Abstract
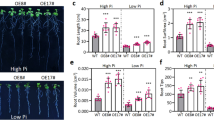
The effective reversion of hyperhydricity (HH) in Dianthus chinensis L. facilitated efficient in vitro production of hyperhydricity-free plantlets. Under routine sub-culture practice, the problem of HH arises after third sub-culture in agar (0.85%) gelled Murashige and Skoog (MS) medium containing 2.5 µM 6-benzyladenine (BA). To confirm the role of ethylene on hyperhydricity induction, an ethylene releasing compound ethephon (5 µM) was used in combination with 2.5 µM BA and demonstrated 100% HH with reduced stomatal aperture. Supplementation of 10 µM silver nitrate (AgNO3) to 2.5 µM BA containing medium resulted HH reversion with reduced shoot number (19.0); however, addition of 5 µM cobalt chloride (CoCl2) produced highest microshoots (202.0). The combination effect of AgNO3 (10 µM), CoCl2 (5 µM), and BA (2.5 µM) showed complete HH reversion and upheld normal microshoots (55.0) with reduced relative water content (78.3%). The Ag and Co salts regulate ethylene biosynthesis and thereby 50% reductions in H2O2 content characterized by formation of green healthy shoots with proper stomatal morphology. The gene expression profile of 1-Amminocyclopropane-1-carboxylase synthase (ACS1) and 1-Amminocyclopropane-1-carboxylic acid oxidase (ACO1) showed reduced expression after the retroversion of microshoots in anti-ethylene reversion medium compared to hyperhydric shoot. In vitro raised shoots were rooted (93.3%) ex vitro by 10 mM IBA treatment and 92.2% plants were survived. The genetic stability of micropropagated plants were analyzed and proved that addition of low levels of heavy metal salt in the medium does not cause any variation in banding pattern. The protocol forwards a novel method to revert HH of in vitro cultures by adopting intermittent exposure of anti-ethylene compounds added in the medium and the procedure can be applied to many other plants facing similar HH problems.






Similar content being viewed by others
Abbreviations
- MS:
-
Murashige and Skoog
- HH:
-
Hyperhydricity
- CoCl2 :
-
Cobalt chloride
- AgNO3 :
-
Silver nitrate
- RWC:
-
Relative water content
- ISSR:
-
Inter-simple sequence repeat
References
Ahmadian M, Babaei A, Shokri S, Hessami S (2017) Micropropagation of carnation (Dianthus caryophyllus L.) in liquid medium by temporary immersion bioreactor in comparison with solid culture. J Genet Eng Biotechnol 15:309–315. https://doi.org/10.1016/j.jgeb.2017.07.005
Al-Mayahi AMW (2014) Effect of copper sulphate and cobalt chloride on growth of the in vitro culture tissues for date palm (Phoenix dactylifera l.) cv. Ashgar. Am J Agricult Biol Sci 9:6–18. https://doi.org/10.3844/ajabssp.2014.6.18
Amoo SO, Finnie JF, Van Staden J (2011) The role of meta-topolins in alleviating micropropagation problems. Plant Growth Regul 63:197–206. https://doi.org/10.1007/s10725-010-9504-7
Bernard F, Moghadam NN, Mirzajani F (2015) The effect of colloidal silver nanoparticles on the level of lignification and hyperhydricity syndrome in Thymus daenensis in vitro shoots: a possible involvement of bonded polyamines. In Vitro Cell Dev Biol Plant 51:546–553. https://doi.org/10.1007/s11627-015-9700-2
Bhatia R, Singh KP, Sharma TR, Jhang T (2011) Evaluation of the genetic fidelity of in vitro-propagated gerbera (Gerbera jamesonii Bolus) using DNA-based markers. Plant Cell Tiss Org Cult 104:131–135. https://doi.org/10.1007/s11240-010-9806-5
Borkowska B (2001) Morphological and physiological characteristics of micro propagated strawberry plants rooted in vitro or ex vitro. Sci Hort 89:195–206. https://doi.org/10.1016/S0304-4238(00)00230-2
Bornet B, Branchard M (2001) Nonanchored inter simple sequence repeat (ISSR) markers: reproducible and specific tools for genome fingerprinting. Plant Mol Biol Rep 19:209–215. https://doi.org/10.1007/BF02772892
Buah JN, Danso E, Taah KJ, Abole EA, Bediako EA, Asiedu J, Baidoo R (2010) The effects of different concentrations cytokinins on the in vitro multiplication of plantain (Musa sp.). Biotechnol 9:343–347. https://doi.org/10.3923/biotech.2010.343.347
Chakrabarty D, Park S, Ali M, Shin K, Paek K (2006) Hyperhydricity in apple: ultrastructural and physiological aspects. Tree Physiol 26:377–388. https://doi.org/10.1093/treephys/26.3.377
De D, De B (2003) Effect of ethephon on antioxidant enzymes and diosgenin production in seedlings of Trigonella foenum-graecum. Food Chem 82:211–216. https://doi.org/10.1016/S0308-8146(02)00514-9
Desikan R, Harrett LK, Williams R, Tagliavia C, Harter K, Hooley R, Hancock JT, Neill SJ (2006) Ethylene-induced stomatal closure in Arabidopsis occurs via Atrboh F-mediated hydrogen peroxide synthesis. Plant J 47:907–916. https://doi.org/10.1111/j.1365-313X.2006.02842.x
Doyle JJ, Doyle JL (1990) Isolation of plant DNA from fresh tissue. Focus 12:13–15
Duncan DB (1955) Multiple range and multiple F tests. Biometrics 11:1–42. https://www.jstor.org/stable/3001478
Fu X, Yang S, Bao M (2008) Factors affecting somatic embryogenesis in anther cultures of Chinese pink (Dianthus chinensis L.). In Vitro Cell Dev Biol Plant 44:194–202. https://doi.org/10.1007/s11627-008-9107-4
Gao H, Xia X, An L, Xin X, Liang Y (2017a) Reversion of hyperhydricity in pink (Dianthus chinensis L.) plantlets by AgNO3 and its associated mechanism during in vitro culture. Plant Sci 254:1–11. https://doi.org/10.1016/j.plantsci.2016.10.008
Gao H, Xu P, Li J, Ji H, An L, Xia X (2017b) AgNO3 prevents the occurrence of hyperhydricity in Dianthus chinensis L. by enhancing water loss and antioxidant capacity. In Vitro Cell Dev Biol Plant 53:561–570. https://doi.org/10.1007/s11627-017-9871-0
Ge XM, Cai HL, Lei X, Zhou X, Yue M, He JM (2015) Heterotrimeric G protein mediates ethylene-induced stomatal closure via hydrogen peroxide synthesis in Arabidopsis. Plant J 82:138–150. https://doi.org/10.1111/tpj.12799
Gong Y, Gao F, Tang K (2005) In vitro high frequency direct root and shoot regeneration in sweet potato using the ethylene inhibitor silver nitrate. S Afr J Bot 71:110–113. https://doi.org/10.1016/S0254-6299(15)30159-9
Hassanein AM, Salem JM, Faheed FA, El-nagish A (2018) Effect of anti-ethylene compounds on isoenzyme patterns and genome stability during long term culture of Moringa oleifera. Plant Cell Tiss Organ Cult 132:201–212. https://doi.org/10.1007/s11240-017-1326-0
He JM, Ma XG, Zhang Y, Sun TF, Xu FF, Chen YP, Liu X, Yue M (2013) Role and interrelationship of Gα protein, hydrogen peroxide, and nitric oxide in ultraviolet B-induced stomatal closure in Arabidopsis leaves. Plant Physiol 161:1570–1583. https://doi.org/10.1104/pp.112.211623
Ivanova M, Van Staden J (2011) Influence of gelling agent and cytokinins on the control of hyperhydricity in Aloe polyphylla. Plant Cell Tiss Org Cult 104:13–21. https://doi.org/10.1007/s11240-010-9794-5
Jain A, Kantia A, Kothari SL (2001) De novo differentiation of shoot buds from leaf-callus of Dianthus caryophyllus L. and control of hyperhydricity. Scient Horticult 87:319–326. https://doi.org/10.1016/S0304-4238(00)00185-0
Jethwani V, Kothari SL (1996) Phenylacetic acid induced organogenesis in cultured leaf segments of Dianthus chinensis. Plant Cell Rep 15:869–872. https://doi.org/10.1007/BF00233159
Kaeppler SM, Heidi F, Rhee KY (2000) Epigenetic aspects of somaclonal variation in plants. Plant Mol Biol 43:179–188. https://doi.org/10.1023/A:1006423110134
Kantia A, Kothari SL (2002) High efficiency adventitious shoot bud formation and plant regeneration from leaf explants of Dianthus chinensis L. Sci Hort 96:205–212. https://doi.org/10.1016/S0304-4238(02)00081-X
Karimi S, Yadollahi A, Nazari-Moghadam R, Imani A, Arzani K (2012) In vitro screening of almond (Prunus dulcis Mill.) genotypes for drought tolerance. J Biol Environ Sci 6:263–270
Kerstiens G (1997) In vivo manipulation of cuticular water permeance and its effect on stomatal response to air humidity. New Phytol 137:473–480. https://doi.org/10.1046/j.1469-8137.1997.00847.x
Kharrazi M, Nemati H, Tehranifar A, Bagheri A, Sharifi A (2011) In vitro culture of carnation (Dianthus caryophyllus L.) focusing on the problem of vitrification. J Biol Environ Sci 5:1–6
Krishnaraj C, Jagan EG, Ramachandran R, Abirami SM, Mohan N, Kalaichelvan PT (2012) Effect of biologically synthesized silver nanoparticles on Bacopa monnieri (Linn.) Wettst. Plant Growth Metabol Process Biochem 47:651–658. https://doi.org/10.1016/j.procbio.2012.01.006
Kumar V (2009) AgNO3 - a potential regulator of ethylene activity and plant growth modulator. Mol Biol Genet 12:2. https://doi.org/10.2225/vol12-issue2-fulltext-1
Kumar GP, Sivakumar S, Siva G, Vigneswaran M, Kumar TS, Jayabalan N (2016) Silver nitrate promotes high-frequency multiple shoot regeneration in cotton (Gossypium hirsutum L.) by inhibiting ethylene production and phenolic secretion. In Vitro Cell Dev Biol Plant 52:408–418. https://doi.org/10.1007/s11627-016-9782-5
Kumawat S, Kachhwaha S, Kothari SL (2013) Micronutrient optimization in the basal medium improves in vitro plant regeneration in carnation (Dianthus caryophyllus L) and Chinese pink (Dianthus chinensis L.) Propagation of Ornamental Plants 13: 3–11
Lai CC, Lin HM, Nalawade SM, Fang W, Tsay HS (2005) Hyperhydricity in shootcultures of Scrophularia yoshimurae can be effectively reduced by ventilation of culture vessels. J Plant Physiol 162:355–361. https://doi.org/10.1016/j.jplph.2004.07.015
Lau OL, Yang SF (1976) Inhibition of ethylene production by cobaltous ion. Plant Physiol 58:114–117
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods Enzymol 148:350–382. https://doi.org/10.1016/0076-6879(87)48036-1
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-∆∆CT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Mayor M, Nestares G, Zorzoli R, Picardi L (2003) Reduction of hyperhydricity in sunflower tissue culture. Plant Cell Tiss Org Cult 72:99–103. https://doi.org/10.1023/A:1021216324757
Mirza F, Uliaie ED, Hagh AB (2015) Stimulation effect of AgNO3 and CoCl2 as ethylene inhibitors on in vitro organogenesis of sunflower (Helianthus annuus L.). Yyu J Agr Sci 25:113–118
Muneer S, Soundararajan P, Jeong BR (2016) Proteomic and antioxidant analysis elucidates the underlying mechanism of tolerance to hyperhydricity stress in in vitro shoot cultures of Dianthus caryophyllus. J Plant Growth Regul 35:667–679. https://doi.org/10.1007/s00344-015-9569-7
Muneer S, Park YG, Jeong BR (2018) Red and blue light emitting diodes (LEDs) participate in mitigation of hyperhydricity in in vitro-grown carnation genotypes (Dianthus caryophyllus). J Plant Growth Regul 37:370–379. https://doi.org/10.1007/s00344-017-9733-3
Neill S, Desikan R, Hancock J (2002) Hydrogen peroxide signaling. Curr Opinion Plant Biol 5:388–395
Ozturk OL, Kufrevioglu I, Demir Y (2008) In vivo and in vitro effects of ethephon on some oxidative enzymes in spinach leaves. Acta Physiol Plant 30:105–110. https://doi.org/10.1007/s11738-007-0096-4
Pareek A, Kantia A, Kothari SL (2004) In vitro cloning of ornamental species of Dianthus. Indian J Biotechnol 3:263–266
Petruş-Vancea A (2018) Cell ultrastructure and chlorophyll pigments in hyperhydric and non-hyperhydric Beta vulgaris var. conditiva plantlets, treated with deuterium depleted water. Plant Cell Tiss Org Cult 135:13–21. https://doi.org/10.1007/s11240-018-1439-0
Saher S, Piqueras A, Hellin E, Olmos E (2005) Prevention of hyperhydricity in micropropagated carnation shoots by bottom cooling: implications of oxidative stress. Plant Cell Tiss Org Cult 81:149–158. https://doi.org/10.1007/s11240-004-4043-4
Sarmast MK, Niazi A, Salehi H, Abolimoghadam A (2014) Silver nanoparticles affect ACS expression in Tecomella undulata in vitro culture. Plant Cell Tissu Org Cult 1212:227–236. https://doi.org/10.1007/s11240-014-0697-8
Simms D, Cizdziel PE, Chomczynski P (1993) TRIZOTM: a new reagent for optimal single-step isolation of RNA. Focus 15:99–103
Snedecor GW, Cochran WG (1962) Statistical methods, 4th edn. The Iowa State University Press, Ames
Sreedhar RV, Venkatachalam L, Neelwarne B (2009) Hyperhydricity-related morphologic and biochemical changes in Vanilla (Vanilla planifolia). J Plant Growth Regul 28:46–57. https://doi.org/10.1007/s00344-008-9073-4
Sreelekshmi R, Siril EA (2020) Influence of polyamines on hyperhydricity reversion and its associated mechanism during micropropagation of China pink (Dianthus chinensis L.). Physiol Mol Biol Plants 26:2035–2045. https://doi.org/10.1007/s12298-020-00885-0
Sreelekshmi R, Siril EA (2021) Investigation on in vitro bouquets and flower longevity of micropropagated Dianthus chinensis L. Sci Hortic 275:109708. https://doi.org/10.1016/j.scienta.2020.109708
Sreelekshmi R, Siril EA, Muthukrishnan S (2021) Role of biogenic silver nanoparticles on hyperhydricity reversion in Dianthus chinensis L. an in vitro model culture. J Plant Growth Regul. https://doi.org/10.1007/s00344-020-10276-0
Tamimi SM (2015) Effects of ethylene inhibitors, silver nitrate (AgNO3), cobalt chloride (CoCl2) and aminooxyacetic acid (AOA), on in vitro shoot induction and rooting of banana (Musa acuminata L.). Afr J Biotechnol 32:2510–2516. https://doi.org/10.5897/AJB2015.14788
Tian J, Jiang F, Wu Z (2015) The apoplastic oxidative burst as a key factor of hyperhydricity in garlic plantlet in vitro. Plant Cell Tiss Org Cult 120:571–584. https://doi.org/10.1007/s11240-014-0623-0
Turhan H (2004) The effect of silver nitrate (ethylene inhibitor) on in vitro shoot development in potato (Solanum tuberosum L.). Biotechnol 3:72–74. https://doi.org/10.3923/biotech.2004.72.74
Van den Dries V, Giannì S, Czerednik A, Krens FA, De Klerk GJM (2013) Flooding of the apoplast is a key factor in the development of hyperhydricity. J Exp Bot 64:5221–5230. https://doi.org/10.1093/jxb/ert315
Vinoth A, Ravindhran R (2015) Reduced hyperhydricity in watermelon shoot cultures using silver ions. In Vitro Cell Dev Biol Plant. 51:258–264. https://doi.org/10.1007/s11627-015-9698-5
Yadav MK, Gaur AK, Garg GK (2003) Development of suitable protocol to overcome hyperhydricity in carnation during micropropagation. Plant Cell Tiss Org Cult 72:153–156. https://doi.org/10.1023/A:1022236920605
Yang SF, Hoffman NE (1984) Ethylene biosynthesis and its regulation in higher plants. Annu Rev Plant Physiol 35:155–189. https://doi.org/10.1146/annurev.pp.35.060184.001103
Yasuta T, Satoh S, Minamisawa K (1999) New assay for rhizobitoxine based on inhibition of 1-aminocyclopropane-1-carboxylate synthase. Appl Environ Microbiol 65:849–852
Ziv M, Ariel T (1994) Vitrification in relation to stomatal deformation and malfunction in carnation leaves in vitro. In: Lumsden PJ, Nicholas JR, Davies WJ (eds) Physiology, growth and development of plants in culture. Springer, Dordrecht, pp 143–154. https://doi.org/10.1007/978-94-011-0790-7_15
Acknowledgements
The authors are grateful to Dr. S Suhara Beevy, Professor and Head, Department of Botany, University of Kerala, Kariavattom, Thiruvananthapuram, for providing the facilities. One of the authors RS also thanks University of Kerala for financial support in the form of Junior Research Fellowship (No. AcVI(1)/715/BOT/13289/2015).
Author information
Authors and Affiliations
Contributions
RS is responsible for designing and conducting the experiments under the advice of EAS. Data analysis and manuscript editing has been performed by EAS.
Compliance with ethical standards
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Research involving human and animal participants
No human participants and/or animals are involved in the research.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sreelekshmi, R., Siril, E.A. Effective reversal of hyperhydricity leading to efficient micropropagation of Dianthus chinensis L.. 3 Biotech 11, 95 (2021). https://doi.org/10.1007/s13205-021-02645-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-02645-7