Abstract
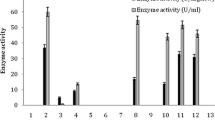
The present study entails the usefulness of thermophilic amidase-producing bacterium in the biotransformation of benzamide to benzohydroxamic acid (BHA). A bacterium Bacillus smithii IIIMB2907 was isolated from a soil sample collected from hot springs of Manikaran, Himachal Pradesh, India. The whole cells of the bacterium displayed versatile substrate specificity by exhibiting significant activity with a diverse range of amides. In addition, amidase from thermophilic bacterium was induced by adding Ɛ-caprolactam in the mineral base media. The optimum temperature and pH of acyltransferase activity of amidase enzyme were found to be 50 °C and 7.0, respectively. Interestingly, half-life (t1/2) of this enzyme was 17.37 h at 50 °C. Bench-scale production and purification of BHA was carried out at optimized conditions which resulted in the recovery of 64% BHA with a purity of 96%. Owing to this, the reported process in the present study can be considered of immense industrial significance for the production of BHA.








Similar content being viewed by others
Availability of data and materials
Not applicable.
Code availability
Not applicable.
References
Agarwal S, Choudhury B (2014) Presence of multiple acyltransferases with diverse substrate specificity in Bacillus smithii strain IITR6b2 and characterization of unique acyltransferase with nicotinamide. J Mol Catal B Enzymatic 107:64–72. https://doi.org/10.1016/j.molcatb.2014.05.017
Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402. https://doi.org/10.1093/nar/25.17.3389
Babu V, Choudhury B (2013) Methods of cell lysis and effect of detergents for the recovery of nitrile metabolizing enzyme from Amycolatopsis sp. IITR215. J Gen Engg Biotechnol 11:117–122. https://doi.org/10.1016/j.jgeb.2013.05.002
Babu V, Shilpi, Choudhury B (2010) Nitrile-metabolizing potential of Amycolatopsis sp. IITR215. Process Biochem 45:866–873. https://doi.org/10.1016/j.procbio.2010.02.008
Bertrand S, Helesbeux JJ, Larcher G (2013) Hydroxamate, a key pharmacophore exhibiting a wide range of biological activities. Mini-Rev Med Chem 13:1311–1326. https://doi.org/10.2174/13895575113139990007
Bhalla TC, Kumar V, Kumar V, Thakur N, Savitri (2018) Nitrile metabolizing enzymes in biocatalysis and biotransformation. Appl Biochem Biotechnol 185:925–946. https://doi.org/10.1007/s12010-018-2705-7
Bhatia RK, Bhatia SK, Mehta PK (2013a) Bench scale production of benzohydroxamic acid using acyl transfer activity of amidase from Alcaligenes sp. MTCC 10674. J Ind Microbiol Biotechnol 40:21–27. https://doi.org/10.1007/s10295-012-1206-x
Bhatia SK, Mehta PK, Bhatia RK (2013b) An isobutyronitrile-induced bienzymatic system of Alcaligenes sp. MTCC 10674 and its application in the synthesis of α-hydroxyisobutyric acid. Bioprocess Biosys Eng 36:613–625. https://doi.org/10.1007/s00449-012-0817-y
Cha M, Chambliss GH (2011) Characterization of acrylamidase isolated from a newly isolated acrylamide-utilizing bacterium, Ralstonia eutropha AUM-01. Curr Microbiol 62:671–678. https://doi.org/10.1007/s00284-010-9761-8
Dhagat S, Jujjavarapu S (2020) Isolation of a novel thermophilic bacterium capable of producing high-yield bioemulsifier and its kinetic modelling aspects along with proposed metabolic pathway. Braz J Microbiol 51:135–143. https://doi.org/10.1007/s42770-020-00228-x
Fournand D, Bigey F, Arnaud A (1998) Acyl transfer activity of an amidase from Rhodococcus sp. strain R312: formation of a wide range of hydroxamic acids. Appl Environ Microbiol 64:2844–2852. https://doi.org/10.1128/AEM.64.8.2844-2852.1998
Galmes MA, García-Junceda E, Swiderek K, Moliner V (2020) Exploring the origin of amidase substrate promiscuity in CALB by a computational approach. ACS Catal 10:1938–1946. https://doi.org/10.1021/acscatal.9b04002
Guan Cj, Ji Yj, Hu Jl et al (2017) Biotransformation of rutin using crude enzyme from Rhodopseudomonas palustris. Curr Microbiol 74:431–436. https://doi.org/10.1007/s00284-017-1204-3
Kumari P, Chand D (2017) Immobilization of whole resting cell of Bacillus sp. APB-6 exhibiting amido-transferase activity on sodium alginate beads and its comparative study with whole resting cells. J Innov Pharm Biol Sci 4:121–127
Makhongela HS, Glowacka AE, Agarkar VB (2007) A novel thermostable nitrilase superfamily amidase from Geobacillus pallidus showing acyl transfer activity. Appl Microbiol Biotechnol 75:801–811. https://doi.org/10.1007/s00253-007-0883-2
Mehta PK, Bhatia SK, Bhatia RK, Bhalla TC (2016) Enhanced production of thermostable amidase from Geobacillus subterraneus RL-2a MTCC 11502 via optimization of physicochemical parameters using Taguchi DOE methodology. 3 Biotech 6:66. https://doi.org/10.1007/s13205-016-0390-1
Pandey D, Patel SKS, Singh R, Kumar P, Thakur V, Chand D (2019) Solvent-tolerant acyltransferase from Bacillus sp. APB-6: purification and characterization. Indian J Microbiol 59:500–507. https://doi.org/10.1007/s12088-019-00836-8
Ruan L, Zheng R, Zheng Y (2016) A novel amidase from Brevibacterium epidermidis ZJB-07021: gene cloning, refolding and application in butyrylhydroxamic acid synthesis. J Ind Microbiol Biotechnol 43:1071–1083. https://doi.org/10.1007/s10295-016-1786-y
Sharma M, Sharma NN, Bhalla TC (2012) Biotransformation of acetamide to acetohydroxamic acid at bench scale using acyl transferase activity of amidase of Geobacillus pallidus BTP-5x MTCC 9225. Indian J Microbiol 52:76–82. https://doi.org/10.1007/s12088-011-0211-5
Sharma H, Singh RV, Raina C, Babu V (2018) Amide hydrolyzing potential of amidase from halotolerant bacterium Brevibacterium sp. IIIMB2706. Biocatal Biotransfor 37:59–65. https://doi.org/10.1080/10242422.2018.1494733
Singh R, Sharma H, Koul A, Babu V (2018) Exploring a broad spectrum nitrilase from moderately halophilic bacterium Halomonas sp. IIIMB2797 isolated from saline lake. J Basic Microbiol 58:867–874. https://doi.org/10.1002/jobm.201800168
Singh RV, Sharma H, Gupta P, Kumar A, Babu V (2019) Green synthesis of acetohydroxamic acid by thermophilic amidase of Bacillus smithii IIIMB2907. Indian J Biochem Bio 56:373–377
Sogani M, Mathur N, Bhatnagar P (2012) Biotransformation of amide using Bacillus sp.: isolation strategy, strain characteristics and enzyme immobilization. Int J Environ Sci Technol 9:119–127. https://doi.org/10.1007/s13762-011-0005-7
Suzuki Y, Ohta H (2006) Identification of a thermostable and enantioselective amidase from the thermoacidophilic archaeon Sulfolobus tokodaii strain 7. Protein Expr Purif 45:368–373. https://doi.org/10.1016/j.pep.2005.06.017
Xi L, Tan W, Li J, Qu J, Liu J (2021) Cloning and characterization of a novel thermostable amidase, Xam, from Xinfangfangia sp. DLY26. Biotech Lett 43:1395–1402. https://doi.org/10.1007/s10529-021-03124-y
Funding
The authors are thankful for the financial assistance from the Science and Engineering Research Board, Department of Science and Technology, Government of India (Grant no-SB/YS/LS-15/2014 and LAB Projects (MLP 1003 MLP1008). A Fellowship grant by the Council of Scientific and Industrial Research (CSIR) to Hitesh Sharma is also acknowledged. In addition, the Indian Council of Medical Research (ICMR) is also appreciated for the fellowship grant of Ananta Ganjoo (Letter no. 5/3/8/ITR-F/2020 Dated: 28/02/2020). IIIM communication no. CSIR-IIIM/IPR/00129, Dated: 11/27/2019.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflicts of interest in this work.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
13205_2022_3109_MOESM1_ESM.tiff
Supplementary Fig S1. Zymogram of (L1) Total cell lysate and; (L2) Partially purified amidase (Brown colour band indicating amidase of Bacillus sp. IIIMB2907) (TIFF 1118 KB)
Rights and permissions
About this article
Cite this article
Sharma, H., Singh, R.V., Ganjoo, A. et al. Development of effective biotransformation process for benzohydroxamic acid production using Bacillus smithii IIIMB2907. 3 Biotech 12, 44 (2022). https://doi.org/10.1007/s13205-022-03109-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-022-03109-2