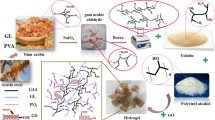
Abstract
Hydrogel development using natural and synthetic polymers for biomedical applications have shown promising properties for drug delivery due to virtuous stimuli response and improvement in mechanical strength. Biomaterial hydrogel was synthesized using combination of arabic gum (AG), a natural polymer and polyethylene glycol dimethacrylate (PEGDMA), a synthetic polymer. The synthesis process follows chemical cross linking by free radical polymerization. Fourier transform infrared spectroscopy (FTIR), field emission scanning electron microscopy (FESEM) and differential scanning calorimetry (DSC) were used to characterize the hydrogels. Product yield, gel content and gelation time depends solely on the variable reacting concentrations and the mechanical properties of hydrogel depends on its compositions. Swelling studies reveal high swelling ratio in buffer solution pH 1.2 compare to buffer solution pH 7.4, and deionized water at pH 6.7. Swelling ratio raises with increase in PEGDMA concentration, but the presences of salt/ions solutions decreases the swelling ratio significantly and it depends on the charge of the ions. The kinetics of the swelling follows a second order non-linear rate equation. The rate of degradation revealed significant degradation in acidic medium and insignificant degradation was observed in basic medium. The encapsulation efficiency of quercetin introduced directly in the hydrogels during synthesis was found in the range of 85–94%. The release profile indicates the dependence of arabic gum grafted PEGDMA hydrogels on pH and compositions. The characteristic properties exhibited by the formulated hydrogels suggest the suitability of them to be applied as drug carriers.

Similar content being viewed by others
References
N. Annabi, A. Tamayol, J. A. Uquillas, M. Akbari, L. E. Bertassoni, C. Cha, G. Camci-Unal, M. R. Dokmeci, N. A. Peppas, and A. Khademhosseini, Adv. Mater., 26, 85 (2014).
D. M. García, J. L. Escobar, N. Bada, J. Casquero, E. Hernáez, and I. Katime, Eur. Polym. J., 40, 1637 (2004).
Y. Qiu and K. Park, Adv. Drug Deliv. Rev., 64, 49 (2012).
A. K. Jha, K. M. Tharp, J. Ye, J. L. Santiago-Ortiz, W. M. Jackson, A. Stahl, D. V. Schaffer, Y. Yeghiazarians, and K. E. Healy, Biomaterials, 47, 1 (2015).
B. Cursaru, A.-L. Radu, F.-X. Perrin, A. Sarbu, M. Teodorescu, A.-M. Gavrilă, C.-M. Damian, T. Sandu, T.-V. Iordache, and A. Zaharia, Macromol. Res., 28, 211 (2020).
A. Vashist, A. Kaushik, A. Vashist R. D. Jayant, A. Tomitaka, S. Ahmad, Y. K. Gupta, and M. Nair, Biomater. Sci., 4, 1535 (2016).
J. Wu, X. Zhao, D. Wu, and C.-C. Chu, J. Mater. Chem. B, 2, 6660 (2014).
M. K. Sinha, J. Gao, C. E. T. Stowell, and Y. Wang, Regen. Biomater., 2, 177 (2015).
G. M. Cruise, D. S. Scharp, and J. A. Hubbell, Biomaterials, 19, 1287 (1998).
D. K. Han and J. A. Hubbell, Macromolecules, 30, 6077 (1997).
H. Lin, T. Kai, B. D. Freeman, S. Kalakkunnath, and D. S. Kalika, Macromolecules, 38, 8381 (2005).
P. van de Wetering, A. T. Metters, R. G. Schoenmakers, and J. A. Hubbell, J. Control Release, 102, 619 (2005).
R. N. Puglisi, L. Strande, M. Santos, G. Schulte, C. W. Hewitt, and T. V. Whalen, J. Surg. Res., 65, 115 (1996).
J. L. Hill-West, S. M. Chowdhury, A. S. Sawhney, C. P. Pathak, R. C. Dunn, and J. A. Hubbell, Obstet. Gynecol., 83, 59 (1994).
A. S. Sawhney, C. P. Pathak, J. J. van Rensburg, R. C. Dunn, and J. A. Hubbell, J. Biomed. Mater Res., 28, 831 (1994).
A. Mahardian, Biocompatible Hydrogel Film of Polyethylene Oxide-Polyethylene Glycol Dimetacrylate for Wound Dressing Application, in IOP Conference Series: Materials Science and Engineering, IOP Publishing, 2018.
J. A. Killion, L. M. Geever, D. M. Devine, J. E. Kennedy, and C. L. Higginbotham, J. Mech. Behav. Biomed. Mater., 4, 1219 (2011).
K. Arcaute, B. K. Mann, and R. B. Wicker, Ann. Biomed. Eng., 34, 1429 (2006)
V. Barron, J. A. Killion, L. Pilkington, G. Burke, L. M. Geever, J. G. Lyons, E. McCullagh, and C. L. Higginbotham, Eur. Polym. J., 75, 25 (2016).
J. Zhu, Biomaterials, 31, 4639 (2010).
N. R. Patel, A. K. Whitehead, J. J. Newman, and M. E. Caldorera-Moore, ACS Biomater. Sci. Eng., 3, 1494 (2017).
Y. Wang, L. Chen, L. Tan, Q. Zhao, F. Luo, Y. Wei, and Z. Qian, Biomaterials, 35, 6972 (2014).
F. P. Seib, M. Tsurkan, U. Freudenberg, D. L. Kaplan, and C. Werner, ACS Biomater. Sci. Eng., 2, 2287 (2016).
M. Li, H. Li, X. Li, H. Zhu, Z. Xu, L. Liu, J. Ma, and M. Zhang, ACS Appl. Mater. Interfaces, 9, 22160 (2017).
P. Sarika and N. R. James, Int. J. Biol. Macromol., 76, 181 (2015).
V. O. Fasiku, B. A. Aderibigbe, E. R. Sadiku, Y. Lemmer, S. J. Owonubi, S. S. Ray, and E. Mukwevho, Polym. Bull., 76, 5011 (2019).
S. Giray, T. Bal, A. M. Kartal, S. Kızılel, and C. Erkey, J. Biomed. Mater. Res. A, 100, 1307 (2012).
G. Jiang, J. Sun, and F. Ding, J. Biomater. Sci. Polym. Ed., 25, 241 (2014).
B. Singh and A. Dhiman, Curr. Drug Deliv., 14, 981 (2017).
M. Shaikh, M. S. Lonikar, and S. V. Lonikar, Asian J. Res. Chem., 7, 407 (2014).
G.-Q. Huang, L.-Y. Cheng, J.-X. Xiao, S.-Q. Wang, and X.-N. Han, J. Biomater. Appl., 31, 193 (2016).
J. Elisseeff, K. Anseth, D. Sims, W. McIntosh, M. Randolph, M. Yaremchuk, and R. Langer, Plast. Reconstr. Surg., 104, 1014 (1999).
A. C. Karmaker, A. T Dibenedetto, and A. J. Goldberg, J. Mater. Sci., Mater Med., 8, 369 (1997).
S. J. Bryant and K. S. Anseth, J. Biomed. Mater Res. A, 64, 70 (2003).
S. Bashir, Y. Y. Teo, S. Ramesh, and K. Ramesh, Polymer, 92, 36 (2016).
R. A. McBath and D. A. Shipp, Polym. Chem., 1, 860 (2010).
S. Bashir, Y. Y. Teo, S. Ramesh, and K. Ramesh, Polymer, 147, 108 (2018).
F. Zonatto, E. C. Muniz, E. B. Tambourgi, and A. T. Paulino, Int. J. Biol. Macromol., 105, 363 (2017).
X. Bao, X. Si, X. Ding, L. Duan, and C. Xiao, J. Polym. Res., 26, 278 (2019).
D. S. Lee, J. I. Kang, B. H. Hwang, and K. M. Park, Macromol. Res., 27, 205 (2019).
B. Mandal and S. K. Ray, Carbohydr. Polym., 98, 257 (2013).
H. S. Samanta and S. K. Ray, Carbohydr. Polym., 99, 666 (2014).
B. Mandal, A. P. Rameshbabu, S. Dhara, and S. Pal, Polymer, 120, 9 (2017).
S. Pacelli, P. Paolicelli, F. Pepi, S. Garzoli, A. Polini, B. Tita, A. Vitalone, and M. A. Casadei, J. Polym. Res., 21, 409 (2014).
S. Saengthong, T. Piroonpan, T. Tangthong, and W. Pasanphan, Macromol. Res., 22, 717 (2014).
Y.-H. Wu, H. B. Park, T. Kai, B. D. Freeman, and D. S. Kalika, J. Membrane Sci., 347, 197 (2010).
V. A. Kusuma, B. D. Freeman, M. A. Borns, and D. S. Kalika, J. Membrane Sci., 327, 195 (2009).
A. C. Sagle, H. Ju, B. D. Freeman, and M. M. Sharma, Polymer, 50, 756 (2009).
M. A. Ghalia and Y. Dahman, J. Polym. Res., 22, 218 (2015).
M. J. Zohuriaan-Mehr, Z. Motazedi, K. Kabiri, and A. Ershad-Langroudi, J. Macromol. Sci. Pure Appl. Chem., A42, 1655 (2005).
M. J. Zohuriaan-Mehr, Z. Motazedi, K. Kabiri, A. Ershad-Langroudi, and I. Allahdadi, J. Appl. Polym. Sci., 102, 5667 (2006).
P. Stagnaro, I. Schizzi, R. Utzeri, E. Marsano, and M. Castellano, Carbohydr. Polym., 185, 56 (2018).
G. Burke, V. Barron, T. Geever, L. Geever, D. M. Devine, and C. L. Higginbotham, J. Mech. Behav. Biomed. Mater., 99, 1 (2019).
D. S. Seeli and M. Prabaharan, Carbohydr. Polym., 158, 51 (2017).
S. Nesrinne and A. Djamel, Arabian J. Chem., 10, 539 (2017).
D. Selli, S. Motta, and C. Di Valentin, J. Colloid Interface Sci., 555, 519 (2019).
I. M. Barszczewska-Rybarek, Materials, 12, 4057 (2019).
P. R. S. Reddy, K. M. Rao, K. S. V. K. Rao, Y. Shchipunov, and C.-S. Ha, Macromol. Res., 22, 832 (2014).
T. Caykara, S. Küçüktepe, and E. Turan, Polym. Int., 56, 532 (2007).
H. Yi, M. Seong, K. Sun, I. Hwang, K. Lee, C. Cha, T.-i. Kim, and H. E. Jeong, Adv. Funct. Mater., 28, 1706498 (2018).
Y. Zhao, H. Su, L. Fang, and T. Tan, Polymer, 46, 5368 (2005).
S. A. Mahdavi, S. M. Jafari, E. Assadpoor, and D. Dehnad, Int. J. Biol. Macromol., 85, 379 (2016).
X. Hu, L. Feng, A. Xie, W. Wei, S. Wang, J. Zhang, and W. Dong, J. Mater. Chem. B, 2, 3646 (2014).
M. J. Ramazani-Harandi, M. J. Zohuriaan-Mehr, A. A. Yousefi, A. Ershad-Langroudi, and K. Kabiri, J. Appl. Polym. Sci., 113, 3676 (2009).
K. S. Anseth, C. N. Bowman, and L. Brannon-Peppas, Biomaterials, 17, 1647 (1996).
M. J. Moura, M. M. Figueiredo, and M. H. Gil, Biomacromolecules, 8, 3823 (2007).
Y. Li and J. C. Kwak, Langmuir, 20, 4859 (2004).
G. Burke, Z. Cao, D. M. Devine, and I. Major, Polymers, 11, 1339 (2019).
Q. Tan, W. Liu, C. Guo, and G. Zhai, Int. J. Nanomedicine, 6, 1621 (2011).
M. C. I. M. Amin, N. Ahmad, N. Halib, and I. Ahmad, Carbohydr. Polym., 88, 465 (2012).
P. Patel, A. Mandal, V. Gote, D. Pal, and A. K. Mitra, J. Polym. Res., 26, 131 (2019).
P. L. Ritger and N. A. Peppas, J. Control. Release, 5, 37 (1987).
E. P. Holowka and S. K. Bhatia, Smart Drug Delivery Systems, in Drug Delivery, Springer, New York, 2014, pp 265–316.
M. R. Guilherme, A. V. Reis, S. H. Takahashi, A. F. Rubira, J. P. A. Feitosa, and E. C. Muniz, Carbohydr. Polym., 61, 464 (2005).
S. A. Agnihotri and T. M. Aminabhavi, Int. J. Pharm., 324, 103 (2006).
Acknowledgment
The research work was supported by University of Malaya, Malaysia. It was financially supported by RU Grant (ST035-2019).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ibrahim, S.M., Yin, T.Y. & Misran, M. Arabic Gum Grafted PEGDMA Hydrogels: Synthesis, Physico-Chemical Characterization and In-vitro Release of Hydrophobic Drug. Macromol. Res. 28 (Suppl 1), 1220–1231 (2020). https://doi.org/10.1007/s13233-020-8166-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-020-8166-1