Abstract
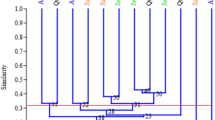
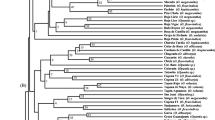
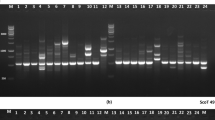
Ornamental purslanes (Portulaca L.) are a popular annual bedding and container plant for landscaping. Little information is available concerning the genetic characterization of ornamental purslane resources thus far. The purpose of this study was to investigate the genetic diversity and relationships present in a collection of ornamental purslanes from Portulaca umbraticola and P. grandiflora cultivated in China, using sequence-related amplified polymorphism (SRAP) markers. The genotyping showed that 16 SRAP primer combinations totally produced 261 informative fragments and averaged 16.31 per primer combination. The major allele frequency and Nei’s gene diversity was calculated at 0.78 and 0.31 across the loci, indicative of a moderate low diversity. Both unweighted pair group method with arithmetic average (UPGMA) clustering and a Bayesian-based approach apparently assigned the whole accessions into two sub-groups: P. umbraticola and P. grandiflora, well concordant with the botanical classification and flower type. The findings provide a brandnew understanding of genetic diversity and population structure present in ornamental purslane, and benefit a sound design of breeding programs in future.



Similar content being viewed by others
References
Aneja B, Yadav NR, Chawla V, Yadav RC (2012) Sequence-related amplified polymorphism (SRAP) molecular marker system and its implications in crop improvement. Mol Breed 30:1635–1648
Bar C, Doğanlar S, Frary A (2015) Genetic relationships among Eurasian Puccinellia distans genotypes. Biochem Syst Ecol 62:20–24
Carolin RG (1993) Portulacaceae. In: Kubitzki K, Rhower JB, Bittrich V (eds) The families and genera of vascular plants. flowering plants—Dicotyledons 2. Springer, Berlin
Chan K, Islam MW, Kamil M, Radhakrishnan R, Zakaria MN, Habibullah M, Attas A (2000) The analgesic and anti-inflammatory effects of Portulaca oleracea L. subsp. sativa (Haw.) Celak. J Ethnopharmacol 73:445–451
Coelho AAOP, Giulietti AM, Harley RM, Yesilyurt JC (2010) Synonymies and typifications in Portulaca (Portulacaeae) of Brazil. Kew Bull 65:37–43
Eggli U, Ford-Werntz D (2002) Illustrated handbook of succulent plants—Dicotyledons. Portulacaceae. Springer, New York
El Jack AE (2004) Portulaca oleracea L. In: Gubben GJH, Denton OA (eds) Plant resources of tropical Africa 2: vegetables/Légumes. Backhuys Publishers, Wageningen, pp 426–428
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791
Geesink R (1969) An account of the genus Portulaca in Indo-Australia and the Pacific (Portulacaceae). Blumea 17:275–307
Hubisz MJ, Falush D, Stephens M, Pritchard JK (2009) Inferring weak population structure with the assistance of sample group information. Mol Ecol Resour 9:1322–1332
Khanuja SPS, Shasany AK, Darokar MP, Kumar S (1999) Rapid isolation of DNA from dry and fresh samples of plants producing large amounts of secondary metabolites and essential oils. Plant Mol Biol Rep 17:74
Klie M, Menz I, Linde M, Debener T (2013) Lack of structure in the gene pool of the highly polyploidy ornamental chrysanthemum. Mol Breed 32:339–348
Li G, Quiros CF (2001) Sequence-related amplified polymorphism (SRAP), a new marker system based on a simple PCR reaction: its application to mapping and gene tagging in Brassic. Theor Appl Genet 103:455–461
Li P, Zhang F, Chen S, Jiang J, Wang H, Su J, Fang W, Guan Z, Chen F (2016) Genetic diversity, population structure and association analysis in cut chrysanthemum (Chrysanthemum morifolium Ramat.). Mol Genet Genom 291:1117–1125
Lim YY, Quah EPL (2006) Antioxidant properties of different cultivars of Portulaca oleracea. Food Chem 103:734–740
Liu K, Muse SV (2005) PowerMarker: an integrated analysis environment for genetic marker analysis. Bioinformatics 21:2128–2129
Mihailovic N, Andrejić G, Dželetović Ž (2015) Tolerance of Portulaca grandiflora to individual and combined application of Ni, Pb and Zn. Bull Environ Contam Toxicol 94:103–107
Nei M, Li WH (1979) Mathematical model for studying genetic variation in terms of restriction endonucleases. Proc Natl Acad Sci USA 76:5269–5273
Ocampo G, Columbus JT (2012) Molecular phylogenetics, historical biogeography, and chromosome number evolution of Portulaca (Portulacaceae). Mol Phylogenet Evol 63:97–112
Ohsaki A, Shibata K, Kubota T, Tokoroyama T (1999) Phylogenetic and chemotaxonomic significance of diterpenes in some Portulaca species (Portulacaceae). Biochem Syst Ecol 27:289–296
Pritchard JK, Falus D (2009) Documentation for STRUCTURE Software: version 2.3. The University of Chicago Press, Chicago
Rahdari P, Hoseini SM (2012) Effect of different levels of drought stress (PEG 6000 concentrations) on seed germination and inorganic elements content in purslane (Portulaca oleracea L.) leaves. J Stress Physiol Biochem 8:51–61
Rohlf FJ (2005) NTSYS-pc numerical taxonomy and multivariate analysis system. Version 2.2. Exeter Software, New York
Simsek O, Curuk P, Aslan F, Bayramoglu M, Izgu T, da Silva JAT, Kacar YA, Mendi YY (2017) Molecular characterization of Cyclamen species collected from different parts of Turkey by RAPD and SRAP markers. Biochem Genet 55:87–102
Teixeira M, Carvalho IS (2009) Effects of salt stress on purslane (Portulaca oleracea) nutrition. Ann Appl Biol 154:77–86
Thangavel P, Subburam V (1998) Effect of trace metals on the restoration potential of leaves of the medicinal plant, Portulaca oleracea Linn. Biol Trace Elem Res 61:313–321
Valdez-Ojeda R, James-Kay A, Ku-Cauich JR, Escobedo-GraciaMedrano RM (2014) Genetic relationships among a collection of Musa germplasm by fluorescent-labeled SRAP. Tree Genet Genomes 10:465–476
Venu-Babu P, Ogale VK, Mishra SD (1996) Rosetteness in Portulaca grandiflora: an altered genetic expression. Mol Biol Rep 23:119–121
Wickramasinghe P, Harrison DK, Johnston ME (2009) Reproductive biology and intergeneric breeding compatibility of ornamental purslanes and Calandrinia (Portulacaceae). Aust J Bot 57:697–707
Yezici I, Türkan I, Sekmen AH, Demiral T (2007) Salinity tolerance of purslane (Portulaca oleracea L.) is achieved by enhances antioxidative system, lower level of lipid peroxidation and proline accumulation. Environ Exp Bot 61:49–57
Yu C, Yin Y, Creech DL, Lu Z, Xu J (2016) Morphological characters and SRAP analysis of two hybrids between Hibiscus dasycalyx and Hibiscus ‘Moy Grande’. Sci Hortic 198:118–124
Zhang F, Chen S, Chen F, Fang W, Chen Y, Li F (2011) SRAP-based mapping and QTL detection for inflorescence-related traits in chrysanthemum (Dendranthema morifolium). Mol Breed 27:11–23
Zhang F, Ge Y, Wang W, Shen X, Liu X, Liu J, Tian D, Yu X (2012) Genetic diversity and population structure of cultivated bromeliad accessions assessed by SRAP markers. Sci Hortic 141:1–6
Acknowledgements
The present research was partly financed by Top-notch Academic Programs Project of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Jia, S., Yan, Z., Wang, Y. et al. Genetic diversity and relatedness among ornamental purslane (Portulaca L.) accessions unraveled by SRAP markers. 3 Biotech 7, 241 (2017). https://doi.org/10.1007/s13205-017-0881-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-017-0881-8