Abstract
The use of low-cost carbon substrates such as agricultural residues can drastically lower the cost of biosurfactant production. In this study, rice bran oil extracted from agricultural waste was used as a renewable carbon source for biosurfactant production using Pseudomonas aeruginosa PTCC 1340. The biosurfactant was characterized as a glycolipid derivative by thin-layer chromatography and Fourier transform infrared spectroscopy. The yields of biosurfactant from rice bran oil (YRL/S) and biosurfactant to biomass (YRL/X) were 0.246 and 2.81 (g/g), respectively. In addition, the surface activity of the produced biosurfactant was studied using dynamic surface tension measurements and a mono-exponential decay model by estimating the relaxation time of the biosurfactants at the interface. The biosurfactant exhibited acceptable performance in reducing surface tension, as confirmed by examining the dynamic surface tension state and the lowest adsorption time without being affected by the type of salt or concentration. It was found that the adsorption/relaxation of biosurfactants at the interface was considerably affected by the biosurfactant concentration. The produced biosurfactant by the strain considerably reduced the surface tension of water from 70.46 to 25.86 mN/m with a critical micelle concentration (CMC) of 0.09 g/L with rice bran oil as a carbon source. The biosurfactant was also found to be highly effective in suppressing one of the most destructive pathogenic fungi, Macrophomina phaseolina, in terms of its environmental impact. The enhanced physicochemical properties of biosurfactants, such as potential antifungal properties, oil displacement properties, and surface tension-reducing ability, demonstrate the potential of this biosurfactant as a bio-adjuvant and perfect replacement for chemical surfactants in addressing oil spills and environmental decontamination processes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Biosurfactants are secondary metabolites with valuable properties such as high surface activity, biodegradability, low toxicity, and good performance over a wide range of temperature and pH conditions (Geetha et al. 2018). Biosurfactants, like other chemical surfactants, include glycolipids, neutral lipids, fatty acids, phospholipids, lipopeptides, and lipopolysaccharides (Jahan et al. 2020). It should be noted that glycolipids are known as the most familiar class of microbially produced surface-active compounds. They consist of long-chain aliphatic acids, hydroxyl aliphatic acids, and hydrophilic carbohydrates. Among the glycolipid groups, rhamnolipids are the most important and extensively studied biosurfactants (Henkel et al. 2012). Owing to the capabilities of biosurfactants (such as reducing the surface tension or emulsion power), they have the potential to be used in a variety of fields, including chemicals, the microbial enhanced oil recovery (MEOR) branch of petroleum (Hajibagheri et al. 2017, Ashish and Debnath 2018, Liu et al. 2018), oil spill bioremediation (Chen et al. 2020), crude oil biodegradation (Ray et al. 2022), pest control (Ben Khedher et al. 2017), corrosion inhibition potential (Verma et al. 2023), food and feed packaging (Zouari et al. 2016), wettability of the compost surface of food waste (Heidarzadeh et al. 2022), and biochemistry, as well as other biosurfactant applications and advanced technology areas (Mozaffari et al. 2021; Ghasemi et al. 2022; Sarubbo et al. 2022).
The production of biosurfactants is not industrialized because of the high cost of substrates (Geetha et al. 2018). The use of inexpensive natural renewable materials, agricultural and industrial waste, and industrial and municipal wastewater as alternative substrates helps prevent the accumulation of these substances in nature and contributes to the production of value-added materials (Henkel et al. 2012). This topic has attracted the attention of several researchers (Henkel et al. 2012; Geetha et al. 2018). Vegetable oils, by-products, and renewable agricultural wastes have been used as substrates for biosurfactant production (Gaur et al. 2022). Among the various possible substrates for biosurfactant production, vegetable oil is one of the most commonly examined (Jadhav et al. 2019; Ghorbani et al. 2022). In general, vegetable oils are the major source of lipids, which often comprise relative proportions of saturated and unsaturated fatty acids. In many countries, only a small portion of rice bran is used for the production of edible oil because of the high levels of free fatty acids produced by the enzymatic activity of lipase immediately after rice milling (Zúñiga-Diaz et al. 2017). Accordingly, several types of vegetable oils (including oils extracted from canola, corn, sunflower, safflower, olive, grape seed, palm, coconut, fish, and soy) have been examined for the production of biosurfactants over the past two decades (Henkel et al. 2012; Zúñiga-Diaz et al. 2017; Gaur et al. 2022). Amani et al. (2013) investigated the surface tension of P. aeruginosa MM1011, which was grown in 250 g/L of sunflower oil as the sole carbon source, and discovered that it decreased by 26 mN/m in a cell-free broth containing a biosurfactant with a CMC of 0.120 g/L. Mishra et al. (2021) optimized the production of rhamnolipid biosurfactant prepared from P. aeruginosa MTCC 424 using rice bran oil as a carbon source. The results showed that the CMC of the optimized biosurfactant was 28.5 mN/m, observed at 120 mg/L rhamnolipid biosurfactant. They reported rhamnolipid production of 6.2 g/L. Moya Ramírez et al. (2016) investigated fermentation studies with a strain of P. aeruginosa grown in olive mill waste as the sole carbon source and reported biosurfactant production of 29.5 mg/L.
On the other hand, Macrophomina phaseolina is one of the most harmful plant pathogens, infecting many plants. These fungi have been found in peanuts, cabbages, peppers, peas, soybeans, sunflowers, sweet potatoes, alfalfa, sesame, potatoes, sorghum, wheat, and maize (Pandey et al. 2020). Several studies have demonstrated the antifungal properties of biosurfactants (Goswami and Deka 2019). The identification of M. phaseolina is usually based on its morphology and attempts to divide the pathogen into subspecies. However, most of these criteria are unreliable because of important variations within the isolation phenotype (Pandey et al. 2020). The misidentification of M. phaseolina using conventional culture-based morphological techniques has led scientists to improve molecular approaches based on highly sensitive nucleic acids and specific polymerase chain reaction (PCR) methods (Mirmajlessi et al. 2015). M. phaseolina affects the fibrovascular system of roots and basal internodes of the host, preventing water and nutrients from moving to the upper parts of plants. Progressive dejection, early death, loss of power, and loss of function are the hallmarks of M. phaseolina (Mirmajlessi et al. 2015). Sathi Reddy et al. (2016) investigated mango kernel oil as a substrate for P. aeruginosa DR1 biosurfactant production and assessed the antifungal efficacy of the biosurfactant against M. phaseolina. They reported a visible mycelial growth inhibition of 60.46% against M. phaseolina. Biocontrol activity may vary based on the differences in the congeners present in the biosurfactant mixture produced by P. aeruginosa from several carbon sources.
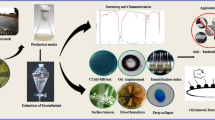
In this study, the effects of carbon sources (including rice bran oil from agricultural waste extracted in the laboratory, industrial rice bran oil, and sunflower oil) on the production of a biosurfactant from Pseudomonas aeruginosa, a common rhamnolipid-producing microorganism (Meng et al. 2019), were investigated (see the flowchart of the current study in Fig. 1). The molecular structure of the biosurfactant was evaluated using Fourier transform infrared spectroscopy (FTIR) and thin-layer chromatography (TLC). In addition, dynamic surface tension was measured to evaluate the surface activity of the biosurfactant. It is well known that surface tension changes with time, known as dynamic surface tension, which directly influences the performance of surface-active compounds. Because most researchers have considered the equilibrium surface tension as a function of biosurfactant concentration, ample space in the area of dynamic surface tension measurement is still available for further research. The dynamic behavior of the surface tension of any surfactant is caused by the polar part of the compounds with the adsorption kinetics of surface-active molecules at the interface, which is controlled by the movement of molecules from the bulk phase to the interface, or vice versa. In addition to measuring the equilibrium surface tension and determining the CMC of the biosurfactant, this study aimed to determine the effects of the biosurfactant concentration on the dynamic surface tension. In addition, the measured dynamic surface tensions were modeled using a monoexponential decay model. To the best of our knowledge, there are no reports in the literature on the evaluation of surface activity of rhamnolipid biosurfactants through dynamic surface tension. Finally, the antifungal activity of the produced biosurfactant (rhamnolipid) against M. phaseolina was investigated considering the environmental considerations.
Materials and methods
Carbon source
Crude rice bran oil extracted from agricultural waste was used as a carbon source for biosurfactant production. Rice bran was collected after milling at a local mill (Babol, Mazandaran, Iran). Rice bran oil and sunflower oil (food grade; Oila, Tehran, Iran) were used as comparative carbon sources. The solution was then autoclaved at 121 °C for 20 min.
Extraction of rice bran oil
In the first stage, the collected rice bran was cleaned and rinsed, and then dried in an air-circulating oven for approximately 48 h. The prepared bran was then mixed with hexane for 72 h at a mass ratio of 1:3 bran to hexane. After mixing, the mixture was placed in a Soxhlet apparatus for 12 h. The extracted oil was vigorously mixed with 1% w/w water to remove resinous materials by centrifugation at 4700 rpm for 30 min (Z 326 K, HERMLE, Germany) (Henkel et al. 2012; Zúñiga-Diaz et al. 2017), and rice bran oil was measured gravimetrically.
Microorganisms
In this study, the microorganism of P. aeruginosa PTCC 1340 was obtained from the Persian-type culture collection (IROST, Tehran, Iran). P. aeruginosa is a common organism that produces rhamnolipids. P. aeruginosa is an aerobic and gram-negative organism that exists in water, soil, and vegetation. This organism can grow and survive in most conditions, making it a good candidate for various processes.
Production of the rhamnolipid biosurfactant
Pre-cultivation condition
LB broth (MILLER) was used for precultivation (Walter 2009). First, two loops of bacteria were transferred to 100 mL of LB broth medium in a 500 mL Erlenmeyer flask under sterile conditions. The flask was then incubated in an incubator shaker at 150 rpm and 37 °C for 24 h (Amani 2015).
Cultivation condition
For the seed culture, the composition (g/L) of a Ca-free mineral salt medium (MSM) is MgSO4 ·7H2O (0.05), KCl (0.1), NaNO3 (1.5), Na2HPO4·2H2O (0.7862), and NaHPO4·H2O (1.1627). The production medium, adjusted to pH 6.8, was autoclaved at 121 °C for 20 min. In addition, one mL of a trace element solution was filter-sterilized through a 0.22 μm membrane filter (Merck, Germany). The used trace element solution consists of 2.0 g Na3C6H5O7·2H2O, 0.3 g FeCl3·6H2O, 1.4 g ZnSO4·7H2O, 1.2 g CoCl2·6H2O, 1.2 g CuSO4·5H2O, 0.8 g MnSO4·H2O, and pH of 7 in 1 L of distilled water. Analytical grade chemicals were supplied by Merck (Darmstadt, Germany). After this period, 5% of the preculture and 50 g/L of carbon source were added to the medium to make 100 mL of solution in a 500 mL flask under sterile conditions, and cultivation was carried out at 150 rpm and 37 °C. Samples were collected for analysis every 24 h for 168 h (Amani 2015).
Extraction and purification of rhamnolipid
The prepared samples were thoroughly mixed with n-hexane (1:1 v/v). This was followed by centrifugation at 4700 rpm and 4 °C for 30 min to remove residual plant oil. The cell-oil-free broth was then adjusted to an acidic pH values of 2–3 by adding 10% H3PO4. The precipitate was then extracted with equal amounts of ethyl acetate, and rhamnolipid substances were obtained by evaporating the ethyl acetate (Amani 2015).
Measurement
Measurement of the rhamnolipid concentration
The concentration of rhamnolipids produced in rhamnose (g/L) was measured using a colorimetric method with the aid of an orcinol reaction (Rahman et al. 2010; Raza et al. 2014). The orcinol reagent was prepared from 0.19% 5-methyl resorcinol in 53% sulfuric acid. The samples were centrifuged at 4700 rpm and 4 °C for 30 min, followed by a 50-fold dilution. Next, 4.5 mL of orcinol reagent was added to the diluted sample (0.5 mL), which was then placed in a water bath at 80 °C for 30 min. The prepared solution was cooled to room temperature and its absorbance was measured using a spectrophotometer at 421 nm. Rhamnose concentrations were calculated using a standard curve prepared using rhamnose sugar. To obtain the concentration of rhamnolipids produced by P. aeruginosa, the amount of rhamnose is usually multiplied by a factor of 3.4, which shows the relationship between rhamnose and rhamnolipids (Rahman et al. 2010; Raza et al. 2014).
Measurement of dry biomass
The samples were mixed with equal amounts of n-hexane and centrifuged at 4700 rpm and 4 °C for 30 min to remove residual plant oil. The cell pellets obtained after the first centrifugation step were carefully washed with a 0.9% sodium chloride solution (4700 rpm, 30 min). After the evaporation of water in an oven at 100 °C for 24 h, the dry biomass was gravimetrically measured (Müller et al. 2010).
Emulsification index (E24)
The emulsification index (E24) was determined by mixing equal volumes of cell-free supernatant with hexadecane. The mixture was vortexed for 2 min. After 24 h, emulsification activity was determined as follows (Datta et al. 2018).
E24% = (Height of the emulsified layer ⁄ Total height of the liquid column) × 100 (1)
Oil displacement test
An oil displacement test was performed to determine the potential of the biosurfactant in generating a clean zone. In the Petri plate, 30 mL of distilled water was poured and a thin coating of oil was created over the surface of the water. Then, 20 µL of rhamnolipid solution was added and a clean zone appeared very quickly. Twenty microliters of distilled water were used in the control experiments (Datta et al. 2018).
Kinetic study of biosurfactant production
A growth profile analysis was carried out in MSM using rice bran oil as a carbon source to examine the change in the bacterial cell population over time in an aerobic culture (at 37 ºC, 180 rpm agitation). The optical density of the culture was determined by measuring the absorbance of the oil-free broth sample at 600 nm (OD600) using a spectrophotometer. Surface tension reduction (biosurfactant production) and rice bran oil utilization were measured for 160 h at 10 h intervals. Rice bran oil utilization was expressed as the percentage of weight loss of rice bran oil divided by initial weight. Each data point was repeated at least three times to guarantee the repeatability and accuracy of the tests, and the average value was reported.
Chemical characterization of the biosurfactant
The molecular structure of the produced material was evaluated using FTIR (TENSOR 27, Bruker, Germany), rhamnolipids were separated on paper (TLC; No.105554, Merck) according to their hydrophobicity using specific reagents. A mixture containing chloroform, methanol, and acetic acid at a volume ratio of 65:15:2 was used as the detector and mobile phases. A binary mixture of acetic acid and sulfuric acid at a volume ratio of 100:2 was used as the resting phase (Amani et al. 2010a).
Evaluation of the surface activity of the produced biosurfactant
Surface tension measurement
One of the most important properties of a biosurfactant is the reduction in surface tension. In this study, automatic surface tension measurement equipment (Atiyeh Pouyandegan Exir Technologies Co. [Apex Technologies Co.], Arak, Iran) was used. Dynamic surface tension was measured using the pendant drop method, which is one of the most accurate surface tension measurement techniques (Yang et al. 2014). The shape of the pendant drop containing biosurfactant was monitored using a capture system. The captured image of the pendant drop was analyzed using an online image processing software according to the following equation (Berry et al. 2015):
g is the acceleration due to gravity and Δρ is the difference between the air and drop densities. In addition, H is a shape-dependent parameter that depends on the shape factor value, that is, S = d/D (Berry et al. 2015), which was experimentally measured using image processing. It should be noted that D is the equatorial diameter of the pendant drop, and d is the diameter at a distance of D from the top of the drop. To check the repeatability of the measurements, the average of at least three independent measurements was reported as the equilibrium surface tension value. Notably, the surface tension values decreased with time (dynamic surface tension) before reaching the equilibrium point, that is, the equilibrium surface tension was unaffected by the interface aging time. The adsorption time was calculated from the dynamic surface tension values and mono-exponential decay model.
Mono-exponential decay model
Generally, biosurfactant molecules can migrate from the bulk toward the interface as a function of time and consequently affect the dynamic and equilibrium surface tension behavior, which is related to high adsorption activation energy barriers. Generally, diffusion can control the adsorption of active agents when the diffusive part is larger than the kinetic part. When the adsorption is limited by the activation energy, a decay model has been proposed to model the surface tension and interfacial tension values versus time (Lashkarbolooki et al. 2017). The decay model is frequently used to assess dynamic surface activity (Lashkarbolooki et al. 2016; Hamidian et al. 2019).
In this equation, γe, γt, and γ0 are the surface tensions at equilibrium, time t, and initial (t = 0), respectively. The adjustable parameter of the model, τ, is a parameter with the unit of time known as the adsorption time and can be obtained by fitting to the experimental dynamic surface tension values.
Critical micelle concentration
One of the key characteristics of a biosurfactant is its CMC, which denotes the start of micelle formation. Surface tension cannot be reduced by adding additional biosurfactants beyond the CMC value. The surface tension values of biosurfactant solutions of various concentrations were tested to calculate the CMC value (Kłosowska-Chomiczewska et al. 2017).
Effect of salt type and concentration on biosurfactant activity
The effects of different salts (MgSO4, Na2SO4, KCl, and NaCl) and their concentrations on the surface activity of rhamnolipids were also investigated. Different concentrations of each salt (2–12% w/v) were added to the supernatants containing rhamnolipids and their surface tensions were measured.
Antifungal effect of the biosurfactant produced by rice bran oil
M. phaseolina is one of the most harmful seeds and soil-borne plant pathogens. Studies have demonstrated that this species can survive in soil for up to three years (Kaur et al. 2012). Therefore, the antifungal activity of biosurfactants capable of controlling M. phaseolina has also been investigated, mainly for applications in food and pharmaceutical industries. Potato Dextrose Agar (PDA; 3.9 g) was dissolved in 100 mL of distilled water and autoclaved at 121 °C for 20 min. Then, the biosurfactant at different concentrations of 0.2, 0.1, and 0.05 g/L was filter-sterilized through a 0.22 μm membrane. The filtrate was added to molten PDA medium and incubated at 25 °C for 24 h. After ensuring that the plates were not contaminated, the mycelial plugs of M. phaseolina were cultured in the center of Petri dishes. The dishes were incubated at 25 °C for four days. The antifungal activity of rhamnolipids against M. phaseolina was evaluated. All assays were performed in triplicate (Santos et al. 2021).
Results and discussion
Extraction of rice bran oil
In the first stage of this study, rice bran oil was produced using agricultural waste as a substrate for rhamnolipid production. The resulting materials were separated by centrifugation to obtain rice bran oil. The results revealed that the oil produced from rice bran was approximately 11.54 g per 100 g of dry bran. The changes in the culture medium from the first day to the last day depict the main emulsification of rice bran oil. This indicates the breakdown of rice bran oil and a reduction in free fatty acids over time (Lan et al. 2015).
Figure 2 shows the molecular structure of rice bran oil produced using FTIR. The IR spectrum indicated the stretching vibration of the cis-olefin double bonds (=C–H) at about 3006.11 cm−1, the asymmetric methylene stretching band at 2923.11 cm−1, and the symmetrical methylene stretching band at 2855.29 cm−1. The spectrum also gave vibrational stretching bands for CH3 at 2855.29 cm−1, the C=O group at 1743.16 cm−1 for triglycerides, the stretch (–C–H) vibrations of the CH2 group at 1458.15 cm−1, and the stretching –C–O–O bands at 1162.38 cm−1 (Zúñiga-Diaz et al. 2017).
Evaluation of the effect of different carbon sources on biosurfactant production
The obtained results revealed that it would be possible to produce a 7.45 g/L rhamnolipid after seven days using rice bran oil as a carbon source. For better comparison, industrial rice bran oil and sunflower oil were also used as carbon sources besides rice bran oil, which led to the production of the rhamnolipid at concentrations of 4.14 and 5.28 g/L, respectively. Regarding the results obtained for the efficiency of rhamnolipid production, it can be concluded that rice bran oil extracted from waste (an inexpensive and biodegradable carbon source) is superior to industrial oils. A closer examination also revealed that rice bran oil consumption was more obvious than that of other carbon sources, leading to a more considerable color change in the culture medium and foam creation. Table 1 summarizes rhamnolipid production from different types of waste and compares the results with those reported in the literature.
Vedaraman and Venkatesh (2011) obtained surfactin using frying oils (sunflower and rice bran) at concentrations up to 0.650 g/L crude extract. Ghorbani et al. (2022) studied the surface tension of P. aeruginosa PTCC 1340, which was grown in 125 g/L of sunflower oil as the sole carbon source, and discovered that it decreased by 26.8 mN/m in a cell-free broth containing a biosurfactant with a CMC of 0.123 g/L. In another study, Patowary et al. (2016) investigated the production of rhamnolipid biosurfactant prepared from P. aeruginosa SR17 using paneer whey as a carbon source. Their results showed that the CMC was 26.5 mN/m, observed at 110 mg/L rhamnolipid biosurfactant. They reported rhamnolipid production of 2.7 g/L. Amani et al. (2010b) also studied the growth kinetics and biosurfactant production by P. aeruginosa with 250 g/L sunflower oil. They reported biosurfactant production of 0.7 g/L after 72 h of cultivation. Chen et al. (2018) investigated fermentation studies with a strain of P. aeruginosa grown in kitchen waste oil as the sole carbon source and reported biosurfactant production of 2.47 g/L. Nagat Abdalla et al. (2019) also reported a minimum surface tension value of 30.2 mN/m by P. aeruginosa grown in petroleum oil wastes as the sole carbon substrate for biosurfactant production of 2.7 g/L. In our study, the biosurfactant produced with a CMC of 0.09 g/L exhibited effective surface activity because it significantly reduced the surface tension of water from 70.46 to 25.86 mN/m. Figure 3 shows the production rates of rhamnolipids and biomass. As shown in Fig. 3A, the maximum biomass concentration obtained was 2.85 g/L after 120 h of incubation when rice bran oil extracted in the laboratory was used as a carbon source. Various oil-degrading microorganisms form stable emulsions and some produce surface-active biomolecules. Some yeasts and bacteria share these properties (Santos et al. 2013). The results showed that P. aeruginosa PTCC 1340 was able to produce biosurfactants that could form stable emulsions, and reduce surface tension.
Growth kinetics of P. aeruginosa, production and characterization of biosurfactant
Figure 4 depicts the bacterial growth profile at 37 °C and 180 rpm, and it can be seen that the bacterial count grew somewhat in the first few hours as it adapted to the new environment. During the exponential period of the aerobic growth curve (10–47 h), the bacteria expanded quickly and then entered the stationary phase. The specific growth rate is 0.053 h−1. The association between bacterial growth under aerobic conditions and surface tension is shown in Fig. 4. The lowest surface tension (25.72 mN/m) was obtained at the end of the logarithmic phase (46 h). Several bacteria have been shown to reduce the surface tension of media as a consequence of biosurfactant generation during the transition between exponential and stationary phases (Nitschke and Pastore 2006Ghojavand et al. 2008; Yin et al. 2009; Najafi et al. 2010). Maximum biosurfactant synthesis (7.45 g/L) was obtained by P. aeruginosa at 72 h. At the same time, rice bran oil utilization reached approximately 84%. The yields of rhamnolipid to rice bran oil utilization (YRL/S) and rhamnolipid to biomass (YRL/X) were 0.246 and 2.81 (g/g), respectively.
Chemical properties of the produced biosurfactant
Four rhamnolipid homologs have been identified and characterized (Henkel et al. 2012). They contain one or two rhamnose molecules as well as one or two β-hydroxy fatty acids. FTIR and TLC analyses were performed to identify rhamnolipids in a very high-producing strain of P. aeruginosa.
The FTIR spectra of the biosurfactant from P. aeruginosa PTCC 1340, using rice bran oil as a carbon source, are shown in Fig. 5. Stretching C–O bands were detected in the IR spectrum at 1043, 1126, and 1170 cm–1, a symmetric O–C–O band at 1043 cm–1, a deformation C–OH band at 1384 cm–1, CH3 rocking at 985 cm–1, deformation C–H bands for RL at 1455, 1245, and 809 cm–1, and a strong symmetric stretching C=O band of the carboxylate group at 1732 cm–1. The spectrum showed the stretch (–C–H) vibrations of CH2 and CH3 groups at 2924 and 2857 cm−1. A broad peak at 3397 cm−1 for symmetric O–H stretching was observed.
These characteristic adsorption bands show that the biosurfactant produced by P. aeruginosa PTCC 1340 using rice bran oil belongs to the glycolipid group, and the FTIR spectra of the biosurfactant are in good agreement with the typical FTIR spectrum of rhamnolipids from P. aeruginosa (Rahman et al. 2010).
In Fig. 6, TLC analysis was performed in triplicate. The pure biosurfactant showed two spots on the TLC plate: mono-rhamnolipid and di-rhamnolipid, with retention factors (RF) of 0.69 and 0.34, respectively. According to previous reports (Silva et al. 2010; Lan et al. 2015), the sample was considered rhamnolipid.
Dynamical surface tensions of aqueous solutions containing different concentrations of the biosurfactant
The measured dynamic surface tension of cell-free broth containing rhamnolipids produced by P. aeruginosa PTCC 1340 using rice bran oil extracted from agricultural waste as a carbon source revealed different dynamic behaviors of the biosurfactant as a function of concentration (see Fig. 7). The measured data revealed that although no significant surface tension variation could be observed as a function of time for water, the biosurfactants migrated from the bulk to the interface as a function of time, consequently reducing the surface tension.
A monoexponential decay model (Eq. 2) was used to explain the surface tension functionality over time for a more comprehensive understanding of the surface activity of the rhamnolipids produced. When comparing the results of the experimental dynamic surface tensions and modeled ones at three different biosurfactant concentrations (Fig. 8), it was shown that there was a satisfactory match between the experimental and modeled data. In addition, the standard deviation (SD) of the measured equilibrium surface tension and average absolute relative deviation (AARD %) error of the predicted surface tension values revealed the good accuracy of the proposed model.
In the next step, the adsorption time and differences between initial and equilibrium surface tension (\({\Delta \gamma =(\gamma }_{0}-{\gamma }_{e})\)) are shown in Fig. 9A, B; these figures show two distinct concentration ranges. In the first interval (rhamnolipid concentration \(<\) 0.025 g/L), the adsorption time and \(\Delta \gamma\) increased as a function of rhamnolipid concentration. The maximum difference between the initial and equilibrium surface tension values (27.99 mN/m) was obtained at a concentration of 0.025 g/L, and approximately 130 s were required for the migration of rhamnolipids from the bulk to the interface. In the second interval (concentration higher than 0.025 g/L), the reverse trend was observed, and even less time was required to reach equilibrium because of the higher surface activity of rhamnolipids at higher concentrations (their lower initial surface tension (see Fig. 9). In other words, the maximum surface activity of the biosurfactant at the maximum concentration of rhamnolipids did not require time to reach equilibrium. Therefore, the initial and equilibrium surface tensions were equal.
Physical characteristics of the produced rhamnolipid using rice bran oil
As shown in Fig. 10, the CMC was calculated from a plot of the surface tension for different rhamnolipid concentrations. The obtained results showed that the produced biosurfactant with a CMC of 0.09 g/L had effective surface activity since it reduced the surface tension of water from 70.46 to 25.86 mN/m. Rice bran oil extracted from agricultural waste is considered an inexpensive carbon source for producing biodegradable rhamnolipids with an acceptable amount of rhamnolipid production (7.45 g/L). A 7.4 cm clean zone was measured during the oil displacement assay. E24 was also found to be the highest (89%) for rhamnolipid biosurfactants produced using rice bran oil as a carbon source. The emulsification index remained stable for at least four days without any stabilizing additives. Furthermore, the present biosurfactant exhibited a higher emulsification activity than sodium dodecyl sulfate (SDS) surfactant (80%) against hexadecane (Datta et al. 2018). Mishra et al. (2021) optimized the production of rhamnolipid biosurfactants prepared from Pseudomonas aeruginosa MTCC 424 using waste rice bran oil as a carbon source. The results showed that the CMC of the optimized biosurfactant was 28.5 mN/m, observed at 120 mg/L rhamnolipid biosurfactant.
The CMC of SDS was 2100 mg/L (Zhao et al. 2019). The biosurfactant produced possessed excellent surface activity compared to that of traditional chemosynthetic surfactants.
Effect of salt type and concentration on the surface tension of bio-products
Different concentrations of salts were added to the supernatant containing the biosurfactant at CMC, and the surface tension of the solutions was measured (see Fig. 11). Studies have shown that increasing the salt concentration up to 12% w/v has no significant effect on the biosurfactant performance in reducing surface tension. Sathi Reddy et al. (2016) investigated the effect of salinity concentrations (2, 6, 10, 15, and 20% w/v) on the stability of rhamnolipid biosurfactants produced by P. aeruginosa DR1 using mango kernel oil. They observed that the surface tension gradually increased from 30 to 38 mN/m as the salt concentration increased from 6 to 20%.
As shown in Fig. 11, the surface activity of rhamnolipids was independent of salt concentration. It can be concluded that the biosurfactant produced with a carbon source of rice bran oil has good performance in reducing surface tension with a low adsorption time and a good ability to reduce surface tension without being considerably affected by salt type. Because of these properties, biosurfactants can be used instead of chemical surfactants in various industries such as petroleum, pharmaceuticals, cosmetics, and food.
Antifungal results of the biosurfactant produced using rice bran oil
In terms of environmental considerations, the antifungal activity of the biosurfactants was evaluated as an important parameter. Figure 12 shows that rhamnolipids produced by P. aeruginosa had an excellent ability to to inhibit the growth of M. phaseolina. According to the tests performed with different rhamnolipid concentrations, mycelial growth inhibition of 100% was obtained at a concentration of 0.2 g/L. The rhamnolipids produced showed strong antifungal activity against M. phaseolina.
Sathi Reddy et al. (2016) studied mango kernel oil as a carbon source for rhamnolipid production by P. aeruginosa DR1 and evaluated the antifungal activity of the rhamnolipids produced against M. phaseolina. They reported a mycelial growth inhibition of 60.46% with a rhamnolipid concentration of 0.3 g/L against M. phaseolina. The rhamnolipid biosurfactant has shown great potential as a biocontrol agent for several plant pathogens by adopting various mechanisms, such as the loss of outer membrane permeability due to the formation of new ion channels in the cell plasma membrane (Liu et al. 2012; Sathi Reddy et al. 2016). Biocontrol activity may vary based on differences in the congeners present in the biosurfactant mixture produced by P. aeruginosa from several carbon sources.
Conclusions
In this study, rice bran oil extracted from agricultural waste was considered a renewable substrate for producing biodegradable biosurfactants, and the following main results were obtained:
-
An acceptable amount of rhamnolipid (7.45 g/L) from Pseudomonas aeruginosa was produced, with rhamnolipid yields to rice bran oil utilization (YRL/S) and rhamnolipid to biomass (YRL/X) of 0.246 and 2.81 (g/g), respectively.
-
The rhamnolipids produced using rice bran oil had acceptable performance in reducing surface tension, as confirmed by examining the dynamic surface tension state and adsorption time.
-
The biosurfactant produced with a critical micelle concentration (CMC) of 0.09 g/L exhibited effective surface activity because it significantly reduced the surface tension of water from 70.46 to 25.86 mN/m.
-
When comparing the results of the experimentally determined dynamic surface tensions with the modeled ones at different biosurfactant concentrations, it was shown that there was a satisfactory match between the experimental and modeled data. The results revealed that the adsorption and relaxation of the biosurfactants at the interface were considerably affected by the biosurfactant concentration.
-
In terms of environmental impact, the rhamnolipids produced demonstrated potent antifungal activity against Macrophomina phaseolina. According to tests performed with different rhamnolipid concentrations, mycelial growth inhibition of 100% was obtained at a concentration of 0.2 g/L.
-
In summary, the biosurfactant produced using rice bran oil as a carbon source showed excellent performance in reducing surface tension with a short adsorption time without being affected by the type of salt or the concentration for a variety of applications.
Abbreviations
- AARD %:
-
Average absolute relative deviation percent
- D:
-
Equatorial diameter of the pendant drop (mm)
- d:
-
Diameter at distance D from the top of the drop (mm)
- E24 :
-
Emulsification index
- FTIR:
-
Fourier transform infrared spectroscopy
- g:
-
Acceleration of gravity (9.8 m/s2)
- H:
-
Shape-dependent parameter
- MEOR:
-
Microbial enhanced oil recovery
- MSM:
-
Mineral salt medium
- PDA:
-
Potato dextrose agar
- RF:
-
Retention factor
- SD:
-
Standard deviation
- SDS:
-
Sodium dodecyl sulphate
- TLC:
-
Thin-layer chromatography
- t:
-
Time (s)
- YRL/S :
-
The yield of the rhamnolipid to rice bran oil utilization
- YRL/X :
-
The yield of the rhamnolipid to biomass
- γ0 :
-
Surface tension at initial (mN/m)
- γe :
-
Surface tension at the equilibrium (mN/m)
- γt :
-
Surface tension at time t (mN/m)
- Δρ:
-
Difference between air and drop densities (kg/m3)
- τ:
-
Relaxation/adsorption time (s)
References
Amani H (2015) Study of enhanced oil recovery by rhamnolipids in a homogeneous 2D micromodel. J Petrol Sci Eng 128:212–219. https://doi.org/10.1016/j.petrol.2015.02.030
Amani H, Mehrnia MR, Sarrafzadeh MH, Haghighi M, Soudi MR (2010a) Scale up and application of biosurfactant from bacillus subtilis in enhanced oil recovery. Appl Biochem Biotechnol 162(2):510–523. https://doi.org/10.1007/s12010-009-8889-0
Amani H, Sarrafzadeh MH, Haghighi M, Mehrnia MR (2010b) Comparative study of biosurfactant producing bacteria in MEOR applications. J Petrol Sci Eng 75(1):209–214. https://doi.org/10.1016/j.petrol.2010.11.008
Amani H, Müller MM, Syldatk C, Hausmann R (2013) Production of microbial rhamnolipid by pseudomonas aeruginosa MM1011 for ex situ enhanced oil recovery. Appl Biochem Biotechnol 170(5):1080–1093. https://doi.org/10.1007/s12010-013-0249-4
AshishDebnath M (2018) Application of biosurfactant produced by an adaptive strain of C.tropicalis MTCC230 in microbial enhanced oil recovery (MEOR) and removal of motor oil from contaminated sand and water. J Petrol Sci Eng 170:40–48. https://doi.org/10.1016/j.petrol.2018.06.034
Ben Khedher S, Boukedi H, Dammak M, Kilani-Feki O, Sellami-Boudawara T, Abdelkefi-Mesrati L, Tounsi S (2017) Combinatorial effect of Bacillus amyloliquefaciens AG1 biosurfactant and Bacillus thuringiensis Vip3Aa16 toxin on Spodoptera littoralis larvae. J Invertebr Pathol 144:11–17. https://doi.org/10.1016/j.jip.2017.01.006
Berry JD, Neeson MJ, Dagastine RR, Chan DYC, Tabor RF (2015) Measurement of surface and interfacial tension using pendant drop tensiometry. J Colloid Interface Sci 454:226–237. https://doi.org/10.1016/j.jcis.2015.05.012
Chen C, Sun N, Li D, Long S, Tang X, Xiao G, Wang L (2018) Optimization and characterization of biosurfactant production from kitchen waste oil using Pseudomonas aeruginosa. Environ Sci Pollut Res 25(15):14934–14943. https://doi.org/10.1007/s11356-018-1691-1
Chen W, Kong Y, Li J, Sun Y, Min J, Hu X (2020) Enhanced biodegradation of crude oil by constructed bacterial consortium comprising salt-tolerant petroleum degraders and biosurfactant producer. Int Biodeterior Biodegrad 154:105047. https://doi.org/10.1016/j.ibiod.2020.105047
Datta P, Tiwari P, Pandey LM (2018) Isolation and characterization of biosurfactant producing and oil degrading Bacillus subtilis MG495086 from formation water of Assam oil reservoir and its suitability for enhanced oil recovery. Biores Technol 270:439–448. https://doi.org/10.1016/j.biortech.2018.09.047
Gaur VK, Sharma P, Sirohi R, Varjani S, Taherzadeh MJ, Chang J-S, Yong Ng H, Wong JWC, Kim S-H (2022) Production of biosurfactants from agro-industrial waste and waste cooking oil in a circular bioeconomy: an overview. Bioresour Technol 343:126059. https://doi.org/10.1016/j.biortech.2021.126059
Geetha SJ, Banat IM, Joshi SJ (2018) Biosurfactants: production and potential applications in microbial enhanced oil recovery (MEOR). Biocatal Agric Biotechnol 14(January):23–32. https://doi.org/10.1016/j.bcab.2018.01.010
Ghasemi H, Mozaffari S, Mohammadghasemi H, Jemere AB, Nazemifard N (2022) Microfluidic platforms for characterization of crude oil emulsion stability. Can J Chem 100(7):484–494. https://doi.org/10.1139/cjc-2021-0248
Ghojavand H, Vahabzadeh F, Roayaei E, Shahraki AK (2008) Production and properties of a biosurfactant obtained from a member of the Bacillus subtilis group (PTCC 1696). J Colloid Interface Sci 324(1):172–176. https://doi.org/10.1016/j.jcis.2008.05.001
Ghorbani M, Hosseini M, Najafpour G, Hajimohammadi R (2022) Synthesis and characterization of rhamnolipid biosurfactant produced by Pseudomonas aeruginosa PTCC 1340 for emulsification of oil sludge in oil storage tank. Arab J Sci Eng 47(1):219–226. https://doi.org/10.1007/s13369-021-05872-5
Goswami M, Deka S (2019) Biosurfactant production by a rhizosphere bacteria Bacillus altitudinis MS16 and its promising emulsification and antifungal activity. Colloids Surf B 178:285–296. https://doi.org/10.1016/j.colsurfb.2019.03.003
Hajibagheri F, Lashkarbolooki M, Ayatollahi S, Hashemi A (2017) The synergic effects of anionic and cationic chemical surfactants, and bacterial solution on wettability alteration of carbonate rock: an experimental investigation. Colloids Surf A 513:422–429. https://doi.org/10.1016/j.colsurfa.2016.11.010
Hamidian R, Lashkarbolooki M, Amani H (2019) Ion type adjustment with emphasize on the presence of NaCl existence; measuring interfacial tension, wettability and spreading of crude oil in the carbonate reservoir. J Petrol Sci Eng 182:106266. https://doi.org/10.1016/j.petrol.2019.106266
Heidarzadeh MH, Amani H, Najafpour Darzi G (2022) Accurate investigation of the mechanism of rhamnolipid biosurfactant effects on food waste composting: a comparison of in-situ and ex-situ techniques. J Environ Manage 322:116090. https://doi.org/10.1016/j.jenvman.2022.116090
Henkel M, Müller MM, Kügler JH, Lovaglio RB, Contiero J, Syldatk C, Hausmann R (2012) Rhamnolipids as biosurfactants from renewable resources: concepts for next-generation rhamnolipid production. Process Biochem 47(8):1207–1219. https://doi.org/10.1016/j.procbio.2012.04.018
Jadhav JV, Pratap AP, Kale SB (2019) Evaluation of sunflower oil refinery waste as feedstock for production of sophorolipid. Process Biochem 78:15–24. https://doi.org/10.1016/j.procbio.2019.01.015
Jahan R, Bodratti AM, Tsianou M, Alexandridis P (2020) Biosurfactants, natural alternatives to synthetic surfactants: physicochemical properties and applications. Adv Colloid Interface Sci 275:102061. https://doi.org/10.1016/j.cis.2019.102061
Kaur S, Dhillon GS, Brar SK, Vallad GE, Chand R, Chauhan VB (2012) Emerging phytopathogen Macrophomina phaseolina: biology, economic importance and current diagnostic trends. Crit Rev Microbiol 38(2):136–151. https://doi.org/10.3109/1040841X.2011.640977
Kłosowska-Chomiczewska IE, Mędrzycka K, Hallmann E, Karpenko E, Pokynbroda T, Macierzanka A, Jungnickel C (2017) Rhamnolipid CMC prediction. J Colloid Interface Sci 488:10–19. https://doi.org/10.1016/j.jcis.2016.10.055
Lan G, Fan Q, Liu Y, Chen C, Li G, Liu Y, Yin X (2015) Rhamnolipid production from waste cooking oil using Pseudomonas SWP-4. Biochem Eng J 101:44–54. https://doi.org/10.1016/j.bej.2015.05.001
Lashkarbolooki M, Riazi M, Ayatollahi S (2016) Investigation of effects of salinity, temperature, pressure, and crude oil type on the dynamic interfacial tensions. Chem Eng Res Des 115:53–65. https://doi.org/10.1016/j.cherd.2016.09.020
Lashkarbolooki M, Riazi M, Ayatollahi S (2017) Effect of CO2 and natural surfactant of crude oil on the dynamic interfacial tensions during carbonated water flooding: experimental and modeling investigation. J Petrol Sci Eng 159:58–67. https://doi.org/10.1016/j.petrol.2017.09.023
Liu Z, Zeng Z, Zeng G, Li J, Zhong H, Yuan X, Liu Y, Zhang J, Chen M, Liu Y, Xie G (2012) Influence of rhamnolipids and Triton X-100 on adsorption of phenol by Penicillium simplicissimum. Biores Technol 110:468–473. https://doi.org/10.1016/j.biortech.2012.01.092
Liu C, Zhang Y, Sun S, Huang L, Yu L, Liu X, Lai R, Luo Y, Zhang Z, Zhang Z (2018) Oil recovery from tank bottom sludge using rhamnolipids. J Petrol Sci Eng 170:14–20. https://doi.org/10.1016/j.petrol.2018.06.031
Meng L, Li W, Bao M, Sun P (2019) Effect of surfactants on the solubilization, sorption and biodegradation of benzo (a) pyrene by Pseudomonas aeruginosa BT-1. J Taiwan Inst Chem Eng 96:121–130. https://doi.org/10.1016/j.jtice.2019.01.007
Mirmajlessi SM, Destefanis M, Gottsberger RA, Mänd M, Loit E (2015) PCR-based specific techniques used for detecting the most important pathogens on strawberry: a systematic review. Syst Rev 4(1):9. https://doi.org/10.1186/2046-4053-4-9
Mishra A, Sawood GM, Gautam SB, Trivedi RK (2021) Optimization of process inputs for the synthesis of waste rice bran oil isolated Pseudomonas aeruginosa MTCC 424 biosurfactant using response surface methodology for oil recovery applications. Bioresour Technol Rep 14:100653. https://doi.org/10.1016/j.biteb.2021.100653
Moya Ramírez I, Altmajer Vaz D, Banat IM, Marchant R, Jurado Alameda E, García Román M (2016) Hydrolysis of olive mill waste to enhance rhamnolipids and surfactin production. Biores Technol 205:1–6. https://doi.org/10.1016/j.biortech.2016.01.016
Mozaffari S, Ghasemi H, Tchoukov P, Czarnecki J, Nazemifard N (2021) Lab-on-a-chip systems in asphaltene characterization: a review of recent advances. Energy Fuels 35(11):9080–9101. https://doi.org/10.1021/acs.energyfuels.1c00717
Müller MM, Hörmann B, Syldatk C, Hausmann R (2010) Pseudomonas aeruginosa PAO1 as a model for rhamnolipid production in bioreactor systems. Appl Microbiol Biotechnol 87(1):167–174. https://doi.org/10.1007/s00253-010-2513-7
Nagat Abdalla M, Aghareed MT, Olfat Abdalla M, Rania F (2019) Biodegradation of petroleum oil effluents and production of biosurfactants: effect of initial oil concentration. J Surfactants Deterg 22(2):385–394. https://doi.org/10.1002/jsde.12240
Najafi AR, Rahimpour MR, Jahanmiri AH, Roostaazad R, Arabian D, Ghobadi Z (2010) Enhancing biosurfactant production from an indigenous strain of Bacillus mycoides by optimizing the growth conditions using a response surface methodology. Chem Eng J 163(3):188–194. https://doi.org/10.1016/j.cej.2010.06.044
Nitschke M, Pastore GM (2006) Production and properties of a surfactant obtained from Bacillus subtilis grown on cassava wastewater. Biores Technol 97(2):336–341. https://doi.org/10.1016/j.biortech.2005.02.044
Pandey AK, Burlakoti RR, Rathore A, Nair RM (2020) Morphological and molecular characterization of Macrophomina phaseolina isolated from three legume crops and evaluation of mungbean genotypes for resistance to dry root rot. Crop Protect 127:104962. https://doi.org/10.1016/j.cropro.2019.104962
Patowary R, Patowary K, Kalita MC, Deka S (2016) Utilization of Paneer Whey waste for cost-effective production of rhamnolipid biosurfactant. Appl Biochem Biotechnol 180(3):383–399. https://doi.org/10.1007/s12010-016-2105-9
Radzuan MN, Banat IM, Winterburn J (2017) Production and characterization of rhamnolipid using palm oil agricultural refinery waste. Biores Technol 225:99–105. https://doi.org/10.1016/j.biortech.2016.11.052
Rahman KSM, Rahman TJ, McClean S, Marchant R, Banat IM (2002) Rhamnolipid biosurfactant production by strains of pseudomonas aeruginosa using low-cost raw materials. Biotechnol Prog 18(6):1277–1281. https://doi.org/10.1021/bp020071x
Rahman PKSM, Pasirayi G, Auger V, Ali Z (2010) Production of rhamnolipid biosurfactants by Pseudomonas aeruginosa DS10-129 in a microfluidic bioreactor. Biotechnol Appl Biochem 55(1):45–52. https://doi.org/10.1042/BA20090277
Ray M, Kumar V, Banerjee C (2022) Kinetic modelling, production optimization, functional characterization and phyto-toxicity evaluation of biosurfactant derived from crude oil biodegrading Pseudomonas sp. IITISM 19. J Environ Chem Eng 10(2):107190. https://doi.org/10.1016/j.jece.2022.107190
Raza ZA, Ahmad N, Kamal S (2014) Multi-response optimization of rhamnolipid production using grey rational analysis in Taguchi method. Biotechnol Rep 3:86–94. https://doi.org/10.1016/j.btre.2014.06.007
Santos DKF, Rufino RD, Luna JM, Santos VA, Salgueiro AA, Sarubbo LA (2013) Synthesis and evaluation of biosurfactant produced by Candida lipolytica using animal fat and corn steep liquor. J Petrol Sci Eng 105:43–50. https://doi.org/10.1016/j.petrol.2013.03.028
Santos AP, Muratore LN, Solé-Gil A, Farías ME, Ferrando A, Blázquez MA, Belfiore C (2021) Extremophilic bacteria restrict the growth of Macrophomina phaseolina by combined secretion of polyamines and lytic enzymes. Biotechnol Rep 32:e00674. https://doi.org/10.1016/j.btre.2021.e00674
Sarubbo LA, Silva MDGC, Durval IJB, Bezerra KGO, Ribeiro BG, Silva IA, Twigg MS, Banat IM (2022) Biosurfactants: production, properties, applications, trends, and general perspectives. Biochem Eng J 181:108377. https://doi.org/10.1016/j.bej.2022.108377
Sathi Reddy K, Yahya Khan M, Archana K, Gopal Reddy M, Hameeda B (2016) Utilization of mango kernel oil for the rhamnolipid production by Pseudomonas aeruginosa DR1 towards its application as biocontrol agent. Biores Technol 221:291–299. https://doi.org/10.1016/j.biortech.2016.09.041
Sharma S, Datta P, Kumar B, Tiwari P, Pandey LM (2019) Production of novel rhamnolipids via biodegradation of waste cooking oil using Pseudomonas aeruginosa MTCC7815. Biodegradation 30(4):301–312. https://doi.org/10.1007/s10532-019-09874-x
Silva SNRL, Farias CBB, Rufino RD, Luna JM, Sarubbo LA (2010) Glycerol as substrate for the production of biosurfactant by Pseudomonas aeruginosa UCP0992. Colloids Surf B 79(1):174–183. https://doi.org/10.1016/j.colsurfb.2010.03.050
Tripathi V, Gaur VK, Dhiman N, Gautam K, Manickam N (2020) Characterization and properties of the biosurfactant produced by PAH-degrading bacteria isolated from contaminated oily sludge environment. Environ Sci Pollut Res 27(22):27268–27278. https://doi.org/10.1007/s11356-019-05591-3
Varjani S, Upasani VN (2019) Evaluation of rhamnolipid production by a halotolerant novel strain of Pseudomonas aeruginosa. Bioresour Technol 288:121577. https://doi.org/10.1016/j.biortech.2019.121577
Vedaraman N, Venkatesh N (2011) Production of surfactin by Bacillus subtilis MTCC 2423 from waste frying oils. Braz J Chem Eng 28:175–180. https://doi.org/10.1590/S0104-66322011000200001
Verma C, Hussain CM, Quraishi MA, Alfantazi A (2023) Green surfactants for corrosion control: design, performance and applications. Adv Colloid Interface Sci 311:102822. https://doi.org/10.1016/j.cis.2022.102822
Yang Z, Li M, Peng B, Lin M, Dong Z, Ling Y (2014) Interfacial tension of CO2 and organic liquid under high pressure and temperature. Chin J Chem Eng 22(11):1302–1306. https://doi.org/10.1016/j.cjche.2014.09.042
Yin H, Qiang J, Jia Y, Ye J, Peng H, Qin H, Zhang N, He B (2009) Characteristics of biosurfactant produced by Pseudomonas aeruginosa S6 isolated from oil-containing wastewater. Process Biochem 44(3):302–308. https://doi.org/10.1016/j.procbio.2008.11.003
Zhao F, Jiang H, Sun H, Liu C, Han S, Zhang Y (2019) Production of rhamnolipids with different proportions of mono-rhamnolipids using crude glycerol and a comparison of their application potential for oil recovery from oily sludge. RSC Adv 9(6):2885–2891. https://doi.org/10.1039/C8RA09351B
Zouari R, Besbes S, Ellouze-Chaabouni S, Ghribi-Aydi D (2016) Cookies from composite wheat–sesame peels flours: dough quality and effect of Bacillus subtilis SPB1 biosurfactant addition. Food Chem 194:758–769. https://doi.org/10.1016/j.foodchem.2015.08.064
Zúñiga-Diaz J, Reyes-Dorantes E, Quinto-Hernandez A, Porcayo-Calderon J, Gonzalez-Rodriguez JG, Martinez-Gomez L (2017) Oil extraction from “Morelos Rice” bran: kinetics and raw oil stability. J Chem 2017:3837506. https://doi.org/10.1155/2017/3837506
Acknowledgements
The authors are delighted to acknowledge the Babol Noshirvani University of Technology, Faculty of Chemical Engineering, for funding the project (grant number: BNUT/964215021/2019).
Funding
The authors are delighted to acknowledge the Babol Noshirvani University of Technology, Faculty of Chemical Engineering, for funding the project (grant number: BNUT/964215021/2019).
Author information
Authors and Affiliations
Contributions
PS: Investigation, Software, Writing—Original Draft. MH: Supervision. ML: Supervision, Conceptualization, Methodology, Software, Formal analysis, Writing—Review & Editing. MG: Investigation, Methodology, Formal analysis, Validation, Writing—Original Draft, Writing—Review & Editing. GND: Writing—Review & Editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors report that there are no competing interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Safari, P., Hosseini, M., Lashkarbolooki, M. et al. Evaluation of surface activity of rhamnolipid biosurfactants produced from rice bran oil through dynamic surface tension. J Petrol Explor Prod Technol 13, 2139–2153 (2023). https://doi.org/10.1007/s13202-023-01660-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13202-023-01660-z