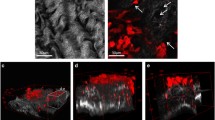
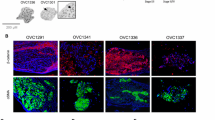
Abstract
Ovarian cancer progression is frequently associated with the development of malignant ascites. Multicellular aggregates of carcinoma cells (spheroids) found within ascites are thought to be able to promote peritoneal carcinomatosis. We have previously demonstrated the involvement of the vitronectin/αv integrin adhesive system in the dissemination of ovarian cancer cells and continue to investigate the influence of these molecules by studying their role(s) in spheroid behavior. The aim of this study was to generate ovarian cancer multicellular aggregates and to focus on the role of vitronectin and αv integrins in their initiation. IGROV1 cancer cells cultured in the absence of adhesive substratum formed multicellular aggregates comparable to spheroids. After 21 days, a fraction of the cells within clusters remained viable and proliferated recurrently. Within the multicellular aggregates, vitronectin and αv integrins were co-localized at intercellular sites, suggesting their involvement in cell-cell interactions. Initial formation of IGROV1 aggregates was inhibited using anti-vitronectin and anti-αv integrin blocking antibodies or the cyclic peptide cRGDfV. Vitronectin expression persisted during cluster disaggregation on fibronectin. These results demonstrate the ability of IGROV1 cells to generate multicellular aggregates and point to a contributory role for the vitronectin/αv integrin system in the initial step of this process. These events could represent a prerequisite for further dissemination.





Similar content being viewed by others
References
Auersperg N, Wong AS, Choi KC, Kang SK, Leung PC. Ovarian surface epithelium: biology, endocrinology, and pathology. Endocr Rev. 2001;22:255–88.
Allen HJ, Porter C, Gamarra M, Piver MS, Johnson EA. Isolation and morphologic characterization of human ovarian carcinoma cell clusters present in effusions. Exp Cell Biol. 1987;55:194–208.
Shield K, Riley C, Quinn MA, Rice GE, Ackland ML, Ahmed N. Alpha2beta1 integrin affects metastatic potential of ovarian carcinoma spheroids by supporting disaggregation and proteolysis. J Carcinog. 2007;6:11.
Burleson KM, Boente MP, Pambuccian SE, Skubitz AP. Disaggregation and invasion of ovarian carcinoma ascites spheroids. J Transl Med. 2006;4:6.
Puiffe ML, Le Page C, Filali-Mouhim A, Zietarska M, Ouellet V, Tonin PN, et al. Characterization of ovarian cancer ascites on cell invasion, proliferation, spheroid formation, and gene expression in an in vitro model of epithelial ovarian cancer. Neoplasia. 2007;9:820–9.
Yoshida Y, Kurokawa T, Nishikawa Y, Orisa M, Kleinman HK, Kotsuji F. Laminin-1-derived scrambled peptide AG73T disaggregates laminin-1-induced ovarian cancer cell spheroids and improves the efficacy of cisplatin. Int J Oncol. 2008;32:673–81.
Fishman DA, Kearns A, Chilukuri K, Bafetti LM, O'Toole EA, Georgacopoulos J, et al. Metastatic dissemination of human ovarian epithelial carcinoma is promoted by alpha2beta1-integrin-mediated interaction with type I collagen. Invasion Metastasis. 1998;18:15–26.
Casey RC, Burleson KM, Skubitz KM, Pambuccian SE, Oegema Jr TR, Ruff LE, et al. Beta 1-integrins regulate the formation and adhesion of ovarian carcinoma multicellular spheroids. Am J Pathol. 2001;159:2071–80.
Hapke S, Kessler H, Luber B, Benge A, Hutzler P, Hofler H, et al. Ovarian cancer cell proliferation and motility is induced by engagement of integrin alpha(v)beta3/Vitronectin interaction. Biol Chem. 2003;384:1073–83.
Leroy-Dudal J, Demeilliers C, Gallet O, Pauthe E, Dutoit S, Agniel R, et al. Transmigration of human ovarian adenocarcinoma cells through endothelial extracellular matrix involves alphav integrins and the participation of MMP2. Int J Cancer. 2005;114:531–43.
Carreiras F, Rigot V, Cruet S, Andre F, Gauduchon P, Marvaldi J. Migration properties of the human ovarian adenocarcinoma cell line IGROV1: importance of alpha(v)beta3 integrins and vitronectin. Int J Cancer. 1999;80:285–94.
Burleson KM, Casey RC, Skubitz KM, Pambuccian SE, Oegema Jr TR, Skubitz AP. Ovarian carcinoma ascites spheroids adhere to extracellular matrix components and mesothelial cell monolayers. Gynecol Oncol. 2004;93:170–81.
Landen CN, Kim TJ, Lin YG, Merritt WM, Kamat AA, Han LY, et al. Tumor-selective response to antibody-mediated targeting of alphavbeta3 integrin in ovarian cancer. Neoplasia. 2008;10(11):1259–67.
Preissner KT. Structure and biological role of vitronectin. Annu Rev Cell Biol. 1991;7:275–310.
Schvartz I, Seger D, Shaltiel S. Vitronectin. Int J Biochem Cell Biol. 1999;31:539–44.
Carreiras F, Cruet S, Staedel C, Sichel F, Gauduchon P. Human ovarian adenocarcinoma cells synthesize vitronectin and use It to organize their adhesion. Gynecol Oncol. 1999;72:312–22.
Heyman L, Kellouche S, Fernandes J, Dutoit S, Poulain L, Carreiras F. Vitronectin and its receptors partly mediate adhesion of ovarian cancer cells to peritonel mesothelium in vitro. Tumour Biol. 2008;29:231–44.
Leroy-Dudal J, Heyman L, Gauduchon P, Carreiras F. Adhesion of human ovarian adenocarcinoma IGROV1 cells to endothelial cells is partly mediated by the alphav integrins-vitronectin adhesive system and induces an alteration of endothelial integrity. Cell Biol Int. 2005;29:482–8.
Bardies M, Thedrez P, Gestin JF, Marcille BM, Guerreau D, Faivre-Chauvet A, et al. Use of multi-cell spheroids of ovarian carcinoma as an intraperitoneal radio-immunotherapy model: uptake, retention kinetics and dosimetric evaluation. Int J Cancer. 1992;50:984–91.
Zietarska M, Maugard CM, Filali-Mouhim A, Alam-Fahmy M, Tonin PN, Provencher DM, et al. Molecular description of a 3D in vitro model for the study of epithelial ovarian cancer (EOC). Mol Carcinog. 2007;46:872–85.
Bénard J, Da Silva J, De Blois MC, Boyer P, Duvillard P, Chiric E, et al. Characterization of a human ovarian adenocarcinoma line, IGROV1, in tissue culture and in nude mice. Cancer Res. 1985;45:4970–9.
Poulain L, Lincet H, Duigou F, Deslandes E, Sichel F, Gauduchon P, et al. Acquisition of chemoresistance in a human ovarian carcinoma cell is linked to a defect in cell cycle control. Int J Cancer. 1998;78:454–63.
Jin ZH, Josserand V, Razkin J, Garanger E, Boturyn D, Favrot MC, et al. Noninvasive optical imaging of ovarian metastases using Cy5-labeled RAFT-c(-RGDfK-)4. Mol Imaging. 2006;5:188–97.
Yatohgo T, Izumi M, Kashiwagi H, Hayashi M. Novel purification of vitronectin from human plasma by heparin affinity chromatography. Cell Struct Funct. 1988;13:281–92.
Poulouin L, Gallet O, Rouahi M, Imhoff JM. Plasma fibronectin: three steps to purification and stability. Protein Expr Purif. 1999;17(1):146–52.
Villedieu M, Deslandes E, Duval M, Heron JF, Gauduchon P, Poulain L. Acquisition of chemoresistance following discontinuous exposures to cisplatin is associated in ovarian carcinoma cells with progressive alteration of FAK, ERK and p38 activation in response to treatment. Gynecol Oncol. 2006;101:507–19.
Darzynkiewicz Z, Bruno S, Del Bino G, Gorczyca W, Hotz MA, Lassota P, et al. Features of apoptotic cells measured by flow cytometry. Cytometry. 1992;13:795–808.
Carreiras F, Denoux Y, Staedel C, Lehmann M, Sichel F, Gauduchon P. Expression and localization of alpha v integrins and their ligand vitronectin in normal ovarian epithelium and in ovarian carcinoma. Gynecol Oncol. 1996;62:260–7.
Hoffmann S, He S, Jin M, Ehren M, Wiedemann P, Ryan SJ, et al. A selective cyclic integrin antagonist blocks the integrin receptors alphavbeta3 and alphavbeta5 and inhibits retinal pigment epithelium cell attachment, migration and invasion. BMC Ophthalmol. 2005;5:16.
Shield K, Ackland ML, Ahmed N, Rice GE. Multicellular spheroids in ovarian cancer metastases: biology and pathology. Gynecol Oncol. 2009;113:143–8.
Kenny HA, Kaur S, Coussens LM, Lengyel E. The initial steps of ovarian cancer cell metastasis are mediated by MMP-2 cleavage of vitronectin and fibronectin. J Clin Invest. 2008;118(4):1367–79.
Desoize B, Gimonet D, Jardillier JC. Multicellular resistance: another mechanism for multidrug resistance? Bull Cancer. 1998;85(9):785.
Frisch SM, Francis H. Disruption of epithelial cell-matrix interactions induces apoptosis. J Cell Biol. 1994;124(4):619–26.
Davidson B, Berner A, Nesland JM, Risberg B, Berner HS, Tropè CG, et al. E-cadherin and alpha-, beta-, and gamma-catenin protein expression is up-regulated in ovarian carcinoma cells in serous effusions. J Pathol. 2000;192(4):460–9.
Davidson B, Goldberg I, Reich R, Tell L, Dong HP, Trope CG, et al. AlphaV- and beta1-integrin subunits are commonly expressed in malignant effusions from ovarian carcinoma patients. Gynecol Oncol. 2003;90:248–57.
Zanetti A, Conforti G, Hess S, Martin-Padura I, Ghibaudi E, Preissner KT, et al. Clustering of vitronectin and RGD peptides on microspheres leads to engagement of integrins on the luminal aspect of endothelial cell membrane. Blood. 1994;84:1116–23.
Cruet-Hennequart S, Maubant S, Luis J, Gauduchon P, Staedel C, Dedhar S. alpha(v) integrins regulate cell proliferation through integrin-linked kinase (ILK) in ovarian cancer cells. Oncogene. 2003;22:1688–702.
Acknowledgments
We thank Pr JF Héron, oncogynecologist of the Centre F. Baclesse (CFB) and Dr F. Morvan, head of the oncology service of the Centre R. Dubos (CRD) for their constant support. This work was supported by the Ligue Contre le Cancer (Comité du Val d’Oise) and the University of Cergy-Pontoise. We thank C. Blanc-Fournier (CFB), V. Villefranques (CRD) for providing ascites I. Zimmerman (UMR CNRS 6143) for his technical support in scanning microscopy and M. Duval (plateforme cytométrie, IFR 146 ICORE). J. Fernandes is the recipient of a fellowship from the Ligue Contre le Cancer (Comité du Val d’Oise).
Author information
Authors and Affiliations
Corresponding author
Additional information
Grant sponsor
Ligue Contre le Cancer (Comité du Val d’Oise) and Cergy-Pontoise University
Sabrina Kellouche and Julien Fernandes contributed equally to this work.
Rights and permissions
About this article
Cite this article
Kellouche, S., Fernandes, J., Leroy-Dudal, J. et al. Initial formation of IGROV1 ovarian cancer multicellular aggregates involves vitronectin. Tumor Biol. 31, 129–139 (2010). https://doi.org/10.1007/s13277-010-0017-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-010-0017-9