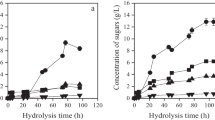
Abstract
One of the tannase isoforms produced by the fungus Aspergillus fumigatus CAS-21 under submerged fermentation (SbmF) was purified 4.9-fold with a 10.2% recovery. The glycoprotein (39.1% carbohydrate content) showed an estimated molecular mass of 60 kDa. Optimum temperature and pH for its activity were 30–40 °C and 5.0, respectively. It showed a half-life (t50) of 60 min at 45 and 50 °C, and it was stable at pH 5.0 and 6.0 for 3 h. The tannase activity was insensitive to most salts used, but it reduced in the presence of Fe2(SO4)3 and FeCl3. On contrary, in presence of SDS, Triton-X100, and urea the enzyme activity increased. The Km value indicated high affinity for propyl gallate (3.61 mmol L−1) when compared with tannic acid (6.38 mmol L−1) and methyl gallate (6.28 mmol L−1), but the best Kcat (362.24 s−1) and Kcat/Km (56.78 s−1 mmol−1 L) were obtained for tannic acid. The purified tannase reduced 89 and 25% of tannin content of the leather tannery effluent generated by manual and mechanical processing, respectively, after 2-h treatment. The total phenolic content was also reduced. Additionally, the enzyme produced propyl gallate, indicating its ability to do the transesterification reaction. Thus, A. fumigatus CAS-21 tannase presents interesting properties, especially the ability to degrade tannery effluent, highlighting its potential in biotechnological applications.





Similar content being viewed by others
References
Abdel-Nabey MA, El-Tanash AB, Sherief ADA (2016) Structural characterization, catalytic, kinetic and thermodynamic properties of Aspergillus oryzae tannase. Int J Biol Macromol 92:803–811
Agostini-Costa TS, Garruti DS, Lima L, Freire S, Abreu FAP, Feitosa T (1999) Avaliação de metodologias para determinação de taninos no suco de caju. Bol Cent Pesqui Process Aliment 17:167–176
Aguilar CN, Rodríguez R, Gutiérrez-Sánchez G, Augur C, Favela-Torres E, Prado-Barragan LA, Ramírez-Coronel A, Contreras-Esquivel JC (2007) Microbial tannases: advances and perspectives. Appl Microbiol Biotechnol 76:47–59
Beena PS, Soorej MB, Elyas KK, Sarita GB, Chandrasekaran M (2010) Acidophilic tannase from marine Aspergillus awamori BTMFW032. Int J Microbiol Biotechnol 20:1403–1414
Beniwal V, Kumar A, Sharma J, Chhokar V (2013) Recent advances in industrial application of tannases: a review. Recent Pat Biotechnol 7:228–233
Böer E, Breuer FS, Weniger M, Denter S, Piontek M, Kunze G (2011) Large-scale production of tannase using the yeast Arxula adeninivorans. Appl Microbiol Biotechnol 92:105–114
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Candiano G, Bruschi M, Musante L, Ghiggeri GM, Carnemolla B, Orecchia P, Zardi L, Righetti PG (2004) Blue silver: a very sensitive colloidal Coomassie G-250 staining for proteome analysis. Electrophoresis 25:1327–1333
Chhokar V, Sangwan M, Beniwal V, Nehra K, Nehra KS (2010) Effect of additives on the activity of tannase from Aspergillus awamori MTCC9299. Appl Biochem Biotechnol 160:2256–2264
Costa AM, Ribeiro WX, Kato E, Monteiro ARG, Peralta RM (2008) Production of tannase by Aspergillus tamarii in submerged cultures. Braz Arch Biol Technol 51:399–404
Costa AM, Kadowaki MK, Minozzo MC, Souza CGM, Boer CG, Bracht A, Peralta RM (2012) Production, purification and characterization of tannase from Aspergillus tamarii. Afr J Biotechnol 11:391–398
Dixit S, Yadav A, Dwivedi PD, Das M (2015) Toxic hazards of leather industry and technologies to combat threat: a review. J Clean Prod 87:39–49
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Durai G, Rajasimman M (2011) Biological treatment of tannery wastewater—a review. J Environ Sci Technol 4:1–17
El-Fouly MZ, El-Awamry Z, Shahin AAM, El-Bialy HA, Naeem E, El-Saed G (2010) Biosynthesis and characterization of Aspergillus niger AUMC 4301 tannase. J Am Sci 6:709–721
Fernandez-Lorente G, Bolivar JM, Rocha-Martin J, Curiel JA, Muñoz R, Rivas B, Carrascos AV, Guisan JM (2011) Synthesis of propyl gallate by transesterification of tannic acid in aqueous media by derivatives of tannase from. Lactobacillus plantarum Food Chem 128:214–217
Goel G, Puniya AK, Aguilar CN, Sing K (2005) Interaction of gut microflora with tannins in feeds. Sci Nat 92:497–503
Gonçalves HB, Riul AJ, Terenzi HF, Jorge JA, Guimarães LHS (2011) Extracellular tannase from Emericella nidulans showing hypertolerance to temperature and organic solvents. J Mol Catal B Enzym 71:29–35
Gonçalves HB, Riul AJ, Quiapin AC, Jorge JA, Guimarães LHS (2012) Characterization of a thermostable extracellular tannase produced under submerged fermentation by Aspergillus ochraceus. Electron J Biotechnol 15:1–12
Hagerman AE, Butler LG (1978) Protein precipitation method for the quantitative determination of tannins. J Agric Food Chem 26:809–812
Hatamoto O, Watarai T, Kikuchi M, Mizusawa M, Sekine H (1996) Cloning and sequencing of the gene encoding tannase and a structural study of the tannase subunit from Aspergillus oryzae. Gene 175:215–221
Kar B, Banerjee R, Bhattacharyya BC (1999) Microbial production of gallic acid by modified solid-state fermentation. J Ind Microbiol Biotechnol 23:173–177
Kasieczka-Burnecka M, Karina K, Kalinowska H, Knap M, Turkiewicz M (2007) Purification and characterization of two cold-adapted extracellular tannin acyl hydrolases from an Antarctic strain Verticillium sp. P9. Appl Microbiol Biotechnol 77:77–89
Kumar S, Beniwal V, Kumar N, Kumar A, Chhokar V, Khalet TP (2015) Biochemical characterization of immobilized tannase from Aspergillus awamori. Biocatal Agric Biotechnol 4:398–403
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lal D, Gardner JJ (2012) Production, characterization and purification of tannase from Aspergillus niger. Eur J Exp Biol 2:1430–1438
Lekha PK, Lonsane BK (1997) Production and application of tannin acyl hydrolase: state of the art. Adv Appl Microbiol 44:215–260
Leone FA, Degreve L, Baranauskas JA (1992) SIGRAF: a versatile computer program for fitting enzyme kinetic data. Biochem Mol Biol Educ 20:94–96
Liu TPS, Costa RMPB., Freitas DJV, Nascimento CO, Motta CMS, Bezerra RP, Herculano PN, Porto ALF (2017) Tannase from Aspergillus melleus improves the antioxidant activity of green tea: purification and biochemical characterization. Int J Food Sci Technol 52:652–661
Lofrano G, Meriç S, Zengin GE, Orhon D (2013) Chemical and biological treatment technologies for leather tannery chemicals and wastewaters: a review. Sci Total Environ 461–462:265–281
Mahapatra K, Nanda RK, Baq SS, Banerjee R, Pandev A, Szakacs G (2005) Purification, characterization and some studies on secondary structure of tannase from Aspergillus awamori nakazawa. Process Biochem 40:3251–3254
Mata-Gomez M, Rodríguez LV, Ramos EL, Renovato J, Cruz-Hernandez MA, Rodríguez R, Contreras J, Aguilar CN (2009) A novel tannase from the xerophilic fungus Aspergillus niger GH1. J Microbiol Biotechnol 19:987–996
Mizuno T, Shiono Y, Koseki T (2014) Biochemical characterization of Aspergillus oryzae native tannase and the recombinant enzyme expressed in Pichia pastoris. J Biosci Bioeng 118:392–395
Mukherjee G, Banerjee R (2006) Effects of temperature, pH and additives on the activity of tannase produced by a co-culture of Rhizopus oryzae and Aspergillus foetidus. World J Microbiol Biotechnol 22:207–212
Murugan K, Al-Sohaibani SA (2010) Biocompatible removal of tannin and associated color from tannery effluent using the biomass and tannin acyl hydrolase (EC 3.1.20) enzymes of mango industry solid waste isolate Aspergillus candidus MTTC 9628. Res J Microbiol 5:262–271
Pinto GAS, Gouri S, Leite SGF, Brito ES (2005) Tanase: conceitos, produção e aplicação. Bol Cent Pesqui Process Aliment 23(2):435–462
Qiu YU, Niu H, Huang W, He Y, Wu X-H (2011) Properties and secondary structure of tannase from Penicillium herquei. Biotechnol Bioprocess Eng 16:858–866
Riul AJ, Gonçalves HB, Jorge JA, Guimarães LHS (2013) Characterization of a glucose- and solvent-tolerant extracellular tannase from Aspergillus phoenicis. J Mol Catal B Enzym 85/86:126–133
Rodríguez-Durán LV, Valdivia-Urdiales B, Contreras-Esquivel JC, Rodríguez-Herrera R, Aguilar CN (2011) Novel strategies for upstream and downstream processing of tannin acyl hydrolase. Enzym Res 2011:1–20
Romero-Dondiz EM, Almazán JE, Rajal VB, Castro-Vidaurre EF (2015) Removal of vegetable tannins to recover water in the leather industry by ultrafiltration polymeric membranes. Chem Eng Res Des 93:727–735
Sabu A, Kiran GS, Pandey A (2005) Purification and characterization of tannin acyl hydrolase from Aspergillus niger ATCC 16620. Food Technol Biotechnol 43:133–138
Salgado JM, Abrunhosa L, Venancio A, Domínguez JM, Belo I (2016) Combined bioremediation and enzyme production by Aspergillus sp. In olive mill and winery wastewaters. Int Biodeterior Biodegrad 110:16–23
Santos SC, Mello JPC (2001) Taninos In: Simões CMO (ed) Em Farmacognosia: da planta ao medicamento, 3rd edn. UFSC, Porto Alegre, pp 615–655
Sena AR, Santos ACB, Gouveia MJ, Mello MRFM., Leite TCC, Moreira KA, Assis SA (2014) Production of tannase by Pestalotiopsis guepinii. Food Technol Biotechnol 52:459–467
Sharma S, Saxena RK (2012) Evaluation of the versatility of the tannases produced from Aspergillus niger and Penicillium variable with respect to gallic acid production, gallate ester synthesis, animal feed improvement, tannery effluent degradation and tannin stain removal. Res Biotechnol 3:9–20
Sharma S, Bhat TK, Dawra RK (1999) Isolation, purification and properties of tannase from Aspergillus niger van Tieghem. World J Microbiol Biotechnol 15:673–677
Sharma S, Bhat TK, Dawra RK (2000) A spectrophotometric method for assay of tannase using rhodamine. Anal Biochem 279:85–89
Sharma S, Argawal L, Saxena RK (2008) Purification, immobilization and characterization of tannase from Penicillium variable. Bioresour Technol 99:2544–2551
Valera LS, Jorge JA, Guimarães LHS (2015) Characterization of a multi-tolerant tannin acyl hydrolase II from Aspergillus carbonarius produced under solid-state fermentation. Electron J Biotechnol 18:464–470
Yao J, Guo GS, Ren GH, Liu YH (2014) Production, characterization and applications of tannase. J Mol Catal B Enzym 101:137–147
Acknowledgements
The authors gratefully acknowledge the financial support from Fundação de Amparo à Pesquisa do Estado de São Paulo—FAPESP (2016/11311-5) and the research scholarships from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES). This manuscript is a part of the Master Degree dissertation from R.M.F.C. We also thank Maurício de Oliveira and Dr. Eduardo J. Crevelin for the technical assistance, and Carlos Ambrosio from ARTEZA, who provided the effluent samples.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Cavalcanti, R.M.F., Jorge, J.A. & Guimarães, L.H.S. Characterization of Aspergillus fumigatus CAS-21 tannase with potential for propyl gallate synthesis and treatment of tannery effluent from leather industry. 3 Biotech 8, 270 (2018). https://doi.org/10.1007/s13205-018-1294-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-018-1294-z