Abstract
Introduction
The aim of this study was to objectively analyze the correlation between dietary components and blood glucose variation by means of continuous glucose monitoring (CGM).
Methods
Patients with type 1 diabetes mellitus (T1DM) who received CGM to manage their blood glucose levels were enrolled into the study, and the components of their total caloric intake were analyzed. Glycemic variation parameters were calculated, and dietary components, including percentages of carbohydrate, protein and fat in the total dietary intake, were analyzed by a dietitian. The interaction between parameters of glycemic variability and dietary components was analyzed.
Results
Sixty-one patients with T1DM (33 females, 28 males) were enrolled. The mean age of the participants was 34.7 years, and the average duration of diabetes was 14 years. Glycated hemoglobin before CGM was 8.54%. Participants with a carbohydrate intake that accounted for < 50% of their total caloric intake had a longer DM duration and a higher protein and fat intake than did those with a carbohydrate intake that accounted for ≥ 50% of total caloric intake, but there was no between-group difference in total caloric intake per day. The group with a carbohydrate intake that accounted for < 50% of their total caloric intake also had lower nocturnal continuous overlapping net glycemic action (CONGA) 1, − 2 and − 4 values. The percentage of protein intake had a slightly negative correlation with mean amplitude of glycemic excursions (MAGE) (r = − 0.286, p < 0.05) and a moderately negative correlation with coefficient of variation (CV) (r = 0.289, p < 0.05). One additional percentage of protein calories of total calories per day decreased the MAGE to 4.25 mg/dL and CV to 0.012 (p < 0.05). The optimal dietary protein percentage for MAGE < 140 mg/dL was 15.13%. The performance of predictive models revealed the beneficial effect of adequate carbohydrate intake on glucose variation when combined with protein consumption.
Conclusions
Adequate carbohydrate consumption—but not more than half the daily total calories—combined with protein calories that amount to approximately 15% of the daily caloric intake is important for glucose stability and beneficial for patients with T1DM.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The 2019 Standards of Medical Care in Diabetes published by the American Diabetes Association recommends glycated hemoglobin (HbA1c) as the gold standard for the assessment of blood glucose (BG) control in patients with diabetes mellitus (DM) [1]. However, HbA1c level merely demonstrates the average BG level—it does not represent the current BG level or glycemic variability. Indeed, whether or not glycemic variability or the average BG level is the more crucial parameter with respect to diabetic complications is a matter of debate [2, 3]. Long-term glycemic variability may be the etiology of microvascular and macrovascular complications associated with DM [4]. The glycemic variability index has been found to be associated with the frequency of hypoglycemia events [5], and frequent hypoglycemia events in persons with either type 1 DM or type 2 DM (T1DM, T2DM, respectively) are also associated with a number of adverse outcomes, including increased mortality rate, cardiovascular disease and cognitive dysfunction [6]. In addition, the glycemic variability index is correlated with oxidative stress. Monnier et al. [7] collected the 24-h urinary excretion of free 8-iso prostaglandin F2 (8-iso PGF2), a free radical-mediated oxidation product of arachidonic acid, in 21 patients with DM and 21 controls. A continuous glucose monitoring (CGM) system was used to measure the BG level, and glucose fluctuations were calculated as the mean amplitude of glycemic excursions (MAGE). The result showed that MAGE had significant correlation with urinary 8-iso PGF2. Roberts and Morrow also found the same result [8]. In critically ill patients, increasing glycemic variability contributes to a higher mortality rate [9]. Taken together, these observations indicate that reduced glycemic variability is important for diabetes control.
Diet plays an important role in glycemic variation. The World Health Organization (WHO) recommends that in a healthy diet for the general population, < 30% of total energy intake should be derived from fats and that unsaturated fats are a better source of energy than saturated fats [10]. With respect to carbohydrate intake, although the WHO does not recommend an absolute daily total energy intake, it does suggest that adequate and individualized amounts of carbohydrates be consumed [11,12,13]. There is a trend to recommend a low carbohydrate diet for better BG control in patients with T1DM or T2DM [14,15,16]; however, the long-term benefits of such a diet are not persistent [17], and there are differing opinions on the possibility of side effects [18, 19]. There are additional concerns about potential adverse effects of a low carbohydrate diet in persons with T1DM, including diabetic ketoacidosis, worsening of the lipid profile and an uncertainty regarding interference with growth in children, although despite the heterogenous results from recent studies, it would seem that there are also some benefits to such a diet, such as reduced insulin dose, decreased HbA1c and borderline body weight change [20].
With advances in technology, CGM has become a widely accepted tool in the management of DM. CGM can assist physicians in collecting up-to-date information on glycemic changes in their patients with DM. In 2003, Brynes et al. [21] evaluated nine healthy subjects who received CGM for 2 weeks and a low glycemic index (GI) diet intervention for 1 of these weeks; the purpose of this study design was to compare the difference in BG levels. The reported effect of BG control with a low GI diet highlighted the benefit of CGM in BG monitoring [21]. CGM can therefore be a tool in the physican’s arsenal to achieve better glucose control, but the use of CGM in DM patients as a means to observe the effect of food on BG level has not been well studied.
Given this lack of data on the use of CGM to monitor the effect of diet on BG level in persons with diabetes, we enrolled 61 patients with T1DM who received CGM in our study. These patients continued receiving CGM and agreed to maintain a comprehensive food diary. The dietary components of the diets, including carbohydrate, protein and fat, were analyzed by a qualified dietitian. The primary endpoint of this study was to identify which nutrient intake components have the greatest impact on glycemic variability.
Methods
Participants
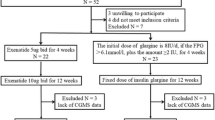
A total of 61 patients with T1DM who received CGM at Chang Gung Memorial Hospital (CGMH), a medical center in Taiwan, from November 2007 to July 2018 were enrolled. Participants were not asked to change their diet and amount of exercise. Inclusion criteria were: diagnosis of T1DM; ambulatory status; and willingness to receive CGM and cooperate in keeping a food diary, whether by the patient or by a family member. Exclusion criteria included recent history of drug or alcohol abuse; serious cardiovascular disorders; participation in another clinical investigation study; and ongoing influenza, autoimmune disease or other metabolic disorders.
Permission was obtained from the Institutional Review Board (IRB) and ethics committees of CGMH (no. 200800097B0) for a retrospective review of the medical records of the study subjects. The IRB waived the requirement for obtaining informed consent. Confidentiality of the research subjects was maintained in accordance with the requirements of the IRB of CGMH (Taipei, Taiwan). The study conformed with the Helsinki Declaration of 1964, as revised in 2013, concerning human and animal rights.
Diet Records
The amount and types of food consumed during the day, including at breakfast, lunch and dinner, fruits, snacks and beverages, were self-recorded by the participants or by a family member after being informed by a certificated dietitian. The nutrient elements of the diet, including carbohydrates, protein, fats and calories, were analyzed and calculated by the professional dietitian so we could have confidence in the accuracy of this analysis. Food was predominantly Asian and cooked well.
Glucose Monitoring
Participants were monitored by the Continuous Glucose Monitoring System (CGMS) Gold (MiniMed CGMS MMT-7102-W; Medtronic, Inc., Northridge, CA, USA). The data were collected at 24-h intervals (from midnight to midnight the next day); therefore, there was a short average duration of 3.1 days to allow time for sensor wetting. Data were not used when values were above the mean absolute relative difference threshold of 8.7%, in accordance with the manufacturer. The CGMS sensor was implanted subcutaneously in the abdomen, buttocks or arm. It detected the interstitial fluid glucose level every 10 s, with an average value output every 5 min. The CGMS recognizes the glucose level of interstitial fluid in the range of 40 to 400 mg/dL; if the glucose level falls below or rises above the detectable range, the value is presented as 40 or 400 mg/dL, respectively.
Outcome Measures
At the end of the study period, the CGMS data were downloaded using the MiniMed Solutions CGMS sensor (MMT-7310, version 3.0B [3.0.116]; Medtronic, Inc.). Several parameters were calculated for all glucose values in order to provide a full picture of glucose variability, including the standard deviation (SD; [21]), percentage coefficient of variation (%CV; [22]) and MAGE [7]. Patients with T1DM have a greater glucose variability than those without DM [23], with an average MAGE level of around 140 mg/dL; therefore, we used a MAGE of 140 mg/dL as the cutoff value for high glucose variability. The area under the curve (AUC) of a glucose level > 180 mg/dL (AUC180) and < 70 mg/dL (AUC70) were designated as hyperglycemic and hypoglycemic periods, respectively. The AUCt and AUCn represent the total and normal (70–180 mg/dL) AUCs of glucose levels, respectively. The risk of hypoglycemia and hyperglycemia events were calculated as a low blood glucose index (LBGI) and high blood glucose index (HBGI) [24, 25], respectively. The M-value was calculated to evaluate glycemic variability [24, 26]. The continuous overlapping net glycemic action (CONGA) was used as an index to assess intraday glycemic variability [2]. CONGA(n) represents the SD of all valid differences between a current observation and an observation (n) hours earlier [27]. The index formulas are reported in Table 1 [2, 4, 24, 28].
Statistics
All statistical analyses were performed using Statistical Analyses Package Program (SPSS) version 20.0 (IBM Corp., Armonk, NY, USA) and Stata/SE 9.0 for Windows (StataCorp, College Station, TX, USA). Differences between groups of continuous variables were calculated using a paired Student’s t test, and correlation between two continuous variables were analyzed by linear regression. The odds ratio (OR) was determined by logistic regression. A Chi-square test was used to analyze differences within nominal variables groups. ORs and 95% confidence intervals (CIs) for MAGE ≥ 140 mg/dL were derived from logistic regression models using subjects with MAGE < 140 mg/dL as the reference group (OR 1). Three predictive models were constructed based on multiple logistic regression models using the regression coefficients as the weight for the dependent variables. Age, body mass index (BMI), gender, and DM duration, carbohydrate, protein and fat content (%) and total calories were included in different predictive models. The diagnostic performance was evaluated by the AUC. The optimal cutoff point was derived from the receiver operator characteristic (ROC) curve with the shortest distance to sensitivity = 1 and 1 − specificity = 0. The sensitivity was the probability that the prediction would be positive for subjects with a MAGE ≥ 140 mg/dL, and the specificity was the probability that the prediction would be negative for subjects without a MAGE ≥ 140 mg/dL. A p value < 0.05 was considered to indicate statistical significance, and 59 participants would provide 0.90 power.
Results
Demographic Characteristics
A total of 61 patients (33 women, 28 men) with T1DM who received CGM assessments at Chang Gung Memorial Hospital (CGMH) from November 2007 to July 2018 were enrolled in the study. The age of the participants ranged from 9.9 to 80.7 years. The demographic characteristics are summarized in Table 2. Participants with a carbohydrate intake that accounted for < 50% of their total caloric intake had a longer DM duration and a higher protein and fat intake than did those with a carbohydrate intake that accounted for ≥ 50% of total caloric intake, but there was no between-group difference in total caloric intake per day. The former (< 50%) group also had lower nocturnal CONGA1, − 2 and − 4 values. The most common reason for receiving CGM was high glucose excursion. With respect to daily nutrient intake, patients with T1DM consumed on average 1641.74 kcal/day, which after correction for body weight was 28.19 kcal/day; carbohydrate, protein and fat intake was 3.46, 1.11 and 1.10 g/kg/day, respectively.
CGM Index
The computerized analysis of glycemic variability indexes (Table 3) revealed that the MAGE, which is most representative index of glycemic variation, was 142.19 in the participants. MAGE, SD and CV, which are affected by nutrient intake, are presented in Table 4. The impact of nutrient components on time in range is given in Electronic Supplementary Material Table 1. Pearson’s correlations of MAGE, SD and CV with age, BMI, DM duration, HbA1c and calories according to percentage of carbohydrate, protein, and fat each day are presented in Table 5.
Predictive Models
The average MAGE level of the participants was determined to be approximately 140 mg/dL; therefore, the study patients were divided into two groups based on the MAGE level (≥ 140 mg/dL and < 140 mg/dL). Based on the logistic regression model, age, BMI, male gender, DM duration, baseline HbA1c, percentage of calories as carbohydrate, percentage of calories as fat and total caloric intake were calculated as a risk factor of a higher MAGE. Different factors were used to construct the three most feasible models, and the results are presented in Table 6. In model 1, age and DM duration were significant risk factors of higher MAGE (OR 0.944, p = 0.034 and OR 1.092, p = 0.033, respectively). In model 2, age and percentage of protein calories intake were significant risk factors of higher MAGE (OR 0.938, p = 0.024 and 0.691, p = 0.011, respectively). In model 3, age, baseline HbA1c, percentage of carbohydrate in diet and fat intake were significant risk factors of higher MAGE (OR 0.936, p = 0.037, OR 1.926, p = 0.039, OR 1.568, p = 0.009 and OR 1.739, p = 0.006, respectively).
A ROC curve was used to analyze the best cutoff value of the percentage of dietary protein and MAGE < 140 mg/dL. The best discrimination point determined by the Younden index was 15.13%, with a sensitivity of 55.6% and a specificity of 81.8% (p = 0.019; Fig. 1).
The receiver operator characteristic (ROC) curve analysis determines the best discrimination point of percentage of dietary protein and mean amplitude of glycemic excursions (MAGE) < 140 mg/dL. The best discrimination point of dietary protein percentage, as determined by the Younden index was 15.13%, with a sensitivity of 55.6% and a specificity of 81.8%. Area under the ROC curve was 0.689 with a 95% confidence interval of 0.546–0.831; p = 0.019; standard error = 0.073
As shown in Table 7 and Fig. 2, age, BMI, gender, DM duration, percentage of caloric intake as carbohydrate, protein and fat and total calories intake per day were analyzed as factors contributing to the risk for high BG variation. The three predictive models derived from these variables showed a good performance in calculating the risk of MAGE ≥ 140 mg/dL. Using optimal cutoff values, the sensitivity ranged from 70.4 to 85.2% and the specificity ranged from 66.7 to 78.8%. Similar results were validated by the leave-one-out cross-validation method, which had a sensitivity of 51.9–66.7% and specificity of 63.6–75.8%.
Discussion
Principal Findings
The current study was a pilot study in which CGM was used to determine the interstitial glucose level with the aim to objectively study the relationship between nutrient component intake and glycemic variability. The results showed that in persons with T1DM, an adequate amount of carbohydrate and protein intake is better for stable glycemic variability.
Interpretation and Research Implications of Results
Regarding the parameters we used to evaluate glycemic variability, the MAGE was defined as the level of glucose fluctuation. The normal range of glucose fluctuation is 22–60 mg/dL; glucose fluctuation of > 120 mg/dL is consistent with poorly controlled diabetes [29]. In the Chinese population, MAGE of < 70 mg/dL is recommended as the normal reference range [30]. T2DM patients have higher glucose variability [23], with an average MAGE level around 140 mg/dL; therefore, in our study we used MAGE of 140 mg/dL as the high glucose variability cutoff value.
Although the 2019 American Diabetes Association (ADA) guideline does not contain a recommended dietary carbohydrate ratio, the 2015–2020 Dietary Guidelines for Americans (8th edn; https://health.gov/dietaryguidelines/2015/guidelines/) recommends that at least 50% of daily total caloric intake be in the form of carbohydrates. Giugliano et al. reported that people with metabolic disorder should choose a diet of the appropriate carbohydrates that provide between 40 and 50% of the daily energy requirement [31]. Based on these recommendations, we further grouped the participants by proportion of carbohydrate calorie intake with 50% of calorie intake as the cutoff value. We found that the group who had a low carbohydrate intake obtained their total daily calories by consuming a higher proportion of protein and fat; consequently, there was no difference in total daily calorie intake between the two groups.
Carbohydrate-restricted diets with a reliance on protein and fat as energy sources for health maintenance and BG control have been shown not to have a great benefit in terms of managing glucose fluctuations. In the current study, we constructed three models to predict the risk for MAGE ≥ 140 mg/dL that included age, BMI, gender, DM duration, baseline HbA1c, percentages of carbohydrate, protein and fat in daily dietary intake and total calories. The results suggested that carbohydrate is necessary for BG stability. Snorgaard et al. [17] systemically reviewed ten randomized trials that included 1376 participants with T2DM who were divided into a low-to-moderate carbohydrate diet (LCD; energy < 45%) group and high-carbohydrate diet (HCD) group. In the first year, the patients on the LCD initially had a lower HbA1c level, such that the lower the carbohydrate intake, the better the HbA1c level. After 1 year of follow-up, however, the HbA1c level was similar in the two groups [17]. However, few studies have assessed the effect of a low-carbohydrate diet on BG variability in persons with T1DM. Turton et al. reviewed nine studies and concluded that a low-carbohydrate diet might reduce HbA1c in persons with T1DM, but their result was controversial [32]. In the present study, SD was modestly correlated with HbA1c, and the result was compatible with the finding by Kuenen et al. [33] that in T1DM, high SD, which is representative of high glucose variability, is associated with higher HbA1c.
Several complications of a ketogenic diet have been noted, including renal stones, gallstones, dehydration, gastrointestinal disturbances, hypercholesterolemia, metabolic acidosis, insulin resistance, vascular inflammation, liver dysfunction and cerebral abnormalities [34]. In mice fed a ketogenic diet for 22 weeks, no benefit of weight loss was noted, despite there being an initial weight loss; furthermore, hepatic steatosis, glucose intolerance and decreased β- and α-cells were reported [35].
In contrast, the traditional diet of the Okinawan population, a group which has one of the longest lifespans in the world, is rich in carbohydrates, with sweet potato as one of the main components; up to 80% of the daily total energy intake of this population is in the form of carbohydrate. The rates of age-related diseases, such as coronary artery disease, prostate cancer, breast cancer and lymphoma, are lower than those in age-matched Americans [36]. Therefore, it appears to be that quality and not quantity is important in terms of carbohydrate-related BG control. The ADA recommends that dietary carbohydrates as an energy source be derived from whole grains, vegetables, fruits, legumes and dairy products, with a preference for foods high in fiber, and that free sugar be avoided [12].
As glycemic variability marker, nocturnal CONGA1, -2 and -4 were found to be significantly lower in persons on a low-carbohydrate diet [37]. Tay et al. found that patients with T2DM on an energy-restricted, low-carbohydrate, high-unsaturated fat diet had lower CONGA1 and CONGA4 than those on a high-carbohydrate, low-fat diet [38]. Noakes et al. also compared glucose variability in persons with T2DM on a high-carbohydrate diet with those on a low-carbohydrate diet; these authors noted a lower CONGA1 in the low-carbohydrate group [39]. The current focus of studies on the effect of diet on glycemic variability is on T2DM, and few studies have assessed the effect of diet on glycemic variability in persons with T1DM. Ranjan et al. grouped ten patients with T1DM into a high-carbohydrate diet group and a low-carbohydrate diet group, respectively, and noted lower glycemic variability parameters, including SD, CV, MAGE and LBGI, in the low-carbohydrate diet group [40].
In our study, one additional percentage increase in the daily intake of calories in the form of protein decreased the MAGE to 4.25 mg/dL and CV to 0.012 (p < 0.05). The relationship between glycemic variability and protein intake in T1DM has not been well studied to date, but a number of studies have looked at the effect of protein intake on glucose variability in T2DM. Gannon et al. [41] enrolled 12 patients with untreated DM and divided them into two groups [high-protein diet (protein:carbohydrate:fat, 30:40:30) and a control diet (protein:carbohydrate:fat, 15:55:30)]. After 5 weeks, the high-protein diet group showed a 40% decrease in the mean 24-h integrated glucose levels and a > 0.5% decrease in the HbA1c level (0.8 vs. 0.3%). Alish et al. [42] demonstrated that there is a lower MAGE level in the diabetic-specific formula with a higher percentage of protein. Fabricatore et al. [43] also showed that MAGE was inversely associated with protein intake.
We also found that the longer the duration of diabetes, the lower the intake of carbohydrates by the participants. We suggest that this was due to better education on diet with a longer duration of DM, which led directly to the participants themselves decreasing their carbohydrate intake.
Strengths and Limitations
This study had several limitations. First, the patients had to have a CGM device and maintain a daily diet diary, which led to a relatively small sample size. Second, it was a cross-sectional study. Third, the participant self-reported on diet composition, although a dietitian would recheck the record and confirm it. Fourth, the subtypes of carbohydrate were not analyzed. Fifth, each patient followed his/her normal daily routine regarding physical activity; therefore, we could not rule out the effect of excessive exercise on glucose variability in the analysis.
The strengths of this study are that it examines the impact of food components on glucose variability, which has been only infrequently studied, and that it is the first to access data on dietary consumption and link this to CGM data among patients with DM in Taiwan.
Conclusions
In conclusion, adequate carbohydrate consumption—but not more than half the daily total calories—combined with protein calories that amount to approximately 15% of the daily caloric intake is important for glucose stability and beneficial for patients with T1DM.
References
American Diabetes Association. 6. Glycemic targets: standards of medical care in diabetes—2019. Diabetes Care. 2019;42[Suppl 1]:S61–70.
Siegelaar SE, Holleman F, Hoekstra JBL, DeVries JH. Glucose variability; does it matter? Endocr Rev. 2010;31(2):171–82.
Brownlee M, Hirsch IB. Glycemic variability: a hemoglobin A1c-independent risk factor for diabetic complications. JAMA. 2006;295(14):1707–8.
Jung HS. Clinical implications of glucose variability: chronic complications of diabetes. Endocrinol Metab (Seoul). 2015;30(2):167–74.
Rama Chandran S, Tay WL, Lye WK, et al. Beyond HbA1c: comparing glycemic variability and glycemic indices in predicting hypoglycemia in type 1 and type 2 diabetes. Diabetes Technol Ther. 2018;20(5):353–62.
Yun JS, Ko SH. Risk factors and adverse outcomes of severe hypoglycemia in type 2 diabetes mellitus. Diabetes Metab J. 2016;40(6):423–32.
Monnier L, Mas E, Ginet C, et al. Activation of oxidative stress by acute glucose fluctuations compared with sustained chronic hyperglycemia in patients with type 2 diabetes. JAMA. 2006;295(14):1681–7.
Roberts LJ II, Morrow JD. Isoprostanes. Novel markers of endogenous lipid peroxidation and potential mediators of oxidant injury. Ann N Y Acad Sci. 1994;744:237–42.
Krinsley JS. Glycemic variability: a strong independent predictor of mortality in critically ill patients. Crit Care Med. 2008;36(11):3008–13.
Hooper L, Abdelhamid A, Moore HJ, Douthwaite W, Skeaff CM, Summerbell CD. Effect of reducing total fat intake on body weight: systematic review and meta-analysis of randomised controlled trials and cohort studies. BMJ. 2012;345:e7666.
He M, van Dam RM, Rimm E, Hu FB, Qi L. Whole-grain, cereal fiber, bran, and germ intake and the risks of all-cause and cardiovascular disease-specific mortality among women with type 2 diabetes mellitus. Circulation. 2010;121(20):2162–8.
American Diabetes Association, Bantle JP, Wylie-Rosett J et al. Nutrition recommendations and interventions for diabetes. A position statement of the American Diabetes Association. Diabetes Care. 2008;31[Suppl 1]:S61–78.
[No authors listed]. Summary of revisions: standards of medical care in diabetes—2019. Diabetes Care. 2019;42[Suppl 1]:S4–S6.
Nielsen JV, Jonsson E, Nilsson AK. Lasting improvements of hyperglycemia and bodyweight: low-carbohydrate diet in type 2 diabetes. Ups J Med Sci. 2005;110:69–74.
Nielsen JV, Joensson EA. Low-carbohydrate diet in type 2 diabetes: stable improvement of bodyweight and glycemic control during 44 months follow-up. Nutr Metab. 2008;5(1):14.
Meng Y, Bai H, Wang S, Li Z, Wang Q, Chen L. Efficacy of low carbohydrate diet for type 2 diabetes mellitus management: a systematic review and meta-analysis of randomized controlled trials. Diabetes Res Clin Pract. 2017;131:124–31.
Snorgaard O, Poulsen GM, Andersen HK, Astrup A. Systematic review and meta-analysis of dietary carbohydrate restriction in patients with type 2 diabetes. BMJ Open Diabetes Res Care. 2017;5(1):e000354.
Nilsson J, Ericsson M, Joibari MM, et al. A low-carbohydrate high-fat diet decreases lean mass and impairs cardiac function in pair-fed female C57BL/6J mice. Nutr Metab (Lond). 2016;13:79.
Lamont BJ, Waters MF, Andrikopoulos S. A low-carbohydrate high-fat diet increases weight gain and does not improve glucose tolerance, insulin secretion or beta-cell mass in NZO mice. Nutr Diabetes. 2016;6:e194.
Bolla AM, Caretto A, Laurenzi A, Scavini M, Piemonti L. Low-carb and ketogenic diets in type 1 and type 2 diabetes. Nutrients. 2019;11(5):962.
Brynes AE, Adamson J, Dornhorst A, Frost GS. The beneficial effect of a diet with low glycaemic index on 24 h glucose profiles in healthy young people as assessed by continuous glucose monitoring. Br J Nutr. 2005;93(2):179–82.
Rodbard D. Hypo- and hyperglycemia in relation to the mean, standard deviation, coefficient of variation, and nature of the glucose distribution. Diabetes Technol Ther. 2012;14(10):868–76.
Sechterberger MK, van Steen SCJ, Boerboom EMN, et al. Higher glucose variability in type 1 than in type 2 diabetes patients admitted to the intensive care unit: a retrospective cohort study. J Crit Care. 2017;38:300–3.
Hill NR, Oliver NS, Choudhary P, Levy JC, Hindmarsh P, Matthews DR. Normal reference range for mean tissue glucose and glycemic variability derived from continuous glucose monitoring for subjects without diabetes in different ethnic groups. Diabetes Technol Ther. 2011;13(9):921–8.
Kovatchev BP, Cox DJ, Kumar A, Gonder-Frederick L, Clarke WL. Algorithmic evaluation of metabolic control and risk of severe hypoglycemia in type 1 and type 2 diabetes using self-monitoring blood glucose data. Diabetes Technol Ther. 2003;5(5):817–28.
Schlichtkrull J, Munck O, Jersild M. The M value, an index of blood-sugar control in diabetics. Acta Med Scand. 1965;177:95–102.
Rawlings RA, Shi H, Yuan LH, Brehm W, Pop-Busui R, Nelson PW. Translating glucose variability metrics into the clinic via Continuous Glucose Monitoring: a Graphical User Interface for Diabetes Evaluation (CGM-GUIDE(c)). Diabetes Technol Ther. 2011;13(12):1241–8.
Service FJ. Glucose variability. Diabetes. 2013;62(5):1398–404.
Service FJ, Molnar GD, Rosevear JW, Ackerman E, Gatewood LC, Taylor WF. Mean amplitude of glycemic excursions, a measure of diabetic instability. Diabetes. 1970;19(9):644–55.
Zhou J, Li H, Ran X, Yang W, Li Q, Peng Y, et al. Establishment of normal reference ranges for glycemic variability in Chinese subjects using continuous glucose monitoring. Med Sci Monit. 2011;17(1):CR9–13.
Giugliano D, Maiorino MI, Bellastella G, Esposito K. More sugar? No, thank you! The elusive nature of low carbohydrate diets. Endocrine. 2018;61(3):383–7.
Turton JL, Raab R, Rooney KB. Low-carbohydrate diets for type 1 diabetes mellitus: a systematic review. PLoS ONE. 2018;13(3):e0194987.
Kuenen JC, Borg R, Kuik DJ, et al. Does glucose variability influence the relationship between mean plasma glucose and HbA1c levels in type 1 and type 2 diabetic patients? Diabetes Care. 2011;34(8):1843.
Kanikarla-Marie P, Jain SK. Hyperketonemia and ketosis increase the risk of complications in type 1 diabetes. Free Radic Biol Med. 2016;95:268–77.
Ellenbroek JH, van Dijck L, Tons HA, et al. Long-term ketogenic diet causes glucose intolerance and reduced beta- and alpha-cell mass but no weight loss in mice. Am J Physiol Endocrinol Metab. 2014;306(5):E552–8.
Willcox BJ, Willcox DC, Todoriki H, et al. Caloric restriction, the traditional Okinawan diet, and healthy aging: the diet of the world’s longest-lived people and its potential impact on morbidity and life span. Ann N Y Acad Sci. 2007;1114:434–55.
Tay J, Thompson CH, Brinkworth GD. Glycemic variability: assessing glycemia differently and the implications for dietary management of diabetes. Annu Rev Nutr. 2015;35(1):389–424.
Tay J, Thompson CH, Luscombe-Marsh ND, et al. Effects of an energy-restricted low-carbohydrate, high unsaturated fat/low saturated fat diet versus a high-carbohydrate, low-fat diet in type 2 diabetes: a 2-year randomized clinical trial. Diabetes Obes Metab. 2018;20(4):858–71.
Noakes M, Brinkworth GD, Luscombe-Marsh ND, et al. Comparison of low- and high-carbohydrate diets for type 2 diabetes management: a randomized trial. Am J Clin Nutr. 2015;102(4):780–90.
Ranjan A, Schmidt S, Damm-Frydenberg C, Holst JJ, Madsbad S, Nørgaard K. Short-term effects of a low carbohydrate diet on glycaemic variables and cardiovascular risk markers in patients with type 1 diabetes: a randomized open-label crossover trial. Diabetes Obes Metab. 2017;19(10):1479–84.
Gannon MC, Nuttall FQ, Saeed A, Jordan K, Hoover H. An increase in dietary protein improves the blood glucose response in persons with type 2 diabetes. Am J Clin Nutr. 2003;78(4):734–41.
Alish CJ, Garvey WT, Maki KC, et al. A diabetes-specific enteral formula improves glycemic variability in patients with type 2 diabetes. Diabetes Ther. 2010;12(6):419–25.
Fabricatore AN, Ebbeling CB, Wadden TA, Ludwig DS. Continuous glucose monitoring to assess the ecologic validity of dietary glycemic index and glycemic load. Am J Clin Nutr. 2011;94(6):1519–24.
Acknowledgements
Funding
This research was supported by grants from the Ministry of Science and Technology, ROC (MOST 105-2628-B-182A-007-MY3) and Chang Gung Memorial Hospital (CMRPG3H0401, CMRPG3H0941, CORPG5F0011). The Rapid Service Fee was funded by the authors.
Medical Writing and/or Editorial Assistance
MedCom Asia, Inc. provided medical editing for this manuscript which was funded by Chang Gung Memorial Hospital (CMRPG3H0401, CMRPG3H0941, CORPG5F0011).
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author’s Contributions
Yi-Hsuan Lin wrote the manuscript and researched data; Yu-Yao Huang researched data; Hsin-Yun Chen analyzed the data; Sheng-Hwu Hsieh collected the data; Jui-Hung Sun collected the data; Szu-Tah Chen researched data; Chia-Hung Lin reviewed/edited the manuscript. All authors were involved in the interpretation of data, critical revision and approval of the manuscript. Chia-Hung Lin is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Disclosures
Yi-Hsuan Lin, Yu-Yao Huang, Hsin-Yun Chen, Sheng-Hwu Hsieh, Jui-Hung Sun, Szu-Tah Chen and Chia-Hung Lin have nothing to disclose.
Compliance with Ethics Guidelines
Permission was obtained from the Institutional Review Board (IRB) and ethics committees of CGMH for a retrospective review of the medical records of study subjects (200800097B0). The IRB waived the requirement for obtaining informed consent. Confidentiality of the research subjects was maintained in accordance with the requirements of the IRB of CGMH (Taipei, Taiwan). The study conformed with the Helsinki Declaration of 1964, as revised in 2013, concerning human and animal rights.
Data Availability
The datasets generated during and/or analyzed during the current study are not publicly available due to patients’ privacy but are available from the corresponding author on reasonable request.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced Digital Features
To view enhanced digital features for this article go to https://doi.org/10.6084/m9.figshare.9913898.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Lin, YH., Huang, YY., Chen, HY. et al. Impact of Carbohydrate on Glucose Variability in Patients with Type 1 Diabetes Assessed Through Professional Continuous Glucose Monitoring: A Retrospective Study. Diabetes Ther 10, 2289–2304 (2019). https://doi.org/10.1007/s13300-019-00707-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-019-00707-x