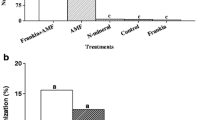
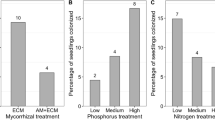
Abstract
Few studies have focused on analyzing the effect of native inoculated ectomycorrhizal (ECM) fungal strains on seedlings under field conditions in temperate forests. However, it is crucial to verify that the positive effects of ECM under nursery conditions also occur in the field, favoring their performance. We evaluated the short-term effect of ECM on three-year-old seedlings of Pinus hartwegii and Abies religiosa in central Mexico by subjecting them to four treatments: inoculation with Inocybe splendens, inoculation with Suillus brevipes (both native strains), inoculation with forest soil, and non-inoculated plants. Percentage of ECM colonization, plant growth (shoot height and stem diameter), and physiological (osmotic potential, stomatal conductance, CO2 assimilation and water use efficiency) responses were evaluated. We found that these two ECM species were partial (P. hartwegii) or totally (A. religiosa) replaced after one and a half years in the field. P. hartwegii seedlings increased their water use efficiency during the dry season, but in A. religiosa seedlings, a clear strategy for avoiding water stress was not detected. This ECM replacement had a negative effect on the physiological performance Of A. religiosa. Our results emphasize the importance of selecting compatible fungal-host species combinations for nursery inoculation and of using sources of inoculum adapted to the environmental conditions of the transplant site, ensuring root colonization prior to field transplanting to minimize seedling mortality due to water stress.





Similar content being viewed by others
References
Agerer R (1997) Colour atlas of Ectomycorrhizae, vol 1-140. Einhorn-Verlag, Schwäbisch Gmünd
Almeida-Leñero L, Nava M, Ramos A, Espinosa M, Ordoñez M, Jujnovsky J (2007) Ecosystem services in the Magdalena River, Mexico City. Gaceta ecológica, Instituto Nacional de Ecología 84-85:53–64
Aranda I, Gil L, Pardos J (2001) Effects of thinning in a Pinus sylvestris L. stand on foliar water relations of Fagus sylvatica L. seedlings planted within the pinewood. Trees 15:358–364. doi:10.1007/s004680100109
Ávila-Akerberg VD (2010) Forest quality in the southwest of México City. Assessment towards ecological restoration of ecosystem services. PhD Dissertation, Albert-Ludwigs-Universitat
Bauman JM, Keiffer CH, Hiremath S (2011) The influence of inoculated and native ectomycorrhizal fungi on morphology, physiology and survival of American chestnut. In: Barnhisel RI (ed) Proceedings of the 2011 National meeting of the American Society of Mining and Reclamation. Reclamation., vol 11-16. Bismarck, ND. ASMR, Lexington, pp 16-37
Bazzaz FA, Grace J (1997) Plant resource allocation. Academic Press, San Diego
Brown DJ, Mali I, Forstner MRJ (2014) Wildfire and postfire restoration action effects on microclimate and seedling pine tree survivorship. J Fish Wildl Manag 5:174–182. doi:10.3996/072013-jfwm-051
Brundrett M, Bougher N, Dell B, Grove T, Malajczuk N (1996) Working with mycorrhizas in forestry and agriculture. Australian Centre for International Agricultural Research, Canberra
Buckley TN (2005) The control of stomata by water balance. New Phytol 168:275–292. doi:10.1111/j.1469-8137.2005.01543.x
Calvaruso C, Turpault M-P, Uroz S, Leclerc E, Kies A, Frey-Klett P (2010) Laccaria bicolor S238N improves scots pine mineral nutrition by increasing root nutrient uptake from soil minerals but does not increase mineral weathering. Plant Soil 328:145–154. doi:10.1007/s11104-009-0092-0
Challenger A (2003) Conceptos generales acerca de los ecosistemas templados de montaña de México y su estado de conservación. In: Sánchez O, Vega E, Peters E, Monroy–Vilchis O (eds) Conservación de ecosistemas templados de montaña en México. Instituto Nacional de Ecología (INE, SEMARNAT)/ SyG, Mexico, D. F., pp 17–44
Clark CJ, Poulsen JR, Levey DJ, Osenberg CW (2007) Are plant populations seed limited? A critique and meta-analysis of seed addition experiments. Am Nat 170:128–142. doi:10.1086/518565
CONAFOR-SEMARNAT (2004) Protección, restauración y conservación de suelos forestales. Manual de obras y prácticas. Comisión Nacional Forestal and Secretaría de Medio Ambiente y Recursos Naturales
Dahlberg A, Finlay RD (1999) Suillus. In: Cairney JWG, Chambers SM (eds) Ectomycorrhizal fungi key genera in profile. Springer, Verlag, Berlin, pp 33–55
Dunk C, Lebel T, Keane P (2012) Characterisation of ectomycorrhizal formation by the exotic fungus Amanita muscaria with Nothofagus Cunninghamii in Victoria, Australia. Mycorrhiza 22:135–147. doi:10.1007/s00572-011-0388-9
Farjon A, Filer D (2013) An atlas of the World's conifers: an analysis of their distribution, biogeography, diversity and conservation status. Brill, Oxford
Gehring CA, Cobb N, Whitham Thomas G (1997) Three-way interactions among ectomycorrhizal mutualists, scale insects, and resistant and susceptible pinyon pines. Am Nat 149:824–841. doi:10.1086/286026
Guerin-Laguette A, Conventi S, Ruiz G, Plassard C, Mousain D (2003) The ectomycorrhizal symbiosis between Lactarius deliciosus and Pinus sylvestris in forest soil samples: symbiotic efficiency and development on roots of a rDNA internal transcribed spacer-selected isolate of L-deliciosus. Mycorrhiza 13:17–25. doi:10.1007/s00572-002-0191-8
Hart MM, Antunes PM, Abbott LK (2017) Unknown risks to soil biodiversity from commercial fungal inoculants. Nat Ecol Evol 1:0115
Honrubia M, de Murcia U (1995) Biotecnología forestal: técnicas de micorrización y micropropagación de plantas. Universidad de Murcia, Murcia
Hopkins WG (1999) Introduction to plant physiology, 2nd edn. Wiley, New York
Johnson NC et al (2006) From Lilliput to Brobdingnag: extending models of mycorrhizal function across scales. Bioscience 56:889–900. doi:10.1641/0006-3568(2006)56[889:FLTBEM]2.0.CO;2
Jones MD, Durall DM, Cairney JWG (2003) Ectomycorrhizal fungal communities in young forest stands regenerating after clearcut logging. New Phytol 157:399–422
Jujnovsky J, Almeida-Leñero L, Bojorge-García M, Monges JL, Cantoral-Uriza EA, Mazari-Hiriart M (2010) Hydrologic ecosystem services: water quality and quantity in the Magdalena River, Mexico City. Hidrobiológica 20:113–126
Jujnovsky J, González-Martínez T, Cantoral-Uriza E, Almeida-Leñero L (2012) Assessment of water supply as an ecosystem Service in a Rural-Urban Watershed in southwestern Mexico City. Environ Manag 49:690–702. doi:10.1007/s00267-011-9804-3
Koide RT, Xu B, Sharda J, Lekberg Y, Ostiguy N (2005) Evidence of species interactions within an ectomycorrhizal fungal community. New Phytol 165:305–316. doi:10.1111/j.1469-8137.2004.01216.x
Kramer PJ, Boyer JS (1995) Water relations of plants and soils. Academic Press, San Diego
LoBuglio KF (1999) Cenococcum. In: Cairney JWG, Chambers SM (eds) Ectomycorrhizal fungi key genera in profile. Springer, Berlin, pp 287–309
Marjanović Ž, Nehls U (2008) Ectomycorrhiza and water transport. In: Varma A (ed) Mycorrhiza. Springer, Berlin Heidelberg, pp 149–159. doi:10.1007/978-3-540-78826-3_8
Menkis A, Vasiliauskas R, Taylor AFS, Stenlid J, Finlay R (2007) Afforestation of abandoned farmland with conifer seedlings inoculated with three ectomycorrhizal fungi—impact on plant performance and ectomycorrhizal community. Mycorrhiza 17:337–348. doi:10.1007/s00572-007-0110-0
Molina R, Trappe JM (1982) Patterns of ectomycorrhizal host specificity and potential among Pacific northwest conifers and fungi. For Sci 28:423–458
Molina R, Massicote H, Trappe J (1992) Specificity phenomena in mycorrhizal symbioses: community-ecological consequences and practical implications. In: Allen M (ed) Mycorrhizal functioning –an integrative plant-fungal process. Chapman and Hall, New York, pp 357–423
Nannipieri P, Ascher J, Ceccherini MT, Landi L, Pietramellara G, Renella G (2003) Microbial diversity and soil functions. Eur J Soil Sci 54:655–670. doi:10.1046/j.1351-0754.2003.0556.x
Nara K (2006) Ectomycorrhizal networks and seedling establishment during early primary succession. New Phytol 169:169–178
Nations U (2008) World Urbanization Prospects. The 2007 Revision. Department of Economic and Social Affairs. New York
O’Hanlon R (2012) Below-ground ectomycorrhizal communities: the effect of small scale spatial and short term temporal variation. Symbiosis 57:57–71. doi:10.1007/s13199-012-0179-x
Paluch JG (2005) The influence of the spatial pattern of trees on forest floor vegetation and silver fir (Abies alba Mill.) regeneration in uneven-aged forests. For Ecol Manag 205:283–298. doi:10.1016/j.foreco.2004.10.010
Pena R, Simon J, Rennenberg H, Polle A (2013) Ectomycorrhiza affect architecture and nitrogen partitioning of beech (Fagus sylvatica L.) seedlings under shade and drought. Environ Exp Bot 87:207–217. doi:10.1016/j.envexpbot.2012.11.005
Peñuelas J, Ocaña L (2000) Cultivo de plantas forestales en contenedor. Mundi-Prensa, Madrid
PUEC-UNAM (2008) University Study Program of the City-National Autonomous University of Mexico. Integrated Diagnostic Proposal for the Magdalena River Basin. In: Master Plan for Comprehensive Management and Sustainable Use of the Magdalena River Basin. SMA-GDF, UNAM. PUEC-GDF., Mexico, D. F
Pyke DA, Thompson JN (1986) Statistical analysis of survival and removal rate experiments. Ecology 67:240–245. doi:10.2307/1938523
Rincón A, Ruíz-Díez B, Fernández-Pascual M, Probanza A, Pozuelo JM, de Felipe MR (2006) Afforestation of degraded soils with Pinus halepensis Mill.: effects of inoculation with selected microorganisms and soil amendment on plant growth, rhizospheric microbial activity and ectomycorrhizal formation. Appl Soil Ecol 34:42–51. doi:10.1016/j.apsoil.2005.12.004
Rincon A, de Felipe MR, Fernandez-Pascual M (2007) Inoculation of Pinus halepensis Mill. With selected ectomycorrhizal fungi improves seedling establishment 2 years after planting in a degraded gypsum soil. Mycorrhiza 18:23–32. doi:10.1007/s00572-007-0149-y
Ruiz-Lozano JM (2003) Arbuscular mycorrhizal symbiosis and alleviation of osmotic stress. New perspectives for molecular studies. Mycorrhiza 13:309–317. doi:10.1007/s00572-003-0237-6
Rzedowski J (2006) Vegetación de México. First digital edn. Comisión Nacional para el Conocimiento y Uso de la Biodiversidad, México
Sade N, Gebremedhin A, Moshelion M (2012) Risk-taking plants. Plant Signal Behav 7:767–770. doi:10.4161/psb.20505
Sandoval I (2010) Efecto de los hongos ectomicorrizógenos en el crecimiento y supervivencia de plántulas de Pinus hartwegii Lindl. y Abies religiosa (Kunth Schltdl. et Cham.): Un enfoque para la restauración de ambientes deteriorados en la Cuenca del Río Magdalena D.F. Master's Dissertation, Universidad Nacional Autónoma de México
Smith SE, Read DJ (2008) Mycorrhizal symbiosis, Third edn. Academic Press, Oxford. doi:10.1016/B978-012652840-4/50001-2
Tammi H, Timonen S, Sen R (2001) Spatiotemporal colonization of scots pine roots by introduced and indigenous ectomycorrhizal fungi in forest humus and nursery Sphagnum peat microcosms. Can J For Res/Rev Can Rech For 31:746–756. doi:10.1139/x01-011
Trocha LK, Kalucha L, Stasińska M, Nowak W, Dabert M, Leski T, Rudawska M, Oleksyn J (2012) Ectomycorrhizal fungal communities of native and non-native Pinus and Quercus species in a common garden of 35-year-old trees. Mycorrhiza 22:121–134. doi:10.1007/s00572-011-0387-x
Uroz S, Frey-Klett P (2011) Linking diversity to function: highlight on the mineral weathering bacteria. Cent Eur J Biol 6:817–820. doi:10.2478/s11535-011-0053-5
Varela L, Estrada-Torres A (1997) Diversity and potential use of mycorrhizae for sustainable development in Mexico. In: Palm ME, Chapela IH (eds) Mycology in sustainable development: expanding concepts. Vanishing Borders. Parkway Publishers, Inc., pp 160–1882
Verslues PE, Agarwal M, Katiyar-Agarwal S, Zhu J, Zhu J-K (2006) Methods and concepts in quantifying resistance to drought, salt and freezing, abiotic stresses that affect plant water status. Plant J 45:523–539. doi:10.1111/j.1365-313X.2005.02593.x
WRB IWG (2006) World reference base for soil resources 2006 vol 103. World Soil Resources Reports. FAO, Rome
Zhang J, Nguyen HT, Blum A (1999) Genetic analysis of osmotic adjustment in crop plants. J Exp Bot 50:291–302. doi:10.1093/jxb/50.332.291
Zhang HH, Tang M, Chen H, Zheng CL (2010) Effects of inoculation with ectomycorrhizal fungi on microbial biomass and bacterial functional diversity in the rhizosphere of Pinus tabulaeformis seedlings. Eur J Soil Biol 46:55–61. doi:10.1016/j.ejsobi.2009.10.005
Acknowledgements
We are very thankful to Dr. Gema Galindo for her help identifying the mycorrhizae, to Dr. Oscar Briones for his advice on the osmotic potential analysis and mostly to Dr. Margarita Collazo for her help with the infrared gas analyzer. We are also grateful to Juan Carlos Peña, Lizbeth Guzmán and Irene Sánchez Gallén for technical and field assistance. Thanks to Salvador Castro chief of the San Luis Tlaxialtemalco nursery. Additionally, we are grateful to the referees and the manuscript editor for their very constructive input. This project was funded by two grants: “Managing Ecosystems and Human Development in the Magdalena River Basin” (SEDEI-PTID-02), and Programa de Apoyo para la Investigación e Innovación Tecnológica de la Universidad Nacional Autónoma de México (PAPIIT-UNAM IN-200906). The Mexican Council of Science and Technology (CONACyT) awarded a scholarship to Dulce Flores-Rentería for her studies in the Biological Sciences Graduate Program of the Universidad Nacional Autónoma de México.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(PDF 516 kb)
Rights and permissions
About this article
Cite this article
Flores-Rentería, D., Barradas, V.L. & Álvarez-Sánchez, J. Ectomycorrhizal pre-inoculation of Pinus hartwegii and Abies religiosa is replaced by native fungi in a temperate forest of central Mexico. Symbiosis 74, 131–144 (2018). https://doi.org/10.1007/s13199-017-0498-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13199-017-0498-z