Abstract
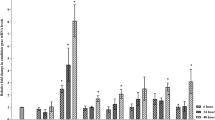
The biocontrol function of the repressor of cellulase expression I (ACE1) in Trichoderma atroviride was verified through constructing Δace1 mutant strain by Agrobacterium tumefaciens-mediated transformation. The activities of cell wall-degrading enzymes (cellulase, xylanase, chitinase, β-1,3-glucanase, and protease) in the supernatant of Δace1 mutant strain were distinctly higher than those of control strain, followed with the elevation of related genes transcript levels. Besides, the Δace1 mutant resulted in an elevating transcript level of xyr1, but no obvious change in the expression of cre1, which suggested that ACE1 was negative regulator of the xyr1 transcription, but not involved in cre1 transcription. On core polyketide synthases of four biosynthesis gene clusters for antibiotic secondary metabolites, only the transcription levels of encoding genes Try83179/TryH and Aza79482/AzaJ in Δace1 mutant strain were significantly higher than that in wild-type during antagonizing with pathogenic fungi Fusarium oxysporum and Rhizoctonia solani (with the inhibition rate of 30.7 and 19.8%, respectively). The biocontrol function of Δace1 mutant strain was remarkably enhanced. The results indicated that ACE1, indeed, acted as a repressor for cell wall-degrading enzymes and PKSs expression in T. atroviride, and the Δace1 mutant strain effectively made related enzymes activities improved with potential enhancement of biocontrol potency.







Similar content being viewed by others
References
Aro N, Ilmén M, Saloheimo A, Penttilä M (2003) ACEI of Trichoderma reesei is a repressor of cellulase and xylanase expression. Appl Environ Microbiol 69:56–65
Bailey MJ, Biely P, Poutanen K (1992) Interlaboratory testing of methods for assay of xylanase activity. J Biotechnol 23:257–270
Baker SE, Perrone G, Richardson NM, Gallo A, Kubicek CP (2012) Phylogenomic analysis of polyketide synthase-encoding genes in Trichoderma. Microbiology 158:147–154
Becker J, Liermann JC, Opatz T, Anke H, Thines E (2012) GKK1032A2, a secondary metabolite from Penicillium sp. IBWF-029-96, inhibits conidial germination in the rice blast fungus Magnaporthe oryzae. J Antibiot (Tokyo) 65:99–102
Bigelis R, He H, Yang HY, Chang LP, Greenstein M (2006) Production of fungal antibiotics using polymeric solid supports in solid-state and liquid fermentation. J Ind Microbiol Biotechnol 33:815–826
Bushley KE, Turgeon BG (2010) Phylogenomics reveals subfamilies of fungal nonribosomal peptide synthetases and their evolutionary relationships. BMC Evol Biol 10:26
Chen J, Zeng X, Yang YL, Xing YM, Zhang Q, Li JM, Ma K, Liu HW, Guo SX (2017) Genomic and transcriptomic analyses reveal differential regulation of diverse terpenoid and polyketides secondary metabolites in Hericium erinaceus. Sci Rep 7:10151
Chiang YM, Oakley BR, Keller NP, Wang CC (2010) Unraveling polyketide synthesis in members of the genus Aspergillus. Appl Microbiol Biotechnol 86:1719–1736
Coradetti ST, Craig JP, Xiong Y, Shock T, Tian C, Glass NL (2012) Conserved and essential transcription factors for cellulase gene expression in ascomycete fungi. Proc Natl Acad Sci USA 109:7397–7402
De Groot MJ, Bundock P, Hooykaas PJ, Beijersbergen AG (1998) Agrobacterium tumefaciens-mediated transformation of filamentous fungi. Nat Biotechnol 16:839–842
Gao X, Chooi YH, Ames BD, Wang P, Walsh CT, Tang Y (2011) Fungal indole alkaloid biosynthesis: genetic and biochemical investigation of the tryptoquialanine pathway in Penicillium aethiopicum. J Am Chem Soc 133:2729–2741
He H, Yang HY, Bigelis R, Solum EH, Greenstein M, Carter G (2002) Pyrrocidines A and B, new antibiotics produced by a filamentous fungus. Tetrahedron Lett 43:1633–1636
Isaka M, Rugseree N, Maithip P, Kongsaeree P, Prabpai S, Thebtaranonth Y (2005) Hirsutellones A–E, antimycobacterial alkaloids from the insect pathogenic fungus Hirsutella nivea BCC 2594. Tetrahedron 61:5577–5583
Isaka M, Prathumpai W, Wongsa P, Tanticharoen M (2006) Hirsutellone F, a dimer of antitubercular alkaloids from the seed fungus Trichoderma species BCC 7579. Org Lett 8:2815–2817
Kubicek CP, Herrera-Estrella A, Seidl-Seiboth V, Martinez DA, Druzhinina IS, Thon M, Zeilinger S, Casas-Flores S, Horwitz BA (2011) Comparative genome sequence analysis underscores mycoparasitism as the ancestral life style of Trichoderma. Genome Biol 12:R40
Li C, Yang Z, Zhang RH, Zhang D, Chen S, Ma L (2013a) Effect of pH on cellulase production and morphology of Trichoderma reesei and the application in cellulosic material hydrolysis. J Biotechnol 168:470–477
Li XW, Ear A, Nay B (2013b) Hirsutellones and beyond: figuring out the biological and synthetic logics toward chemical complexity in fungal PKS–NRPS compounds. Natl Prod Rep 30:765–782
Li Q, Ng WT, Wu JC (2014) Isolation, characterization and application of a cellulose-degrading strain Neurospora crassa S1 from oil palm empty fruit bunch. Microb Cell Fact 13:157
Li Y, Sun R, Yu J, Saravanakumar K, Chen J (2016) Antagonistic and biocontrol potential of Trichoderma asperellum ZJSX5003 against the Maize Stalk Rot Pathogen Fusarium graminearum. Indian J Microbiol 56:318–327
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408
Mach-Aigner AR, Pucher ME, Steiger MG, Bauer GE, Preis SJ, Mach RL (2008) Transcriptional regulation of xyr1, encoding the main regulator of the xylanolytic and cellulolytic enzyme system in Hypocrea jecorina. Appl Environ Microbiol 74:6554–6562
Martinez D, Berka RM, Henrissat B, Saloheimo M, Arvas M, Baker SE, Chapman J, Chertkov O, Coutinho PM (2008) Genome sequencing and analysis of the biomass-degrading fungus Trichoderma reesei (syn. Hypocrea jecorina). Nat Biotechnol 26:553–560
Mukherjee PK, Buensanteai N, Moran-Diez ME, Druzhinina IS, Kenerley CM (2012) Functional analysis of non-ribosomal peptide synthetases (NRPSs) in Trichoderma virens reveals a polyketide synthase (PKS)/NRPS hybrid enzyme involved in the induced systemic resistance response in maize. Microbiology 158:155–165
Noronha EF, Ulhoa CJ (2000) Characterization of a 29-kDa beta-1,3-glucanase from Trichoderma harzianum. FEMS Microbiol Lett 183:119–123
Oda S, Kameda A, Okanan M, Sakakibara Y, Ohashi S (2015) Discovery of secondary metabolites in an extractive liquid-surface immobilization system and its application to high-throughput interfacial screening of antibiotic-producing fungi. J Antibiot (Tokyo) 68:691–697
Oikawa H (2003) Biosynthesis of structurally unique fungal metabolite GKK1032A(2): indication of novel carbocyclic formation mechanism in polyketide biosynthesis. J Org Chem 68:3552–3557
Osmanova N, Schultze W, Ayoub N (2010) Azaphilones: a class of fungal metabolites with diverse biological activities. Phytochem Rev 9:315–342
Saloheimo A, Aro N, Ilmén M, Penttilä M (2000) Isolation of the ace1 gene encoding a Cys(2)-His(2) transcription factor involved in regulation of activity of the cellulase promoter cbh1 of Trichoderma reesei. J Biol Chem 275:5817–5825
Saxena A, Raghuwanshi R, Singh HB (2015) Trichoderma species mediated differential tolerance against biotic stress of phytopathogens in Cicer arietinum L. J Basic Microbiol 55:195–206
Schmoll M, Kubicek CP (2003) Regulation of Trichoderma cellulase formation: lessons in molecular biology from an industrial fungus. A review. Acta Microbiol Immunol Hung 50:125–145
Schuster A, Schmoll M (2010) Biology and biotechnology of Trichoderma. Appl Microbiol Biotechnol 87:787–799
Strauss J, Mach RL, Zeilinger S, Hartler G, Stöffler G, Wolschek M, Kubicek CP (1995) Cre1, the carbon catabolite repressor protein from Trichoderma reesei. FEBS Lett 376:103–107
Viterbo A, Wiest A, Brotman Y, Chet I, Kenerley C (2007) The 18mer peptaibols from Trichoderma virens elicit plant defence responses. Mol Plant Pathol 8:737–746
Zheng F, Cao Y, Wang L, Lv X, Meng X, Zhang W, Chen G, Liu W (2017) The mating type locus protein MAT1-2-1 of Trichoderma reesei interacts with Xyr1 and regulates cellulase gene expression in response to light. Sci Rep 7:17346
Acknowledgements
We really appreciate all the useful comment and support from our colleagues in Jiangxi University of Technology, China.
Funding
This work was supported by Jiangxi Provincial Science and Technology Project of Ministry of Education (Project No.: GJJ161137) and Natural Science Project of Jiangxi Science and Technology College (Project No.: 16ZRYB03).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no competing interests.
Ethical statement
All procedures of this study were complied with local experimental operation specification. In addition, no relevant ethical statement was needed to be declared.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fang, C., Chen, X. Potential biocontrol efficacy of Trichoderma atroviride with cellulase expression regulator ace1 gene knock-out. 3 Biotech 8, 302 (2018). https://doi.org/10.1007/s13205-018-1314-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-018-1314-z