Abstract
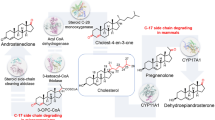
The metabolism of host cholesterol by Mycobacterium tuberculosis is an important factor for both its virulence and pathogenesis. However, the rationale for this cholesterol metabolism has not been fully understood yet. In the present study, we characterized several previously undescribed acyl-CoA synthetases that are involved in the steroid side-chain degradation in Mycobacterium smegmatis, and an analogue of intermediate from steroid degradation, 5′-O-(lithocholoyl sulfamoyl) adenosine (LCA-AMS), was successfully designed and synthesized to be used as a specific anti-mycobacterial agent. The acyl-CoA synthetases exhibited strong preferences for the length of side chain. FadD19 homologs, including FadD19 (MSMEG_5914), FadD19-2 (MSMEG_2241), and FadD19-4 (MSMEG_3687), are unanimously favorable cholesterol with a C8 alkanoate side chain. FadD17 (MSMEG_5908) and FadD1 (MSMEG_4952) showed high preferences for steroids, containing a C5 alkanoate side chain. FadD8 (MSMEG_1098) exhibited specific activity toward cholestenoate with a C8 alkanoate side chain. An acylsulfamoyl analogue of lithocholate, 5′-O-(lithocholoyl sulfamoyl) adenosine (LCA-AMS), was designed and synthesized. As expected, the intermediate analogue not only specifically inhibited those steroid-activated acyl-CoA synthetases, but also selectively inhibited the growth of mycobacterial species, including M. tuberculosis, M. smegmatis, and Mycobacterium neoaurum. Overall, our research advanced our understanding of mycobacterial steroid degradation and provided new insights to develop novel mechanism-based anti-mycobacterial agents.






Similar content being viewed by others
References
Abuhammad A (2017) Cholesterol metabolism: a potential therapeutic target in Mycobacteria. Br J Pharmacol 174(14):2194–2208
Arora P, Goyal A, Natarajan VT, Rajakumara E, Verma P, Gupta R, Yousuf M, Trivedi OA, Mohanty D, Tyagi A (2009) Mechanistic and functional insights into fatty acid activation in Mycobacterium tuberculosis. Nat Chem Biol 5(3):166–173
Bernson VSM (1976) Acetyl-CoA hydrolase; activity, regulation and physiological, significance of the enzyme in brown adipose tissue from hamster. Eur J Biochem 67(2):403–410
Capyk JK, Kalscheuer RG (2009) Mycobacterial cytochrome p450 125 (cyp125) catalyzes the terminal hydroxylation of C27 steroids. J Biol Chem 284(51):35534–35542
Casabon I, Crowe AM, Liu J, Eltis LD (2013) FadD3 is an acyl-CoA synthetase that initiates catabolism of cholesterol rings C and D in actinobacteria. Mol Microb 87(2):269–283
Casabon I, Swain K, Crowe AM, Eltis LD, Mohn WW (2014) Actinobacterial acyl coenzyme A synthetases involved in steroid side-chain catabolism. J Bacteriol 196(3):579–587
Crowe AM, Casabon I, Brown KL, Jie L, Lian J, Rogalski JC, Hurst TE, Snieckus V, Foster LJ, Eltis LD (2017) Catabolism of the last two steroid rings in Mycobacterium tuberculosis and other bacteria. Mbio 8(2):e00321-17
Fernández Cabezón L, Galán B, García JL (2017) Molecular characterization of a new gene cluster for steroid degradation in Mycobacterium smegmatis. Environ Microbiol 19(7):2546–2563
Ferreras JA, Ryu JS, Lello FD, Tan DS, Quadri LEN (2005) Small-molecule inhibition of siderophore biosynthesis in Mycobacterium tuberculosis and Yersinia pestis. Nat Chem Biol 1(1):29–32
Ferreras JA, Stirrett KL, Lu X, Ryu JS, Soll CE, Tan DS, Quadri LEN (2008) Mycobacterial phenolic glycolipid virulence factor biosynthesis: mechanism and small-molecule inhibition of Polyketide chain initiation. Chem Biol 15(1):51–61
Ge F, Li W, Chen G, Liu Y, Zhang G, Yong B, Wang Q, Wang N, Huang Z, Li W (2011) Draft genome sequence of Gordonia neofelifaecis NRRL B-59395, a cholesterol-degrading actinomycete. J Bacteriol 193(18):5045–5046
Geize RVD, Yam K, Heuser T, Wilbrink MH, Hara H, Anderton MC, Sim E, Dijkhuizen L, Davies JE, Mohn WW (2007) A gene cluster encoding cholesterol catabolism in a soil actinomycete provides insight into Mycobacterium tuberculosis survival in macrophages. Proc Natl Acad Sci USA 104(6):1947
Gibson DT, Wang KC, Sih CJ, Whitlock H Jr (1966) Mechanisms of steroid oxidation by microorganisms. IX. On the mechanism of ring A cleavage in the degradation of 9,10-seco steroids by microorganisms. J Biol Chem 241(3):551–559
Griffin JE, Pandey AK, Gilmore SA, Mizrahi V, Mckinney JD, Bertozzi CR, Sassetti CM (2012) Cholesterol catabolism by Mycobacterium tuberculosis requires transcriptional and metabolic adaptations. Chem Biol 19(2):218–227
Groot PH, Scholte HR, Hülsmann WC (1976) Fatty acid activation: specificity, localization, and function. Adv Lipid Res 14:75–126
Guo F, Zhang H, Fritzler JM, Rider SD Jr, Xiang L, Mcnair NN, Mead JR, Zhu G (2014) Amelioration of Cryptosporidium parvum infection in vitro and in vivo by targeting parasite fatty acyl-coenzyme A synthetases. J Infect Dis 209(8):1279–1287
Johnston JB, Ouellet H, Montellano PR (2010) Functional redundancy of steroid C26-monooxygenase activity in Mycobacterium tuberculosis revealed by biochemical and genetic analyses. J Biol Chem 285(47):36352–36360
Kendall SL, Withers M, Soffair CN, Moreland NJ, Gurcha S, Sidders B, Frita R, Bokum AT, Besra GS, Lott JS (2007) A highly conserved transcriptional repressor controls a large regulon involved in lipid degradation in Mycobacterium smegmatis and Mycobacterium tuberculosis. Mol Microbiol 65(3):684–699
Léger M, Gavalda S, Guillet V, Rest BVD, Slama N, Montrozier H, Mourey L, Quémard A, Daffé M, Marrakchi H (2009) The dual function of the Mycobacterium tuberculosis FadD32 required for mycolic acid biosynthesis. Chem Biol 16(5):510–519
Liu Y, Chen G, Ge F, Li W, Zeng L, Cao W (2011) Efficient biotransformation of cholesterol to androsta-1,4-diene-3,17-dione by a newly isolated actinomycete Gordonia neofelifaecis. World J Microbiol Biotechnol 27(4):759–765
Lovewell RR, Sassetti CM, Vanderven BC (2016) Chewing the fat: lipid metabolism and homeostasis during M. tuberculosis infection. Curr Opin Microbiol 29:30–36
Meng Y, Rui L, Guja KE, Wipperman M, Clair JRS, Bonds AC, Garciadiaz M, Sampson NS (2015) Unraveling cholesterol catabolism in Mycobacterium tuberculosis: ChsE4-ChsE5 α2β2 Acyl-CoA dehydrogenase initiates β-oxidation of 3-Oxo-cholest-4-en-26-oyl CoA. Acs Infect Dis 1(2):110–125
Mohn WW, Van DGR, Stewart GR, Okamoto S, Liu J, Dijkhuizen L, Eltis LD (2008) The actinobacterial mce4 locus encodes a steroid transporter. J Biol Chem 283(51):35368–35374
Mohn WW, Wilbrink MH, Casabon I, Stewart GR, Liu J, Van DGR, Eltis LD (2012) Gene cluster encoding cholate catabolism in Rhodococcus spp. J Bacteriol 194(24):6712–6719
Nesbitt NM, Yang X (2010) A thiolase of Mycobacterium tuberculosis is required for virulence and production of androstenedione and androstadienedione from cholesterol. Infect Immun 78(1):275–282
Ouellet H, Johnston JB, de Montellano PR (2011) Cholesterol catabolism as a therapeutic target in Mycobacterium tuberculosis. Trends Microbiol 19(11):530–539
Pandey AK, Sassetti CM (2008) Mycobacterial persistence requires the utilization of host cholesterol. Proc Natl Acad Sci USA 105(11):4376–4380
Pei Y, Parreira V, Nicholson VM, Prescott JF (2007) Mutation and virulence assessment of chromosomal genes of Rhodococcus equi 103. Can J Vet Res 71:1–7
Rodríguezgarcía A, Fernándezalegre E, Morales A, Solalanda A, Lorraine J, Macdonald S, Dovbnya D, Smith MC, Donova M, Barreiro C (2016) Complete genome sequence of ‘Mycobacterium neoaurum’ NRRL B-3805, an androstenedione (AD) producer for industrial biotransformation of sterols. J Biotechnol 224:64–65
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Shinnick TM, Good RC (1994) Mycobacterial taxonomy. Eur J Clin Microbiol Infect Dis 13(11):884–901
Soto-Ramirez MD, Aguilar-Ayala DA, Garcia-Morales L, Rodriguez-Peredo SM, Badillo-Lopez C, Rios-Muñiz DE, Meza-Segura MA, Rivera-Morales GY, Leon-Solis L, Cerna-Cortes JF (2017) Cholesterol plays a larger role during Mycobacterium tuberculosis in vitro dormancy and reactivation than previously suspected. Tuberculosis 103:1–9
Sun B, Zeng Q, Luo Z, Yang HJ, Feng Y (2017) Synthesis of (25R)-cholestenoic acid. Chin J Pharm 2017(48):174–178
Trivedi OA, Arora P, Sridharan V, Tickoo R, Mohanty D, Gokhale RS (2004) Enzymic activation and transfer of fatty acids as acyl-adenylates in mycobacteria. Nat 428(6981):441–445
Uhía I, Galán B, Kendall SL, Stoker NG, García JL (2012) Cholesterol metabolism in Mycobacterium smegmatis. Environ Microbiol Rep 4(2):168–182
Vanderven BC, Fahey RJ, Lee W, Liu Y, Abramovitch RB, Memmott C, Crowe AM, Eltis LD, Perola E, Deininger DD (2015) Novel inhibitors of cholesterol degradation in Mycobacterium tuberculosis reveal how the bacterium’s metabolism is constrained by the intracellular environment. PLoS Pathog 11(2):e1004679
Wilbrink MH, Petrusma M, Dijkhuizen L, Geize RVD (2011) FadD19 of Rhodococcus rhodochrous DSM43269, a steroid-coenzyme A ligase essential for degradation of C-24 branched sterol side chains. Appl Environ Microbiol 77(13):44554464
Yang X, Nesbitt NM, Dubnau E, Smith I, Sampson NS (2009) Cholesterol metabolism increases the metabolic pool of propionate in M. tuberculosis. Biochem 48(18):3819–3821
Zhuravleva E, Gut H, Hynx D, Marcellin D, Bleck CK, Genoud C, Cron P, Keusch JJ, Dummler B, Esposti MD (2012) Acyl coenzyme A thioesterase Them5/Acot15 is involved in cardiolipin remodeling and fatty liver development. Mol Cell Biol 32(14):2685–2697
Acknowledgements
This study was funded by the National Natural Science Foundation of China (no. 31571285), the Sichuan provincial Science & Technology Department (2012JY0067), and the Sichuan Provincial Department of Education (17ZA0323), College student innovation, and entrepreneurship training program (201310636026).
Author information
Authors and Affiliations
Contributions
YN, FG, and YY did all the biological and analytical work on acyl-coenzyme A synthetases. YR and XZ did the synthetic work. WL conceived and designed the project. GC, DW, FL, and XY prepared the purified acyl-coenzyme A synthetases. All the authors participated in writing the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest with the contents of this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
13205_2019_1703_MOESM1_ESM.jpg
Fig. S1 Synthesis of LCA-AMS. TBDSC = tert-butyldimethylsilyl chloride; CDI = 1,1-carbonyldiimidazole; DBU =1,8-diazabicyclo [5.4.0] undec-7-ene; TBAF = tetrabutylammonium; THF = tetrahydrofuran (JPEG 63 kb)
Rights and permissions
About this article
Cite this article
Niu, Y., Ge, F., Yang, Y. et al. Biochemical characterization of acyl-coenzyme A synthetases involved in mycobacterial steroid side-chain catabolism and molecular design: synthesis of an anti-mycobacterial agent. 3 Biotech 9, 169 (2019). https://doi.org/10.1007/s13205-019-1703-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-019-1703-y