Abstract
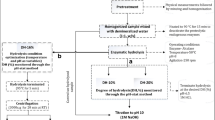
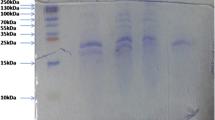
The study aimed to evaluate the extent of hydrolysis on the functional properties of fish protein hydrolysates from fish wastes. Fish protein hydrolysate (FPH) was prepared from the visceral waste of Rohu (Labeo rohita) using Alcalase®. Independent variables of the hydrolysis conditions, namely, time, temperature and enzyme–substrate ratio were optimized through response surface methodology using a completely randomized factorial design. Functional properties of FPH, such as antioxidant activity, metal chelating activity, emulsifying and foaming capacity were studied. Pre-heating of the raw viscera resulted 83% reduction of lipid content in the FPH. The DH was found to be significantly influenced by the hydrolysis conditions studied. The gel electrophoretic study showed that FPH contained peptides in the range of < 10–25 kDa. Optimum degree of hydrolysis (DH) was obtained as 34.7% at 87.5 min, 48.64 °C and enzyme concentration of 0.99%. The results demonstrated that the antioxidant and metal chelating properties of FPH increased with the increase of DH. The surface active properties like emulsifying and foaming capacity were found to be decreased with the increased levels of hydrolysis. These results suggest that hydrolysates from visceral waste of Rohu could find potential use as supplement in animal feed.



Similar content being viewed by others
References
Adler-Nissen J (1986) Enzymic hydrolysis of food proteins. Elsevier Applied Science Publishers, Amsterdam
AOAC (2002) Official methods of analysis, 17th edn. Association of Official Analytical Chemists, Gaithersburg, Maryland, USA
Batista I, Ramos C, Coutinho J, Bandarra NM, Nunes ML (2010) Characterization of protein hydrolysates and lipids obtained from black scabbard fish (Aphanopus carbo) by-products and antioxidative activity of the hydrolysates produced. Process Biochem 45:18–24
Benjakul S, Morrissey MT (1997) Protein hydrolysates from Pacific whiting solid wastes. J Agric Food Chem 45(9):3423–3430
Bernardi Don LS, Pilosof AMR, Bartholomai GB (1991) Enzymatic modification of soy protein concentrates by fungal and bacterial proteases. J Am Oil Chem Soc 68:102–105
Bhaskar N, Modi VK, Govindaraju K, Radha C, Lalitha RG (2007) Utilisation of meat industry byproducts: protein hydrolysate from sheep visceral mass. Biores Technol 98:388–394
Bhaskar N, Benila T, Radha C, Lalitha RG (2008) Optimization of enzymatic hydrolysis of visceral waste proteins of Catla (Catla catla) for preparing protein hydrolysate using a commercial protease. Biores Technol 99(2):335–343
Chalamaiah M, Jyothirmayi T, Bhaskarachary K, Vajreswari A, Hemalatha R, Kumar BD (2013) Chemical composition, molecular mass distribution and antioxidant capacity of rohu (Labeo rohita) roe (egg) protein hydrolysates prepared by gastrointestinal proteases. Food Res Int 52(1):221–229
Chalamaiah M, Hemalatha R, Jyothirmayi T, Diwan P V, Bhaskarachary K, Vajreswari A, Kumar B D (2015) Chemical composition and immunomodulatory effects of enzymatic protein hydrolysates from common carp (Cyprinus carpio) egg. Nutrition 31(2):388–398
Decker EA, Welch B (1990) Role of ferritin as a lipid oxidation catalyst in muscle food. J Agric Food Chem 38(3):674–677
Elavarasan K, Naveen Kumar V, Shamasundar BA (2012) Antioxidant and functional properties of fish protein hydrolysates from fresh water carp (Catla catla) as influenced by the nature of enzyme. J Food Process Pres 38(3):1207–1214
FAO (2006) The State of World Fisheries and Aquaculture, 2006
Guerard F, Dufosse L, De La Broise D, Binet A (2001) Enzymatic hydrolysis of proteins from yellowfin tuna (Thunnus albacares) wastes using Alcalase. J Mol Catal B-Enzym 11(4):1051–1059
Guerard F, Guimas L, Binet A (2002) Production of tuna waste hydrolysates by a commercial neutral protease preparation. J Mol Catal B-Enzym 19:489–498
Hoyle NT, Merrltt JOHN (1994) Quality of fish protein hydrolysates from herring (Clupea harengus). J Food Sci 59(1):76–79
Intarasirisawat R, Benjakul S, Visessanguan W, Wu J (2012) Antioxidative and functional properties of protein hydrolysate from defatted skipjack (Katsuwonous pelamis) roe. Food Chem 135(4):3039–3048
Klompong V, Benjakul S, Kantachote D, Shahidi F (2007) Antioxidant activity and functional properties of protein hydrolysate of yellow stripe trevally (Selaroides leptolepis) as influenced by the degree of hydrolysis and enzyme type. Food Chem 102(4):1317–1327
Kristinsson HG, Rasco BA (2000) Fish protein hydrolysates: production, biochemical, and functional properties. Crit Rev Food Sci Nutr 40(1):43–81
Kulkarni AP, Aradhya SM (2005) Chemical changes and antioxidant activity in pomegranate arils during fruit development. Food Chem 93(2):319–324
Laemmli UK (1970) SDS-page Laemmli method. Nature 227:680–685
Linder M, Fanni J, Parmentier M (1996) Functional properties of veal bone hydrolysates. J Food Sci 61(4):712–716
Loizzo MR, Tundis R, Bonesi M, Menichini F, Mastellone V, Avallone L, Menichini F (2012) Radical scavenging, antioxidant and metal chelating activities of Annonacherimola Mill (Cherimoya) peel and pulp in relation to their total phenolic and total flavonoid contents. J Food Compos Anal 25:179–184
Molla AE, Hovannisyan HG (2011) Optimization of enzymatic hydrolysis of visceral waste proteins of beluga Huso huso using Protamex. Int Aquatic Res 3:93–99
Nasri R, Younes I, Jridi M, Trigui M, Bougatef A, Nedjar-Arroume N, Karra-Châabouni M (2013) ACE inhibitory and antioxidative activities of Goby (Zosterissessorophio cephalus) fish protein hydrolysates: effect on meat lipid oxidation. Food Res Int 54(1):552–561
Ovissipour MR, Ghomi MR (2009) Biotechnology in seafood production. Islamic Azad University Publication, Tehran, Iran, pp 1–198
Ovissipour M, Abedian A, Motamedzadegan A, Rasco B, Safari R, Shahiri H (2009) The effect of enzymatic hydrolysis time and temperature on the properties of protein hydrolysates from Persian sturgeon (Acipenser persicus) viscera. Food Chem 115(1):238–242
Ovissipour M, Benjakul S, Safari R, Motamedzadegan A (2010) Fish protein hydrolysates production from yellowfin tuna Thunnus albacares head using Alcalase and Protamex. Int Aquat Res 2(2):87–95
Pasupuleti VK, Braun S (2008) State of the art manufacturing of protein hydrolysates. In: Pasupuleti V, Demain A (eds) Protein hydrolysates in biotechnology. Springer, Dordrecht, pp 11–32
Rakesh J, Metz A (1973) Acid precipitated fish protein isolate exhibits good functional properties. Food Prod Dev 7:18–24
Shankar TJ, Sokhansanj S, Bandyopadhyay S, Bawa AS (2008) A case study on optimization of biomass flow during single-screw extrusion cooking using genetic algorithm (GA) and response surface methodology (RSM). Food Bioproc Technol 3(4):498–510. https://doi.org/10.1007/s11947-008-0172-9
Vidotti RM, Viegas EMM, Carneiro DJ (2003) Amino acid composition of processed fish silage using different raw materials. Anim Feed Sci Technol 105:199–204
Vijayalakshmi MA, Lemieux L, Amiot J (1986) High performance size exclusion liquid chromatography of small molecular weight peptides from protein hydrolysates using methanol as a mobile phase additive. J Liq Chromatogr Relat Technol 9(16):3559–3576
Yang JI, Ho HY, Chu YJ, Chow CJ (2008) Characteristic and antioxidant activity of retorted gelatine hydrolysates from cobia (Rachycentron canadum) skin. Food Chem 110(1):128–136
You L, Zhao M, Cui C, Zhao H, Yang B (2009) Effect of degree of hydrolysis on the antioxidant activity of loach (Misgurnus anguillicaudatus) protein hydrolysates. Innov Food Sci Emerg Technol 10(2):235–240
Zynudheen AA (2012) Utilization of fishery waste in India. CIFT Publication, Central Institute of Fisheries Technology, Cochin
Acknowledgement
The authors would like to thank the Dean, College of Fisheries (CAU), Lembucherra, Tripura (W), and India for providing facilities to conduct this research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mohanty, U., Majumdar, R.K., Mohanty, B. et al. Influence of the extent of enzymatic hydrolysis on the functional properties of protein hydrolysates from visceral waste of Labeo rohita. J Food Sci Technol 58, 4349–4358 (2021). https://doi.org/10.1007/s13197-020-04915-3
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-020-04915-3