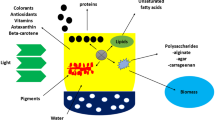
Abstract
The pulsed light (PL) technique is used for food and surface decontamination widely. The sterilization effect of PL is well known and identified as the photo-chemical effect. Besides, PL is used to inactivate enzymes, reduce the immunoreactivity of proteins, and change protein function properties at a laboratory level. The current study aims to review the effect of PL on proteins by highlighting the differences between proteins in buffer solutions or food systems. Although PL is known as a non-thermal technique, most studies done on food systems, food temperature raised considerably. Therefore, PL inactivated many enzymes in buffer solution non-thermally, while mostly with a high increase in temperature of a food system. PL reduced food allergens several folds in some foods. However, immunoreactivity responses of some protein were increased after PL treatment. Also, the current study covers the conformational changes of proteins that occur because of PL treatment. Therefore, some techniques used to follow proteins structural changes such as polyacrylamide gel electrophoresis (SDS-PAGE), high-performance liquid chromatography (HPLC), Fourier-transform infrared spectroscopy (FTIR), etc. were defined. Studies reported that PL altered proteins structure differently. For example, some studies reported that PL degraded some proteins, while other studies suggested that PL aggregated proteins. Also, there were contrary results regarding α-helix and ß-sheet concentration for the treated proteins. In conclusion, some techniques, such as amino acid sequencing, specially when some small new fragments proteins appeared on SDS-PAGE, should be used to detect the effect of PL on proteins precisely.
Similar content being viewed by others
References
Aguilar K, Ibarz R, Garvín A, Ibarz A (2016) Effect of UV–vis irradiation on enzymatic activities and the physicochemical properties of nectarine juices from different varieties. LWT—Food Sci 65:969–977
Aguiló-Aguayo I, Charles F, Renard CMGC, Page D, Carlin F (2013) Pulsed light effects on surface decontamination, physical qualities and nutritional composition of tomato fruit. Postharvest Biol Technol 86:29–36
Aitken A, Learmonth MP (2009) Protein determination by UV absorption. The protein protocols handbook. Humana Press, Totowa, NJ, pp 3–6
Alhendi A (2016) Inactivation of Lipoxygenase in soymilk and soybean by pulsed light. University of Florida, Gainesville, FL, p 2016
Alhendi A (2020) A Review: protein identification by LC-MS: principles, instrumentation and applications. Iraqi J Sci 61(10):2428–2466
Alhendi A, Yang W, Sarnoski PJ (2018) The effect of solution properties on the photochemical ability of pulsed light to inactivate soybean lipoxygenase. IJFE 14:5–6
Alhendi A, Yang W, Goodrich-Schneider R, Sarnoski PJ (2017) Inactivation of lipoxygenase in soymilk by pulsed light. IJFE 13:12
Alhendi A, Yang W, Goodrich-Schneider R, Sarnoski PJ (2018) Total inactivation of lipoxygenase in whole soya bean by pulsed light and the effect of pulsed light on the chemical properties of soya milk produced from the treated soya beans. Int J Food Sci Tech 53(2):457–466
Alhendi A, Yang W, Goodrich-Schneider R, Sims C, Marshall S, Sarnoski PJ (2018) Sensory evaluation and flavour analysis of soymilk produced from lipoxygenase-free soya beans after modified processes and pulsed light treatment. Int J Food Sci Tech 53(6):1434–1441
Anthon GE, Barrett DM (2002) Kinetic parameters for the thermal inactivation of quality-related enzymes in carrots and potatoes. J Agric Food Chem 50(14):4119–4125. https://doi.org/10.1021/jf011698i
Antonio Pellicer J, Navarro P, Gómez-López VM (2019) Pulsed light inactivation of polygalacturonase. Food Chem 271:109–113
Aravindan R, Anbumathi P, Viruthagiri T (2007) Lipase applications in food industry. IJBT 6(2):141–158
Battais F, Mothes T, Moneret-Vautrin DA, Pineau F, Kanny G, Popineau Y, Bodinier M, Denery-Papini S (2005) Identification of IgE-binding epitopes on gliadins for patients with food allergy to wheat. Allergy 60(6):815–821
Burks AW, Jones SM, Wood RA, Fleischer DM, Sicherer SH, Lindblad RW, Stablein D, Henning AK, Vickery BP, Liu AH (2012) Oral immunotherapy for treatment of egg allergy in children. N Engl J Med 367(3):233–243
Carbonaro M, Maselli P, Nucara A (2012) Relationship between digestibility and secondary structure of raw and thermally treated legume proteins: a fourier transform infrared (FT-IR) spectroscopic study. Amino Acid 43(2):911–921
Chang SKC, Zhang Y (2017) Protein analysis. In: Nielsen SS (ed) Food analysis. Springer International Publishing, New york
Chisari M, Barbagallo RN, Spagna G (2008) Characterization and role of polyphenol oxidase and peroxidase in browning of fresh-cut melon. J Agric Food Chem 56(1):132–138
Chung S-Y, Yang W, Krishnamurthy K (2008) Effects of pulsed UV-light on peanut allergens in extracts and liquid peanut butter. J Food Sci 73(5):C400–C404
Ciou J-Y, Lin H-H, Chiang P-Y, Wang C-C, Charles AL (2011) The role of polyphenol oxidase and peroxidase in the browning of water caltrop pericarp during heat treatment. Food Chem 127(2):523–527
del Castillo-Santaella T, Sanmartín E, Cabrerizo-Vílchez MA, Arboleya JC, Maldonado-Valderrama J (2014) Improved digestibility of β-lactoglobulin by pulsed light processing: a dilatational and shear study. Soft Matter 10(48):9702–9714
Deslauriers M, Eidhin D, ní, Lamonde, L., & Mouton, C. (1990) SDS-PAGE analysis of protein and lipopolysaccharide of extracellular vesicules and sarkosyl-insoluble membranes from bacteroides gingivalis. Oral Microbiol Immunol 5(1):1–7. https://doi.org/10.1111/j.1399-302X.1990.tb00217.x
Dunn JE, Clark RW, Asmus JF, Pearlman JS, Boyer K, Painchaud F, & Hofmann GA (1989) Methods for preservation of foodstuffs. Google Patents.
Elmnasser N, Dalgalarrondo M, Orange N, Bakhrouf A, Haertlé T, Federighi M, Chobert J-M (2008) Effect of pulsed-light treatment on milk proteins and lipids. J Agric Food Chem 56(6):1984–1991
Elmnasser N, Guillou S, Leroi F, Orange N, Bakhrouf A, Federighi M (2007) Pulsed-light system as a novel food decontamination technology: a review. Can J Microbiol 53(7):813–821
Fachin D, Smout C, Verlent I, Ly Nguyen B, Van Loey AM, Hendrickx ME (2004) Inactivation kinetics of purified tomato polygalacturonase by thermal and high-pressure processing. J Agric Food Chem 52(9):2697–2703. https://doi.org/10.1021/jf030531h
Falguera V, Garza S, Pagán J, Garvín A, Ibarz A (2013) Effect of UV–Vis irradiation on enzymatic activities and physicochemical properties of four grape musts from different varieties. Food Bioproc Tech 6(8):2223–2229
Fernández E, Artiguez ML, de Marañón IM, Villate M, Blanco FJ, Arboleya J-C (2012) Effect of pulsed-light processing on the surface and foaming properties of β-lactoglobulin. Food Hydrocoll 27(1):154–160
Gessendorfer B, Koehler P, Wieser H (2009) Preparation and characterization of enzymatically hydrolyzed prolamins from wheat, rye, and barley as references for the immunochemical quantitation of partially hydrolyzed gluten. Anal Bioanal Chem 395(6):1721
Gómez-López VM, Ragaert P, Debevere J, Devlieghere F (2007) Pulsed light for food decontamination: a review. Trends in Food Sci Tech 18(9):464–473
Greenfield NJ (2006) Using circular dichroism spectra to estimate protein secondary structure. Nat Protoc 1(6):2876–2890. https://doi.org/10.1038/nprot.2006.202
Jackson M, Mantsch HH (1991) Protein secondary structure from FT-IR spectroscopy: correlation with dihedral angles from three-dimensional ramachandran plots. Can J Chem 69(11):1639–1642
Janve BA, Yang W, Marshall MR, Reyes-De-Corcuera JI, Rababah TM (2014) Nonthermal inactivation of soy (glycine max sp.) lipoxygenase by pulsed ultraviolet light. J Food Sci 79(1):C8–C18
Jeon M-S, Park K-M, Yu H, Park J-Y, Chang P-S (2019) Effect of intense pulsed light on the deactivation of lipase: enzyme-deactivation kinetics and tertiary structural changes by fragmentation. Enzyme Microb Technol 124:63–69
John D, Ramaswamy HS (2018) Pulsed light technology to enhance food safety and quality: a mini-review. Curr Opin Food Sci 23:70–79
Knapp MJ, Klinman JP (2003) Kinetic studies of oxygen reactivity in soybean lipoxygenase-1. Biochemistry 42(39):11466–11475
Kobayashi Y, Mayer SG, Park JW (2017) FT-IR and Raman spectroscopies determine structural changes of tilapia fish protein isolate and surimi under different comminution conditions. Food Chem 226:156–164
Kumar V, Sharma VK, Kalonia DS (2005) Second derivative tryptophan fluorescence spectroscopy as a tool to characterize partially unfolded intermediates of proteins. Int J Pharm 294(1–2):193–199
Li Y, Yang W, Chung S-Y, Chen H, Ye M, Teixeira AA, Gregory JF, Welt BA, Shriver S (2013) Effect of pulsed ultraviolet light and high hydrostatic pressure on the antigenicity of almond protein extracts. Food Bioproc Tech 6(2):431–440
Li Y-Q, Chen Q, Liu X-H, Chen Z-X (2008) Inactivation of soybean lipoxygenase in soymilk by pulsed electric fields. Food Chem 109(2):408–414
Lobley A, Whitmore L, Wallace BA (2002) DICHROWEB: An interactive website for the analysis of protein secondary structure from circular dichroism spectra. Bioinformatics 18(1):211–212
Manzocco L (2015) Photo-induced modification of food protein structure and functionality. Food Eng Rev 7(3):346–356. https://doi.org/10.1007/s12393-015-9110-6
Manzocco L, Panozzo A, Nicoli MC (2013a) Effect of pulsed light on selected properties of egg white. IFSET 18:183–189
Manzocco L, Panozzo A, Nicoli MC (2013b) Inactivation of polyphenoloxidase by pulsed light. J Food Sci 78(8):E1183–E1187
Manzocco L, Quarta B, Dri A (2009) Polyphenoloxidase inactivation by light exposure in model systems and apple derivatives. IFSET 10(4):506–511. https://doi.org/10.1016/j.ifset.2009.02.004
Meinlschmidt P, Ueberham E, Lehmann J, Reineke K, Schlüter O, Schweiggert-Weisz U, Eisner P (2016) The effects of pulsed ultraviolet light, cold atmospheric pressure plasma, and gamma-irradiation on the immunoreactivity of soy protein isolate. IFSET 38:374–383
Morales-Blancas EF, Chandia VE, Cisneros-Zevallos L (2002) Thermal inactivation kinetics of peroxidase and lipoxygenase from broccoli, green asparagus and carrots. J Food Sci 67(1):146–154. https://doi.org/10.1111/j.1365-2621.2002.tb11375.x
Nguyen TD, Leung K-F, Liong M, Liu Y, Stoddart JF, Zink JI (2007) Versatile supramolecular nanovalves reconfigured for light activation. Adv Funct Mater 17(13):2101–2110
Nooji JK (2011) Reduction of wheat allergen potency by pulsed ultraviolet light, high hydrostatic pressure, and non-thermal plasma [PhD Thesis]. University of Florida.
Ogawa T, Samoto M, Takahashi K (2000) Soybean allergens and hypoallergenic soybean products. J Nutr Sci Vitaminol 46(6):271–279
Oms-Oliu G, Martín-Belloso O, Soliva-Fortuny R (2010) Pulsed light treatments for food preservation. A Review Food Bioproc Tech 3(1):13–23
Queiroz JA, Tomaz CT, Cabral JMS (2001) Hydrophobic interaction chromatography of proteins. J Biotechnol 87(2):143–159
Panozzo A, Manzocco L, Lippe G, Nicoli MC (2016) Effect of pulsed light on structure and immunoreactivity of gluten. Food Chem 194:366–372
Pellicer JA, Gómez-López VM (2017) Pulsed light inactivation of horseradish peroxidase and associated structural changes. Food Chem 237:632–637
Pellicer JA, Navarro P, Gómez-López VM (2018) Pulsed light inactivation of mushroom polyphenol oxidase: a fluorometric and spectrophotometric study. Food Bioproc Tech 11(3):603–609
Pellicer JA, Navarro P, Hernández Sánchez P, Gómez-López VM (2019) Structural changes associated with the inactivation of lipoxygenase by pulsed light. LWT 113:108332. https://doi.org/10.1016/j.lwt.2019.108332
Schmid F-X (2001) Biological macromolecules: UV-visible spectrophotometry. ACS, In ELS. https://doi.org/10.1038/npg.els.0003142
Sharma VK, Kalonia DS (2003) Steady-state tryptophan fluorescence spectroscopy study to probe tertiary structure of proteins in solid powders. J Pharm Sci 92(4):890–899. https://doi.org/10.1002/jps.10354
Shriver SK, Yang WW (2011) Thermal and nonthermal methods for food allergen control. Food Eng Rev 3(1):26–43
Shriver S, Yang W, Chung S-Y, Percival S (2011) Pulsed ultraviolet light reduces immunoglobulin E binding to Atlantic white shrimp (Litopenaeus setiferus) extract. Int J Environ Res 8(7):2569–2583
Siddique MAB, Maresca P, Pataro G, Ferrari G (2016) Effect of pulsed light treatment on structural and functional properties of whey protein isolate. Food Res Int 87:189–196
Singh MB, Knox RB (1985) Grass pollen allergens: antigenic relationships detected using monoclonal antibodies and dot blotting immunoassay. Int Arch Allergy Immunol 78(3):300–304. https://doi.org/10.1159/000233901
Taylor SC, Berkelman T, Yadav G, Hammond M (2013) A defined methodology for reliable quantification of western blot data. Mol Biotechnol 55(3):217–226. https://doi.org/10.1007/s12033-013-9672-6
Terefe NS, Yang YH, Knoerzer K, Buckow R, Versteeg C (2010) High pressure and thermal inactivation kinetics of polyphenol oxidase and peroxidase in strawberry puree. IFSET 11(1):52–60
Neves-Petersen MT, Gajula GP, Petersen SB (2012) UV light effects on proteins: from photochemistry to nanomedicine. In: Molecular photochemistry-various aspects, pp 125–158
Tiwari RS, Venkatachalam M, Sharma GM, Su M, Roux KH, Sathe SK (2010) Effect of food matrix on amandin, almond (Prunus dulcis L) major protein, immunorecognition and recovery. LWT—Food Sci 43(4):675–683
van den Broeck HC, van Herpen TW, Schuit C, Salentijn EM, Dekking L, Bosch D, Hamer RJ, Smulders MJ, Gilissen LJ, van der Meer IM (2009) Removing celiac disease-related gluten proteins from bread wheat while retaining technological properties: A study with Chinese Spring deletion lines. BMC Plant Biol 9(1):41
Vickery BP, Lin J, Kulis M, Fu Z, Steele PH, Jones SM, Scurlock AM, Gimenez G, Bardina L, Sampson HA (2013) Peanut oral immunotherapy modifies IgE and IgG4 responses to major peanut allergens. J Allergy Clin Immunol 131(1):128–134
Wachholz PA, Dearman RJ, Kimber I (2005) Detection of allergen-specific IgE antibody responses. J Immunotoxicol 1(3–4):189–199
Wang B, Zhang Y, Venkitasamy C, Wu B, Pan Z, Ma H (2017) Effect of pulsed light on activity and structural changes of horseradish peroxidase. Food Chem 234:20–25
Wang Y, Li D, Ren W, Liu Z, Dong S, Wang E (2008) Ultrasensitive colorimetric detection of protein by aptamer-Au nanoparticles conjugates based on a dot-blot assay. Chem Comm 22:2520. https://doi.org/10.1039/b801055b
Wihodo M, Moraru CI (2015) Effect of Pulsed Light treatment on the functional properties of casein films. LWT—Food Sci 64(2):837–844
Yang WW, Chung S-Y, Ajayi O, Krishnamurthy K, Konan K, Goodrich-Schneider R (2010) Use of pulsed ultraviolet light to reduce the allergenic potency of soybean extracts. IJFE. https://doi.org/10.2202/1556-3758.1876
Yang WW, Mwakatage NR, Goodrich-Schneider R, Krishnamurthy K, Rababah TM (2012) Mitigation of major peanut allergens by pulsed ultraviolet light. Food Bioproc Tech 5(7):2728–2738
Zhao X, Yang W, Chung S-Y, Sims CA, Otwell SW, Rababah TM (2014) Reduction of IgE immunoreactivity of whole peanut (Arachis hypogaea L) after pulsed light illumination. Food Bioproc Tech 7(9):2637–26
Acknowledgement
The author thanks all the authors that worked on the effect of PL on proteins. Without their studies, this review cannot found. Also, the author appreciates all the reviewer suggestions that improve the manuscript considerably.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
This author declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Alhendi, A.S. Effect of pulsed light treatment on enzymes and protein allergens associated with their structural changes: a review. J Food Sci Technol 58, 2853–2862 (2021). https://doi.org/10.1007/s13197-020-04882-9
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-020-04882-9