier findings on the reduction of HF hospitalization by SGLT2 inhibition and confirms that these agents also improve renal outcomes (composite renal endpoint HR 0.62, 95% CI 0.43–0.90, P = 0.013) and reduce all-cause death (pooled HR 0.87, 95% CI 0.77–0.98, P = 0.018) and cardiovascular death (pooled HR 0.86, 95% CI 0.76–0.98, P = 0.027) in patients with HFrEF. It is noteworthy to mention that the pooled treatment effects showed consistent benefits for subgroups based on age, sex, diabetes, treatment with an ARNI, and baseline eGFR [29].
SGLT2i in Patients with HFpEF, with and without T2D
Although there is no universal conception of HFpEF, and the effects of SGLT2i are considered to be related to the studies’ design, SGLT2i are the only drugs that have shown efficacy in patients with HFpEF. Very recently, in the EMPEROR-Preserved Trial carried out in 5988 patients with HF and EF > 40%, empagliflozin was also demonstrated to reduce the combined risk of cardiovascular death or hospitalization for HF in patients with HFpEF (HR 0.79, 95% CI 0.69–0.90, P < 0.001), regardless of the presence or absence of diabetes, with an NNT of 31. This effect was mainly related to a lower risk of hospitalization for HF in the SGLT2 inhibitor (empagliflozin 10 mg) group [30]. Recently, the US Food and Drug Administration (FDA) approved empagliflozin for the treatment of adults with HF regardless of the phenotype of the condition [31].
Taking into account these results, the unique mechanism of action of SGLT2i across the EF spectrum, and the collective experience from prior HFpEF trials showing that treatment effect with currently available therapies declined with increasing LVEF, the DELIVER trial has been designed to determine the efficacy and safety of the SGLT2 inhibitor dapagliflozin, added to conventional therapy, in patients with HF and preserved and mildly reduced EF. In this study, a total of 6263 patients with or without diabetes, with signs and symptoms of HF, an LVEF > 40%, elevation in NPs, and evidence of structural heart disease are eligible. The primary endpoint is time to first cardiovascular death or worsening HF event (HF hospitalization or urgent HF visit), and will be assessed in dual primary analyses—the full population and in those with LVEF < 60%. This study will provide complementary information to DAPA-HF, and the results of both studies will be pooled to assess the effects of dapagliflozin across the EF spectrum [32].
Finally, patients with HF and HFpEF have a high burden of symptoms and functional limitations, and have a poor quality of life. In this regard, a recent multicenter, randomized trial of patients with HFpEF (PRESERVED-HF) showed that 12 weeks of dapagliflozin treatment, by targeting cardiometabolic abnormalities, significantly improve patient-reported symptoms [improved KCCQ-CS 5.8 points (95% CI 2.3–9.2, P = 0.001)], physical limitations [improved 5.3 points (95% CI 0.7–10.0; P = 0.026)], and exercise function [improved 6MWT 20.1 m (95% CI 5.6–34.7, P = 0.007)] and was well tolerated in chronic HFpEF [33].
SGLT2i in Hospitalized Patients with T2D with Decompensated HF (HFrEF and HFpEF)
Another recent study, the SOLOIST-WHF trial (effect of sotagliflozin on cardiovascular events in patients with type 2 diabetes post worsening heart failure), focused on a different clinical scenario. This study analyzed the safety and efficacy of SGLT2i when initiated soon after an episode of decompensated HF. Patients with T2D recently hospitalized for worsening HF were randomly assigned to receive sotagliflozin or placebo and were followed up for an average of 9.0 months. In these patients with T2D with newly diagnosed, worsening heart failure, sotagliflozin therapy started before or shortly after discharge resulted in a significantly lower total number of deaths from cardiovascular causes and hospitalizations and urgent visits for HF than placebo (HR 0.67, 95% CI 0.52–0.85, P < 0.001). This result was consistent regardless of EF at study entry, including patients with EF > 50%. The percentage of patients with low blood pressure was comparable in both the sotagliflozin and placebo groups (6.0% and 4.6%, respectively), as was the percentage with acute kidney injury (4.1% and 4.4%, respectively). The benefits derived from sotagliflozin therapy were consistent in the prespecified subgroups of patients stratified according to the timing of the first dose [34].
Addressing the vulnerable period of hospital admission for worsening HF, a meta-analysis including SOLOIST and SCORED data found a reduction in the primary endpoint (total death, hospitalized HF, urgent HF visit) independent of the ejection function [HFrEF (< 40%), HFmrEF (40–50%) or HFp (> 50%)] and an NNT of 9 as well as the existence or not of a previous history. This highlights the benefits of SGLT2i across the cardiorenal continuum, with different molecules and in different clinical settings [35].
New Guidelines: Moving towards an Intensive and Multidimensional Approach for HF Treatment
On the basis of this clinical evidence, several HF guidelines have introduced SGLT2i as a first-line HF treatment [36,37,38]. Specifically, the latest ESC 2021 guidelines [36] and ACC guidelines update (in which the SOLOIST data have not yet been included) [37] include the recommendation for the use of this group of drugs in all patients with reduced ejection fraction since the beginning of HF (Fig. 2), regardless of whether they have diabetes or not, favoring an intensive and multidimensional approach for HF treatment.

Adapted from: McDonagh TA, et al. 2021. Eur Heart J. 2021 [36]
Management of heart failure with reduced ejection fraction to reduce mortality for all patients.
Likewise, the Canadian guidelines [38] recommend that, in the absence of contraindications, patients with HFrEF be treated with combination therapy from the beginning, including one evidenced-based medication from each of the categories (ARNI, B-blocker, MRA, and SGLT2i) (strong recommendation; moderate level of evidence). The main recommendations on the use of SGLT2i stated in the main guidelines are summarized in Table 3.
Future Perspectives in the Treatment of HF for Patients with and without T2D
On the basis of these promising findings in patients with chronic HF, it would be interesting to develop studies that assess the early use of SGLT2i in the acute phase of admission for HF and personalize treatment for the most significant benefit. Recently, the EMPULSE-HF study has shown that, among patients with acute decompensated HF, empagliflozin versus placebo is associated with significant clinical benefits at 90 days. This trial enrolled participants regardless of EF or diabetes status, and there was no evidence for treatment interaction based on either of these variables. Empagliflozin versus placebo was also associated with fewer deaths, improved quality of life, and greater reduction in body weight [39]. Currently, other studies are underway that assess the possible advantages of other SGLT2i in this context, such as the DICTATE-AHF (https://clinicaltrials.gov/ct2/show/NCT04298229) and DAPA-ACT HF-TIMI 68 (https://clinicaltrials.gov/ct2/show/NCT04363697) studies with dapagliflozin.
Some authors suggest the importance of evaluating the clinical efficacy and safety of the SGLT2i in nondiabetic patients with acute decompensated HF [40]. Acutely decompensated HF is the leading cause of hospitalization in the elderly and is associated with a high risk of mortality (in-hospital and post-discharge), rehospitalization, and health care costs [41].
Safety in hospitalized patients in an unstable clinical condition could be a concern. For example, in the DARE-19 trial, treatment with dapagliflozin did not result in a statistically significant risk reduction in organ dysfunction, death, or improvement in clinical recovery in patients with coronavirus disease 2019 (COVID-19) [42]. However, the DARE-19 trial suggests some lessons. First, dapagliflozin was well tolerated and can be safely used in patients with cardiometabolic risk factors who were hospitalized with COVID-19 pneumonia, regardless of their diabetes status. Second, although findings of dapagliflozin failed to show efficacy in the COVID-19 acute setting, SGLT2i have a promising future not only in patients with diabetes but also in patients with chronic cardiovascular disease or chronic kidney disease, and perhaps even in asymptomatic people [43].
The SOLOIST, SCORED, and DARE-19 trials lay the foundations for the use of SGLT2i in the hospital setting, although its implementation will require the design of safety and effectiveness monitoring strategies within multidisciplinary care teams.
Unlike in chronic HF, there are currently no therapies that convincingly improve clinical outcomes in acute HF, apart from those treating underlying causes, and SGLT2i could be an option.
Interestingly, even in the absence of T2D, insulin sensitivity decreases as HF progresses, indicating a possible link between HF and glucometabolic alterations and a possible role for SGLT2i in HF and metabolic syndrome prevention. SGLT2i are also a pharmacologic therapy with promising potential to alter clinical practice and influence practice guidelines to prevent CAD. They have demonstrated great potential in reducing major adverse cardiac events in patients with T2D and CAD [44]. Moreover, among people with T2D and CAD, SGLT2 inhibition with empagliflozin has been associated with a significant reduction in LV mass indexed to body surface area after 6 months [45].
Using these pharmacologic agents as a part of optimal medical therapy may narrow the gap between revascularization and optimal medical therapy alone in patients with T2D and multivessel disease. Future studies on the role of SGLT2i with regard to HF outcomes are needed to elucidate the mechanisms and clinical effects in all these vulnerable populations.
Conclusions
In HF, morbidity and mortality remain high despite advances in therapy, and new therapies are needed to improve the prognosis of these patients, mainly when T2D is also present. Patients with T2D show a very high risk of developing HF, and those with HF are at higher risk of developing T2D and worse prognoses when both pathologies are present.
Interestingly, SGLT2i have been shown to target various mechanisms underpinning the HF pathogenesis that go beyond the glucose-lowering effect and metabolic control of diabetes. These pleiotropic mechanisms of action of SGLT2i place them at the intersection of hemodynamic, metabolic, and neurohumoral pathways that impact the heart, the kidney, and the peripheral vasculature, providing them with a regulatory action on the main mechanisms involved in the pathogenesis of HF. In fact, several recent clinical studies (DAPA-HF, EMPEROR-Reduced, EMPEROR-Preserved, SOLOIST-WHF trial) and meta-analyses have demonstrated that SGLT2i reduce the risk of HF hospitalization, improve QOL, and increase survival in HF across the whole LVEF spectrum.
In light of the favorable results obtained in the studies as mentioned above, SGLT2i are among the first antidiabetic drugs capable of reducing cardiovascular risks related to HF and consistently improving the prognosis in different populations and settings. On the basis of this clinical evidence, several HF guidelines have introduced SGLT2i as a first-line treatment for HF from the start, and several studies are underway to specify the role of SGLT2i with regard to HF outcomes in different vulnerable populations, including the hospital setting.
References
Benjamin EJ, Blaha MJ, Chiuve SE, et al. Heart disease and stroke statistics-2017 update: a report from the American Heart Association. Circulation. 2017;135(10):e146–603.
Cook C, Cole G, Asaria P, Jabbour R, Francis DP. The annual global economic burden of heart failure. Int J Cardiol. 2014;171(3):368–76.
Rawshani A, Rawshani A, Franzén S, et al. Risk factors, mortality, and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2018;379(7):633–44.
Crespo-Leiro MG, Barge-Caballero E, Segovia-Cubero J, et al. Pronóstico al año en pacientes con insuficiencia cardiaca en España Registro ESC-EORP-HFA heart failure long-term. REC CardioClin. 2020;55(4):207–16.
Jones NR, Roalfe AK, Adoki I, Hobbs FDR, Taylor CJ. Survival of patients with chronic heart failure in the community: a systematic review and meta-analysis. Eur J Heart Fail. 2019;21(11):1306–25.
Dharmarajan K, Hsieh AF, Kulkarni VT, et al. Trajectories of risk after hospitalization for heart failure, acute myocardial infarction, or pneumonia: retrospective cohort study. BMJ. 2015;350:h411–h411.
Echouffo-Tcheugui JB, Greene SJ, Papadimitriou L, et al. Population risk prediction models for incident heart failure. Circ Heart Fail. 2015;8:438–47.
McDonagh TA, Metra M, Adamo M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42(36):3599–726.
Kemp CD, John V, Conte JV. The pathophysiology of heart failure. Cardiovasc Pathol. 2012;21(5):365–71.
Bayes-Genis A, Pascual-Figal D, Núñez J. The pre-HFpEF stage: a new entity that requires proper phenotyping for better management. Eur J Prev Cardiol. 2020;28:935–6.
Gori M, Lam CS, D’Elia E, et al. Integrating natriuretic peptides and diastolic dysfunction to predict adverse events in high-risk asymptomatic subjects. Eur J Prev Cardiol. 2020;28:937–45.
Tromp J, Westenbrink BD, Ouwerkerk W, et al. Identifying pathophysiological mechanisms in heart failure with reduced versus preserved ejection fraction. J Am Coll Cardiol. 2018;72(10):1081–90.
American Diabetes Association. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes-2018. Diabetes Care. 2018;41:S73–85.
De Couto G, Ouzounian M, Liu PP. Early detection of myocardial dysfunction and heart failure. Nat Rev Cardiol. 2010;7(6):334–44.
Faden G, Faganello G, De Feo S, et al. The increasing detection of asymptomatic left ventricular dysfunction in patients with type 2 diabetes mellitus without overt cardiac disease: data from the SHORTWAVE study. Diabetes Res Clin Pract. 2013;101(3):309–16.
Seferović PM, Petrie MC, Filippatos GS, et al. Type 2 diabetes mellitus and heart failure: a position statement from the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2018;20(5):853–72.
Packer M. Critical examination of mechanisms underlying the reduction in heart failure events with SGLT2 inhibitors: identification of a molecular link between their actions to stimulate erythrocytosis and to alleviate cellular stress. Cardiovasc Res. 2021;117(1):74–84.
Packer M, Anker SD, Butler J, Filippatos G, Zannad F. Effects of sodium-glucose cotransporter 2 inhibitors for the treatment of patients with heart failure: proposal of a novel mechanism of action. JAMA Cardiol. 2017;2(9):1025–9.
Packer M. Autophagy-dependent and -independent modulation of oxidative and organellar stress in the diabetic heart by glucose-lowering drugs. Cardiovasc Diabetol. 2020;19(1):62.
Packer M. Cardioprotective effects of sirtuin-1 and its downstream effectors: potential role in mediating the heart failure benefits of SGLT2 (sodium–glucose cotransporter 2) inhibitors. Circ Heart Fail. 2020;13(9): e007197.
Masson W, Lavalle-Cobo A, Nogueira JP. Effect of SGLT2-inhibitors on epicardial adipose tissue: a meta-analysis. Cells. 2021;10(8):2150.
McMurray JJV, Solomon SD, Inzucchi SE, et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med. 2019;381(21):1995–2008.
Berg DD, Jhund PS, Docherty KF, et al. Time to clinical benefit of dapagliflozin and significance of prior heart failure hospitalization in patients with heart failure with reduced ejection fraction. JAMA Cardiol. 2021;6(5):499–507.
Petrie MC, Verma S, Docherty KF, et al. Effect of dapagliflozin on worsening heart failure and cardiovascular death in patients with heart failure with and without diabetes. JAMA. 2020;323:1353–68.
Docherty KF, Jhund PS, Inzucchi SE, et al. Effects of dapagliflozin in DAPA-HF according to background heart failure therapy. Eur Heart J. 2020;41:2379–92.
Inzucchi SE, Docherty KF, Køber L, et al. Dapagliflozin and the incidence of type 2 diabetes in patients with heart failure and reduced ejection fraction: an exploratory analysis from DAPA-HF. Diabetes Care. 2021;44(2):586–94.
Packer M, Anker SD, Butler J, et al. Cardiovascular and renal outcomes with empagliflozin in heart failure. N Engl J Med. 2020;383(15):1413–24.
Anker SD, Butler J, Filippatos G, et al. Effect of empagliflozin on cardiovascular and renal outcomes in patients with heart failure by baseline diabetes status: results from the EMPEROR-reduced trial. Circulation. 2021;143(4):337–49.
Zannad F, Ferreira JP, Pocock SJ, et al. SGLT2 inhibitors in patients with heart failure with reduced ejection fraction: a meta-analysis of the EMPEROR-Reduced and DAPA-HF trials. Lancet. 2020;396(10254):819–29.
Anker SD, Butler J, Filippatos G, et al. Empagliflozin in heart failure with a preserved ejection fraction. N Engl J Med. 2021;385(16):1451–61.
Food and Drug Administration. Accessed at: https://pace-cme.org/2022/02/28/sglt2i-approved-by-fda-for-treatment-of-hf-regardless-of-lvef/
Solomon SD, de Boer RA, DeMets D, et al. Dapagliflozin in heart failure with preserved and mildly reduced ejection fraction: rationale and design of the DELIVER trial. Eur J Heart Fail. 2021;23(7):1217–25.
Nassif ME, Windsor SL, Borlaug BA, et al. The SGLT2 inhibitor dapagliflozin in heart failure with preserved ejection fraction: a multicenter randomized trial. Nat Med. 2021;27:1954–60.
Bhatt DL, Szarek M, Steg PG, et al. Sotagliflozin in patients with diabetes and recent worsening heart failure. N Engl J Med. 2021;384(2):117–28.
Bhatt DL, et al. Benefits of SGLT1/2 inhibition with sotagliflozin in heart failure with preserved ejection fraction. In ACC Congress 2021.
McDonagh TA, Metra M, Adamo M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42(36):3599–726.
Maddox TM, Januzzi JL Jr, et al. Update to the 2017 ACC expert consensus decision pathway for optimization of heart failure treatment: answers to 10 pivotal issues about heart failure with reduced ejection fraction: a report of the American College of Cardiology solution set oversight committee. J Am Coll Cardiol. 2021;77(6):772–810.
McDonald M, Virani S, Chan M, et al. CCS/CHFS heart failure guidelines update: defining a new pharmacologic standard of care for heart failure with reduced ejection fraction. Can J Cardiol. 2021;37(4):531–46.
Voors AA. American Heart Association Virtual Annual Scientific Sessions (AHA 2021), November 14, 2021.
Tromp J, Ponikowski P, Salsali A, et al. Sodium–glucose co-transporter 2 inhibition in patients hospitalized for acute decompensated heart failure: rationale for and design of the EMPULSE trial. Eur J Heart Fail. 2021;23:826–34.
Reeves GR, Whellan DJ, Duncan P, et al. Rehabilitation therapy in older acute heart failure patients (REHAB-HF) trial: design and rationale. Am Heart J. 2017;185:130–9.
Kosiborod MN, Esterline R, Furtado RHM, et al. Dapagliflozin in patients with cardiometabolic risk factors hospitalised with COVID-19 (DARE-19): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Diabetes Endocrinol. 2021;9(9):586–94.
Rossello X, Caimari F. Dapagliflozin in patients with COVID-19: truth or dare. Lancet Diabetes Endocrinol. 2021;9(9):550–1.
Sharma UC, Farkouh ME. The role of SGLT-2 inhibitors as part of optimal medical therapy in improving cardiovascular outcomes in patients with diabetes and coronary artery disease. Cardiovasc Drugs Ther. 2017;31(3):311–8.
Verma S, Mazer CD, Yan AT, et al. Effect of empagliflozin on left ventricular mass in patients with type 2 diabetes mellitus and coronary artery disease: the EMPA-HEART CardioLink-6 randomized clinical trial. Circulation. 2019;140(21):1693–702.
Acknowledgements
Funding
AstraZeneca funded the writing assistance provided by Springer Healthcare Ibérica SL and the journal’s Rapid Service Fee. This supplement has been sponsored by AstraZeneca.
Editorial and Other Assistance
Editorial assistance in the preparation of this article was provided by Ana Ortega, PhD, on behalf Springer Healthcare Ibérica and was funded by AstraZeneca. The authors want to express their gratitude to José Juan Aparicio-Sánchez from the Medical Affairs department of AstraZeneca Spain for his continuous scientific support during the development of this manuscript.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
All authors equally participated in the development of the manuscript, had full editorial control, reviewed and edited various drafts, and provided their final approval of all content and submission for publication.
Disclosures
Elías Delgado has received unrestricted research support from AstraZeneca, Novo Nordisk, Sanofi, Pfizer, and Roche and has received consulting fees and/or honoraria for membership on advisory boards and speaker´s bureau from AstraZeneca, Novo Nordisk, Lilly, Sanofi, GlaxoSmithKline, Pfizer, Almirall, Novartis, Abbott Laboratories, Esteve, and Merck Sharp & Dohme. Esteban Jódar has received grants from Novo Nordisk, MSD, Lilly, Janssen, Pfizer, AstraZeneca, Sanofi, UCB, Amgen, Boehringer, FAES, and Shire; and consultancy and speaker fees from Mundipharma, Amgen, AstraZeneca, FAES, Helios-Fresenius, Italfármaco, Lilly, MSD, Novo Nordisk, UCB, and Viatris. Pedro Mezquita-Raya has consulted for Abbott, AstraZeneca, FAES and Novo Nordisk; reports speaker fees from AstraZeneca, Eli Lilly, FAES, Fresenius and Novo Nordisk; and received grant support from Abbott and Novo Nordisk, outside the submitted work. Óscar Moreno-Pérez has consulted for AstraZeneca and Novo Nordisk; reports speaker fees from AstraZeneca, Eli Lilly, Boehringer and Novo Nordisk; and received grant support from Eli Lilly and Boehringer, outside the submitted work.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Delgado, E., Jódar, E., Mezquita-Raya, P. et al. Benefits of SGLT2i for the Treatment of Heart Failure Irrespective of Diabetes Diagnosis: A State-of-the-Art Review. Diabetes Ther 13 (Suppl 1), 19–34 (2022). https://doi.org/10.1007/s13300-022-01278-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-022-01278-0