Abstract
Climate change is likely to have large effects on the Baltic Sea ecosystem. Simulations indicate 2–4 °C warming and 50–80 % decrease in ice cover by 2100. Precipitation may increase ~30 % in the north, causing increased land runoff of allochthonous organic matter (AOM) and organic pollutants and decreased salinity. Coupled physical–biogeochemical models indicate that, in the south, bottom-water anoxia may spread, reducing cod recruitment and increasing sediment phosphorus release, thus promoting cyanobacterial blooms. In the north, heterotrophic bacteria will be favored by AOM, while phytoplankton production may be reduced. Extra trophic levels in the food web may increase energy losses and consequently reduce fish production. Future management of the Baltic Sea must consider the effects of climate change on the ecosystem dynamics and functions, as well as the effects of anthropogenic nutrient and pollutant load. Monitoring should have a holistic approach, encompassing both autotrophic (phytoplankton) and heterotrophic (e.g., bacterial) processes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Baltic Sea is exposed to many stressors, e.g., eutrophication, organic pollutants, overfishing, invasive species, and acidification. Of these disturbances, eutrophication is presently considered to have the most severe effects in the Baltic proper, while organic pollutants are the largest environmental problem in the Gulf of Bothnia. Climate change may worsen these problems and it is thus a challenge to try to understand how different basins of the Baltic Sea may be influenced and how to appropriately manage this vulnerable ecosystem.
Climate change, induced by anthropogenic emissions of greenhouse gasses, is expected to have a significant impact on the Baltic Sea (BACC I author team 2008; BACC II author team 2015). A warming trend is already evident in the Baltic region and will continue through the twenty first century. Due to the large variability of the climate system, only temperature and directly related variables, such as ice conditions, are likely to show statistically significant changes in the next few decades, whereas significant effects in the water cycle can only be expected later in the century.
The impact of climate change on the Baltic Sea environment can be estimated with the help of coupled physical–biogeochemical models in conjunction with downscaling techniques that link projected global climate change to regional scales (e.g., Meier et al. 2011a). However, Baltic Sea ecosystem projections suffer from the biases of global and regional climate models, uncertainty in greenhouse gas emissions, nutrient load scenarios, and ecosystem responses, as well as natural climate variability. Hence, ensemble simulations are essential to estimate uncertainties in the projections (e.g., Meier et al. 2006, 2011a).
When assessing the effects of climate change on marine ecosystems, it is important to understand the driving mechanisms for biological and chemical processes. These drivers can influence the marine ecosystem in a variety of ways, altering food web community structure, overall productivity, and the transport of pollutants. Such changes are of great significance for the health and sustainability of the marine ecosystem. In this study, we have therefore combined information from field and experimental studies with modeling simulations, to try to understand the effects of climate change on the Baltic Sea ecosystems.
Physical changes
Model simulations indicate that the surface water layer will warm more than the deep water in all sub-basins (Meier et al. 2012b). Sea surface temperature (SST) changes are projected to be the largest in the Bothnian Bay and the Bothnian Sea during summer and in the Gulf of Finland during spring. According to Meier et al. (2012a), the mean summer SST will increase by about 2 °C in the southern and 4 °C in the northern Baltic Sea by the end of this century. The larger warming in the north is caused at least partly by the ice-albedo feedback (Meier et al. 2011b). Under a more optimistic scenario, the average SST may increase by only 1 °C (Neumann 2010).
The future reduction of the ice cover depends mainly on projected air temperature changes over the Baltic Sea in winter, whereas the other drivers, like wind speed, are less important (e.g., Meier et al. 2004). Despite substantial uncertainties, all available scenario simulations indicate a 50–80 % decrease in sea ice extent by 2100 (Meier 2006). This increase in open water conditions will influence winds, wave climate, and underwater light conditions (Eilola et al. 2013). Wave height is projected to increase in spring in large parts of the Gulf of Finland, the Bothnian Sea, and the Bothnian Bay; and mean spring irradiance may increase in previously ice-covered areas. Significantly increased well-mixed layer depths are expected in most of the Bothnian Bay and the Gulf of Finland.
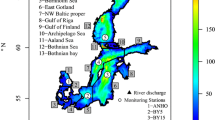
Sea surface salinity (SSS) will change less in the northern and eastern Baltic Sea (least in the Bothnian Bay) and most in the Danish straits region, especially the Belt Sea (Fig. 1) (Meier et al. 2012a), through shifting salinity fronts in the transition zone. The SSS changes are rather uniform across the seasons. In the ensemble mean, salinity in the strongly stratified Bornholm and Gotland basins decreases by 1.5–2 salinity units at all depths (Meier et al. 2012b). In the less strongly stratified Gulf of Finland and Bothnian Bay, salinity changes are larger in the deep water, reducing the vertical stability. These results are rather consistent among various Baltic Sea models. The largest, model-related uncertainty was found for projected halocline depth in the Baltic proper (Meier et al. 2012b).
Projected seasonal (DJF = December to February, MAM = March to May, JJA = June to August, SON = September to November) and annual mean ensemble average sea surface salinity changes (in g kg−1) from 1978–2007 to 2069–2098 (from Meier et al. 2012a)
Salinity changes (Fig. 1) are caused by changes in runoff, which in the depicted scenario simulations is projected to increase by 15–22 % (Meier et al. 2012a). Although all scenario simulations suggest either unchanged or decreased salinity compared to present climate (Meier et al. 2006), projections have large uncertainties due to variability among regional Baltic climate models in water balance (e.g., Kjellström and Lind 2009), wind projections (Kjellström et al. 2011; Nikulin et al. 2011), runoff projections using various hydrological models, and also bias in correction methods for air temperature and precipitation (Meier et al. 2012b; Donnelly et al. 2014).
Overall stratification changes in the Baltic proper are expected to be small because the greater freshwater supply will increase recirculation of brackish surface waters and consequently reduce saltwater influx to the Baltic Sea (Meier 2005). Changes in wind-induced mixing are more important for stratification changes in the Baltic proper than changes in runoff (Meier 2005).
Saltwater inflows from the Kattegat influence average salinity, vertical stratification, and deep water oxygen conditions in the Baltic Sea (e.g., Meier and Kauker 2003). Still Gräwe et al. (2013) found no clear tendency for salt water transport to change in the future climate, either during medium or major inflow events. Schimanke et al. (2014) estimated that atmospheric events favorable for major Baltic inflows may become slightly more common. All published scenario simulations share the weakness that they have not considered the effect of global sea-level rise on saltwater inflows. Unpublished research by H.E.M. Meier et al. indicates that if the global mean sea level rises by 1.5 m, then the effect on salinity and stratification cannot be neglected.
Pelagic food web structure from north to south
To evaluate the effects of climate change on the marine food web, it is crucial to understand the mechanisms regulating food web structure and productivity. Bacteria and phytoplankton are key organisms at the base of the marine food web, providing biomass production on which the rest of the food web relies. They take up nutrients via diffusion and are thus the first organisms to respond to changes in nutrient availability. Their interaction strongly influences the structure and efficiency of the food web and hence production at higher trophic levels.
Phytoplankton primary production increases strongly basin by basin from north to south in Baltic Sea offshore waters and is almost tenfold higher in the Baltic proper than in the Bothnian Bay (Fig. S1, Supplementary Material). The low primary production in the Bothnian Bay is caused by a combination of strong phosphorus (P) limitation (Andersson et al. 1996), a short productive season and a poor light climate for phytoplankton. The Bothnian Bay water is relatively brown, with twice the concentration of humic substances and ~70 % lower phosphorus concentration compared to the Baltic proper (Fig. S1).
The annual bacterial biomass production, on the other hand, is rather uniform from north to south, although its relative share of the total production varies among the basins. In offshore waters of the Bothnian Bay, the Bothnian Sea, and the Baltic proper, the bacterial production equals 70, 30, and 12 % of the primary production, respectively (Fig. S1). The large importance of bacteria in the north may partly be due to high concentrations of allochthonous organic matter (AOM), e.g., humic substances, which fuel bacteria with external organic matter (Sandberg et al. 2004). Another contributing factor can be low nutrient availability (P), as phytoplankton might exude more of their photosynthetic products when nutrients are scarce. These photosynthetic exudates are then channeled into bacterial production.
Drivers of coastal production
Field studies were performed during the spring period in three estuaries in the Baltic Sea, in order to find out how the river inflow influences primary and bacterial production. In the Öre estuary, Gulf of Bothnia, primary production was stimulated by increased phosphorus availability and hampered by high concentration of dissolved organic carbon (DOC) (Table S1). DOC, humic substances, and colored dissolved organic matter (CDOM) are all measures of allochthonous organic matter, which is transported from land to the Baltic Sea via rivers, reducing light penetration in coastal waters (Pettersson et al. 1997). In the nitrogen-limited Emån estuary, in the southern Baltic Sea, the spring primary production was negatively related to phosphorus and temperature (Table S1).
Bacterial production was mainly controlled by factors that are likely to be influenced by climate change. A stimulation by humic substances and DOC was observed in the coastal areas of the Gulf of Bothnia (Råne and Öre estuaries) (Table S1). Even though the bioavailable fraction of the AOM in short-term experiments is small (Stepanauskas et al. 2002; Lignell et al. 2008), the large AOM export via rivers to the coastal zone, combined with increased bioavailability by, e.g., photochemical processes over longer time scales, makes AOM a major driver of bacterial production in the northern Baltic Sea (Sandberg et al. 2004). In the study area in the southern Baltic Sea, however, regression analysis indicated temperature as the major factor influencing bacterial production (Table S1). This agrees with previous studies in the Baltic Sea, indicating that bacterial production is temperature limited below +6 to +8 °C, but substrate limited (by inorganic nutrients or organic C) at higher temperatures (e.g., Autio 1998).
Effects of increased dissolved organic matter inflow on coastal production
An integrated understanding of the effects of the projected increase in runoff in the northern Baltic Sea is required. The fertilizing effect of nitrogen and phosphorus discharged by rivers has been the main issue in recent research (Smith 2006; Finkel et al. 2010). Nutrient-rich freshwater discharge can lead to eutrophication, with increased phytoplankton growth, oxygen consumption, hypoxia, and reduced recreational value of the coastal zone. However, freshwater discharge also has other potential effects on the hydrography, chemistry, and biology of the marine environment. Dissolved organic matter (DOM) is a major chemical constituent of river water, with potential effects in coastal areas. Colored humic substances degrade the light climate, heat the near-surface water (Cole et al. 1992; Howarth et al. 2000; Ask et al. 2009), and can also act as a carbon source for bacterioplankton production (Wikner et al. 1999).
Using a 13-year ecological time-series from the Bothnian Bay, the Bothnian Sea, and the Öre estuary, Wikner and Andersson (2012) studied the effect on the trophic balance of a 4-year period (1998–2001) with elevated river discharge. The ratio between phytoplankton and bacterial production was used as an index of trophic balance. Correlation analysis indicated that increased freshwater discharge of colored DOM reduced the primary production, while bacterial production remained stable. Previous studies have shown that part of the riverine DOM can be assimilated by bacterioplankton (Zweifel et al. 1995; Wikner et al. 1999). This suggested a dual effect of riverine DOM on the trophic balance, by reducing light (i.e., energy) supply to photosynthetic organisms, and simultaneously stimulating heterotrophic organisms by providing an alternative carbon and energy source. It is likely that the food web efficiency decreased during this period, since bacteria-based food webs generally have more trophic levels than those based on phytoplankton (Berglund et al. 2007). However, in a climate change experiment combining increased input of humic substances with higher temperature, the food web efficiency was not reduced when the planktivorous fish was the three-spine stickleback (Gasterosteus aculeatus), which is able to adapt to high temperature and can exploit the system efficiently (Lefébure et al. 2013).
Benthic zones are often important contributors to overall production in shallow coastal waters. During the 1998–2001 period of elevated river flow and lower pelagic primary production, the native benthic amphipod Monoporeia affinis declined drastically in the Gulf of Bothnia (Eriksson-Wiklund and Andersson 2014), probably due to food shortage, since settling phytoplankton is its main food source. In the virtual absence of this amphipod, the non-native polychaete Marenzelleria spp. invaded the area. Such changes in species composition alter food web structure and resource use and can modify the transport and release of pollutants.
Effects of increased DOC inflow on organic pollutant sorption and bioaccumulation
The seasonal export of hydrophobic organic contaminants from soils to river water peaks during the spring flood, and is connected to processes determining DOC release at the soil–water interface (Bergknut et al. 2010). Climate-induced increases in precipitation and DOC release are therefore likely to cause increased inflow of contaminants to coastal areas.
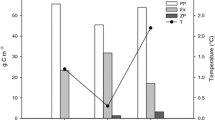
Higher concentrations of DOC in the seawater may also affect the fate of the pollutants, e.g., by changing the effects on their solubility, volatilization, long-range transport, transformations, and bioavailability (e.g., Kukkonen et al. 1990; Poerschmann and Kopinke 2001). In a spiking experiment, we demonstrated that the sorption of structurally diverse pollutants to dissolved organic matter in seawater varies between areas of the Baltic Sea (Fig. 2) (Ripszam et al. 2015). The distribution constant between water and DOC (log KDOC) for hexachlorobenzene and phenanthrene, representing halogenated aromatic and polycyclic aromatic compounds, respectively, decreased from north to south in the Baltic Sea, while tributyl phosphate, representing compounds with polar functional groups and relatively high water solubility, showed no geographical variation. The salinity gradient in the study, from 2.8 to 6.6, probably had only a minor effect on the partitioning of the tested organic pollutants (Engebretson and von Wandruszka 1994; Kuivikko et al. 2010). Considering the small variation of DOC concentration in the offshore Baltic Sea (4.2–5.2 mg C/l), we propose that differences in quality, i.e., the terrestrial component of the DOC, caused the differences in log KDOC values.
Log KDOC values for hexachlorobenzene (black), phenanthrene (squared), and tributyl phosphate (striped) at different locations in the Baltic Sea. Water samples were collected along a north–south gradient in August 2013, filtered to retrieve the dissolved fraction, and spiked with different organic pollutants. Error bars represent the standard deviation of data from four sampling points in each basin
Scenarios for ecosystem changes
The combined influence of changing climate, eutrophication, acidification, and overfishing on the marine ecosystem has been studied with coupled physical–biogeochemical–carbonate models (e.g., Neumann 2010; Meier et al. 2011a, b; Neumann et al. 2012; Omstedt et al. 2012) and food web models (e.g., Niiranen et al. 2013). However, models coupling lower and higher trophic levels do not exist, and our knowledge of the effects of changing climate and other anthropogenic drivers on the marine ecosystem is still very limited. Eilola et al. (2011) compared three physical–biogeochemical Baltic Sea models and identified four major sources of uncertainty: (1) uncertain initial conditions, (2) unknown bioavailability of nutrients in land runoff, (3) unreliable parameterization of sediment fluxes and turnover of nutrients in the sediments, and (4) lack of process understanding in the Gulf of Bothnia. In addition, the possible influence of genetic adaptation is currently unpredictable. Nevertheless, projected future hydrographic conditions such as mixing depth, light penetration, vertical exchange of nutrients and oxygen, and deep water ventilation will very likely affect biogeochemical cycles and consequently also the entire ecosystem. Given the projected changes in the abiotic environment and biogeochemical processes, we cautiously suggest some likely future changes in the marine ecosystem.
Ice cover is expected to decrease in the northern Baltic Sea, causing an earlier onset of the spring bloom by up to 1 month (Fig. 3), and increased wind- and wave-induced resuspension will hasten the transport of nutrients from the coastal zone to the open sea (Eilola et al. 2013). Increased river runoff will lead to higher concentrations of AOM, which reduces light penetration in the water and potentially also primary production (Wikner and Andersson 2012; Lefébure et al. 2013). The AOM provides an alternate carbon source for heterotrophic bacteria as compared to phytoplankton-derived substrates, which may increase bacterial activity, making bacteria outcompete phytoplankton for inorganic nutrients. The poorer light climate and increased competition from bacteria may decrease phytoplankton production (Lefébure et al. 2013). Although the spring bloom occurs earlier due to earlier ice break-up, primary production does not necessarily increase according to modeling results (Eilola et al. 2013). Should the yearly primary production decrease, it is likely that the production will decrease also at higher trophic levels. The food web will get an additional intermediate trophic level, which will cause increased respiration and excretion losses (Fig. 5), most likely causing decreased production of zooplankton, benthos, and fish. Because water absorbs less oxygen at higher temperature, oxygen concentration in the water will be reduced, and increased AOM will stimulate bacterial respiration, further reducing the oxygen concentration (Panigrahi et al. 2013). Decreased light availability may favor tactile over visual predators. Taken together, field observations in the coastal zone (data presented here), experimental studies (e.g., Lefébure et al. 2013), and long-term, large-scale studies (Wikner and Andersson 2012) indicate that the base of the food web, i.e., the phytoplankton and bacteria, can become significantly altered if climate change increases inputs of AOM to the Gulf of Bothnia.
Simplified schematic view of climate-altered food webs in the Bothnian Bay (upper), southern Bothnian Sea (mid), and Baltic proper (lower) in summer. Green arrows represent autochthonous and brown arrows allochthonous production. Organisms included in the food webs and their trophic position (in parenthesis); bacteria (1, 2), phytoplankton (1), flagellates (2, 3), ciliates (3, 4), zooplankton (2, 4, 5), and fish (3, 5, 6). Illustration by Mats Minnhagen
In the Baltic proper, increased nutrient loads and higher temperatures may intensify internal nutrient cycling (Meier et al. 2011a), potentially increasing both primary production and oxygen consumption (Fig. 4). This may possibly increase phosphorus mobility and reduce denitrification efficiency (Meier et al. 2012b). Without drastic nutrient load abatements, hypoxic and anoxic areas are projected to increase (Meier et al. 2011a) and cause intensified exchange of nutrients between shallow and deeper waters (Eilola et al. 2012).
Scenario simulations suggest that the rising atmospheric CO2 will control future pH changes in the surface water and that eutrophication will not affect the mean pH (Omstedt et al. 2012). Climate warming may lead to earlier and more frequent cyanobacterial blooms, as already observed for surface accumulations of cyanobacteria (Kahru and Elmgren 2014) and perhaps also to increased nitrogen fixation (Meier et al. 2012b; Neumann et al. 2012; Hense et al. 2013). Nitrogen-fixing cyanobacteria will supply the ecosystem with plant-available nitrogen, but, as they are of poor food quality for consumers, the efficiency of energy transfer to higher trophic levels may be reduced (Fig. 5).
A lower salinity may reduce or eliminate cod spawning areas, reducing the value of the fish catch (Fig. 4). Anoxia will reduce the production of benthic fish food. A lower salinity will eliminate many marine species, but allow more freshwater species to colonize the ecosystem. A decrease in salinity will inevitably change species composition and therefore ecosystem function (e.g., through the loss of filter-feeding bivalves). Most food-web and fish population models indicate that cod fishing will remain an important determinant of the cod stock in the future, although climate effects may be substantial. Eutrophication has, however, been suggested to be of minor importance for the cod stock size (Niiranen et al. 2013).
Climate change may directly benefit invasive species by providing conditions nearer to those in their native ranges, e.g., warmer temperatures. Furthermore, invasive species tolerant of low-oxygen conditions may be favored, such as Marenzelleria spp. (Maximov 2011). As these polychaetes burrow deep into sediments (30 cm), they can stimulate release and subsequent bioaccumulation of buried organic pollutants (Josefsson et al. 2010, 2011).
In a future climate, halogenated aromatic and polycyclic aromatic pollutants may be sorbed to DOC to a greater extent. Whether this will increase or reduce their availability to marine organisms is unknown. Since DOC is partly utilized by bacteria as a food source, we hypothesize an increased transport of DOC-associated pollutants up the food web via microbes (Wallberg et al. 2001). On the other hand, high concentrations of humic substances in lakes have been shown to make persistent organic pollutants less available for uptake by fish (Larsson et al. 1992).
Implications for management of the Baltic Sea
Considering the complex and interactive alterations climate change may induce in the Baltic Sea ecosystem, it is crucial that future Baltic Sea management takes these aspects into consideration. During recent decades, the Baltic countries have agreed on joint goals for the management of the Baltic Sea environment. Compliance monitoring has been used to classify the ecological state of marine waters (e.g., Anon 2007), but is not yet focused on climate change, which interacts with other ecosystem changes and exposures.
One example is chlorophyll a, a proxy for phytoplankton biomass, which should respond significantly to nutrient loading. The chlorophyll content in algae is, however, highly variable depending on, e.g., species composition and local light climate, which in turn are affected by AOM in the water. An increased AOM load is likely to promote bacterial production, but decrease primary production, as described above. The oxygen concentration may decrease and the ecosystem may become more heterotrophic. The food web will acquire extra trophic levels, leading to greater energy losses and decreased fish production. We therefore suggest that not only primary production but also bacterial production should be monitored in the Baltic Sea.
Management should also consider the effects of temperature and DOC on hypoxia, and thus not only interpret low oxygen levels as a result of enrichment with nitrogen and phosphorus. We found markedly higher temperature sensitivity of bacterioplankton in coastal environments with high concentrations of DOC. Thus, climate-related factors (i.e., temperature and runoff) can influence hypoxia in estuarine waters, and in such cases reduction of nitrogen and phosphorus alone may not be an effective restoration measure.
Climate-induced changes in geochemical and biological conditions will alter the concentration and distribution of semi-persistent and persistent organic pollutants in the sea. The net effect is expected to differ among pollutants depending on their lipophilicity, sorption properties, volatility, and resistance to metabolism. This is important to consider when forecasting food web transfer and human exposure and when designing or modifying monitoring programs for such pollutants (Undeman et al. 2015).
It has been suggested that, because the invasive deep-burrowing polychaete Marenzelleria stimulates the release of legacy pollutants from fiber banks and other hotspots in the Gulf of Bothnia (Josefsson et al. 2010, 2011), it may have played a part in a recent local reproduction failure in white-tailed eagle (Haliaeetus albicilla) (Helander and Bignert 2012). Although the interactions between native food webs, invasive species, pollutant chemistry, and climate change are complex, they clearly need to be considered when managing marine ecosystem.
While a number of management plans have been developed in response to eutrophication (e.g., the HELCOM Baltic Sea Action Plan, http://helcom.fi/baltic-sea-action-plan and the EU water framework directive), monitoring needs to be expanded also to cover secondary producers and other processes and to encompass changes in climate and anthropogenic pollutant loads.
References
Andersson, A., S. Hajdu, P. Haecky, J. Kuparinen, and J. Wikner. 1996. Succession and growth limitation of phytoplankton in the Gulf of Bothnia. Marine Biology 126: 791–801.
Anon. 2007. Classification system for coastal and transition waters. Handbook 2007:4. Appendix B. Stockholm: Swedish Environmental Protection Agency (in Swedish).
Ask, J., J. Karlsson, L. Persson, P. Ask, P. Byström, and M. Jansson. 2009. Terrestrial organic matter and light penetration: Effects on bacterial and primary production in lakes. Limnology and Oceanography 54: 2034–2040.
Autio, R. 1998. Response of seasonally cold-water bacterioplankton to temperature and substrate treatments. Estuarine, Coastal and Shelf Science 46: 465–474.
BACC I author team. 2008. Assessment of climate change for the Baltic Sea basin. Regional climate studies. Berlin: Springer.
BACC II author team. 2015. Second assessment of climate change for the Baltic Sea basin. Regional climate studies. Berlin: Springer.
Bergknut, M., S. Meijer, C. Halsall, A. Ågren, H. Laudon, S. Kohle, K.C. Jones, M. Tysklind, et al. 2010. Modelling the fate of hydrophobic organic contaminants in a boreal forest catchment: A cross-disciplinary approach to assessing diffuse pollution to surface waters. Environmental Pollution 158: 2964–2969.
Berglund, J., U. Muren, U. Båmstedt, and A. Andersson. 2007. Efficiency of a phytoplankton and a bacterial-based food web in a pelagic marine system. Limnology and Oceanography 52: 121–131.
Cole, J.J., N.F. Caraco, and B.L. Peierls. 1992. Can phytoplankton maintain a positive carbon balance in a turbid, freshwater, tidal estuary. Limnology and Oceanography 37: 1608–1617.
Donnelly, C., W. Yang, and J. Dahné. 2014. River discharge to the Baltic Sea in a future climate. Climatic Change 122: 157–170.
Eilola, K., B.G. Gustafson, I. Kuznetsov, H.E.M. Meier, T. Neumann, and O.P. Savchuk. 2011. Evaluation of biogeochemical cycles in an ensemble of three state-of-the-art numerical models of the Baltic Sea. Journal of Marine Systems 88: 267–284.
Eilola, K., E. Almroth-Rosell, C. Dieterich, F. Fransner, A. Höglund, and H.E.M. Meier. 2012. Modeling nutrient transports and exchanges of nutrients between shallow regions and the open Baltic Sea in present and future climate. AMBIO 41: 574–585.
Eilola, K., S. Mårtensson, and H.E.M. Meier. 2013. Modeling the impact of reduced sea ice cover in future climate on the Baltic Sea biogeochemistry. Geophysical Research Letters 40: 1–6.
Engebretson, R.R., and R. von Wandruszka. 1994. Microorganization in dissolved humic acids. Environmental Science and Technology 28: 1934–1941.
Eriksson-Wiklund, A.-K., and A. Andersson. 2014. Benthic competition and population dynamics of Monoporeia affinis and Marenzelleria sp. in the northern Baltic Sea. Estuarine, Coastal and Shelf Science 144: 46–53.
Finkel, Z.V., J. Beardall, K.J. Flynn, A. Quigg, T.A.V. Rees, and J.A. Raven. 2010. Phytoplankton in a changing world: Cell size and elemental stoichiometry. Journal of Plankton Research 32: 119–137.
Gräwe, U., R. Friedland, and H. Burchard. 2013. The future of the western Baltic Sea: Two possible scenarios. Ocean Dynamics 63: 901–921.
Helander, B., and A. Bignert. 2012. White tailed eagle. In Havet 2012, 100–101. Göteborg: Swedish Institute for the Marine Environment (in Swedish).
Hense, I., H.E.M. Meier, and S. Sonntag. 2013. Projected climate change impact on Baltic Sea cyanobacteria. Climatic Change 119: 391–406.
Howarth, R.W., D.P. Swaney, T.J. Butler, and R. Marino. 2000. Climatic control on eutrophication of the Hudson River estuary. Ecosystems 3: 210–215.
Josefsson, S., K. Leonardsson, J. Gunnarsson, and K. Wiberg. 2010. Bioturbation-driven release of buried PCBs and PDBEs from different depths in contaminated sediments. Environmental Science and Technology 44: 7456–7464.
Josefsson, S., K. Leonardsson, J. Gunnarsson, and K. Wiberg. 2011. Influence of contamination burial depth on the bioaccumulation of PCBS and PBDEs by two benthic invertebrates (Monoporeia affinis and Marenzelleria spp.). Chemosphere 85: 1444–1451.
Kahru, M., and R. Elmgren. 2014. Multi-decadal time series of satellite-detected accumulations of cyanobacteria in the Baltic Sea. Biogeosciences 11: 3619–3633.
Kjellström, E., and P. Lind. 2009. Changes in the water budget in the Baltic Sea drainage basin in future warmer climates as simulated by the regional climate model RCA3. Boreal Environment Research 14: 114–124.
Kjellström, E., G. Nikulin, U. Hansson, G. Strandberg, and A. Ullerstig. 2011. 21st century changes in the European climate: Uncertainties derived from an ensemble of regional climate model simulations. Tellus A 63: 24–40.
Kuivikko, M., K. Sorsa, J.V.K. Kukkonen, J. Akkanen, T. Kotiaho, and A.V. Vahatalo. 2010. Partitioning of tetra- and pentabromo diphenyl ether and benzo(a)pyrene among water and dissolved and particulate organic carbon along a salinity gradient in coastal waters. Environmental Toxicology and Chemistry 29: 2443–2449.
Kukkonen, J., J.F. McCarthy, and A. Oikari. 1990. Effects of XAD-8 fractions of dissolved organic carbon on the sortpion and bioavailability of organic micropollutants. Archives of Environmental Contamination and Toxicology 19: 551–557.
Larsson, P., L. Collvin, L. Okla, and G. Meyer. 1992. Lake productivity and water chemistry as governors of the uptake of persistent pollutants in fish. Environmental Science and Technology 26: 346–352.
Lefébure, R., R. Degerman, A. Andersson, S. Larsson, L.-O. Eriksson, U. Båmstedt, and P. Byström. 2013. Impacts of elevated terrestrial nutrient loads and temperature on pelagic food-web efficiency and fish production. Global Change Biology 19: 1358–1372.
Lignell, R., L. Hiokkala, and T. Lahtinen. 2008. Effects of inorganic nutrients, glucose and solar radiation on bacterial growth and exploitation of dissolved organic carbon and nitrogen in the northern Baltic Sea. Aquatic Microbial Ecology 51: 209–221.
Maximov, A.A. 2011. Large-scale invasion of Marenzelleria spp. (Polychaeta; Spionidae) in the eastern Gulf of Finland, Baltic Sea. Russian Journal of Biological Invasions 2: 11–19.
Meier, H.E.M., and F. Kauker. 2003. Modeling decadal variability of the Baltic Sea: 2. Role of freshwater inflow and large-scale atmospheric circulation for salinity. Journal of Geophysical Research 108: 3368.
Meier, H.E.M., R. Döscher, and A. Halkka. 2004. Simulated distributions of Baltic sea-ice in warming climate and consequences for the winter habitat of the Baltic ringed seal. AMBIO 33: 249–256.
Meier, H.E.M. 2005. Modeling the age of Baltic Sea water masses: Quantification and steady state sensitivity experiments. Journal of Geophysical Research 110: C02006.
Meier, H.E.M. 2006. Baltic Sea climate in the late twenty-first century: A dynamical downscaling approach using two global models and two emission scenarios. Climate Dynamics 27: 39–68.
Meier, H.E.M., E. Kjellström, and L.P. Graham. 2006. Estimating uncertainties of projected Baltic Sea salinity in the late 21st century. Geophysical Research Letters 33: L15705.
Meier, H.E.M., H.C. Andersson, K. Eilola, B.G. Gustafsson, I. Kuznetsov, B. Müller-Karulis, T. Neumann, and O.P. Savchuk. 2011a. Hypoxia in future climates—A model ensemble study for the Baltic Sea. Geophysical Research Letters 38: L24608.
Meier, H.E.M., A. Höglund, R. Döscher, H. Andersson, U. Löptien, and E. Kjellström. 2011b. Quality assessment of atmospheric surface fields over the Baltic Sea of an ensemble of regional climate model simulations with respect to ocean dynamics. Oceanologia 53: 193–227.
Meier, H.E.M., K. Eilola, B.G. Gustafsson, I. Kuznetsov, T. Neumann, and O.P. Savchuk. 2012a. Uncertainty assessment of projected ecological quality indicators in future climate. Rapport Oceanografi No. 112, SMHI, Norrköping, Sweden.
Meier, H.E.M., B. Müller-Karulis, H.C. Andersson, C. Dieterich, K. Eilola, B.G. Gustafsson, A. Höglund, R. Hordoir, et al. 2012b. Impact of climate change on ecological quality indicators and biogeochemical fluxes in the Baltic Sea: A multi-model ensemble study. AMBIO 41: 558–573.
Neumann, T. 2010. Climate-change effects on the Baltic Sea ecosystem: A model study. Journal of Marine Systems 81: 213–224.
Neumann, T., K. Eilola, B.G. Gustafsson, B. Müller-Karulis, I. Kuznetsov, H.E.M. Meier, and O.P. Savchuk. 2012. Extreme values of temperature, oxygen and blooms in the Baltic Sea in changing climate. AMBIO 41: 574–585.
Niiranen, S., J. Yletyinen, M.T. Tomczak, T. Blenckner, O. Hjerne, B. MacKenzie, B. Müller-Karulis, T. Neumann, et al. 2013. Combined effects of global climate change and regional ecosystem drivers on an exploited marine food web. Global Change Biology 19: 3327–3342.
Nikulin, G., E. Kjellström, U. Hansson, C. Jones, G. Strandberg, and A. Ullerstig. 2011. Evaluation and future projections of temperature, precipitation and wind extremes over Europe in an ensemble of regional climate simulations. Tellus A 63: 41–55.
Omstedt, A., M. Edman, B. Claremar, P. Frodin, E. Gustafsson, C. Humborg, M. Mörth, A. Rutgersson, et al. 2012. Future changes of the Baltic Sea acid-base (pH) and oxygen balances. Tellus B 64: 19586.
Panigrahi, S., A. Nydahl, P. Anton, and J. Wikner. 2013. Strong seasonal effect of moderate experimental warming on plankton respiration in a temperate estuarine plankton community. Estuarine, Coastal and Shelf Science 135: 269–279.
Pettersson, C., B. Allard, and H. Boren. 1997. River discharge of humic substances and humic-bound metals to the Gulf of Bothnia. Estuarine, Coastal and Shelf Science 44: 533–541.
Poerschmann, J., and F.D. Kopinke. 2001. Sorption of very hydrophobic organic Compounds (VHOCs) on dissolved humic organic matter (DOM). 2. Measurement of sorption and application of a Flory–Huggins concept to interpret the data. Environmental Science and Technology 35: 1142–1148.
Ripszam, M., J. Paczkowska, J. Figueira, C. Veenaas, and P. Haglund. 2015. Dissolved organic carbon quality and sorption of organic pollutants in the Baltic Sea in light of future climate. Environmental Science and Technology 49: 1445–1452.
Sandberg, J., A. Andersson, S. Johansson, and J. Wikner. 2004. Pelagic food web structure and carbon budget in three brackish water environments: Potential importance of terrigenous carbon. Marine Ecology Progress Series 268: 13–29.
Schimanke, S., C. Dieterich, and H.E.M. Meier. 2014. An algorithm based on sea level pressure fluctuations to identify major Baltic inflow events. Tellus A 66: 23452. doi:10.3402/tellusa.v66.23452.
Smith, V.H. 2006. Responses of estuarine and coastal marine phytoplankton to nitrogen and phosphorus enrichment. Limnology and Oceanography 51: 377–384.
Stepanauskas, R., N.O.G. Jorgensen, O.R. Eigaard, A. Zvikas, L. Tranvik, and L. Leonardsson. 2002. Summer inputs of riverine nutrients to the Baltic Sea: Bioavailability and eutrophication relevance. Ecological Monographs 72: 579–597.
Undeman, E., B.G. Gustafsson, C. Humborg, and M.S. McLachlan. 2015. Application of a novel modeling tool with multistressor functionality to support management of organic contaminants in the Baltic Sea. AMBIO. doi:10.1007/s13280-015-0668-2.
Wallberg, P., P. Jonsson, and A. Andersson. 2001. Trophic transfer and passive uptake of a polychlorinated biphenyl (PCB) in experimental marine microbial food webs. Environmental Toxicology and Chemistry 20: 2158–2164.
Wikner, J., R. Quadros, and M. Jansson. 1999. Differences in consumption of allochthonous DOC under limnic and estuarine conditions in a watershed. Aquatic Microbial Ecology 17: 289–299.
Wikner, J., and A. Andersson. 2012. Increased freshwater discharge shifts the trophic balance in the coastal zone of the northern Baltic Sea. Global Change Biology 18: 2509–2519.
Zweifel, U.L., J. Wikner, and Å. Hagström. 1995. Dynamics of dissolved organic carbon in a coastal ecosystem. Limnology and Oceanography 40: 299–305.
Acknowledgments
This study was financed by the Strategic Marine Environmental Research programs Ecosystem dynamics in the Baltic Sea in a changing climate perspective (ECOCHANGE) and Baltic Ecosystem Adaptive Management (BEAM). We thank Umeå Marine Science Centre and the Marine Laboratory in Kalmar for excellent support during field sampling.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Andersson, A., Meier, H.E.M., Ripszam, M. et al. Projected future climate change and Baltic Sea ecosystem management. AMBIO 44 (Suppl 3), 345–356 (2015). https://doi.org/10.1007/s13280-015-0654-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13280-015-0654-8