Abstract
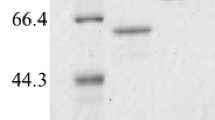
Maltooligosaccharide-forming amylases (MFAses) are promising enzymes for a variety of industrial applications. In this study, a maltooligosaccharide-forming amylase (BkAmy) isolated from Bacillus koreensis HL12 was first heterologous expressed and characterized. According to structural-sequence alignment, BkAmy contained seven conserved regions which are the signature of a novel GH13 subfamily. The gene was expressed in Pichia pastoris KM71 as an extracellular protein with a volumetric activity of 3.38 U/mL culture medium after 72 h induction by 3% (w/v) of methanol. The recombinant BkAmy migrated as a single protein band with an expected size approximately of 55 kDa. BkAmy exhibited the highest catalytic activity on soluble starch with a specific activity of 42.2 U/mg at 40 °C, pH 7.0. The enzyme exhibited 65% relative activity at 30 °C, indicating its advantage on application at moderate reaction temperature desirable for energy saving and reduction of side unwanted reactions. The enzyme exhibited a specific cleavage pattern by releasing maltose (G2), maltotriose (G3) and maltotetraose (G4) from cassava starch with the highest yield of 363 mg/g substrate equivalent to 36% conversion using 40 U/g substrate at 60 min. The work demonstrates the potential of this enzyme on maltooligosaccharide production from starch to create high value-added products in starch processing industries.







Similar content being viewed by others
References
Ben Ali M, Mhiri S, Mezghani M, Bejar S (2001) Purification and sequence analysis of the atypical maltohexaose-forming α-amylase of the B. stearothermophilus US100. Enzyme Microb Technol 28(6):537–542. https://doi.org/10.1016/s0141-0229(01)00294-0
Benson DA, Clark K, Karsch-Mizrachi I, Lipman DJ, Ostell J, Sayers EW (2014) GenBank. Nucleic Acids Res 42(Database issue):D32–D37. https://doi.org/10.1093/nar/gkt1030
Berman HM, Westbrook J, Feng Z, Gilliland G, Bhat TN, Weissig H, Shindyalov IN, Bourne PE (2000) The Protein Data Bank. Nucleic Acids Res 28(1):235–242. https://doi.org/10.1093/nar/28.1.235
Bertoldo C, Antranikian G (2002) Starch-hydrolyzing enzymes from thermophilic archaea and bacteria. Curr Opin Chem Biol 6(2):151–160. https://doi.org/10.1016/s1367-5931(02)00311-3
Cantarel BL, Coutinho PM, Rancurel C, Bernard T, Lombard V, Henrissat B (2009) The carbohydrate-active enzymes database (CAZy): an expert resource for glycogenomics. Nucleic Acids Res 37(Database issue):D233–D238. https://doi.org/10.1093/nar/gkn663
Cavicchioli R, Charlton T, Ertan H, Mohd Omar S, Siddiqui KS, Williams TJ (2011) Biotechnological uses of enzymes from psychrophiles. Microb Biotechnol 4(4):449–460. https://doi.org/10.1111/j.1751-7915.2011.00258.x
Chai KP, Othman NFB, The AH, Ho KL, Chan KG, Shamsir MS, Goh KM, Ng CL (2016) Crystal structure of Anoxybacillus α-amylase provides insights into maltose binding of a new glycosyl hydrolase subclass. Sci Rep 6(1):23126. https://doi.org/10.1038/srep23126
Cihan AC, Yildiz ED, Sahin E, Mutlu O (2018) Introduction of novel thermostable α-amylases from genus Anoxybacillus and proposing to group the Bacillaceae related α-amylases under five individual GH13 subfamilies. World J Microbiol Biotechnol 34(7):95. https://doi.org/10.1007/s11274-018-2478-8
Crooks GE, Hon G, Chandonia JM, Brenner SE (2004) WebLogo: a sequence logo generator. Genome Res 14(6):1188–1190. https://doi.org/10.1101/gr.849004
Dey G, Palit S, Banerjee R, Maiti BR (2002) Purification and characterization of maltooligosaccharide-forming amylase from Bacillus circulans GRS 313. J Ind Microbiol Biotechnol 28(4):193–200. https://doi.org/10.1038/sj/jim/7000220
Doukyu N, Yamagishi W, Kuwahara H, Ogino H, Furuki N (2007) Purification and characterization of a maltooligosaccharide-forming amylase that improves product selectivity in water-miscible organic solvents, from dimethylsulfoxide-tolerant Brachybacterium sp. strain LB25. Extremophiles 11(6):781–788. https://doi.org/10.1007/s00792-007-0096-8
Drula E, Garron ML, Dogan S, Lombard V, Henrissat B, Terrapon N (2022) The carbohydrate-active enzyme database: functions and literature. Nucleic Acids Res 50(D1):D571–D577. https://doi.org/10.1093/nar/gkab1045
Gerday C, Aittaleb M, Bentahir M, Chessa JP, Claverie P, Collins T, D’Amico S, Dumont J, Garsoux G, Georlette D, Hoyoux A, Lonhienne T, Meuwis MA, Feller G (2000) Cold-adapted enzymes: from fundamentals to biotechnology. Trends Biotechnol 18(3):103–107. https://doi.org/10.1016/S0167-7799(99)01413-4
Gupta R, Gigras P, Mohapatra H, Goswami VK, Chauhan B (2003) Microbial α-amylases: a biotechnological perspective. Process Biochem 38(11):1599–1616. https://doi.org/10.1016/S0032-9592(03)00053-0
Hamid B, Mohiddin FA (2018) Cold-active enzymes in food processing. Enzymes Food Technol. https://doi.org/10.1007/978-981-13-1933-4_19
Hashim SO, Delgado OD, Martínez MA, Kaul RH, Mulaa FJ, Mattiasson B (2005) Alkaline active maltohexaose-forming α-amylase from Bacillus halodurans LBK 34. Enzyme Microb Technol 36(1):139–146. https://doi.org/10.1016/j.enzmictec.2004.07.017
Hatada Y, Masuda N, Akita M, Miyazaki M, Ohta Y, Horikoshi K (2006) Oxidatively stable maltopentaose-producing α-amylase from a deep-sea Bacillus isolate, and mechanism of its oxidative stability validated by site-directed mutagenesis. Enzyme Microb Technol 39(6):1333–1340. https://doi.org/10.1016/j.enzmictec.2006.03.022
Hmidet N, Maalej H, Haddar A, Nasri M (2010) A novel α-amylase from Bacillus mojavensis A21: purification and biochemical characterization. Appl Biochem Biotechnol 162(4):1018–1030. https://doi.org/10.1007/s12010-009-8902-7
Jana M, Maity C, Samanta S, Pati BR, Islam SS, Mohapatra PKD, Mondal KC (2013) Salt-independent thermophilic α-amylase from Bacillus megaterium VUMB109: an efficacy testing for preparation of maltooligosaccharides. Ind Crops Prod 41:386–391. https://doi.org/10.1016/j.indcrop.2012.04.048
Janeček Š, Svensson B (2022) How many α-amylase GH families are there in the CAZy database? Amylase 6:1–10. https://doi.org/10.1515/amylase-2022-0001
Janeček Š, Zámocká B (2020) A new GH13 subfamily represented by the α-amylase from the halophilic archaeon Haloarcula hispanica. Extremophiles 24(2):207–217. https://doi.org/10.1007/s00792-019-01147-y
Janeček Š, Kuchtová A, Petrovičová S (2015) A novel GH13 subfamily of α-amylases with a pair of tryptophans in the helix α3 of the catalytic TIM-barrel, the LPDlx signature in the conserved sequence region V and a conserved aromatic motif at the C-terminus. Biologia 70(10):1284–1294. https://doi.org/10.1515/biolog-2015-0165
Jang EY, Ahn Y, Suh HJ, Hong KB, Jo K (2020) Amylase-producing maltooligosaccharide provides potential relief in rats with loperamide-induced constipation. Evid Based Complement Alternat Med 2020:5470268. https://doi.org/10.1155/2020/5470268
Kamon M, Sumitani JI, Tani S, Kawaguchi T (2015) Characterization and gene cloning of a maltotriose-forming exo-amylase from Kitasatospora sp. MK-1785. Appl Microbiol Biotechnol 99(11):4743–4753. https://doi.org/10.1007/s00253-015-6396-5
Kelley LA, Mezulis S, Yates CM, Wass MN, Sternberg MJE (2015) The Phyre2 web portal for protein modeling, prediction and analysis. Nat Protoc 10(6):845–858. https://doi.org/10.1038/nprot.2015.053
Kumar S, Khare SK (2012) Purification and characterization of maltooligosaccharide-forming α-amylase from moderately halophilic Marinobacter sp. EMB8. Bioresour Technol 116:247–251. https://doi.org/10.1016/j.biortech.2011.11.109
Lee YS, Park DJ, Choi YL (2015) Characterization of maltotriose production by hydrolyzing of soluble starch with α-amylase from Microbulbifer thermotolerans DAU221. Appl Microbiol Biotechnol 99(9):3901–3911. https://doi.org/10.1007/s00253-014-6186-5
Li Z, Wu J, Zhang B, Wang F, Ye X, Huang Y, Huang Q, Cui Z (2015) AmyM, a novel maltohexaose-forming α-amylase from Corallococcus sp. strain EGB. Appl Environ Microbiol 81(6):1977–1987. https://doi.org/10.1128/aem.03934-14
Lim JM, Jeon CO, Lee JC, Ju YJ, Park DJ, Kim CJ (2006) Bacillus koreensis sp. nov., a spore-forming bacterium, isolated from the rhizosphere of willow roots in Korea. Int J Syst Evol Microbiol 56(1):59–63. https://doi.org/10.1099/ijs.0.63701-0
Lin L, Zhang L, Cai X, Liu Q, Zhang C, Wei C (2018) The relationship between enzyme hydrolysis and the components of rice starches with the same genetic background and amylopectin structure but different amylose contents. Food Hydrocoll 84:406–413. https://doi.org/10.1016/j.foodhyd.2018.06.029
Maalej H, Ben Ayed H, Ghorbel-Bellaaj O, Nasri M, Hmidet N (2014) Production and biochemical characterization of a high maltotetraose (g4) producing amylase from Pseudomonas stutzeri AS22. Biomed Res Int 2014:156438. https://doi.org/10.1155/2014/156438
Messaoud EB, Ali MB, Elleuch N, Masmoudi NF, Bejar S (2004) Purification and properties of a maltoheptaose- and maltohexaose-forming amylase produced by Bacillus subtilis US116. Enzyme Microb Technol 34(7):662–666. https://doi.org/10.1016/j.enzmictec.2004.03.002
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31(3):426–428. https://doi.org/10.1021/ac60147a030
Mok SC, Teh AH, Saito JA, Najimudin N, Alam M (2013) Crystal structure of a compact alpha-amylase from Geobacillus thermoleovorans. Enzyme Microb Technol 53(1):46–54. https://doi.org/10.1016/j.enzmictec.2013.03.009
Murakami S, Nagasaki K, Nishimoto H, Shigematu R, Umesaki J, Takenaka S, Kaulpiboon J, Prousoontorn M, Limpaseni T, Pongsawasdi P, Aoki K (2008) Purification and characterization of five alkaline, thermotolerant, and maltotetraose-producing α-amylases from Bacillus halodurans MS-2-5, and production of recombinant enzymes in Escherichia coli. Enzyme Microb Technol 43(4):321–328. https://doi.org/10.1016/j.enzmictec.2008.05.006
Nagarajan DR, Rajagopalan G, Krishnan C (2006) Purification and characterization of a maltooligosaccharide-forming α-amylase from a new Bacillus subtilis KCC103. Appl Microbiol Biotechnol 73(3):591–597. https://doi.org/10.1007/s00253-006-0513-4
Nandanwar S, Borkar S, Lee J, Kim HJ (2020) Taking advantage of promiscuity of cold-active enzymes. Appl Sci 10:8128. https://doi.org/10.3390/app10228128
Nielsen H (2017) Predicting secretory proteins with SignalP. Protein function prediction: methods and protocols. Springer, New York, pp 59–73. https://doi.org/10.1007/978-1-4939-7015-5_6
Ogunsona E, Ojogbo E, Mekonnen T (2018) Advanced material applications of starch and its derivatives. Eur Polym J 108:570–581. https://doi.org/10.1016/j.eurpolymj.2018.09.039
Ozturk U, Denizci A, Ogan A, Kazan D (2014) A maltooligosaccharides producing α-amylase from Bacillus subtilis SDP1 isolated from rhizosphere of Acacia cyanophylla Lindley. Food Biotechnol 28(4):309–332. https://doi.org/10.1080/08905436.2014.963600
Pan S, Ding N, Ren J, Gu Z, Li C, Hong Y, Cheng L, Holler TP, Li Z (2017) Maltooligosaccharide-forming amylase: characteristics, preparation, and application. Biotechnol Adv 35(5):619–632. https://doi.org/10.1016/j.biotechadv.2017.04.004
Parashar D, Satyanarayana T (2017) Engineering a chimeric acid-stable α-amylase-glucoamylase (Amy-Glu) for one step starch saccharification. Int J Biol Macromol 99:274–281. https://doi.org/10.1016/j.ijbiomac.2017.02.083
Park JT, Suwanto A, Tan I, Nuryanto T, Lukman R, Wang K, Jane JL (2014) Molecular cloning and characterization of a thermostable α-amylase exhibiting an unusually high activity. Food Sci Biotechnol 23(1):125–132. https://doi.org/10.1007/s10068-014-0017-4
Pettersen EF, Goddard TD, Huang CC, Couch GS, Greenblatt DM, Meng EC, Ferrin TE (2004) UCSF Chimera—a visualization system for exploratory research and analysis. J Comput Chem 25(13):1605–1612. https://doi.org/10.1002/jcc.20084
Puspasari F, Radjasa OK, Noer AS, Nurachman Z, Syah YM, van der Maarel M, Dijkhuizen L, Janeček Š, Natalia D (2013) Raw starch-degrading alpha-amylase from Bacillus aquimaris MKSC 6.2: isolation and expression of the gene, bioinformatics and biochemical characterization of the recombinant enzyme. J Appl Microbiol 114(1):108–120. https://doi.org/10.1111/jam.12025
Rahmani N, Rohana R, Sukarno S, Andriani ADE, Yopi Y (2013) Production of maltooligosaccharides from black potato (Coleus tuberosus) starch by α-amylase from a marine bacterium (Brevibacterium sp.). Microbiol Indones 7(3):129–136. https://doi.org/10.5454/mi.7.3.6
Ranjani V, Janeček Š, Chai KP, Shahir S, Rahman RNZRA, Chan KG, Goh KM (2014) Protein engineering of selected residues from conserved sequence regions of a novel Anoxybacillus α-amylase. Sci Rep 4(1):5850. https://doi.org/10.1038/srep05850
Robert X, Gouet P (2014) Deciphering key features in protein structures with the new ENDscript server. Nucleic Acids Res 42(W1):W320–W324. https://doi.org/10.1093/nar/gku316
Sakac N, Karnas M, Dobsa J, Jozanovic M, Gvozdic V, Kovac-Andric E, Krasevac Sakac M, Sarkanj B (2020) Application of spectrophotometric fingerprint in cluster analysis for starch origin determination. Food Technol Biotechnol 58(1):5–11. https://doi.org/10.17113/ftb.58.01.20.6239
Sarian FD, Janecek S, Pijning T, Ihsanawati NZ, Radjasa OK, Dijkhuizen L, Natalia D, van der Maarel MJ (2017) A new group of glycoside hydrolase family 13 alpha-amylases with an aberrant catalytic triad. Sci Rep 7(1):44230. https://doi.org/10.1038/srep44230
Schwede T, Kopp J, Guex N, Peitsch MC (2003) SWISS-MODEL: an automated protein homology-modeling server. Nucleic Acids Res 31(13):3381–3385. https://doi.org/10.1093/nar/gkg520
Stam MR, Danchin JEG, Rancurel C, Coutinho PM, Henrissat B (2006) Dividing the large glycoside hydrolase family 13 into subfamilies: towards improved functional annotations of α-amylase-related proteins. Protein Eng Des Sel 19(12):555–562. https://doi.org/10.1093/protein/gzl044
Tamura K, Stecher G, Kumar S (2021) MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol 38(7):3022–3027. https://doi.org/10.1093/molbev/msab120
Tester RF, Karkalas J, Qi X (2004) Starch structure and digestibility enzyme-substrate relationship. Worlds Poult Sci J 60(2):186–195. https://doi.org/10.1079/WPS200312
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22(22):4673–4680. https://doi.org/10.1093/nar/22.22.4673
UniProt Consortium T (2018) UniProt: the universal protein knowledgebase. Nucleic Acids Res 46(5):2699. https://doi.org/10.1093/nar/gky092
Wang Y, Pan S, Jiang Z, Liu S, Feng Y, Gu Z, Li C, Li Z (2019) A novel maltooligosaccharide-forming amylase from Bacillus stearothermophilus. Food Biosci 30:100415. https://doi.org/10.1016/j.fbio.2019.100415
Waterhouse A, Bertoni M, Bienert S, Studer G, Tauriello G, Gumienny R, Heer FT, de Beer TAP, Rempfer C, Bordoli L, Lepore R, Schwede T (2018) SWISS-MODEL: homology modelling of protein structures and complexes. Nucleic Acids Res 46(W1):W296–W303. https://doi.org/10.1093/nar/gky427
Yang CH, Liu WH (2007) Cloning and characterization of a maltotriose-producing α-amylase gene from Thermobifida fusca. J Ind Microbiol Biotechnol 34(4):325–330. https://doi.org/10.1007/s10295-006-0200-6
Acknowledgements
The authors acknowledge the support provided by the Department of Biotechnology, Faculty of Science and Technology, Thammasat University. The authors are grateful to Natinee Junmat for soil sample collection. Daran Prongjit was supported by the Thailand Graduate Institute of Science and Technology (TGIST) (Grant number: SCA-CO-2563-12209-TH), National Science and Technology Development Agency.
Funding
This study was financially supported by Thammasat University Research Fund (Contract no. TUFT 042/2563). This study was also financially supported by the National Science and Technology Development Agency (Grant no. P18-52705).
Author information
Authors and Affiliations
Contributions
HL, BB and VC conceived and designed the work. HL, BB, NP and DP performed the experiments, and analyzed the data. HL and BB drafted the manuscript. BB and VC critically revised the manuscript for intellectual content. All authors have read and agree to the submission of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest with the contents of this article.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lekakarn, H., Bunterngsook, B., Pajongpakdeekul, N. et al. A novel low temperature active maltooligosaccharides-forming amylase from Bacillus koreensis HL12 as biocatalyst for maltooligosaccharide production. 3 Biotech 12, 134 (2022). https://doi.org/10.1007/s13205-022-03188-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-022-03188-1