Abstract
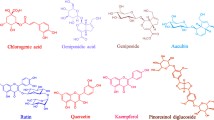
Polysaccharide-rich materials were extracted from the alcohol-insoluble solids of Olea europaea l. **leaves. Structural characteristics were determined by colorimetric techniques, FT-IR, GC–MS, SEC/MALS/VD/DRI, and NMR (1H,13C). The extract and its main macromolecular components were characterized to assess their ability toward antioxidant, α-amylase inhibition, and antiproliferative activities. Results revealed that the ultrasound olive leave extract comprises polysaccharides with uronic acid, galactose, arabinose, and glucose in molar percentages of 11.7%, 11.3%, 7.5%, and 4.9% respectively, constituting 41% of the total mass. In addition, polyphenols (21%) and proteins (9%) are associated with these polysaccharides. Further, the extract showed noticeable ORAC and free radical scavenging abilities, in addition to high in vitro antiproliferative activity against Caco-2 colon carcinoma cell lines. Similarly, the extract exhibited a strong, uncompetitive inhibition of α-amylase by 75% in the presence of the extract with 0.75 µg/mL of concentration. This research concludes that ultrasound extraction method can be used for the extraction of polysaccharide–polyphenol–protein complexes. These conjugates exhibit the potential for combined biological activities resulting from a synergistic effect of its compounds, making them promising ingredients for the development of functional food.






Similar content being viewed by others
References
Acar-Tek N, Aǧagündüz D (2020) Olive Leaf (Olea europaea L. folium): Potential Effects on Glycemia and Lipidemia. Ann Nutr Metab 76:10–15
Ahmad-Qasem MH, Cánovas J, Barrajón-Catalán E, Micol V, Cárcel JA, García-Pérez JV (2013) Kinetic and compositional study of phenolic extraction from olive leaves (var. Serrana) by using power ultrasound. Innovative Food Sci Emerg Technol 17:120–129. https://doi.org/10.1016/j.ifset.2012.11.008
Bermúdez-Oria A, Rodríguez-Gutiérrez G, Alaiz M et al (2019) Polyphenols associated to pectic polysaccharides account for most of the antiproliferative and antioxidant activities in olive extracts. J Funct Foods. https://doi.org/10.1016/j.jff.2019.103530
Bermúdez-Oria A, Rodríguez-Gutiérrez G, Fernández-Prior Á et al (2020) Confirmation by solid-state NMR spectroscopy of a strong complex phenol-dietary fiber with retention of antioxidant activity in vitro. Food Hydrocoll. https://doi.org/10.1016/j.foodhyd.2019.105584
Bermúdez-Oria A, Rodríguez-Juan E, Rodríguez-Gutiérrez G et al (2021) Effect of the olive oil extraction process on the formation of complex pectin–polyphenols and their antioxidant and antiproliferative activities. Antioxidants. https://doi.org/10.3390/antiox10121858
Bernfeld P (1955) Amylases, α and β
Blumenkrantz N, Asboe-Hansen G (1973) New method for quantitative determination of uronic acids. Anal Biochem 54:484–489. https://doi.org/10.1016/0003-2697(73)90377-1
Campos F, Peixoto AF, Fernandes PAR et al (2021) The antidiabetic effect of grape pomace polysaccharide-polyphenol complexes. Nutrients. https://doi.org/10.3390/nu13124495
Chaouch MA, Hafsa J, Rihouey C et al (2016) Effect of extraction conditions on the antioxidant and antiglycation capacity of carbohydrates from Opuntia robusta cladodes. Int J Food Sci Technol 51:929–937. https://doi.org/10.1111/ijfs.13055
Chen XQ, Zhang Y (2007) Ultrasonic-associated extraction of water soluble polysaccharides from defatted Korean pine kernel. J for Res (harbin) 18:133–135. https://doi.org/10.1007/s11676-007-0026-8
Chen Y, Xie M, Li W et al (2012) An effective method for deproteinization of bioactive polysaccharides extracted from lingzhi (Ganoderma atrum). Food Sci Biotechnol 21:191–198. https://doi.org/10.1007/s10068-012-0024-2
Chen W, Lv R, Muhammad AI et al (2019) Fabrication of (-)-epigallocatechin-3-gallate carrier based on glycosylated whey protein isolate obtained by ultrasound Maillard reaction. Ultrason Sonochem. https://doi.org/10.1016/j.ultsonch.2019.104678
Chen X, Wang Z, Kan J (2021) Polysaccharides from ginger stems and leaves: Effects of dual and triple frequency ultrasound assisted extraction on structural characteristics and biological activities. Food Biosci. https://doi.org/10.1016/j.fbio.2021.101166
Dupree R, Simmons TJ, Mortimer JC et al (2015) Probing the molecular architecture of arabidopsis thaliana secondary cell walls using two- and three-dimensional 13C solid state nuclear magnetic resonance spectroscopy. Biochemistry 54:2335–2345. https://doi.org/10.1021/bi501552k
Duque-Soto C, Quirantes-Piné R, Borrás-Linares I et al (2022) Characterization and Influence of Static In Vitro Digestion on Bioaccessibility of Bioactive Polyphenols from an Olive Leaf Extract. Foods. https://doi.org/10.3390/foods11050743
Ebringerová A, Hromádková Z (2010) An overview on the application of ultrasound in extraction, separation and purification of plant polysaccharides. Cent Eur J Chem 8:243–257
El SN, Karakaya S (2009) Olive tree (Olea europaea) leaves: Potential beneficial effects on human health. Nutr Rev 67:632–638. https://doi.org/10.1111/j.1753-4887.2009.00248.x
Encalada AMI, Pérez CD, Flores SK et al (2019) Antioxidant pectin enriched fractions obtained from discarded carrots (Daucus carota L.) by ultrasound-enzyme assisted extraction. Food Chem 289:453–460. https://doi.org/10.1016/j.foodchem.2019.03.078
Englyst HN, Cummings JH (1984) Simplified method for the measurement of total non-starch polysaccharides by gas - liquid chromatography of constituent sugars as alditol acetates. Analyst 109:937–942. https://doi.org/10.1039/an9840900937
Espeso J, Isaza A, Lee JY, et al (2021) Olive leaf waste management. Front Sustain Food Syst 5:660582. https://doi.org/10.3389/fsufs.2021.660582
Fernandes PAR, Le Bourvellec C, Renard CMGC et al (2020a) Interactions of arabinan-rich pectic polysaccharides with polyphenols. Carbohydr Polym. https://doi.org/10.1016/j.carbpol.2019.115644
Fincher GB, Stone BA, Clarke AE (1983) Arabinogalactan-Proteins: Structure, Biosynthesis, and Function. Annu Rev Plant Physiol 34:47–70. https://doi.org/10.1146/annurev.pp.34.060183.000403
Galanakis CM (2021) Functionality of food components and emerging technologies. Foods 10(1):128. https://doi.org/10.3390/foods10010128
Gibson RB (1904) The determination of nitrogen by the kjeldahl method. J Am Chem Soc 26:105–110. https://doi.org/10.1021/ja01991a014
Guo MQ, Hu X, Wang C, Ai L (2017) Polysaccharides: structure and solubility. Solubility Polysaccharides 2:8–21. https://doi.org/10.5772/intechopen.70137
Guo Q, Xiao X, Lu L, Ai L, Xu M, Liu Y, Goff HD (2022) Polyphenol–polysaccharide complex: preparation, characterization, and potential utilization in food and health. Ann Rev Food Sci Technol 13:59–87. https://doi.org/10.1146/annurev-food-052720-010354
Haslam E, Lilley TH (1986) Interactions of natural phenols with macromolecules. Prog Clin Biol Res 213:53–65
Huang D, Ou B, Hampsch-Woodill M et al (2002) High-throughput assay of oxygen radical absorbance capacity (ORAC) using a multichannel liquid handling system coupled with a microplate fluorescence reader in 96-well format. J Agric Food Chem 50:4437–4444. https://doi.org/10.1021/jf0201529
Jilani H, Cilla A, Barberá R, Hamdi M (2020) Antiproliferative activity of green, black tea and olive leaves polyphenols subjected to biosorption and in vitro gastrointestinal digestion in Caco-2 cells. Food Res Int. https://doi.org/10.1016/j.foodres.2020.109317
Jun XP, Xin HL, Hong ZC, Lei ZY (2015) Phenolic compositions, and antioxidant performance of olive leaf and fruit (Olea europaea L.) extracts and their structure-activity relationships. J Funct Foods 16:460–471. https://doi.org/10.1016/j.jff.2015.05.005
Khemakhem I, Abdelhedi O, Trigui I et al (2018) Structural, antioxidant and antibacterial activities of polysaccharides extracted from olive leaves. Int J Biol Macromol 106:425–432. https://doi.org/10.1016/j.ijbiomac.2017.08.037
Lama-Muñoz A, Rodríguez-Gutiérrez G, Rubio-Senent F, Fernández-Bolaños J (2012) Production, characterization and isolation of neutral and pectic oligosaccharides with low molecular weights from olive by-products thermally treated. Food Hydrocoll 28:92–104. https://doi.org/10.1016/j.foodhyd.2011.11.008
Li BW, Cardozo MS (1994) Determination of total dietary fiber in foods and products with little or no starch, nonenzymatic-gravimetric method: collaborative study. J AOAC Int 77:687–689. https://doi.org/10.1093/jaoac/77.3.687
Li N, Wang C, Georgiev MI et al (2021) Advances in dietary polysaccharides as anticancer agents: Structure-activity relationship. Trends Food Sci Technol 111:360–377
Liu F, Ma C, Gao Y, McClements DJ (2017) Food-Grade Covalent Complexes and Their Application as Nutraceutical Delivery Systems: A Review. Compr Rev Food Sci Food Saf 16:76–95. https://doi.org/10.1111/1541-4337.12229
Liu J, Bai R, Liu Y et al (2018) Isolation, structural characterization and bioactivities of naturally occurring polysaccharide–polyphenolic conjugates from medicinal plants—A reivew. Int J Biol Macromol 107:2242–2250. https://doi.org/10.1016/j.ijbiomac.2017.10.097
Liu B, Liu J, Huang D et al (2020) Separation and purification of hydroxytysol and oleuropein from Olea europaea L. (olive) leaves using macroporous resins and a novel solvent system. J Sep Sci 43:2619–2625. https://doi.org/10.1002/jssc.201901227
Liu H, Xu J, Xu X et al (2021) Structure/function relationships of bean polysaccharides: A review. Crit Rev Food Sci Nutr. https://doi.org/10.1080/10408398.2021.1946480
Martín García AI, Moumen A, Yáñez Ruiz DR, Molina Alcaide E (2003) Chemical composition and nutrients availability for goats and sheep of two-stage olive cake and olive leaves. Anim Feed Sci Technol 107:61–74. https://doi.org/10.1016/S0377-8401(03)00066-X
Mehrnia MA, Barzegar H, Hagh L (2020) Optimization of polysaccharide extraction from olive leaves and evaluation of its antioxidant and rheological properties. Ir Food Sci Technol Res J 15:133–144
Miao M, Jiang H, Jiang B et al (2013) Elucidation of structural difference in theaflavins for modulation of starch digestion. J Funct Foods 5:2024–2029. https://doi.org/10.1016/j.jff.2013.09.021
Ortega MLS, Orellana-Palacios JC, Garcia SR et al (2024) Olive leaf protein: Extraction optimization, in vitro digestibility, structural and techno-functional properties. Int J Biol Macromol. https://doi.org/10.1016/j.ijbiomac.2023.128273
Pawlaczyk-Graja I, Balicki S, Wilk KA (2019) Effect of various extraction methods on the structure of polyphenolic-polysaccharide conjugates from Fragaria vesca L. leaf. Int J Biol Macromol 130:664–674. https://doi.org/10.1016/j.ijbiomac.2019.03.013
Paz A, Solisio C, Converti A, Casazza AA (2023) Application of organosolv technology to improve the anaerobic digestion of olive oil pomace. Ind Crops Prod. https://doi.org/10.1016/j.indcrop.2023.117249
Pham D, Nguyen H, Nguyen T Le, et al (2020) Optimization of ultrasound-assisted extraction of flavonoids from celastrus hindsii leaves using response surface methodology and evaluation of their antioxidant and antitumor activities. BioMed Res Int 2020. https://doi.org/10.1155/2020/3497107
Plantago L, Yin J, Lin H et al (2012) Structural characterization of a highly branched polysaccharide from the seeds of. Carbohydr Polym 87:2416–2424. https://doi.org/10.1016/j.carbpol.2011.11.009
Renard CMGC, Watrelot AA, Le Bourvellec C (2017a) Interactions between polyphenols and polysaccharides: Mechanisms and consequences in food processing and digestion. Trends Food Sci Technol 60:43–51
Renard CMGC, Watrelot AA, Bourvellec-samour C Le (2015) Interactions between polyphenols and polysaccharides : mechanisms and consequences. 29th EFFOST conference, Nov 2015, Athenes, Greece
Renard CMGC (2005) Variability in cell wall preparations : quantification and comparison of common methods. Carbohydr Polym 60(4):515–522. https://doi.org/10.1016/j.carbpol.2005.03.002
Rodr G, Guill R, Fern J, Jim A (2007) Antioxidant activity of effluents during the purification of hydroxytyrosol and 3, 4-dihydroxyphenyl glycol from olive oil waste. Eur Food Res Technol 224:733–741. https://doi.org/10.1007/s00217-006-0366-1
Rubio-Senent F, Rodríguez-Gutiérrez G, Lama-Muñoz A et al (2015) Novel pectin present in new olive mill wastewater with similar emulsifying and better biological properties than citrus pectin. Food Hydrocoll 50:237–246. https://doi.org/10.1016/j.foodhyd.2015.03.030
Selim S, Albqmi M, Al-Sanea MM, et al (2022) Valorizing the usage of olive leaves, bioactive compounds, biological activities, and food applications: A comprehensive review. Front Nutr 9:1008349. https://doi.org/10.3389/fnut.2022.1008349
Singleton VL, Rossi JA (1965) Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am J Enol Vitic 16:144–158
Soler-Rivas C, Espiń JC, Wichers HJ (2000) Oleuropein and related compounds. J Sci Food Agric 80:1013–1023
Sun L, Warren FJ, Gidley MJ (2018) Soluble polysaccharides reduce binding and inhibitory activity of tea polyphenols against porcine pancreatic α-amylase. Food Hydrocoll 79:63–70. https://doi.org/10.1016/j.foodhyd.2017.12.011
Surin S, You SG, Seesuriyachan P et al (2020) Optimization of ultrasonic-assisted extraction of polysaccharides from purple glutinous rice bran (Oryza sativa L.) and their antioxidant activities. Sci Rep 10:1–10. https://doi.org/10.1038/s41598-020-67266-1
Šutovská M, Capek P, Kocmálová M et al (2013) Characterization and biological activity of Solidago canadensis complex. Int J Biol Macromol 52:192–197. https://doi.org/10.1016/j.ijbiomac.2012.09.021
Talhaoui N, Taamalli A, Mar A (2015) PT NU. FRIN. https://doi.org/10.1016/j.foodres.2015.09.011
Teka N, Lazreg H, Horchani M et al (2022) Characterization, α - Amylase Inhibition and In Silico Docking Study of Polysaccharides Extracted from Rosy Garlic ( Allium roseum ) Bulbs. Chemistry Africa 5:1997–2009. https://doi.org/10.1007/s42250-022-00497-z
Varner JE, Lin LS (1989) Plant cell wall architecture. Cell 56:231–239. https://doi.org/10.1016/0092-8674(89)90896-9
Vioque J, Girón-Calle J, Torres-Salas V et al (2020) Characterization of Vicia ervilia (bitter vetch) seed proteins, free amino acids, and polyphenols. J Food Biochem 44:e13271. https://doi.org/10.1111/jfbc.13271
Wu Z, Ming J, Gao R et al (2011) Characterization and antioxidant activity of the complex of tea polyphenols and oat β-glucan. J Agric Food Chem 59:10737–10746. https://doi.org/10.1021/jf202722w
Wu DT, Liu W, Xian ML et al (2020) Polyphenolic-protein-polysaccharide complexes from hovenia dulcis: Insights into extraction methods on their physicochemical properties and in vitro bioactivities. Foods. https://doi.org/10.3390/foods9040456
Wu D-T, He Y, Fu M-X, et al (2021) Changes in physicochemical and biological properties of polyphenolic-protein-polysaccharide ternary complexes from hovenia dulcis after in vitro simulated saliva-gastrointestinal digestion. Foods 10(10):2322. https://doi.org/10.3390/foods
Yi Y, Tang HS, Sun Y et al (2022) Comprehensive characterization of lotus root polysaccharide-phenol complexes. Food Chem. https://doi.org/10.1016/j.foodchem.2021.130693
Zeng H, Zhang Y, Lin S et al (2015) Ultrasonic-microwave synergistic extraction (UMSE) and molecular weight distribution of polysaccharides from Fortunella margarita (Lour.) Swingle. Sep Purif Technol 144:97–106. https://doi.org/10.1016/j.seppur.2015.02.015
Zimm BH (1948) The scattering of light and the radial distribution function of high polymer solutions. J Chem Phys 16(12):1093–1099. https://doi.org/10.1063/1.1746738
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors solemnly vow that there are no competing financial interests in this work.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Elayeb, R., Bermúdez-Oria, A., Lazreg Aref, H. et al. Antioxidant polysaccharide-enriched fractions obtained from olive leaves by ultrasound-assisted extraction with α-amylase inhibition, and antiproliferative activities. 3 Biotech 14, 92 (2024). https://doi.org/10.1007/s13205-024-03939-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-024-03939-2