Abstract
Introduction
The sodium-glucose cotransporter 2 inhibitor dapagliflozin and the glucagon-like peptide-1 (GLP-1) receptor agonist exenatide reduce bodyweight via differing and complementary mechanisms. This post hoc analysis investigated the metabolic effects and baseline associations with bodyweight loss on coadministration of dapagliflozin and exenatide once weekly (QW) among adults with obesity and without diabetes.
Methods
In the primary trial, adults with obesity and without diabetes [n = 50; 18–70 years; body mass index (BMI) 30–45 kg/m2] were randomized to double-blind oral dapagliflozin 10 mg (DAPA) once daily plus subcutaneous long-acting exenatide 2 mg QW (ExQW) or placebo over 24 weeks, followed by an open-label extension from 24–52 weeks during which all participants received active treatment. Primary results have been published previously. This analysis evaluated: (1) the effects of DAPA + ExQW on changes in substrates [free fatty acids (FFAs), glycerol, beta-OH-butyrate, and glucose], hormones (glucagon and insulin), and insulin secretion [insulinogenic index (IGI)] via an oral glucose tolerance test (OGTT) and (2) associations between bodyweight loss and baseline characteristics (e.g., BMI), single-nucleotide polymorphisms (SNPs) associated with the GLP-1 pathway, and markers of glucose regulation.
Results
Compared with placebo at 24 weeks, 2-h FFAs post-OGTT increased (mean difference, +20.4 μmol/l; P < 0.05), and fasting glucose, 2-h glucose post-OGTT, and glucose area under the concentration-time curve (AUC) decreased with DAPA + ExQW [mean differences, −0.68 mmol/l [P < 0.001], −2.20 mmol/l (P < 0.01), and −306 mmol/l min (P < 0.001), respectively]. Glucagon, glycerol, beta-OH-butyrate, and IGI did not differ by treatment group at 24 weeks. Over 52 weeks, DAPA + ExQW decreased fasting insulin, 2-h post-OGTT insulin, and insulin AUC. Among DAPA + ExQW-treated participants, for each copy of the SNP variant rs10010131 A allele (gene WFS1), bodyweight decreased by 2.4 kg (P < 0.05). Lower BMI and a lower IGI were also associated with greater bodyweight loss with DAPA + ExQW.
Conclusions
Metabolic effects with DAPA + ExQW included less FFA suppression versus placebo during the OGTT, suggesting compensatory lipid mobilization for energy production when glucose availability was reduced because of glucosuria. The expected increase in glucagon with DAPA did not occur with DAPA + ExQW coadministration. Bodyweight loss with DAPA + ExQW was associated with the SNP variant rs10010131 A allele, lower baseline adiposity (BMI), and lower baseline insulin secretion (IGI). These findings require further validation.
Funding
AstraZeneca
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The World Health Organization statistics for 2016 showed that ~ 39% (1.9 billion) of the global adult population was overweight; of these, ~ 13% (650 million) were obese [1]. Between 1980 and 2016, the prevalence of obesity worldwide more than doubled.
Overweight and obesity are associated with significant comorbidities, such as impaired glucose tolerance, type 2 diabetes mellitus (T2DM), hypertension, and dyslipidemia [2, 3]. Individuals with obesity often have multiple comorbidities [3], placing a substantial health-related burden on both the individual and the healthcare system.
Weight loss among overweight or obese individuals ameliorates impaired glucose tolerance, hypertension, and dyslipidemia [4,5,6]. However, bodyweight loss is often not maintained over the long term [7]. Subsequent bodyweight gain results in these conditions returning to baseline levels or worse [4,5,6], emphasizing the importance of bodyweight loss maintenance when managing these patients.
Bodyweight loss with single-use pharmacotherapies is generally modest and is often limited by physiologic counterregulatory mechanisms. Combination pharmacotherapies, on the other hand, may achieve greater bodyweight loss, particularly if they reduce counterregulation, thus providing additive or even synergistic effects. In addition, they may allow reduced doses of the individual agents, thereby minimizing side effect risk [8].
Clinical trials of some glucose-lowering medications have demonstrated bodyweight loss among patients with T2DM, especially those investigating selective sodium–glucose cotransporter 2 (SGLT2) inhibitors (e.g., dapagliflozin and empagliflozin) and glucagon-like peptide-1 receptor agonists (GLP-1RAs; e.g., exenatide and liraglutide). Importantly, the bodyweight loss achieved with exenatide and dapagliflozin is maintained over the long term, with trials demonstrating sustained reductions from baseline for over 6 years with exenatide and for up to 4 years with dapagliflozin [9,10,11].
The mechanisms by which GLP-1RAs (appetite suppression) and SGLT2 inhibitors (urinary caloric loss secondary to glucosuria) induce bodyweight loss are different and complementary [12,13,14]. The recent DURATION-8 study demonstrated that mean bodyweight loss with the combination of exenatide once weekly (QW) and dapagliflozin once daily (QD) among patients with T2DM was greater than that with exenatide and dapagliflozin alone [15].
The current study, which investigated the combination of exenatide QW and dapagliflozin QD on bodyweight loss among individuals with obesity and without diabetes, found significantly greater mean bodyweight loss, systolic blood pressure reduction, and glycemia reduction with active treatment versus placebo during the initial 24-week double-blind phase, which was maintained at 52 weeks in those continuing active treatment during the open-label extension phase [16, 17]. In addition, individuals initially randomized to receive placebo who switched to active treatment during the open-label extension phase achieved similar bodyweight loss at 52 weeks to those remaining on active treatment throughout the study. However, trajectories of bodyweight change varied substantially between individuals [16, 17].
This post hoc analysis aimed to investigate the metabolic effects of dapagliflozin plus exenatide QW on substrate use and selected hormone levels and to conduct exploratory hypothesis-generating analyses on associations between baseline variables and bodyweight loss over time that might account for the observed interindividual variability.
Methods
Study Design and Participants
This study was a randomized, double-blind, parallel-group, single-center study that investigated the efficacy and safety of dual therapy with dapagliflozin 10 mg QD and exenatide 2 mg QW among individuals with obesity and without diabetes over 24 weeks of double-blind treatment, followed by an optional 28-week open-label extension phase for those who had adhered to the protocol during the first 24 weeks. Here, we report a post hoc analysis focusing on the metabolic effects and baseline and pharmacogenetic markers associated with bodyweight loss.
The details of the study design and inclusion and exclusion criteria, as well as the main results, have been published previously [16, 17]. Briefly, adult participants (aged 18–70 years) with obesity and without diabetes with a body mass index (BMI) of 30–45 kg/m2 were eligible for enrollment and were randomized 1:1 to receive active treatment or placebo. Random assignment to study treatment was stratified by sex and predefined to generate a study population comprising 60% females and 40% males. During the 24-week double-blind phase, the active treatment group received dapagliflozin 10 mg QD orally each morning and exenatide 2 mg QW administered by subcutaneous injection (DAPA + ExQW group), while placebo recipients received matching placebo oral tablets and injection (placebo group). During the 28-week open-label extension phase, participants initially allocated to placebo switched to active treatment (PBO → DAPA + ExQW group), while those initially allocated to active treatment continued on active treatment.
Participants remained on their existing background medications without modification, unless instructed by a physician, and were advised to follow a balanced diet and to undertake moderate exercise.
End Points
The study protocol defined a number of exploratory objectives. For the purposes of the current analysis, these were grouped as follows: (1) to investigate the effects of DAPA + ExQW on changes in substrates (free fatty acids [FFAs], glycerol, beta-OH-butyrate, and glucose), hormones (glucagon and insulin), and insulin secretion and sensitivity (insulinogenic index [IGI] and Matsuda index adjusted for urinary glucose excretion [MI-UGE] [18, 19]) via measurements taken during an oral glucose tolerance test (OGTT); (2) to investigate associations among baseline characteristics, pharmacogenetics, and markers of glucose regulation on bodyweight loss.
OGTTs were conducted at enrollment and at 24 weeks for DAPA + ExQW and placebo groups and at 52 weeks for participants continuing on DAPA + ExQW. Blood samples for analysis were taken at time 0 (before glucose administration) and 15, 30, 60, 90, 120, and 180 min after glucose ingestion. Participants were instructed to take dapagliflozin or placebo 30 min before blood sampling and the OGTT. If exenatide QW was due to be administered on the day of testing, it was to be administered after testing was complete. Glucose and insulin were analyzed at all time points, FFAs and glycerol at 0, 30, 60, and 120 min, and glucagon and beta-OH-butyrate at 0 and 120 min. For amylin and glucagon-like peptide-1 (GLP-1), OGTT measures (sampled at 0, 15, 30, 60, and 120 min) were available at enrollment for the DAPA + ExQW group and at enrollment and 24 weeks for the placebo group (meaning that DAPA + ExQW vs. placebo comparisons at 24 weeks were unavailable for these variables). For interleukin-6 (IL-6), only fasting measures were available at enrollment for the DAPA + ExQW group and at enrollment and 24 weeks for the placebo group. Urine was collected during the 3-h OGTT period for estimation of urinary glucose excretion [16, 17].
Pharmacogenetic Analysis
The aim of the pharmacogenetic analysis was to explore the impact of genetic variations in the GLP-1 pathway on treatment response to DAPA + ExQW among participants with obesity and without diabetes.
Whole blood samples were used for DNA analysis, and seven selected single-nucleotide polymorphisms (SNPs) were identified and classified according to the following categories: (1) SNPs affecting endogenous GLP-1 levels; (2) SNPs affecting the function or activity of the GLP-1 receptor; (3) SNPs affecting GLP-1–mediated downstream pathways/biologic effects (e.g., insulin secretion; central nervous system-mediated effects such as food intake, bodyweight loss, or hypothalamic–pituitary–adrenal axis regulation; and IL-6/IL-6 receptor levels or function). For further information on the SNPs selected for inclusion, see Electronic Supplementary Material (ESM) Table S1. Selected SNPs were then entered into analyses to determine their influence on treatment-related changes in bodyweight.
Statistical Analysis
In this exploratory hypothesis-generating study, all statistical analyses were conducted in R version 3.3.3, and nominal significance and P values were determined at α < 0.05 unadjusted for multiple comparisons. Power calculations for the primary end point (change in bodyweight at 24 weeks) have been previously published [16].
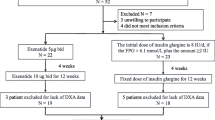
Two analysis sets were defined: (1) the full analysis set (FAS), defined as participants who received ≥ 1 dose of study medication during the entire study period and who had a baseline measurement and ≥ 1 repeated measurement for ≥ 1 efficacy measure (participants with major noncompliance to the protocol were excluded from the FAS [16]); (2) the pooled population of participants exposed to DAPA + ExQW for 24–28 weeks, defined as those with available data at week 24 for the group originally assigned to DAPA + ExQW plus those with available data at week 52 who had switched from placebo to DAPA + ExQW at 24 weeks (ESM Fig. S1).
For OGTT variables, the fasting measurements (time 0), measurements at 120 min, and area under the concentration-time curve (AUC) were analyzed. AUC was calculated using natural splines and integration under the curve. Adjusted least-squares mean (LSM) changes from baseline at 24 weeks, as well as DAPA + ExQW versus placebo LSM differences for changes from baseline at 24 weeks and their associated 95% confidence intervals (CIs), were derived from a mixed-effects model for repeated measures (MMRM), with week, sex, treatment, and week-by-treatment interaction in the 0–24-week FAS as fixed effects. Adjusted LSM changes from baseline at 52 weeks and from 24 to 52 weeks for the DAPA + ExQW group only were derived from an MMRM, with week and sex in the 0–52-week DAPA + ExQW FAS as fixed effects.
Analyses of baseline associations with bodyweight loss proceeded in the following manner. SNP associations with kilogram change in bodyweight were initially evaluated in dominant, recessive, and additive models using the R package SNPassoc [20] among the pooled population of participants exposed to DAPA + ExQW. For other baseline variables (ESM Table S2) potentially associated with kilogram bodyweight loss at 24 weeks in the FAS (including SNP associations identified at the initial step), two contrasting data reduction techniques were employed with the aim of assessing consistency in variable selection: iterative Bayesian model averaging (iBMA) [21] and least absolute shrinkage and selection operator (LASSO) regression [22]. For a complete description of these methods and how missing data were handled, see the ESM Statistical Methods.
Variables selected by iBMA and LASSO were then evaluated together for their respective contributions to kilogram bodyweight loss at 24 weeks in a multivariate generalized linear model (GLM) and individually evaluated in longitudinal MMRM analyses to assess their influence on trajectories of mean bodyweight loss over time. For continuously distributed baseline explanatory covariates, MMRM-derived adjusted LSM changes and differences in bodyweight were estimated at the 25th and 75th percentiles of that covariate; for categorical factors or additive SNP associations, LSM changes and differences in bodyweight were estimated at each factor level or at 0, 1, and 2 alleles, respectively. Treatment-by-covariate-by-week-24 interaction terms assessed whether covariates influenced bodyweight loss at week 24 independently of treatment effect or whether treatment effect was influenced by the covariate.
This article presents findings from pre-planned secondary analyses of a clinical trial on human participants. The primary analyses of this trial, which complied with ethics guidelines, have been published previously [16, 17].
Results
Participants
Characteristics of study participants have been described previously [17]. Briefly, of 50 randomized participants, 23 (92%) and 20 (80%) completed 24 weeks of double-blind treatment with DAPA + ExQW and placebo, respectively. Of these, 21 participants (84%) continued DAPA + ExQW, and 17 (68%) switched from placebo to DAPA + ExQW (ESM Fig. S1).
Effect of DAPA + ExQW on Substrates
Mean ± standard deviation urinary glucose excretion, measured during the 3-h OGTT, was 50.5 mmol/3 h ± 31.4 (9.1 g/3 h ± 5.7) and 44.6 mmol/3 h ± 26.7 (8.0 g/3 h ± 4.8) after 24 and 52 weeks of DAPA + ExQW therapy, respectively. No measurable glucosuria occurred in placebo-treated participants.
FFAs and Glycerol
At 24 weeks, FFA suppression at 120 min of the OGTT (2-h FFAs) was less in the DAPA + ExQW group than in the placebo group (LSM difference, 20.4 μmol/l; 95% CI: 2.8, 38.1; P < 0.05) (Table 1). Over 52 weeks, FFA AUC increased progressively in the DAPA + ExQW group (Fig. 1a; Table 1). No treatment differences were noted for glycerol, but fasting glycerol decreased at 52 weeks in the DAPA + ExQW group (Fig. 1b; Table 1).
Mean plasma concentration–time curves for a free fatty acids, b glycerol, c glucose, and d insulin during an OGTT conducted at baseline (w0), week 24 (w24), and week 52 (w52). DAPA dapagliflozin, ExQW exenatide once weekly, OGTT oral glucose tolerance test, PBO placebo, w week. Error bars represent mean ± standard error
Beta-OH-Butyrate
The fasting beta-OH-butyrate level remained unchanged over time and did not differ by treatment group (Table 1). Change in beta-OH-butyrate at 120 min of the OGTT did not differ between treatment groups at 24 weeks (LSM difference, 0.022 mmol/l; 95% CI: −0.026, 0.071) but declined from 24–52 weeks in the DAPA + ExQW group (Table 1).
Glucose
Fasting glucose, glucose at 120 min of the OGTT (postprandial glucose), and glucose AUC significantly decreased at 24 weeks with DAPA + ExQW compared with placebo (LSM differences, −0.68 mmol/l [P < 0.001], −2.20 mmol/l [P < 0.01], and −306 mmol/l min [P < 0.001], respectively) (Table 1; Fig. 1c). These reductions were maintained at 52 weeks (Table 1).
Effect of DAPA + ExQW on Glucagon and Insulin Secretion and Sensitivity
Glucagon
At 24 weeks, neither changes in fasting glucagon nor changes in glucagon at 120 min of the OGTT significantly differed between treatment groups (Table 1).
Insulin
Fasting insulin, insulin at 120 min of the OGTT, and insulin AUC decreased at 52 weeks versus baseline in the DAPA + ExQW group (Table 1; Fig. 1d), but there were no significant between-treatment differences in changes from baseline at 24 weeks.
Insulin Secretion and Sensitivity
There were no significant changes in the insulin secretion capacity (IGI) from baseline or between treatments; however, IGI was the only variable with a significant DAPA + ExQW versus placebo difference at baseline (Table 1). The MI-UGE increased in the DAPA + ExQW group at 24 weeks, but there were no significant between-treatment differences in change from baseline at 24 weeks, and MI-UGE did not change over 52 weeks (Table 1).
Associations Between Baseline Markers and Bodyweight Loss
Pharmacogenetic Analysis
Summary statistics for the seven SNPs genotyped are shown in ESM Table S3. In the initial evaluation of pharmacogenetic association models, only the SNP rs10010131 (gene WFS1) was associated with kilogram bodyweight loss in dominant and additive models (Table 2). The additive model was associated with the lowest P value; for each copy of the A allele, bodyweight decreased by 2.4 kg (95% CI: 0.3, 4.5; P = 0.0337).
Baseline Associations with Bodyweight Loss at 24 Weeks (Multivariate GLM Analysis)
For detailed results of the iBMA and LASSO variable selection process, including rs10010131, see the ESM Statistical Results. Among variables selected by iBMA and LASSO, GLM analysis identified treatment with DAPA + ExQW, the additive effect of the SNP rs10010131 A allele, lower baseline BMI, a lower baseline IGI, and higher baseline amylin AUC during the OGTT as significantly associated with kilogram bodyweight loss at 24 weeks (Table 3).
Baseline Associations with Bodyweight Loss Trajectories Over Time (MMRM Analyses)
For reference purposes, the primary analysis by treatment group demonstrated bodyweight loss at 24 weeks of 4.5 kg with DAPA + ExQW and 0.4 kg with placebo, with a DAPA + ExQW versus placebo bodyweight loss difference of 4.1 kg (95% CI: 1.8, 6.4; P < 0.001) [16]. Bodyweight loss with DAPA + ExQW was maintained at 52 weeks; following switch from placebo, similar bodyweight loss was achieved in the PBO → DAPA + ExQW group at 52 weeks (Fig. 2a).
Adjusted mean kilogram changes in bodyweight over time for a the primary analysis and by significant baseline associations: b single-nucleotide polymorphism rs10010131 (additive model; 0 vs. 1 copy vs. 2 copies of the A allele); c estimated at BMI 25th percentile (33.4 kg/m2) versus 75th percentile (37.5 kg/m2); d estimated at IGI 25th percentile (17.9) versus 75th percentile (37.9); e estimated at amylin AUC 25th percentile (1380 pM•min) versus 75th percentile (3620 pM min). Data are adjusted least-squares mean kilogram changes in bodyweight and 95% CIs derived from a mixed-effects model for repeated measures, with baseline weight, sex, treatment, week, baseline covariate, treatment-by-week interaction, treatment-by-baseline covariate interaction, and treatment-by-baseline covariate-by-week interaction as fixed effects. Least-squares mean differences and P values are shown in the horizontal annotations. 25P 25th percentile, 75P 75th percentile, AUC area under the concentration-time curve, BMI body mass index, CI confidence interval, D + E dapagliflozin + exenatide QW, DAPA dapagliflozin, ExQW exenatide once weekly, IGI insulinogenic index, PBO placebo
The additive effect of the SNP rs10010131 significantly altered the trajectory of bodyweight change in the DAPA + ExQW group over 24 weeks (LSM difference for 0 vs. 2 A alleles, −5.3%; 95% CI: −9.3, −1.3; P = 0.0106), which was maintained over 52 weeks (Fig. 2b).
Although placebo-treated individuals with two A alleles lost bodyweight at 24 weeks (LSM decrease from baseline, 3.0 kg; 95% CI: −0.1, 6.2; P = 0.0603), this decrease was not significant, and the degree of bodyweight loss with placebo was less than that with DAPA + ExQW at each allele level (Fig. 2b). Thus, among participants with no A alleles (i.e., two G alleles), bodyweight increased by 1.4 kg in the placebo group and decreased by 2.3 kg in the DAPA + ExQW group over 24 weeks, which represented a significant LSM decrease versus placebo of 3.7 kg (95% CI: 0.4, 7.1; P = 0.0295). Within the DAPA + ExQW group, participants with no A alleles lost bodyweight for up to 12 weeks but none thereafter (LSM increase for 24 vs. 12 weeks, 0.7 kg; 95% CI: −0.7, 2.2; P = 0.3115), whereas participants with two A alleles continued to lose bodyweight (LSM decrease for 24 vs. 12 weeks, 2.0 kg; 95% CI: 0.4, 3.6; P = 0.0148). However, the treatment-by-rs10010131-by-week-24 interaction term in the model was not significant (P = 0.9909).
In the PBO → DAPA + ExQW group, change in bodyweight by number of A alleles over 24–52 weeks showed a similar pattern to that with DAPA + ExQW over 0–24 weeks (Fig. 2b). Overall, baseline bodyweight was evenly distributed among rs10010131 genotypes, but baseline bodyweight was higher among participants with the G/G genotype in the DAPA + ExQW group (ESM Fig. S2).
Other baseline variables significantly altering the trajectory of bodyweight loss in response to DAPA + ExQW were BMI and the IGI. DAPA + ExQW-treated participants with lower BMI (estimated at the 25th percentile: 33.4 kg/m2) had greater bodyweight loss than those with higher BMI (estimated at the 75th percentile: 37.5 kg/m2) for up to 52 weeks (Fig. 2c), with a significant treatment-by-BMI-by-week-24 interaction term (P = 0.0266) and significant LSM differences at 24 weeks (−3.2 kg; 95% CI: −5.4, −0.9; P = 0.0063) and 52 weeks (−4.9 kg; 95% CI: −8.8, −1.0; P = 0.0148). Baseline BMI was strongly correlated with baseline total adipose tissue (r = 0.72; P < 0.001) and total lean tissue (r = 0.52; P < 0.001), both measured using magnetic resonance imaging [16]. Similarly, DAPA + ExQW-treated participants with a lower IGI (estimated at the 25th percentile: 17.9) had greater bodyweight loss than those with a higher IGI (estimated at the 75th percentile: 37.9) for up to 52 weeks (Fig. 2d), with a significant treatment-by-IGI-by-week-24 interaction term (P = 0.0158) and significant LSM differences at 24 weeks (−1.5 kg; 95% CI: −2.4, −0.6; P = 0.0019) and 52 weeks (−2.1 kg; 95% CI: −3.8, −0.4; P = 0.0158). Although the baseline amylin AUC was selected as significantly associated with bodyweight loss at 24 weeks in the multivariate GLM analysis, bodyweight loss trajectories with DAPA + ExQW did not meaningfully differ between lower and higher baseline amylin AUC values (Fig. 2e).
Discussion
For individuals with overweight and obesity, achieving and maintaining bodyweight loss present many challenges, including physiologic counterregulatory mechanisms of restoring bodyweight [23], hunger-induced food cravings [24], and ongoing sedentary lifestyles [1]. When administered in combination, the complementary mechanisms of action of an SGLT2 inhibitor (increasing urinary caloric loss) and a GLP-1RA (suppressing appetite) may represent a novel treatment approach for obesity.
Counterregulatory mechanisms in response to SGLT2 inhibitor-induced urinary caloric loss have been described, including compensatory increases in appetite/caloric intake [25, 26]. Therefore, the mechanisms of action of GLP-1RAs, including centrally mediated appetite suppression and possibly also delayed gastric emptying [13], may attenuate dapagliflozin-induced counterregulatory mechanisms resulting in improved bodyweight loss.
Meta-analyses of studies among patients with T2DM evaluating exenatide QW and dapagliflozin administered separately estimate bodyweight loss versus placebo at 24 weeks to range from 0.8 [27] to 1.6 kg [28, 29] and 1.6 [30] to 2.2 kg [27, 31], respectively. Assuming an additive effect with combined administration, bodyweight loss versus placebo at 24 weeks with DAPA + ExQW would be expected to be in the approximate range of 2.4–3.8 kg. Although it is not possible to make direct comparisons with other studies or to confirm additive bodyweight loss because of the lack of single-agent comparator arms, in the current study among participants without diabetes bodyweight loss versus placebo at 24 weeks was 4.1 kg. Although the current study did not assess caloric intake or satiety, the magnitude of bodyweight loss supports the hypothesis that the compensatory increase in caloric intake following SGLT2 inhibition may be attenuated to a degree by the combination with a GLP-1RA. Further support that at least additive weight loss can be achieved by combining an SGLT2 inhibitor with agents that increase satiety comes from a recent study of combined therapy with canagliflozin and phentermine among individuals with obesity and without diabetes. In this study, which, unlike the current study, employed a structured diet and exercise program, combined canagliflozin and phentermine achieved a 6.9% reduction in bodyweight at 28 weeks versus placebo compared with reductions of 1.9 and 4.1% with single administration of canagliflozin or phentermine, respectively [32].
Among patients with T2DM [25], estimated caloric intake is increased by an average of 10–15% during chronic treatment with an SGLT2 inhibitor; this offsets about 9 kg of an expected glucosuria-induced bodyweight loss of 11 kg over 2 years [33]. From the magnitude of bodyweight loss observed in the current study, it might be inferred that food intake was reduced by the addition of exenatide QW, thus counteracting the dapagliflozin-induced increase in food intake. However, the mechanisms involved have not been elucidated, and it is not clear whether the degree of bodyweight loss with DAPA + ExQW achieved among participants with obesity and without diabetes is similar to that achieved among patients with T2DM.
In the current analysis, we first explored possible metabolic adaptations during DAPA + ExQW treatment. These included changes in substrate use (FFAs, glycerol, beta-OH-butyrate, and glucose), insulin and glucagon during OGTTs, and insulin secretion and sensitivity. Change from baseline at 24 weeks in 2-h FFAs was significantly increased versus placebo (Table 1). Although no placebo comparisons were available at 52 weeks, DAPA + ExQW significantly increased 2-h FFAs and significantly decreased fasting glycerol and 2-h beta-OH-butyrate over 52 weeks. Fasting beta-OH-butyrate remained unchanged. The changes in 2-h FFAs and fasting glycerol were similar to those observed among individuals without diabetes receiving the SGLT2 inhibitor empagliflozin [34], supporting the hypothesis that SGLT2-induced glucosuria favors a shift towards greater utilization of lipids for energy production [34, 35]. However, the lack of increase in glycerol during the OGTT could suggest that the observed increase in FFAs during the OGTT might be due to reduced FFA uptake into various tissues rather than increased lipolysis in adipose tissue. Given that other work has identified that raised serum FFAs are associated with the subsequent development of insulin resistance [36], our finding of raised postprandial FFAs is of potential concern. However, no detrimental effects on insulin sensitivity have been found in clinical studies of SGLT2 inhibitors. In the current study, change in insulin sensitivity at 24 weeks with DAPA + ExQW was not significantly different from placebo, and among patients with T2DM, dapagliflozin therapy has been reported to improve insulin sensitivity [37]. Of note, in other studies among individuals with overweight/obesity receiving exenatide alone, no changes in postprandial lipolysis [38], enhanced insulin-mediated suppression of lipolysis and serum FFA levels [39], or suppression of FFA levels during the OGTT [40] were observed, indicating that the increase in 2-h FFAs observed in the current study was driven by the dapagliflozin component. Moreover, neither fasting beta-OH-butyrate nor 2-h beta-OH-butyrate was elevated by DAPA + ExQW, in contrast to elevations observed among individuals without diabetes (mild elevations) and in those with diabetes (greater elevations) treated with empagliflozin alone [34]. Indeed, 2-h beta-OH-butyrate was significantly reduced with DAPA + ExQW. While the finding of reduced 2-h beta-OH-butyrate with DAPA + ExQW may be reassuring with respect to infrequent reports of euglycemic ketoacidosis with SGLT2 inhibitor use [41], the recent hypothesis that increased production of ketone bodies with SGLT2 inhibitor treatment may underpin the cardioprotective effect observed in the EMPA-REG OUTCOME trial should also be considered [42, 43]. The lack of a beta-OH-butyrate increase upon coadministration of dapagliflozin with exenatide QW may be explained by glucagon levels remaining unchanged in the current study. This was likely due to the exenatide component inhibiting the expected increase in glucagon when dapagliflozin [44] is administered alone.
The attenuation of the expected increase in glucagon secretion and associated endogenous glucose production with dapagliflozin [44] by coadministration with exenatide QW, together with the significant reductions in glucose (fasting, 120-min OGTT, and AUC) and insulin (fasting, 120-min OGTT, and AUC) levels over 52 weeks (Table 1), suggest that DAPA + ExQW may have a role in diabetes prevention among individuals with obesity. Indeed, we have previously reported a reduction in prediabetes prevalence at 24 and 52 weeks in this population [16, 17]. However, in these participants without T2DM, we did not observe clear changes in insulin secretion (as measured by the IGI) or insulin sensitivity (as measured by the MI-UGE).
Our analysis exploring baseline associations with bodyweight loss at 24 weeks over 52 weeks demonstrated significant findings regarding SNP rs10010131, BMI, and the IGI. However, we did not find significant associations with markers of GLP-1–related pathways (amylin, GLP-1, and IL-6).
The rs10010131 SNP resides in an intron of the wolframin gene (WFS1). This gene encodes a ubiquitously expressed endoplasmic reticulum membrane glycoprotein that participates in the regulation of cellular calcium homeostasis [45]. WFS1 is mutated in Wolfram syndrome, a rare autosomal recessive disorder characterized by diabetes mellitus, optic atrophy, diabetes insipidus, and deafness [46]. In both humans and mice, wolframin deficiency results in loss of pancreatic β cells, possibly as a consequence of an enhanced endoplasmic reticulum stress response leading to increased β-cell apoptosis [47,48,49]. In the context of diabetes risk in humans, the G allele of the rs10010131 SNP variant of WFS1 is associated with impaired GLP-1–induced insulin secretion and increased diabetes risk [50,51,52], and the A allele of rs10010131 is protective against developing T2DM [53]. In the current study, we found that, for every copy of the A allele in an additive association model, an additional 2.4 kg of bodyweight loss occurred. While some degree of bodyweight loss occurred during the first 24 weeks for placebo-treated participants with two copies of the A allele, which was similar to that achieved among participants with two G alleles receiving DAPA + ExQW, this change was not significantly different from baseline. Thus, it is likely that bodyweight loss with DAPA + ExQW was accentuated among participants with two A alleles and attenuated in those with two G alleles. However, the treatment-by-rs10010131-by-week-24 interaction term was not significant in this analysis, which would rather favor independent effects of the rs10010131 A allele and DAPA + ExQW treatment on bodyweight loss. In a separate investigation, another SNP associated with diabetes risk, rs7903146 in TCF7L2, attenuated bodyweight loss in response to lifestyle intervention among individuals at risk for T2DM [54]. We evaluated rs7903146 in the current study but found no association with bodyweight loss. To the best of our knowledge, this is the first study to describe an association with rs10010131 and bodyweight loss. Further research is required in larger independent samples to confirm/validate whether the rs10010131 variant influences bodyweight loss independently or in response to SGLT2 inhibitors, GLP-1RAs, or combined therapy.
Baseline BMI does not appear to influence the degree of bodyweight loss among patients with T2DM receiving dapagliflozin or exenatide administered alone. However, Ferrannini and colleagues [25] identified that, among patients with T2DM treated with empagliflozin, the estimated compensatory increased caloric intake was inversely correlated with baseline BMI (partial r = −0.34; P < 0.01). Thus, if these findings are equivalent in SGLT2 inhibitor-treated individuals without diabetes, the greater bodyweight loss among individuals with lower baseline BMI receiving DAPA + ExQW might be explained by the exenatide component attenuating the greater degree of dapagliflozin-associated compensatory increase in caloric intake among individuals with lower baseline BMI. This hypothesis is supported by the significant treatment-by-BMI-by-week-24 interaction term in this analysis, suggesting that the treatment effect was influenced by baseline BMI. However, more DAPA + ExQW-treated participants with higher baseline bodyweight had the rs10010131 G/G genotype than placebo-treated participants with higher baseline bodyweight, which may have influenced these findings. Alternatively, the association with lower baseline BMI and greater bodyweight loss observed in the current study may be independent of DAPA + ExQW treatment, as lower baseline BMI is associated with greater bodyweight loss following dietary [55] and surgical [56] interventions.
Individuals with a higher IGI (i.e., more insulin secretion per increase in glucose) showed a poorer bodyweight response with DAPA + ExQW, and the treatment-by-IGI-by-week-24 interaction term was significant in this analysis, suggesting that the treatment effect was influenced by baseline IGI. Although baseline IGI was significantly lower in the DAPA + ExQW versus placebo group, the MMRM analysis of change in bodyweight containing terms for both treatment and baseline IGI adjusted for these differences. This finding is consistent with reports that post-challenge hyperinsulinemia [57], as well as fasting hyperinsulinemia [58], predicts later bodyweight gain. However, other researchers have not replicated this observation [59]. Hyperinsulinemia may not only be a consequence of obesity-associated insulin resistance but may also promote obesity itself.
This study has limitations. First, measures of satiety, appetite, and caloric intake before and during DAPA + ExQW treatment were not undertaken. Second, the small sample size and the presence of missing data for some variables limited the power to detect associations. However, the analytic strategy employing modern variable selection techniques minimized the impact of these limitations. Third, as in any exploratory study without independent validation, statistical overfitting was a potential issue. Consequently, these findings cannot be generalized to future populations undergoing treatment with DAPA + ExQW and should be regarded as hypothesis-generating in nature. Fourth, placebo comparisons were only available from weeks 0–24. While the switch to active treatment at 24 weeks among those who initially received placebo allowed internal validation of the effect of time-invariant covariates, such as the SNP variant rs10010131, which showed a similar pattern of change post-switch as for initial active treatment, this was not the case for time-varying covariates. Because time-varying covariates (e.g., BMI) change over time, the new covariate “baseline” value at 24 weeks among those who switched from placebo to active treatment will vary nonrandomly, making assessments of changes in bodyweight beyond 24 weeks by the baseline value at week 0 unreliable in this group. Fifth, while patients were observed over 52 weeks, obesity is usually a life-long disorder, and longer-term studies are needed to determine the durability of response with DAPA + ExQW as potential attenuation of dapagliflozin-induced compensatory mechanisms by exenatide QW may change over time. Finally, the study design lacked monotherapy arms, so firm conclusions cannot be drawn about potential additive effects of dapagliflozin and exenatide on bodyweight loss.
Conclusions
Metabolic effects following dual therapy with DAPA + ExQW included less suppression of circulating FFAs compared with placebo during the OGTT, suggesting compensatory lipid mobilization for energy production when glucose availability was reduced because of glucosuria. The increase in glucagon secretion previously reported with dapagliflozin alone did not occur with the dual therapy, suggesting that the GLP1 receptor agonist component had an opposite effect to the SGLT2 inhibitor, thus preventing a glucagon rise. Baseline variables associated with greater bodyweight loss during DAPA + ExQW therapy included the A allele of the SNP variant rs10010131, lower baseline adiposity (BMI), and lower glucose-stimulated insulin secretion at baseline. Further research in larger populations with varying baseline demographics and genetic characteristics is required to validate these associations and their potential predictive impact for identification of treatment responders and nonresponders. Such work may also support future efforts in precision medicine within the obesity field.
References
World Health Organization. Obesity and overweight fact sheet. 2017. http://www.who.int/mediacentre/factsheets/fs311/en/. Accessed 5 Feb 2018.
Martin-Rodriguez E, Guillen-Grima F, Marti A, Brugos-Larumbe A. Comorbidity associated with obesity in a large population: the APNA study. Obes Res Clin Pract. 2015;9:435–47.
Sirtori A, Brunani A, Capodaglio P, et al. Patients with obesity-related comorbidities have higher disability compared with those without obesity-related comorbidities: results from a cross-sectional study. Int J Rehabil Res. 2016;39:63–9.
Beavers DP, Beavers KM, Lyles MF, Nicklas BJ. Cardiometabolic risk after weight loss and subsequent weight regain in overweight and obese postmenopausal women. J Gerontol Ser A, Biol Sci Med Sci. 2013;68:691–8.
Beavers KM, Case LD, Blackwell CS, Katula JA, Goff DC Jr, Vitolins MZ. Effects of weight regain following intentional weight loss on glucoregulatory function in overweight and obese adults with pre-diabetes. Obesity Res Clin Pract. 2015;9:266–73.
Kroeger CM, Hoddy KK, Varady KA. Impact of weight regain on metabolic disease risk: a review of human trials. J Obes. 2014;2014:614519.
Greenway FL. Physiological adaptations to weight loss and factors favouring weight regain. Int J Obes. 2015;39:1188–96.
Wilding JP. Combination therapy for obesity. J Psychopharmacol. 2017;31:1503–8.
Henry RR, Klein EJ, Han J, Iqbal N. Efficacy and tolerability of exenatide once weekly over 6 years in patients with type 2 diabetes: an uncontrolled open-label extension of the DURATION-1 study. Diabetes Technol Therapeutics. 2016;18:677–86.
Bolinder J, Ljunggren O, Johansson L, et al. Dapagliflozin maintains glycaemic control while reducing weight and body fat mass over 2 years in patients with type 2 diabetes mellitus inadequately controlled on metformin. Diabetes Obes Metab. 2014;16:159–69.
Del Prato S, Nauck M, Duran-Garcia S, et al. Long-term glycaemic response and tolerability of dapagliflozin versus a sulphonylurea as add-on therapy to metformin in patients with type 2 diabetes: 4-year data. Diabetes Obes Metab. 2015;17:581–90.
Scheen AJ. Pharmacodynamics, efficacy and safety of sodium-glucose co-transporter type 2 (SGLT2) inhibitors for the treatment of type 2 diabetes mellitus. Drugs. 2015;75:33–59.
van Bloemendaal L, Ten Kulve JS, la Fleur SE, Ijzerman RG, Diamant M. Effects of glucagon-like peptide 1 on appetite and body weight: focus on the CNS. J Endocrinol. 2014;221:T1–16.
Busch RS, Kane MP. Combination SGLT2 inhibitor and GLP-1 receptor agonist therapy: a complementary approach to the treatment of type 2 diabetes. Postgraduate medicine. 2017:1–12.
Frias JP, Guja C, Hardy E, et al. Exenatide once weekly plus dapagliflozin once daily versus exenatide or dapagliflozin alone in patients with type 2 diabetes inadequately controlled with metformin monotherapy (DURATION-8): a 28 week, multicentre, double-blind, phase 3, randomised controlled trial. Lancet Diabetes Endocrinol. 2016;4:1004–16.
Lundkvist P, Sjostrom CD, Amini S, Pereira MJ, Johnsson E, Eriksson JW. Dapagliflozin once-daily and exenatide once-weekly dual therapy: a 24-week randomized, placebo-controlled, phase II study examining effects on body weight and prediabetes in obese adults without diabetes. Diabetes Obes Metab. 2017;19:49–60.
Lundkvist P, Pereira MJ, Katsogiannos P, Sjostrom CD, Johnsson E, Eriksson JW. Dapagliflozin once daily plus exenatide once weekly in obese adults without diabetes: Sustained reductions in body weight, glycaemia and blood pressure over 1 year. Diabetes Obes Metab. 2017;19:1276–88.
Matsuda M. Calculation of matsuda index. http://mmatsuda.diabetes-smc.jp/MIndex.html. Accessed 14 Feb 2018.
DeFronzo RA, Hompesch M, Kasichayanula S, et al. Characterization of renal glucose reabsorption in response to dapagliflozin in healthy subjects and subjects with type 2 diabetes. Diabetes Care. 2013;36:3169–76.
Gonzalez JR, Armengol L, Sole X, et al. SNPassoc: an R package to perform whole genome association studies. Bioinformatics. 2007;23:644–5.
Yeung KY, Bumgarner RE, Raftery AE. Bayesian model averaging: development of an improved multi-class, gene selection and classification tool for microarray data. Bioinformatics. 2005;21:2394–402.
Friedman J, Hastie T, Tibshirani R. Regularization paths for generalized linear models via coordinate descent. J Stat Softw. 2010;33:1–22.
Dhurandhar EJ, Kaiser KA, Dawson JA, Alcorn AS, Keating KD, Allison DB. Predicting adult weight change in the real world: a systematic review and meta-analysis accounting for compensatory changes in energy intake or expenditure. Int J Obes. 2015;39:1181–7.
Boswell RG, Kober H. Food cue reactivity and craving predict eating and weight gain: a meta-analytic review. Obes Rev. 2016;17:159–77.
Ferrannini G, Hach T, Crowe S, Sanghvi A, Hall KD, Ferrannini E. Energy balance after sodium-glucose cotransporter 2 inhibition. Diabetes Care. 2015;38:1730–5.
Polidori D, Sanghvi A, Seeley RJ, Hall KD. How strongly does appetite counter weight loss? Quantification of the feedback control of human energy intake. Obesity. 2016;24:2289–95.
Zaccardi F, Htike ZZ, Webb DR, Khunti K, Davies MJ. Benefits and harms of once-weekly glucagon-like peptide-1 receptor agonist treatments: a systematic review and network meta-analysis. Ann Intern Med. 2016;164:102–13.
Potts JE, Gray LJ, Brady EM, Khunti K, Davies MJ, Bodicoat DH. The effect of glucagon-like peptide 1 receptor agonists on weight loss in type 2 diabetes: a systematic review and mixed treatment comparison meta-analysis. PLoS ONE. 2015;10:e0126769.
Sun F, Chai S, Li L, et al. Effects of glucagon-like peptide-1 receptor agonists on weight loss in patients with type 2 diabetes: a systematic review and network meta-analysis. J Diabetes Res. 2015;2015:157201.
Shyangdan DS, Uthman OA, Waugh N. SGLT-2 receptor inhibitors for treating patients with type 2 diabetes mellitus: a systematic review and network meta-analysis. BMJ Open. 2016;6:e009417.
Mearns ES, Sobieraj DM, White CM, et al. Comparative efficacy and safety of antidiabetic drug regimens added to metformin monotherapy in patients with type 2 diabetes: a network meta-analysis. PLoS ONE. 2015;10:e0125879.
Hollander P, Bays HE, Rosenstock J, et al. Coadministration of canagliflozin and phentermine for weight management in overweight and obese individuals without diabetes: a randomized clinical trial. Diabetes Care. 2017;40:632–9.
Hall KD, Sacks G, Chandramohan D, et al. Quantification of the effect of energy imbalance on bodyweight. Lancet. 2011;378:826–37.
Ferrannini E, Baldi S, Frascerra S, et al. Shift to fatty substrate utilization in response to sodium-glucose cotransporter 2 inhibition in subjects without diabetes and patients with type 2 diabetes. Diabetes. 2016;65:1190–5.
Ferrannini E, Muscelli E, Frascerra S, et al. Metabolic response to sodium-glucose cotransporter 2 inhibition in type 2 diabetic patients. J Clin Investig. 2014;124:499–508.
Frohnert BI, Jacobs DR Jr, Steinberger J, Moran A, Steffen LM, Sinaiko AR. Relation between serum free fatty acids and adiposity, insulin resistance, and cardiovascular risk factors from adolescence to adulthood. Diabetes. 2013;62:3163–9.
Mudaliar S, Henry RR, Boden G, et al. Changes in insulin sensitivity and insulin secretion with the sodium glucose cotransporter 2 inhibitor dapagliflozin. Diabetes Technol Therapeutics. 2014;16:137–44.
Camastra S, Astiarraga B, Tura A, et al. Effect of exenatide on postprandial glucose fluxes, lipolysis, and β-cell function in non-diabetic, morbidly obese patients. Diabetes Obes Metab. 2017;19:412–20.
Armstrong MJ, Hull D, Guo K, et al. Glucagon-like peptide 1 decreases lipotoxicity in non-alcoholic steatohepatitis. J Hepatol. 2016;64:399–408.
Gastaldelli A, Gaggini M, Daniele G, et al. Exenatide improves both hepatic and adipose tissue insulin resistance: a dynamic positron emission tomography study. Hepatology. 2016;64:2028–37.
Peters AL, Buschur EO, Buse JB, Cohan P, Diner JC, Hirsch IB. Euglycemic diabetic ketoacidosis: a potential complication of treatment with sodium-glucose cotransporter 2 inhibition. Diabetes Care. 2015;38:1687–93.
Ferrannini E, Mark M, Mayoux E. CV protection in the EMPA-REG OUTCOME trial: a “thrifty substrate” hypothesis. Diabetes Care. 2016;39:1108–14.
Mudaliar S, Alloju S, Henry RR. Can a shift in fuel energetics explain the beneficial cardiorenal outcomes in the EMPA-REG OUTCOME study? A unifying hypothesis. Diabetes Care. 2016;39:1115–22.
Merovci A, Solis-Herrera C, Daniele G, et al. Dapagliflozin improves muscle insulin sensitivity but enhances endogenous glucose production. J Clin Investig. 2014;124:509–14.
Takei D, Ishihara H, Yamaguchi S, et al. WFS1 protein modulates the free Ca(2 +) concentration in the endoplasmic reticulum. FEBS Lett. 2006;580:5635–40.
Online Mendelian Inheritance in Man. Wolfram Syndrome 1 (WFS1): Diabetes Insipidus and Diabetes Mellitus with Optic Atrophy and Deafness (DIDMOAD). https://www.omim.org/entry/222300. Accessed 23 Aug 2017.
Yamada T, Ishihara H, Tamura A, et al. WFS1-deficiency increases endoplasmic reticulum stress, impairs cell cycle progression and triggers the apoptotic pathway specifically in pancreatic beta-cells. Hum Mol Genet. 2006;15:1600–9.
Riggs AC, Bernal-Mizrachi E, Ohsugi M, et al. Mice conditionally lacking the Wolfram gene in pancreatic islet beta cells exhibit diabetes as a result of enhanced endoplasmic reticulum stress and apoptosis. Diabetologia. 2005;48:2313–21.
Karasik A, O’Hara C, Srikanta S, et al. Genetically programmed selective islet beta-cell loss in diabetic subjects with Wolfram’s syndrome. Diabetes Care. 1989;12:135–8.
Sandhu MS, Weedon MN, Fawcett KA, et al. Common variants in WFS1 confer risk of type 2 diabetes. Nat Genet. 2007;39:951–3.
Schafer SA, Mussig K, Staiger H, et al. A common genetic variant in WFS1 determines impaired glucagon-like peptide-1-induced insulin secretion. Diabetologia. 2009;52:1075–82.
Simonis-Bik AM, Nijpels G, van Haeften TW, et al. Gene variants in the novel type 2 diabetes loci CDC123/CAMK1D, THADA, ADAMTS9, BCL11A, and MTNR1B affect different aspects of pancreatic beta-cell function. Diabetes. 2010;59:293–301.
Cheng S, Wu Y, Wu W, Zhang D. Association of rs734312 and rs10010131 polymorphisms in WFS1 gene with type 2 diabetes mellitus: a meta-analysis. Endocrine J. 2013;60:441–7.
Haupt A, Thamer C, Heni M, et al. Gene variants of TCF7L2 influence weight loss and body composition during lifestyle intervention in a population at risk for type 2 diabetes. Diabetes. 2010;59:747–50.
Batterham M, Tapsell LC, Charlton KE. Baseline characteristics associated with different BMI trajectories in weight loss trials: a case for better targeting of interventions. Eur J Clin Nutr. 2016;70:207–11.
Livhits M, Mercado C, Yermilov I, et al. Preoperative predictors of weight loss following bariatric surgery: systematic review. Obes Surg. 2012;22:70–89.
Sigal RJ, El-Hashimy M, Martin BC, Soeldner JS, Krolewski AS, Warram JH. Acute postchallenge hyperinsulinemia predicts weight gain: a prospective study. Diabetes. 1997;46:1025–9.
Erion KA, Corkey BE. Hyperinsulinemia: a cause of obesity? Curr Obes Rep. 2017;6:178–86.
Rebelos E, Muscelli E, Natali A, et al. Body weight, not insulin sensitivity or secretion, may predict spontaneous weight changes in nondiabetic and prediabetic subjects: the RISC study. Diabetes. 2011;60:1938–45.
Acknowledgements
The authors wish to thank the study participants.
Funding
This study was supported by AstraZeneca, who also provided support for journal processing charges. All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analysis.
Medical Writing Assistance
Sheridan Henness of inScience Communications, Springer Healthcare, provided medical writing support. This was funded by AstraZeneca.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
Per Lundkvist has received an honorarium from Merck Sharp & Dohme and a travel grant from AstraZeneca. Julian G. Martins is an employee of inScience Communications, Springer Healthcare, which was contracted by AstraZeneca to provide statistical support for this study. C. David Sjöström is an employee of and owns stock in AstraZeneca. Anna Walentinsson is an employee of and owns stock in AstraZeneca. Eva Johnsson is an employee of and owns stock in AstraZeneca. Volker Schnecke was an employee of AstraZeneca at the time this study was conducted, owns stock in AstraZeneca, and is currently an employee of Novo Nordisk A/S. Jan W. Eriksson has received research grants or honoraria from AstraZeneca, Bristol-Myers Squibb, Merck Sharp & Dohme, Novo Nordisk, and Sanofi. Maria J. Pereira, Prasad G. Kamble, and Joey Lau have nothing to disclose.
Compliance with Ethics Guidelines
This article presents findings from pre-planned secondary analyses of a clinical trial in human participants. The primary analyses of this trial, which complied with ethics guidelines, have been published previously [16, 17].
Data Availability
The data sets generated during and/or analyzed during the current study are available from the corresponding author upon request.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced Digital Features
To view enhanced digital features for this article, go to https://doi.org/10.6084/m9.figshare.6276050.
Volker Schnecke was an employee of AstraZeneca at the time the study was conducted but is currently an employee of Novo Nordisk A/S, Søborg, Denmark.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Pereira, M.J., Lundkvist, P., Kamble, P.G. et al. A Randomized Controlled Trial of Dapagliflozin Plus Once-Weekly Exenatide Versus Placebo in Individuals with Obesity and Without Diabetes: Metabolic Effects and Markers Associated with Bodyweight Loss. Diabetes Ther 9, 1511–1532 (2018). https://doi.org/10.1007/s13300-018-0449-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-018-0449-6