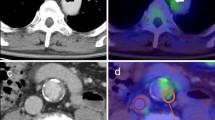
Abstract
Arterial inflammation is an indicator of atheromatous plaque vulnerability to detach and to obstruct blood vessels in the heart or in the brain thus causing heart attack or stroke. To date, it is difficult to predict the plaque vulnerability. This study was aimed to assess the behavior of 18F-sodium fluoride (18F-NaF) and 18F-fluorodeoxyglucose (18F-FDG) uptake in the aorta and iliac arteries as a function of plaque density on CT images. We report metabolically active artery plaques associated to inflammation in the absence of calcification. 18 elderly volunteers were recruited and imaged with computed tomography (CT) and positron emission tomography (PET) with 18F-NaF and 18F-FDG. A total of 1338 arterial segments were analyzed, 766 were non-calcified and 572 had calcifications. For both 18F-NaF and 18F-FDG, the mean SUV values were found statistically significantly different between non-calcified and calcified artery segments. Clustering CT non-calcified segments, excluding blood, resulted in two clusters C1 and C2 with a mean density of 30.63 ± 5.06 HU in C1 and 43.06 ± 4.71 HU in C2 (P < 0.05), and their respective SUV were found statistically different in 18F-NaF and 18F-FDG. The 18F-NaF images showed plaques not detected on CT images, where the 18F-FDG SUV values were high in comparison to artery walls without plaques. The density on CT images alone corresponding to these plaques could be further investigated to see whether it can be an indicator of the active plaques.





Similar content being viewed by others
References
Virmani R, Kolodgie FD, Burke AP et al (2005) Atherosclerotic plaque progression and vulnerability to rupture: Angiogenesis as a source of intraplaque hemorrhage. Arterioscler Thromb Vasc Biol 25:2054–2061. https://doi.org/10.1161/01.ATV.0000178991.71605.18
Kuku KO, Singh M, Ozaki Y et al (2020) Near-Infrared spectroscopy intravascular ultrasound Imaging: state of the art. Front Cardiovasc Med. https://doi.org/10.3389/fcvm.2020.00107
Kramer CM, Anderson JD (2007) MRI of atherosclerosis: diagnosis and monitoring therapy. Expert Rev Cardiovasc Ther 5:69–80. https://doi.org/10.1586/14779072.5.1.69
Naghavi M, Madjid M, Khan MR et al (2001) New developments in the detection of vulnerable plaque. Curr Atheroscler Rep 3:125–135. https://doi.org/10.1007/s11883-001-0048-1
Nighoghossian N, Derex L, Douek P (2005) The vulnerable carotid artery plaque: current imaging methods and new perspectives. Stroke 36:2764–2772. https://doi.org/10.1161/01.STR.0000190895.51934.43
Revkin JH, Shear CL, Pouleur HG et al (2007) Biomarkers in the prevention and treatment of atherosclerosis: need, validation, and future. Pharmacol Rev 59:40–53. https://doi.org/10.1124/pr.59.1.1
Narula J, Nakano M, Virmani R et al (2013) Histopathologic characteristics of atherosclerotic coronary disease and implications of the findings for the invasive and noninvasive detection of vulnerable plaques. J Am Coll Cardiol 61:1041–1051. https://doi.org/10.1016/j.jacc.2012.10.054
Owen DRJ, Lindsay AC, Choudhury RP, Fayad ZA (2011) Imaging of atherosclerosis. Annu Rev Med 62:25–40. https://doi.org/10.1146/annurev-med-041709-133809
Vengrenyuk Y, Carlier S, Xanthos S et al (2006) A hypothesis for vulnerable plaque rupture due to stress-induced debonding around cellular microcalcifications in thin fibrous caps. Proc Natl Acad Sci 103:14678–14683. https://doi.org/10.1073/pnas.0606310103
Hunt JL, Fairman R, Mitchell ME et al (2002) Bone formation in carotid plaques. Stroke 33:1214–1219. https://doi.org/10.1161/01.STR.0000013741.41309.67
Derlin T, Richter U, Bannas P et al (2010) Feasibility of 18F-sodium fluoride PET/CT for imaging of atherosclerotic plaque. J Nucl Med 51:862–865. https://doi.org/10.2967/jnumed.110.076471
Joshi NV, Vesey AT, Williams MC et al (2014) 18F-fluoride positron emission tomography for identification of ruptured and high-risk coronary atherosclerotic plaques: a prospective clinical trial. Lancet 383:705–713. https://doi.org/10.1016/S0140-6736(13)61754-7
Motoyama S, Kondo T, Sarai M et al (2007) Multislice computed tomographic characteristics of coronary lesions in acute coronary syndromes. J Am Coll Cardiol 50:319–326. https://doi.org/10.1016/j.jacc.2007.03.044
Das M, Braunschweig T, Mühlenbruch G et al (2009) Carotid plaque analysis: comparison of dual-source computed tomography (CT) findings and histopathological correlation. Eur J Vasc Endovasc Surg 38:14–19. https://doi.org/10.1016/j.ejvs.2009.03.013
Saremi F, Achenbach S (2015) Coronary plaque characterization using CT. Am J Roentgenol 204:W249–W260. https://doi.org/10.2214/AJR.14.13760
Tarkin JM, Joshi FR, Rudd JHF (2014) PET imaging of inflammation in atherosclerosis. Nat Rev Cardiol 11:443–457. https://doi.org/10.1038/nrcardio.2014.80
Figueroa AL, Abdelbaky A, Truong QA et al (2013) Measurement of arterial activity on routine FDG PET/CT images improves prediction of risk of future CV events. JACC Cardiovasc Imaging 6:1250–1259. https://doi.org/10.1016/j.jcmg.2013.08.006
Izquierdo-Garcia D, Davies JR, Graves MJ et al (2009) Comparison of methods for magnetic resonance-guided [18-F]Fluorodeoxyglucose Positron Emission Tomography in Human carotid arteries. Stroke 40:86–93. https://doi.org/10.1161/STROKEAHA.108.521393
Alberts I, Hünermund JN, Prenosil G et al (2021) Clinical performance of long axial field of view PET/CT: a head-to-head intra-individual comparison of the biograph vision quadra with the biograph vision PET/CT. Eur J Nucl Med Mol Imaging. https://doi.org/10.1007/s00259-021-05282-7
Torizuka T, Nobezawa S, Momiki S et al (2000) Short dynamic FDG-PET imaging protocol for patients with lung cancer. Eur J Nucl Med 27:1538–1542. https://doi.org/10.1007/s002590000312
Derlin T, Tóth Z, Papp L et al (2011) Correlation of inflammation assessed by18F-FDG PET, active mineral deposition assessed by18F-fluoride PET, and vascular calcification in atherosclerotic plaque: a dual-tracer PET/CT study. J Nucl Med. https://doi.org/10.2967/jnumed.111.087452
Agatston AS, Janowitz WR, Hildner FJ et al (1990) Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol 15:827–832. https://doi.org/10.1016/0735-1097(90)90282-T
Liu C-Y, Chen D, Bluemke DA et al (2015) Evolution of aortic wall thickness and stiffness with atherosclerosis. Hypertension 65:1015–1019. https://doi.org/10.1161/HYPERTENSIONAHA.114.05080
Croteau E, Lavallée E, Labbe SM et al (2010) Image-derived input function in dynamic human PET/CT: methodology and validation with 11 C-acetate and 18F-fluorothioheptadecanoic acid in muscle and 18F-fluorodeoxyglucose in brain. Eur J Nucl Med Mol Imaging 37:1539–1550. https://doi.org/10.1007/s00259-010-1443-z
Hofmann Bowman MA, McNally EM (2012) Genetic pathways of vascular calcification. Trends Cardiovasc Med 22:93–98. https://doi.org/10.1016/j.tcm.2012.07.002
Blomberg BA, de Jong PA, Thomassen A et al (2017) Thoracic aorta calcification but not inflammation is associated with increased cardiovascular disease risk: results of the CAMONA study. Eur J Nucl Med Mol Imaging 44:249–258. https://doi.org/10.1007/s00259-016-3552-9
Moss AJ, Adamson PD, Newby DE, Dweck MR (2016) Positron emission tomography imaging of coronary atherosclerosis. Future Cardiol 12:483–496. https://doi.org/10.2217/fca-2016-0017
Lee J-W, Bae S-H, Jeong J-W et al (2004) Hypoxia-inducible factor (HIF-1)α: its protein stability and biological functions. Exp Mol Med 36:1–12. https://doi.org/10.1038/emm.2004.1
Abdelbaky A, Corsini E, Figueroa AL et al (2013) Focal arterial inflammation precedes subsequent calcification in the same location. Circ Cardiovasc Imaging 6:747–754. https://doi.org/10.1161/CIRCIMAGING.113.000382
Derlin T, Richter U, Bannas P et al (2010) Feasibility of 18 F-Sodium fluoride PET/CT for imaging of atherosclerotic plaque. J Nucl Med 51:862–865. https://doi.org/10.2967/jnumed.110.076471
Kitagawa T, Yamamoto H, Toshimitsu S et al (2017) 18F-sodium fluoride positron emission tomography for molecular imaging of coronary atherosclerosis based on computed tomography analysis. Atherosclerosis 263:385–392. https://doi.org/10.1016/j.atherosclerosis.2017.04.024
Vazquez-Figueroa JG, Rinehart S, Qian Z et al (2013) Prospective validation that vulnerable plaque associated with major adverse outcomes have larger plaque volume, less dense calcium, and more non-calcified plaque by quantitative, three-dimensional measurements using intravascular ultrasound with radiofreq. J Cardiovasc Transl Res 6:762–771. https://doi.org/10.1007/s12265-013-9473-0
Wong KKL, Thavornpattanapong P, Cheung SCP et al (2012) Effect of calcification on the mechanical stability of plaque based on a three-dimensional carotid bifurcation model. BMC Cardiovasc Disord. https://doi.org/10.1186/1471-2261-12-7
Achenbach S, Raggi P (2010) Imaging of coronary atherosclerosis by computed tomography. Eur Heart J 31:1442–1448. https://doi.org/10.1093/eurheartj/ehq150
Kitagawa T, Yamamoto H, Horiguchi J et al (2009) Characterization of noncalcified coronary plaques and identification of culprit lesions in patients with acute coronary syndrome by 64-slice computed tomography. JACC Cardiovasc Imaging 2:153–160. https://doi.org/10.1016/j.jcmg.2008.09.015
Li X, Heber D, Gonzalez JC et al (2017) Association between osteogenesis and inflammation during the progression of calcified plaque evaluated by 18F-Fluoride and 18F-FDG. J Nucl Med 58:968–974. https://doi.org/10.2967/jnumed.116.182790
Acknowledgements
AD received a student fellowship from the Faculty of Medicine and Health Sciences, University of Sherbrooke. MSA received a financial support from University of Hail in kingdom of Saudi Arabia.
Funding
This study was funded by the Canadian Institutes of Health Research (CIHR, Grant Number 407104).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Douhi, A., Al‑enezi, M.S., Berrahmoune, N. et al. Non-calcified active atherosclerosis plaque detection with 18F-NaF and 18F-FDG PET/CT dynamic imaging. Phys Eng Sci Med 46, 295–302 (2023). https://doi.org/10.1007/s13246-023-01218-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13246-023-01218-7