Abstract
Introduction
Primary aim was to provide real-world evidence of the outcomes after the switch to glargine 300 U/ml (Gla-300) from other basal insulins (first or second generation) in Italy.
Methods
Multicenter, observational, retrospective study based on electronic medical records.
Results
Overall, 953 T2DM insulin ± OAD treated people switched to Gla-300 or Gla-100 from January 2015 to July 2018. Three clinically relevant cohorts were identified: patients switching to Gla-300 from first-generation basal insulin (cohort 1), patients switching to Gla-300 from degludec-100 (Deg-100) (cohort 2), and those switching to Gla-100 from any basal insulin (cohort 3). The three cohorts differed in terms of age, diabetes duration, and metabolic control. HbA1c changes after 6 months from the switch were − 0.27% (95% CI − 0.38; − 0.16), − 0.06% (95% CI − 0.31; 0.19), and − 0.30% (95% CI − 0.51; − 0.09) in the three cohorts, respectively. FPG significantly decreased in cohort 1 (− 14.07 mg/dl, 95% CI − 20.25; − 7.89), while body weight significantly decreased in cohort 2 (− 1.47 kg, 95% CI − 2.55; − 0.39). Doses of insulin marginally changed during the follow-up (+ 0.89 U in basal insulin daily dose in cohort 1 and + 2.07 U in short-acting insulin daily dose in cohort 2).
Conclusions
Switching to Gla-300 from first-generation basal insulin in the real world is associated with improvements in metabolic control despite a suboptimal titration of both basal and short-acting insulins. Inertia in insulin titration documented in the Gla-100 cohort is also observed with the second-generation basal insulin. The switch to Gla-300 from Deg-100 was associated with a decrease in body weight of − 1.47 kg despite a slight increase in short-acting insulin daily doses of about + 2 U.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Second generation basal insulins provide similar or improved efficacy compared to first generation ones with better safety profile. |
Real world evidence is important to assess the use and the impact of these recent basal insulins in clinical practice. |
Primary aim of the study was to provide real-world evidence of the outcomes after the switch to the second generation basal insulin glargine 300 U/mL (Gla-300) in Italy. |
What was learned from the study? |
A relevant number of people with type 2 diabetes and different clinical profiles switch basal insulin in clinical practice. |
Switching to Gla-300 from 1st generation basal insulin is associated with improvements in metabolic control despite a suboptimal titration of both basal and short acting insulin. |
Switching to Gla-300 from the other available 2nd generation basal insulin (degludec 100 U/ml) is associated with a decrease in body weight. |
Introduction
Type 2 diabetes mellitus (T2DM) is a progressive disease characterized by relative insulin deficiency and eventual need for supplemental insulin therapy [1, 2]. Appropriate initiation and titration of insulin therapy are essential to enhance adherence to therapy and to reach glycemic targets [3]. However, Italian real-life data show that T2DM people treated with insulin are uncontrolled in 50% of cases [4].
New-generation basal insulins provide similar or improved efficacy compared to first-generation (i.e., insulin glargine 100 U/ml or detemir) with a better safety profile [3].
Insulin glargine 300 units/ml (Gla-300) is a second-generation basal insulin analog available in Italy since January 2017. Gla-300 has distinct pharmacokinetic (PK) and pharmacodynamic (PD) profiles compared to glargine 100 units/ml (Gla-100). This new formulation allows for the formation of a more compact subcutaneous depot after injection that results in a more gradual release of insulin providing a more constant and smoother PK profile and longer duration of action on the PD profile versus Gla-100 [5, 6].
In the treat-to-target studies from the EDITION phase 3 clinical program, Gla-300 has been shown to be non-inferior to Gla-100 with respect to glycated hemoglobin (HbA1c) reduction [7,8,9]. Furthermore, a significantly lower percentage of people with T2DM experienced confirmed hypoglycemic events on Gla-300 compared to Gla-100 [7,8,9].
Insulin degludec 100 U/ml (Deg-100) is another second-generation basal insulin available in Italy since 2015. PK-PD data of Deg-100 showed a more stable kinetic profile than Gla-100 [10]. The BEGIN clinical program showed comparable glycemic control vs. Gla-100 in most trials, with lower nocturnal hypoglycemic episodes during the maintaining phase, but a frequency of diurnal hypoglycemia higher with Deg-100 than with Gla-100 [11,12,13]. The new formulation insulin degludec 200 U/ml (Deg-200) provides comparable levels of glycemic control with similar rates of hypoglycemia to Deg-100, but it is not available in Italy [14].
Recently, a PK-PD clinical study of Gla-300 vs. Deg-100 has shown a lower within-day variability of Gla-300 compared to Deg-100 in a once-daily morning dosing regimen of 0.4 U/kg/day [6]. A trial-level meta-analysis using data from BEGIN (Deg-100) and EDITION (Gla-300) suggested that despite greater reductions in fasting plasma glucose, Deg-100 was associated with less improvement in HbA1c versus Gla-100, with an hypoglycemia benefit only evident at night; Gla-300 showed similar HbA1c reduction to Gla-100, accompanied by lower risk of hypoglycemia both at night and at any time of day [15]. The head-to-head BRIGHT trial in a naïve T2DM population has recently documented that Gla-300 and Deg-100 provided similar glycemic control improvements with comparable hypoglycemia incidence during the full study period but lower in favor of Gla-300 during the titration period [16]. A trial involving individuals with T2DM treated with basal insulin showed there was no significant difference in the rate of overall symptomatic hypoglycemia between Deg-200 and Gla-300 in the maintenance period in insulin-treated individuals [17, 18]. In a real-world setting, switching from Gla-100 or detemir to Gla-300 or Deg-100 was associated with similar improvements in glycemic control and hypoglycemia in adult people with T2DM [19].
Starting from these premises, real-world evidence is particularly important to assess the use and the impact of these recent basal insulins in clinical practice. In this respect, retrospective review of electronic medical records (EMRs) represents a new frontier in epidemiologic and clinical research, offering the potential for low-cost, high-volume data on clinical effectiveness [20].
Particularly, no clinical data are available relative to the Italian diabetic population that has switched from other basal insulins to Gla-300 in real-life conditions.
In the Lazio region, a network of seven diabetes centers (ACISMOM) cares for 40% of people with diabetes referred to specialist care. One center of the same network is in the Puglia region. All the centers share the same EMRs system.
The principal aim of this study, which was based on the retrospective review of EMRs adopted in the ACISMOM network, was to assess the clinical outcomes of switching to Gla-300 from first- or other second-generation basal insulins. Secondary endpoints were to assess the most frequent types of switch to Gla-300 or Gla-100, patients’ characteristics associated to the different types of switch and potential reasons for switching.
Methods
This was a multicenter, observational, longitudinal, retrospective study based on pre-existing data derived from EMRs. The study involved eight diabetes clinics of the ACISMOM network.
Eligible patients were those with a diagnosis of T2DM, aged ≥ 18 years, switched to Gla-300 or Gla-100 from other basal insulins (first or second generation) in the time period from January 2015 through December 2017, using the previous basal insulin analogs for at least 6 months before initiating insulin Gla-300 or Gla-100, with availability of a follow-up of at least 3 months after initiating insulin Gla-300 or Gla-100, not switching to another basal insulin analog after the switch to Gla-300 or Gla-100 during the study period.
Study endpoints included within-group changes after 3 and 6 months from the switch in the following parameters: glycated hemoglobin (HbA1c), fasting blood glucose (FBG), body weight, total insulin daily dose, total basal insulin daily dose and total short-acting insulin daily dose. In addition, the number of severe hypoglycemic episodes reported on EMRs and average number of visits during 6 months were assessed. Although the study protocol also included the evaluation of the incidence of non-severe hypoglycemia, this information was not available for most of the patients, since the routine download of self-monitoring of blood glucose (SMBG) values from glucose meters on EMRs was not common practice in participating centers.
To explore the main reasons for switching basal insulin, a site questionnaire was administered to principal investigators of all participating centers. Possible reasons for switching basal insulin in their everyday practice included: better metabolic control, less hypoglycemia, less glycemic variability, better adherence, easier titration, less weight gain, greater flexibility of administration, cost of treatment, frequency of administration, and adverse events.
All centers adopted the same EMRs (MyStar Connect®, Meteda srl, San Benedetto del Tronto, Italy) and an ad hoc software, which was developed for the standardized and anonymous extraction of the following data: age, gender, diabetes duration, diabetes therapy, type of insulin, insulin dose, number of insulin injections/day, HbA1c, FBG, hypoglycemia, body weight, body mass index (BMI), lipid profile (triglycerides, total cholesterol, high-density and low-density lipoprotein), blood pressure, renal function (albuminuria and glomerular filtration rate), antihypertensive and lipid-lowering treatment, and history of major diabetes complications.
Classification of diabetes complications was based on the International Classification of Diseases, 9th revision-Clinical Modification (ICD-9 CM); pharmacologic treatments were based on the Anatomical Therapeutic Chemical Classification System (ATC). Severe hypoglycemia was defined as an episode requiring the assistance of the patient by other persons. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national-Ethics Committee of the ACISMOM network) and with the Helsinki Declaration of 1975, as revised in 2008 (5). Informed consent was obtained from all patients for being included in the study.
Statistical Analysis
Being an exploratory, descriptive study, no formal sample size estimation was performed.
Study outcomes have been investigated on three clinically relevant cohorts, i.e., patients switching to Gla-300 from first-generation basal insulin (cohort 1), patients specifically switching to Gla-300 from second-generation basal insulin, i.e., Deg-100 (cohort 2), and patients switching to Gla-100 from any other basal insulin (first or second generation) (cohort 3).
Patients’ characteristics were summarized by cohort as mean and standard deviation (continuous variables) or counts and percentages (categorical variables). Given the descriptive nature of the study and the substantial differences of patient characteristics in the different cohorts, no between-group comparison was performed.
Within-group changes after 3 (T + 3) and 6 months (T + 6) from the switch in levels of HbA1c, FBG, body weight, and insulin dose were assessed using mixed models for repeated measurements. Results were expressed as estimated mean at each time point with their 95% confidence intervals (95% CI) and estimated mean difference and 95% CI from T0 (switch date) to T + 3 and T + 6. In addition, rate of severe hypoglycemia, proportion of patients with HbA1c < 7.0% and < 8.0%, and mean number of visits after the switch were assessed.
Results
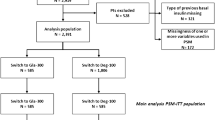
Overall, 953 T2DM insulin ± OAD treated people switched to Gla-300 or Gla-100 from January 2015 to July 2018 (study flow chart is shown in Fig. 1). Characteristics of the patients of each cohort at the time of the basal insulin switch are reported in Table 1. Patients treated with basal and short-acting insulin were 76.6% in cohort 1, 86.9% in cohort 2, and 64.5% in cohort 3, while those treated with basal-bolus insulin scheme (i.e., basal and 3 injections of short-acting insulin) were 62.4%, 78.7%, and 50.8%, respectively. Furthermore, about 60% of patients used insulin in association with other glucose-lowering agents with marked differences in the prevalence of use of the different classes of drugs (Table 1).
Patients in cohort 1 (from first-generation basal insulin to Gla-300) were 59% men and had mean age of 70 years, diabetes duration of 19.6 years, HbA1c of 8.3%, and BMI of 30.9 kg/m2.
Mean total daily dose of insulin prescribed was of 59.6 U (average daily dose of basal insulin of 25.4 U and average daily dose of short-acting insulin of 32.7 U) (Table 1).
Patients in cohort 2 (from Deg-100 to Gla-300) were 53.3% men and had mean age of 69.6 years, diabetes duration of 16.8 years, HbA1c of 8.5%, and BMI of 31.7 kg/m2. Mean total daily dose of insulin prescribed was 72.1 U (average daily dose of basal insulin 32.6 U and average daily dose of short-acting insulin 38.5 U) (Table 1).
Patients in cohort 3 (from any basal insulin to Gla-100) were 53.1% men and had mean age of 73.3 years, diabetes duration of 16.5 years, HbA1c of 8.1%, and BMI of 30.7 kg/m2. Mean total daily dose of insulin prescribed was of 51.2 U (average daily dose of basal insulin 18.6 U and average daily dose of short-acting insulin 31.0 U) (Table 1).
At longitudinal analyses, in cohort 1 HbA1c levels significantly decreased from baseline by − 0.28% (95% CI − 0.38; − 0.18) after 3 months and by − 0.27% (95% CI − 0.38; − 0.16) after 6 months. FBG significantly decreased during 6 months. A slight, non-significant reduction in body weight was also found (Table 2 and Fig. 2).
In cohort 2, HbA1c levels were significantly reduced by − 0.21% (95% CI − 0.43; − 0.01) after 3 months but were unchanged after 6 months. FBG levels did not change after 3 months and were reduced after 6 months, without reaching the statistical significance. In terms of body weight, the switch from Deg-100 to Gla-300 was associated with a statistically significant weight loss of − 1.05 kg (95% − 1.92; − 0.18) after 3 months and − 1.47 kg (95% − 2.55; − 0.39) after 6 months (Table 2 and Fig. 2).
In cohort 3, HbA1c levels significantly decreased by − 0.31% (95% CI − 0.50; 0.12) after 3 months and by − 0.30% (95% CI − 0.51; − 0.09) after 6 months. FBG significantly decreased after 6 months. Body weight was unchanged during the follow-up (Table 2 and Fig. 2).
Changes during the follow-up in total, basal, and short-acting insulin daily doses for the three cohorts are reported in Table 3. In cohort 1, a small increase in basal insulin dose of 0.89 U (95% CI 0.15; 1.63) was found after 6 months, while the short-acting and total daily dose remained unchanged. In cohort 2, the dose of short-acting insulin significantly increased by 2.07 U (95% CI 0.37; 3.77) during 6 months after the switch, while the dose of Gla-300 did not significantly change. In cohort 3, a small increase in basal insulin dose of 1.32 U (95% CI 0.03; 2.61) was found after 3 months, but not after 6 months. No significant changes in short-acting and total daily doses were documented during the follow-up.
The average number of visits after the switch was 2.5 ± 1.3 in cohort 1 and 2.2 ± 1.0 in the other two cohorts.
The site questionnaires highlighted the main reasons for switching in the different cohorts:
-
Hypoglycemia concern, less glucose variability, and better control/efficacy were ranked as the three main reasons for the switch from Deg-100 to Gla-300 and from any other basal insulin to Gla-300.
-
Better control, hypoglycemia concern, efficacy, adverse events, and cost were ranked as the main reasons for the switch from any basal insulin to Gla-100.
Overall, only three severe hypoglycemia episodes were registered in EMRs; all occurred in cohort 1. For this reason, incidence rates of severe hypoglycemia could not be calculated.
Discussion
This study shows that a relevant number of people with T2DM switch basal insulin in clinical practice. Specifically, the study adds important insights for the understanding of the clinical profile of patients switching to Gla-300 from other basal insulins.
Key differences emerged between patients who switched to Gla-300 and Gla-100 and between patients who switched from Deg-100 to Gla-300.
Patients switching to Gla-300 from any first-generation basal insulin had the longest diabetes duration (19.6 years vs. about 16.5 years in the other two cohorts), while patients switching to Gla-100 from any other basal insulin were the oldest ones (73.3 years vs. about 70 years in the other two cohorts). As safety and efficacy of Gla-300 vs. Gla-100 in people with T2DM aged ≥ 65 years were demonstrated in the SENIOR and DELIVER 3 study, where comparable reductions in HbA1c and benefit in hypoglycemia risk with Gla-300 were documented, it would be expected that second-generation basal insulins would be used more often in the oldest people [21, 22].
Data on the cohort switching from Deg-100 to Gla-300 are particularly interesting. These patients showed the worst metabolic control (HbA1c 8.5 ± 1.3% vs. 8.3 ± 1.4% in cohort 1 and 8.1 ± 1.5% in cohort 3) although they were more often treated with four or more insulin injections/day and with the highest insulin doses (72 U/day of total insulin vs. 50–60 U/day in the other two cohorts), identifying a particularly complex patient phenotype. After the switch, metabolic control improved (especially in terms of FBG) and weight decreased although the total insulin dose was slightly increased. No severe hypoglycemic episodes occurred in this cohort.
It is important to underline that observed changes in metabolic control were reasonably due to insulin therapy, since it is recognized that changes in lifestyle (diet and physical activity) seldom occur in the elderly T2DM population [4]. However, we cannot exclude that some reinforcement of education on lifestyle may have occurred together with the switch of basal insulin.
Regarding the Gla-300 titration, the dose increase observed in clinical practice is particularly low (< 1 U during 6 months in all cohorts) with respect to what could be expected based on data from clinical trials and a summary of characteristics of the product. This attitude of participating diabetologists seems to indicate that the therapeutic inertia in terms of insulin titration documented in the Gla-100 cohort is also observed with the new-generation Gla-300 basal insulin. This finding is particularly relevant, as the prompt efficacy of Gla-300 mainly depends on an appropriate and timely dose titration because of its known pharmacologic characteristics and PK/PD profile.
Two recently published prospective real-life comparative studies (REACH and REGAIN) showed no differences in glycemic control with Gla-300 versus standard-of-care basal insulin over 12 months. However, suboptimal insulin titration occurred in both treatment arms suggesting that additional titration instruction/support may be required for patients to fully derive the benefits from newer basal insulin formulations [23].
Short-acting insulin optimization after the switch also seems to be sub-optimal in most patients treated with Gla-300. Thus, the suboptimal titration of insulin doses observed under real-life conditions suggests the lack of adequate education for both patients and physicians.
The paucity of data on SMBG available in EMRs, as also previously documented [24], suggests poor attention to the collection of the data regarding hypoglycemia and glycemic profiles in clinical practice at least in people with T2DM.
The site questionnaire emphasized the role of risk of hypoglycemia, glucose variability, and inadequate metabolic control as the main reasons for switching from any other basal insulin to Gla-300.
Real-world studies provide information that is complementary to randomized clinical trials and may be more generalizable and pertinent to clinicians and healthcare systems.
Data obtained in the DELIVER program [19, 21, 22, 25,26,27] with comparative data in propensity-matched cohorts show that people with T2DM who switched to Gla-300 from other basal insulin analog regimens improved their glycemic control with fewer hypoglycemia events, confirming and complementing the results of the EDITION registration clinical program [7,8,9]. Particularly, the DELIVER 2 and 3 study showed that switching basal insulins in T2DM people with elevated HbA1c levels to Gla-300 was associated with significantly lower risk of hypoglycemia than switching to other basal insulins (mainly first-generation basal insulin), with comparable glycemic control; the lower risk of hypoglycemia-related, diabetes-related, and all-cause emergency room services translates into lower costs in patients using Gla-300 [22, 24].
The non-comparative EU-TREAT study demonstrated that switching patients to Deg-100 from first-generation basal insulins improved glycemic control and significantly reduced the risk of hypoglycemia in routine clinical practice [28]. In the DELIVER D + comparative US study on T2DM high-risk insulin-treated patients, switching from a first- (Gla-100 or detemir) second-generation (Gla-300 or Deg-100) basal insulin analog resulted in similar improvements in glycemic control and in a similar or better effect on hypoglycemia with Gla-300 compared with Deg-100 [19].
Compared to these studies, data from this real-world study confirm the effectiveness and safety of the switch to Gla-300 from other basal insulins, although a major effort regarding timely and efficient titration is needed.
This study has strengths and limitations. Among the strengths, this study provides important complementary information about the effectiveness of Gla-300 in clinical practice. Compared to other studies based on EMRs, this study was able to provide an accurate description of T2DM patient characteristics switched to Gla-300 in real life, data on insulin doses, and physician attitudes towards insulin therapy in Italy.
As for the limitations, the inclusion of a network of centers with homogeneous clinical approaches could limit the generalizability of the results. Furthermore, information about causes and treatment of severe hypoglycemia and data on non-severe hypoglycemia were not found in the database, suggesting the need to improve the systematic revision of SMBG data through EMRs. The overall duration of insulin therapy and other concomitant treatments before the switch was also not available in the database. Finally, the limited number of observations did not allow for propensity score matching, thus precluding the possibility to perform a comparative effectiveness analysis among the cohorts. Therefore, this study has a purely descriptive nature. However, methods applied in this study including EMRs data retrospective review and secure anonymous data transfer represented a pilot phase, used to design two ongoing, larger studies in T1DM and T2DM in European populations, evaluating the switch to Gla-300 or Deg-100 from other basal insulins and comparing their effectiveness and safety after the application of propensity score matching.
Conclusion
In conclusion, this regional, multicenter, non-interventional study based on the retrospective analysis of real-world data from Italy shows that switching to Gla-300 from previous first-generation basal insulins is associated with improvements in HbA1c levels of about − 0.3% despite a suboptimal titration of both basal and short-acting insulin, without an increase in the risk of severe hypoglycemia. The switch from Deg-100 to Gla-300 is associated with a decrease in body weight of − 1.47 kg, despite a slight increase in total insulin daily doses. The study emphasizes the urgent need for optimization of insulin titration to maximize the benefits of basal insulins in these patients, since the therapeutic inertia documented in the Gla-100 cohort is also found with the new-generation Gla-300 basal insulin.
References
UK Prospective Diabetes Study Group. UK prospective diabetes study 16. Overview of 6 years’ therapy of type II diabetes: a progressive disease. Diabetes. 1995;44:1249–58.
UK Prospective Diabetes Study Group. Glycemic control with diet, sulfonylurea, metformin, or insulin in patients with type 2 diabetes mellitus: progressive requirement for multiple therapies (UKPDS 49). JAMA. 1999;281:2005–122.
Davies MJ, D'Alessio DA, Fradkin J, Kernan WN, Mathieu C, Mingrone G, Rossing P, Tsapas A, Wexler DJ, Buse J. Management of hyperglycemia in type 2 diabetes, 2018. A Consensus Report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2018;41:2669–701.
Rossi MC, Candido R, Ceriello A, Cimino A, Di Bartolo P, Giorda C, Esposito K, Lucisano G, Maggini M, Mannucci E, Meloncelli I, Nicolucci A, Pellegrini F, Scardapane M, Vespasiani G. Trends over 8 years in quality of diabetes care: results of the AMD annals continuous quality improvement initiative. Acta Diabetol. 2015;52:557–71.
Becker RH, Dahmen R, Bergmann K, Lehmann A, Jax T, Heise T. New insulin glargine 300 Units · mL−1 provides a more even activity profile and prolonged glycemic control at steady state compared with insulin glargine 100 Units · mL−1. Diabetes Care. 2015;38:637–43.
Bailey TS, Pettus J, Roussel R, Schmider W, Maroccia M, Nassr N, Klein O, Bolli GB, Dahmen R. Morning administration of 0.4U/kg/day insulin glargine 300U/mL provides less fluctuating 24-hour pharmacodynamics and more even pharmacokinetic profiles compared with insulin degludec 100U/mL in type 1 diabetes. Diabetes Metab. 2018;44:15–211.
Riddle M, Bolli GB, Ziemen M, Muehlen-Bartmer I, Bizet F, Home PD. New insulin glargine 300 units/ml versus glargine 100 units/ml in people with type 2 diabetes using basal and mealtime insulin: glucose control and hypoglycemia in a 6-month randomized controlled trial (Edition I). Diabetes Care. 2014;37:2755–62.
Yki-Järvinen H, Bergenstal R, Ziemen M, Wardecki M, Muehlen-Bartmer I, Boelle E, Riddle MC, EDITION 2 Study Investigators. New insulin glargine 300 units/mL versus glargine 100 units/mL in people with type 2 diabetes using oral agents and basal insulin: glucose control and hypoglycemia in a 6-month randomized controlled trial (Edition 2). Diabetes Care. 2014;37:3235–43.
Bolli GB, Riddle MC, Bergenstal RM, Ziemen M, Sestakauskas K, Goyeau H, Home PD, on behalf of the EDITION 3 study investigators. New insulin glargine 300 U/ml compared with glargine 100 U/ml in insulin-naïve people with type 2 diabetes on oral glucose-lowering drugs: a randomized controlled trial (Edition 3). Diabetes Obes Metab. 2015;17:386–94.
Haahr H, Heise T. A review of the pharmacological properties of insulin degludec and their clinical relevance. Clin Pharmacokinet. 2014;5:787–800.
Garber AJ, King AB, Del Prato S, Sreenan S, Balci MK, Munoz-Torres M. Insulin degludec, an ultra-longacting basal insulin, versus insulin glargine in basal-bolus treatment with mealtime insulin aspart in type-2 diabetes (BEGIN Basal-Bolus Type 2): a phase 3, randomised, open-label, treat-to-target noninferiority trial. Lancet. 2012;379:1498–507.
Zinman B, Philis-Tsimikas A, Cariou B, Handelsman Y, Rodbard HW, Johansen T. Insulin degludec versus insulin glargine in insulin-naive patients with type 2 diabetes: a 1-year, randomized, treat-to-target trial (BEGIN Once Long). Diabetes Care. 2012;35:2464–71.
Gough SC, Bhargava A, Jain R, Mersebach H, Rasmussen S, Bergenstal RM. Lowvolume insulin degludec 200 Units/mL once daily improves glycaemic control similarly to insulin glargine with a low risk of hypoglycaemia in insulin-naïve patients with type 2 diabetes: a 26-week, randomized, controlled, multinational, treat-to-target trial: the BEGIN LOW VOLUME trial. Diabetes Care. 2013;36:2536–42.
Bode BW, Chaykin LB, Sussman AM, Warren ML, Niemeyer M, Rabøl R, Rodbard HW. Efficacy and Safety of Insulin Degludec 200 U/mL and Insulin Degludec 100 U/mL in Patients with Type 2 Diabetes (Begin: Compare). Endocr Pract. 2014;20:785–91.
Roussel R, Ritzel R, Boëlle-Le Corfec E, Balkau B, Rosenstock J. Clinical perspectives from the BEGIN and EDITION programmes: Trial-level meta-analyses outcomes with either degludec or glargine 300U/mL vs glargine 100U/mL in T2DM. Diabetes Metab. 2018;44:402–9.
Rosenstock J, Cheng A, Ritzel R, Bosnyak Z, Devisme C, Cali AMG, Sieber J, Stella P, Wang X, Frías JP, Roussel R, Bolli GB. More similarities than differences testing insulin glargine 300 units/mL versus insulin degludec 100 units/mL in insulin-naive type 2 diabetes: the randomized head-to-head BRIGHT trial. Diabetes Care. 2018;41:2147–54.
Philis-Tsimikas A, Klonoff DC, Khunti K, Bajaj HS, Leiter LA, Hansen MV, Troelsen LN, Ladelund S, Heller S, Pieber TR, CONCLUDE Study Group. Risk of hypoglycaemia with insulin degludec versus insulin glargine U300 in insulin-treated patients with type 2 diabetes: the randomised, head-to-head CONCLUDE trial. Diabetologia. 2020;63:698–710.
Del Prato S. How conclusive is the CONCLUDE trial? Diabetologia. 2020;63:692–7.
Sullivan SD, Bailey TS, Roussel R, Zhou FL, Bosnyak Z, Preblick R, Westerbacka J, Gupta RA, Blonde L. Clinical outcomes in real-world patients with type 2 diabetes switching from first- to second-generation basal insulin analogues: comparative effectiveness of insulin glargine 300 units/mL and insulin degludec in the DELIVER D+ cohort study. Diabetes Obes Metab. 2018;20:2148–58.
Berger ML, Sox H, Willke RJ, Brixner DL, Eichler HG, Goettsch W, Madigan D, Makady A, Schneeweiss S, Tarricone R, Wang SV, Watkins J, Mullins CD. Good practices for real-world data studies of treatment and/or aint ISPOR-ISPE Special Task Force on Real-World Evidence in Health Care Decision Making. Value Health. 2017;20:1003–8.
Ritzel R, Harris SB, Baron H, Florez H, Roussel R, Espinasse M, Muehlen-Bartmer I, Zhang N, Bertolini M, Brulle-Wohlhueter C, Munshi M, Bolli GB. A randomized controlled trial comparing efficacy and safety of insulin glargine 300 units/ml versus 100 units/ml in older people with type 2 diabetes: results from the SENIOR study. Diabetes Care. 2018;41:1672–80.
Bailey TS, Wu J, Zhou FL, Gupta RA, Menon AA, Berhanu P, Westerbacka J, Van Vleet J, Blonde L. Switching to insulin glargine 300 units/mL in real-world older patients with type 2 diabetes (DELIVER 3). Diabetes Obes Metab. 2019;21:2384–93.
Freemantle N, Mauricio D, Giaccari A, Bailey T, Roussel R, Franco D, Berthou B, Pilorget V, Westerbacka J, Bosnyak Z, Bonnemaire M, Cali AMG, Nguyên-Pascal ML, Penfornis A, Perez-Maraver M, Seufert J, Sullivan SD, Wilding J, Wysham C, Davies M. Real-world outcomes of treatment with insulin glargine 300 U/mL versus standard-of-care in people with uncontrolled type 2 diabetes mellitus. Curr Med Res Opin. 2020;19:1–11.
Rossi MC, Lucisano G, Ceriello A, Mazzucchelli C, Musacchio N, Ozzello A, Nicolucci A, Di Bartolo P, AMD Annals-SMBG Study Group. Real-world use of self-monitoring of blood glucose in people with type 2 diabetes: an urgent need for improvement. Acta Diabetol. 2018;55:1059–66.
Zhou FL, Ye F, Berhanu P, Gupta VE, Gupta RA, Sung J, Westerbacka J, Bailey TS, Blonde L. Real-world evidence concerning clinical and economic outcomes of switching to insulin glargine 300 units/mL vs other basal insulins in patients with type 2 diabetes using basal insulin. Diabetes Obes Metab. 2018;20:1293–7.
Bailey TS, Zhou FL, Gupta RA, Preblick R, Gupta VE, Berhanu P, Blonde L. Glycaemic goal attainment and hypoglycaemia outcomes in type 2 diabetes patients initiating insulin glargine 300 units/mL or 100 units/mL: real-world results from the DELIVER Naïve cohort study. Diabetes Obes Metab. 2020;21:1596–605.
Sullivan SD, Nicholls CJ, Gupta RA, Menon AA, Wu J, Westerbacka J, Bosnyak Z, Frias JP, Bailey TS. Comparable glycaemic control and hypoglycaemia in adults with type 2 diabetes after initiating insulin glargine 300 units/mL or insulin degludec: The DELIVER Naïve D real-world study. Diabetes Obes Metab. 2019;21:2123–32.
Siegmund T, Tentolouris N, Knudsen ST, Lapolla A, Prager R, Phan TM, Wolden ML, Schultes B, EU-TREAT study group. A European, multicentre, retrospective, non-interventional study (EU-TREAT) of the effectiveness of insulin degludec after switching basal insulin in a population with type 1 or type 2 diabetes. Diabetes Obes Metab. 2018;20:689–97.
Acknowledgements
Meteda (San Benedetto, Italy) developed the software for the data extraction.
Funding
The study was funded by Sanofi SpA, Milano, Italy. Coresearch (Pescara, Italy) was the Clinical Research Organization involved in the data management, statistical analysis, and medical writing of the study. OPIS (Milano, Italy) was the Clinical Research Organization involved in regulatory activities of the study. CORESEARCH will fund the journal’s Rapid Service Fee.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
Monica Larosa is an employee of Sanofi. Antonio Nicolucci and Maria Chiara Rossi received funding for research from Sanofi, NovoNordisk, Alfasigma, Artsana, Astrazeneca, Johnson&Johnson, Medtronic, Shionogi, SOBI, Meteda and Theras. Mauro Ragonese, Stefania Angotti, Stefania Annese, Laura Cruciani, Michela Dainelli, Enrica Salomone, Caterina Saponara, Roberta Semprini, Giuseppe Prosperini, Giuseppe Lucisano, and Michele Sacco have nothing to disclose.
Compliance with Ethics Guidelines
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national - Ethics Committee of the ACISMOM network) and with the Helsinki Declaration of 1975, as revised in 2008 (5). Informed consent was obtained from all patients for being included in the study.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Additional information
Digital Features
To view digital features for this article go to: https://doi.org/10.6084/m9.figshare.12732233.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Ragonese, M., Larosa, M., Angotti, S. et al. Clinical Outcomes of Switching to Insulin Glargine 300 U/ml from Other Basal Insulins in People with Type 2 Diabetes in Italy: A Real-World Study. Diabetes Ther 11, 2283–2298 (2020). https://doi.org/10.1007/s13300-020-00902-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-020-00902-1