Abstract
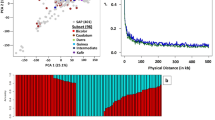
Tuber crops have measurable biological variation in root and stolon phenotyping and thus may be utilized to identify genomic regions associated with these variations. This is the first comprehensive association mapping study related to potato root and stolon traits. A diverse panel of 192 tetraploid potato (Solanum tuberosum L.) genotypes were grown in aeroponics to reveal a biologically significant variation and detection of genomic regions associated with the root and stolon traits. Phenotyping of root traits was performed by image analysis software “WinRHIZO” (a root scanning method), and stolon traits was measured manually, while SolCAP 25K potato array was used for genotyping. Significant variation was observed between the potato genotypes for root and stolon traits along with high heritabilities (0.80 in TNS to 0.95 in SL). For marker-trait associations, Q + K linear mixed model was implemented and 50 novel genomic regions were detected. Significantly associated SNPs with stolon traits were located on chr 4, chr 6, chr 7, chr 9, chr 11 and chr 12, while those linked to root traits on chr 1, chr 2, chr 3, chr 9, chr 11, and chr 12. Structure and PCA analysis grouped genotypes into four sub-populations disclosing population genetic diversity. LD decay was observed at 2.316 Mbps (r2 = 0.29) in the population. The identified SNPs were associated with genes performing vital functions such as root signaling and signal transduction in stress environments (GT-2 factors, protein kinases SAPK2-like and protein phosphatases “StPP1”), transcriptional and post-transcriptional gene regulation (RNA-binding proteins), sucrose synthesis and transporter families (UGPase, Sus3, SuSy, and StSUT1) and PVY resistance (Ry sto). The findings of our study can be employed in future breeding programs for improvement in potato production.








Similar content being viewed by others
References
Achenbach U, Paulo J, Ilarionova E et al (2008) Using SNP markers to dissect linkage disequilibrium at a major quantitative trait locus for resistance to the potato cyst nematode Globodera pallida on potato chromosome V. Theor Appl Genet 118:619. https://doi.org/10.1007/s00122-008-0925-x
Arsenault J-L, Poulcur S, Messier C, Guay R (1995) WinRHlZO™, a root-measuring system with a unique overlap correction method. HortScience 30:906D–906. https://doi.org/10.21273/HORTSCI.30.4.906D
Bahmankar M, Raij MR, Seloki AR, Shirkool K (2014) Assessment of broad sense heritability and genetic advance in safflower. Int J Biosci 4:131–135
Berdugo-Cely J, Valbuena RI, Sánchez-Betancourt E et al (2017) Genetic diversity and association mapping in the Colombian Central Collection of Solanum tuberosum L. Andigenum group using SNPs markers. PLoS ONE 12:e0173039. https://doi.org/10.1371/journal.pone.0173039
Björn B, Paulo MJ, Mank RA et al (2008) Association mapping of quality traits in potato (Solanum tuberosum L.). Euphytica 161:47–60
Björn B, Paulo MJ, Kowitwanich K et al (2010) Population structure and linkage disequilibrium unravelled in tetraploid potato. Theor Appl Genet 121:1151–1170
Blakeslee JJ, Zhou H-W, Heath JT et al (2008) Specificity of RCN1-mediated protein phosphatase 2A regulation in meristem organization and stress response in roots. Plant Physiol 146:539–553. https://doi.org/10.1104/pp.107.112995
Bradshaw JE, Hackett CA, Pande B et al (2008) QTL mapping of yield, agronomic and quality traits in tetraploid potato (Solanum tuberosum subsp. tuberosum). Theor Appl Genet 116:193–211. https://doi.org/10.1007/s00122-007-0659-1
Breseghello F, Sorrells ME (2006) Association mapping of kernel size and milling quality in wheat (Triticum aestivum L.) cultivars. Genetics 172:1165–1177
Byrne S, Meade F, Mesiti F et al (2020) Genome-wide association and genomic prediction for fry color in potato. Agronomy 10:90. https://doi.org/10.3390/agronomy10010090
Çalışkan ME, Onaran H, Arıoğlu H (2010) Overview of the Turkish potato sector: challenges, achievements and expectations. Potato Res 53:255–266
Charlotte P, Vos PG, Paulo MJ et al (2020) Distribution of P1(D1) wart disease resistance in potato germplasm and GWAS identification of haplotype-specific SNP markers. Theor Appl Genet 133:1859–1871. https://doi.org/10.1007/s00122-020-03559-3
Chen YL, Dunbabin VM, Diggle AJ et al (2011) Development of a novel semi-hydroponic phenotyping system for studying root architecture. Funct Plant Biol 38:355–363. https://doi.org/10.1071/FP10241
Cockram J, Scuderi A, Barber T et al (2015) Fine-mapping the wheat Snn1 locus conferring sensitivity to the Parastagonospora nodorum necrotrophic effector SnTox1 using an eight founder multiparent advanced generation inter-cross population. G3: Genes Genomes Genet 5:2257–2266. https://doi.org/10.1534/g3.115.021584
Consortium PGS (2011) Genome sequence and analysis of the tuber crop potato. Nature 475:189
de Dorlodot S, Forster B, Pagès L et al (2007) Root system architecture: opportunities and constraints for genetic improvement of crops. Trends Plant Sci 12:474–481
Earl DA, vonHoldt BM (2012) STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv Genet Resour 4:359–361. https://doi.org/10.1007/s12686-011-9548-7
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software structure: a simulation study. Mol Ecol 14:2611–2620. https://doi.org/10.1111/j.1365-294X.2005.02553.x
FAOSTAT F (2019) Food and Agriculture Organization of the United Nations-Statistic Division https://www.fao.org/faostat/en/#data.QC. Accessed 9 Nov 2020
Felcher KJ, Coombs JJ, Massa AN et al (2012) Integration of two diploid potato linkage maps with the potato genome sequence. PLoS ONE 7:e36347
Feng C, Song X, Tang H (2019) Molecular cloning and expression analysis of GT-2-like genes in strawberry. 3 Biotech 9:105. https://doi.org/10.1007/s13205-019-1603-1
Fita A, Picó B, Nuez F (2006) Implications of the genetics of root structure in melon breeding. J Am Soc Hortic Sci 131:372–379. https://doi.org/10.21273/JASHS.131.3.372
Flint-Garcia SA, Thuillet A-C, Yu J et al (2005) Maize association population: a high-resolution platform for quantitative trait locus dissection. Plant J 44:1054–1064. https://doi.org/10.1111/j.1365-313X.2005.02591.x
Forde BG (2014) Glutamate signalling in roots. J Exp Bot 65:779–787. https://doi.org/10.1093/jxb/ert335
French A, Ubeda-Tomás S, Holman TJ et al (2009) High-throughput quantification of root growth using a novel image-analysis tool. Plant Physiol 150:1784–1795. https://doi.org/10.1104/pp.109.140558
Fu H, Park WD (1995) Sink- and vascular-associated sucrose synthase functions are encoded by different gene classes in potato. Plant Cell 7:1369–1385. https://doi.org/10.1105/tpc.7.9.1369
Gardner KM, Brown P, Cooke TF et al (2014) Fast and cost-effective genetic mapping in apple using next-generation sequencing. G3: Genes Genomes Genet 4:1681–1687. https://doi.org/10.1534/g3.114.011023
Grandellis C, Giammaria V, Bialer M et al (2012) The novel Solanum tuberosum calcium dependent protein kinase, StCDPK3, is expressed in actively growing organs. Planta 236:1831–1848. https://doi.org/10.1007/s00425-012-1732-9
Grossi CEM, Fantino E, Serral F et al (2020) Methylobacterium sp. 2A is a plant growth-promoting rhizobacteria that has the potential to improve potato crop yield under adverse conditions. Front Plant Sci 11:71
Gupta PK, Rustgi S, Kulwal PL (2005) Linkage disequilibrium and association studies in higher plants: present status and future prospects. Plant Mol Biol 57:461–485. https://doi.org/10.1007/s11103-005-0257-z
Hagenblad J, Nordborg M (2002) Sequence variation and haplotype structure surrounding the flowering time locus FRI in Arabidopsis thaliana. Genetics 161:289–298
Hamblin MT, Mitchell SE, White GM et al (2004) Comparative population genetics of the panicoid grasses: sequence polymorphism, linkage disequilibrium and selection in a diverse sample of Sorghum bicolor. Genetics 167:471–483. https://doi.org/10.1534/genetics.167.1.471
Hamilton JP, Hansey CN, Whitty BR et al (2011) Single nucleotide polymorphism discovery in elite North American potato germplasm. BMC Genom 12:1–12
Haverkort AJ, Waart MVD, Bodlaender KBA (1990) The effect of early drought stress on numbers of tubers and stolons of potato in controlled and field conditions. Potato Res 33:89–96. https://doi.org/10.1007/BF02358133
Hill WG, Robertson A (1968) Linkage disequilibrium in finite populations. Theoret Appl Genetics 38:226–231. https://doi.org/10.1007/BF01245622
Hyma KE, Barba P, Wang M et al (2015) Heterozygous mapping strategy (HetMappS) for high resolution genotyping-by-sequencing markers: a case study in grapevine. PLoS ONE 10:e0134880. https://doi.org/10.1371/journal.pone.0134880
Iwama K (2008) Physiology of the potato: new insights into root system and repercussions for crop management. Potato Res 51:333. https://doi.org/10.1007/s11540-008-9120-3
Jannoo N, Grivet L, Dookun A et al (1999) Linkage disequilibrium among modern sugarcane cultivars. Theor Appl Genet 99:1053–1060. https://doi.org/10.1007/s001220051414
Khlestkin VK, Rozanova IV, Efimov VM, Khlestkina EK (2019) Starch phosphorylation associated SNPs found by genome-wide association studies in the potato (Solanum tuberosum L.). BMC Genet 20:29. https://doi.org/10.1186/s12863-019-0729-9
Khlestkin VK, Erst TV, Rozanova IV et al (2020) Genetic loci determining potato starch yield and granule morphology revealed by genome-wide association study (GWAS). PeerJ 8:e10286. https://doi.org/10.7717/peerj.10286
Khu D-M, Lorenzen J, Hackett CA, Love SL (2008) Interval mapping of quantitative trait loci for corky ringspot disease resistance in a tetraploid population of potato (Solanum tuberosum subsp. tuberosum). Am J Pot Res 85:129–139. https://doi.org/10.1007/s12230-008-9016-4
Kohli A, Narciso JO, Miro B, Raorane M (2012) Root proteases: reinforced links between nitrogen uptake and mobilization and drought tolerance. Physiol Plant 145:165–179. https://doi.org/10.1111/j.1399-3054.2012.01573.x
Kraakman ATW, Martínez F, Mussiraliev B et al (2006) Linkage disequilibrium mapping of morphological, resistance, and other agronomically relevant traits in modern spring barley cultivars. Mol Breed 17:41–58. https://doi.org/10.1007/s11032-005-1119-8
Kraft T, Hansen M, Nilsson N-O (2000) Linkage disequilibrium and fingerprinting in sugar beet. Theor Appl Genet 101:323–326. https://doi.org/10.1007/s001220051486
Kühn C, Grof CP (2010) Sucrose transporters of higher plants. Curr Opin Plant Biol 13:287–297. https://doi.org/10.1016/j.pbi.2010.02.001
Kühn C, Hajirezaei M-R, Fernie AR et al (2003) The sucrose transporter StSUT1 localizes to sieve elements in potato tuber phloem and influences tuber physiology and development. Plant Physiol 131:102–113. https://doi.org/10.1104/pp.011676
Kumar J, Pratap A, Kumar S (eds) (2015) Phenomics in crop plants: trends, options and limitations. Springer, New Delhi
Lahlou O, Ledent J-F (2005) Root mass and depth, stolons and roots formed on stolons in four cultivars of potato under water stress. Eur J Agron 22:159–173. https://doi.org/10.1016/j.eja.2004.02.004
Malosetti M, van der Linden CG, Vosman B, van Eeuwijk FA (2007) A mixed-model approach to association mapping using pedigree information with an illustration of resistance to Phytophthora infestans in potato. Genetics 175:879–889. https://doi.org/10.1534/genetics.105.054932
Mosquera T, Alvarez MF, Jiménez-Gómez JM et al (2016) Targeted and untargeted approaches unravel novel candidate genes and diagnostic SNPs for quantitative resistance of the potato (Solanum tuberosum L.) to Phytophthora infestans causing the late blight disease. PLoS ONE 11:e0156254
Na J-K, Kim J-K, Kim D-Y, Assmann SM (2015) Expression of potato RNA-binding proteins StUBA2a/b and StUBA2c induces hypersensitive-like cell death and early leaf senescence in Arabidopsis. J Exp Bot 66:4023–4033. https://doi.org/10.1093/jxb/erv207
Nordborg M, Borevitz JO, Bergelson J et al (2002) The extent of linkage disequilibrium in Arabidopsis thaliana. Nat Genet 30:190–193. https://doi.org/10.1038/ng813
Ogunniyan DJ, Olakojo SA (2014) Genetic variation, heritability, genetic advance and agronomic character association of yellow elite inbred lines of maize (Zea mays L.). Niger J Genetics 28:24–28. https://doi.org/10.1016/j.nigjg.2015.06.005
Oraguzie NC, Wilcox PL, Rikkerink EH, de Silva HN (2007) Linkage disequilibrium. In: Oraguzie NC, Rikkerink EHA, Gardiner SE, de Silva HN (eds) Association mapping in plants. Springer, New York, NY. https://doi.org/10.20900/cbgg20200010
Ortiz R (2020) Genomic-led potato breeding for increasing genetic gains: achievements and outlook. Crop Breed Genet Genom 2(2):e200010. https://doi.org/10.20900/cbgg20200010
País SM, Téllez-Iñón MT, Capiati DA (2009) Serine/threonine protein phosphatases type 2A and their roles in stress signaling. Plant Signal Behav 4:1013–1015. https://doi.org/10.4161/psb.4.11.9783
Phan HTT, Rybak K, Bertazzoni S et al (2018) Novel sources of resistance to Septoria nodorum blotch in the Vavilov wheat collection identified by genome-wide association studies. Theor Appl Genet 131:1223–1238. https://doi.org/10.1007/s00122-018-3073-y
Pierret A, Gonkhamdee S, Jourdan C, Maeght J-L (2013) IJ_Rhizo: an open-source software to measure scanned images of root samples. Plant Soil 373:531–539
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Remington DL, Thornsberry JM, Matsuoka Y et al (2001) Structure of linkage disequilibrium and phenotypic associations in the maize genome. PNAS 98:11479–11484. https://doi.org/10.1073/pnas.201394398
Riaz A, Athiyannan N, Periyannan SK et al (2018) Unlocking new alleles for leaf rust resistance in the Vavilov wheat collection. Theor Appl Genet 131:127–144. https://doi.org/10.1007/s00122-017-2990-5
Ritland K (2005) Multilocus estimation of pairwise relatedness with dominant markers. Mol Ecol 14:3157–3165
Rosyara UR, Jong WSD, Douches DS, Endelman JB (2016) Software for genome-wide association studies in autopolyploids and its application to potato. Plant Genome. https://doi.org/10.3835/plantgenome2015.08.0073
Schiavon M, Macolino S, Leinauer B, Ziliotto U (2016) Seasonal changes in carbohydrate and protein content of seeded Bermudagrasses and their effect on spring green-up. J Agron Crop Sci 202:151–160. https://doi.org/10.1111/jac.12135
Sharma SK, MacKenzie K, McLean K et al (2018) Linkage disequilibrium and evaluation of genome-wide association mapping models in tetraploid potato. G3: Genes Genomes Genet 8:3185–3202. https://doi.org/10.1534/g3.118.200377
Simko I, Costanzo S, Ramanjulu V et al (2006) Mapping polygenes for tuber resistance to late blight in a diploid Solanum phureja × S. stenotomum hybrid population. Plant Breed 125:385–389. https://doi.org/10.1111/j.1439-0523.2006.01232.x
Smith S, De Smet I (2012) Root system architecture: insights from Arabidopsis and cereal crops. The Royal Society
Song Y-S, Schwarzfischer A (2008) Development of STS markers for selection of extreme resistance (Rysto) to PVY and maternal pedigree analysis of extremely resistant cultivars. Am J Pot Res 85:159–170. https://doi.org/10.1007/s12230-008-9012-8
Song A, Wu D, Fan Q et al (2016) Transcriptome-wide identification and expression profiling analysis of chrysanthemum trihelix transcription factors. Int J Mol Sci 17:198. https://doi.org/10.3390/ijms17020198
Spooner DM, McLean K, Ramsay G et al (2005) A single domestication for potato based on multilocus amplified fragment length polymorphism genotyping. Proc Natl Acad Sci 102:14694–14699
Stein O, Granot D (2019) An overview of sucrose synthases in plants. Front Plant Sci. https://doi.org/10.3389/fpls.2019.00095
Stich B, Melchinger AE (2010) An introduction to association mapping in plants. CAB Rev: Perspect Agric Vet Sci Nutr Nat Resour 5:1–9
Stich B, Melchinger AE, Frisch M et al (2005) Linkage disequilibrium in European elite maize germplasm investigated with SSRs. Theor Appl Genet 111:723–730. https://doi.org/10.1007/s00122-005-2057-x
Stich B, Urbany C, Hoffmann P, Gebhardt C (2013) Population structure and linkage disequilibrium in diploid and tetraploid potato revealed by genome-wide high-density genotyping using the SolCAP SNP array. Plant Breed 132:718–724
Szecowka M, Osorio S, Obata T et al (2012) Decreasing the Mitochondrial synthesis of malate in potato tubers does not affect plastidial starch synthesis, suggesting that the physiological regulation of ADPglucose pyrophosphorylase is context dependent. Plant Physiol 160:2227–2238. https://doi.org/10.1104/pp.112.204826
Tamisier L, Szadkowski M, Nemouchi G et al (2020) Genome-wide association mapping of QTLs implied in potato virus Y population sizes in pepper: evidence for widespread resistance QTL pyramiding. Mol Plant Pathol 21:3–16
Thaon P (2018) Genetic analysis of wheat root architecture in aeroponics. Master thesis. Faculté des bioingénieurs, Université catholique de Louvain, Belgium
Trachsel S, Kaeppler SM, Brown KM, Lynch JP (2011) Shovelomics: high throughput phenotyping of maize (Zea mays L.) root architecture in the field. Plant Soil 341:75–87
Trachsel S, Kaeppler SM, Brown KM, Lynch JP (2013) Maize root growth angles become steeper under low N conditions. Field Crops Res 140:18–31
Uitdewilligen JGAML, Wolters A-MA, D’hoop BB et al (2013) A next-generation sequencing method for genotyping-by-sequencing of highly heterozygous autotetraploid potato. PLoS ONE 8:e62355. https://doi.org/10.1371/journal.pone.0062355
Vos PG, Uitdewilligen JGAML, Voorrips RE et al (2015) Development and analysis of a 20K SNP array for potato (Solanum tuberosum): an insight into the breeding history. Theor Appl Genet 128:2387–2401. https://doi.org/10.1007/s00122-015-2593-y
Vos PG, Paulo MJ, Voorrips RE et al (2017) Evaluation of LD decay and various LD-decay estimators in simulated and SNP-array data of tetraploid potato. Theor Appl Genet 130:123–135
Whitt SR, Buckler ES (2003) Using natural allelic diversity to evaluate gene function. In: Grotewold E (ed) Plant functional genomics. Humana Press, Totowa, pp 123–139
Wishart J, George TS, Brown LK et al (2013) Measuring variation in potato roots in both field and glasshouse: the search for useful yield predictors and a simple screen for root traits. Plant Soil 368:231–249. https://doi.org/10.1007/s11104-012-1483-1
Xie Z-M, Zou H-F, Lei G et al (2009) Soybean trihelix transcription factors GmGT-2A and GmGT-2B improve plant tolerance to abiotic stresses in transgenic Arabidopsis. PLoS ONE 4:e6898. https://doi.org/10.1371/journal.pone.0006898
Yu J, Buckler ES (2006) Genetic association mapping and genome organization of maize. Curr Opin Biotechnol 17:155–160. https://doi.org/10.1016/j.copbio.2006.02.003
Zarzyńska K, Boguszewska-Mańkowska D, Nosalewicz A (2017) Differences in size and architecture of the potato cultivars root system and their tolerance to drought stress. Plant Soil Environ 63(2017):159–164. https://doi.org/10.17221/4/2017-PSE
Zhu YL, Song QJ, Hyten DL et al (2003) Single-nucleotide polymorphisms in soybean. Genetics 163:1123–1134
Zhu C, Gore M, Buckler ES, Yu J (2008) Status and prospects of association mapping in plants. Plant Genome. https://doi.org/10.3835/plantgenome2008.02.0089
Zia MAB, Demirel U, Nadeem MA, Caliskan ME (2020) Genome-wide association study identifies various loci underlying agronomic and morphological traits in diversified potato panel. Physiol Mol Biol Plants 26:1003–1020
Acknowledgements
The Scientific and Technological Research Council of Turkey (TUBITAK) was financially supported this study through Project # 115O949. The current paper belongs to the Ph.D. thesis of the corresponding author. I would also like to thank Eric Kuopuobe Naawe for helping in the maintenance of the aeroponic system during research work. Furthermore, an appreciation was acknowledged by all my colleagues during the phenotyping.
Author information
Authors and Affiliations
Contributions
MEÇ: supervised and designed the study, MEÇ and UD: funding acquisition, MFY: set an experiment and phenotypic data acquisition, MFY and MN: phenotyping and genotyping data curation and analysis, UD: formal computational analysis, MFY: drafted the manuscript, MEÇ, UD, MFY and MN review, editing and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Supplementary information
Below is the link to the electronic supplementary material.
13205_2021_2727_MOESM1_ESM.docx
Supplementary data (Table S1, S2, S3 and Figures S1, S2, S3, S4, S5, and S6) of this research article is attached in the additional file section. (DOCX 3779 kb)
Rights and permissions
About this article
Cite this article
Yousaf, M.F., Demirel, U., Naeem, M. et al. Association mapping reveals novel genomic regions controlling some root and stolon traits in tetraploid potato (Solanum tuberosum L.). 3 Biotech 11, 174 (2021). https://doi.org/10.1007/s13205-021-02727-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-02727-6