Abstract
Lipase-catalyzed esterification is an efficient technique in the production of polyunsaturated fatty acid (PUFA) concentrates which are applied for nutrition and health purposes. In this project, a solvent-tolerant lipase from Streptomyces pratensis MV1 was immobilized and purified by a hydrophobic support. The purified lipase revealed enhanced activity and stability towards chemicals, organic solvents, and a broad range of pH values. The production of lipase was enhanced to 7.0 U/mL after optimization by a central composite design. Acylglycerols (AGs) rich in α-linolenic acid (45%, w/w) were produced and a favorable n-6/n-3 free fatty acid (FFA) ratio of 1.1 was achieved in fenugreek seed oil using the immobilized lipase. The ability of S. pratensis lipase in ester synthesis and the improvement of n6/n3 FFA ratio make it a suitable candidate in food production industries.
Similar content being viewed by others
Introduction
Polyunsaturated fatty acids (PUFAs) particularly α-linolenic acid (ALA) are health-promoting fatty acids, because of their role in reducing blood LDL cholesterol, regulation of inflammation, brain functions, fluidity or integrity of membrane bilayer structures, vasodilation, preventing cardiac arrhythmias, and reducing thrombosis by conversion to eicosanoids (Nicholson et al. 2013). Recent studies have shown that PUFAs can strengthen the immune system and increase resistance and recovery from virus diseases such as the Middle East Respiratory Syndrome (MERS) and the Severe Acute Respiratory Syndrome (SARS-CoV-2) infections (Galanakis 2020). The human body does not contain enzymes to biosynthesize ALA, so it should be supplied in the diet. One of the current strategies to develop PUFA functional foods is mainly focused on the addition of one or a combination of PUFAs from natural sources to nutrients to enhance their total content (Granato et al. 2020). Utilization of conventional recovery methods such as thermal technologies are often restricted due to high energy consumption, overheating of the food matrix, loss of functionality and poor stability of the final product (Galanakis 2012). In order to produce functional foods, it is important to explore alternative processing technologies (e.g., high hydrostatic pressure (Kovačević et al. 2018), pulsed electric fields (Barba et al. 2015), ultrasounds (Deng et al. 2015; Zinoviadou et al. 2015a, b), and microwaves (Sarfarazi et al. 2020; Deng et al. 2015; Zinoviadou et al. 2015). These sustainable technologies provide more efficient preservation of native nutrients in food, and consume less energy (Galanakis et al. 2013). PUFAs are very sensitive to autoxidation causing undesirable taste and odor during food processing. To overcome these problems during processing and storage, multiple strategies have been explored such as the use of antioxidants (Galanakis 2018; Galanakis et al. 2018), encapsulation approaches (e.g., freeze-drying, spray-drying, and spray granulation), electrospray, microgels, and enzymatic synthesis. Enzymatic synthesis of PUFA in acylglycerols (AGs) form in the presence of organic solvents provides various industrial advantages such as suppression of water-dependent side reactions, increase in solubility of reactants, and elimination of microbial contamination (Granato et al. 2020; Imanparast et al. 2018; Umesha, 2014).
Lipases (acylglycerol acyl hydrolases, EC 3.1.1.3.) can be successfully used as biocatalysts for hydrolysis, esterification, interesterification, and modification of oils (Gandhi 1997). Lipase-catalyzed esterification can be a suitable method for producing PUFA concentrates as AGs which are applied in nutrition, clinical, health, pharmaceutical, and medical purposes (Akoh and Sista 1995). Productions of lipases are widely reported in animals, plants, and microorganisms, but microbial lipases are commercially applied in biotechnological applications due to their greater stability and wider availability (Chakraborty and Raj 2009).
The concentration of PUFA from a wide range of organisms has been reported in previous studies, such as fish oil, marine alga (Guil-Guerrero et al. 2007), vegetable oil (Senanayake and Shahidi 2004), and some edible oils (López-Martínez et al. 2005). Vegetable oils could be an appropriate source for high concentrates of PUFA owing to their low cost, availability, and absence of heavy metal and hence toxicity concerns (Zahir et al. 2005).
Fenugreek belongs to the family of Fabaceae and is indigenous to southeastern Europe and Western Asia (El Nasri and El Tinay 2007). Fenugreek seeds containing 7.5% oil are a suitable source of PUFA containing large amounts of ALA and linoleic acid with 66–82% and 26–40%, respectively. The concentration of PUFAs in the oil of fenugreek seeds by means of enzyme has not been reported so far. There are several studies on the physiochemical characteristics of fenugreek seeds (El-Sebaiy and El-Mahdy 1983). The ratio of n-6/n-3 free fatty acid (FFA) in the common diet of western people is 10–30. Due to high consumption of plant oils and relatively low-available sources of marine products, a ratio of less than 5 is recommended by nutritionists (Orsavova et al. 2015). Thus, functional foods rich in n-3 PUFAs are suggested mainly for vegetarians, whose blood has characteristically high n-6/n-3 ratios (Imanparast et al. 2018).
The present study was designed to screen and identify a solvent-tolerant lipase producing microorganism followed by simultaneous purification and immobilization of the enzyme. The efficiency of an immobilized solvent-tolerant lipase of Streptomyces pratensis MV1 was investigated for the enzymatic esterification of acylglycerols rich in omega-3 PUFA of fenugreek seed oil. According to Literature, there is no report on the isolation of a solvent-tolerant lipase of this microorganism; hence, this is the first attempt to report a solvent-tolerant lipase from S. pratensis MV1 and its application for production of acylglycerols rich in n-3 PUFA. Furthermore, the immobilized lipase showed high efficiency to n-3 for enzymatic production of functional foods with high content of n-3 PUFAs.
Materials and methods
Chemicals and reagents
Rhodamine B, Pluronics P123, tetraethoxysilane (TEOS), (3-aminopropyl)triethoxysilane (APTES), dimethylformamide (DMF), and p-nitrophenyl palmitate (p-NPP) were supplied from Sigma (St. Louis, MO, USA). Surfactants, bovine serum albumin, sodium dodecyl sulfate (SDS), urea, and media components were purchased from Merck (Darmstadt, Germany). Other chemicals and solvents were of analytical grade.
Isolation and identification of the solvent-tolerant lipase producing strain
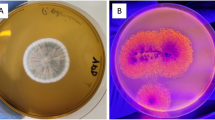
Lipase-producing bacteria were simultaneously isolated and screened by Rhodamine B agar plate from the oil-contaminated regions, petroleum refineries, and coal mines in Iran. The Rhodamine B agar plates were incubated at 28 °C for 48–168 h and the isolates with orange fluorescent halos under UV irradiation (350 nm) were applied for secondary screening by a colorimetric assay (Hasan-Beikdashti et al. 2012). The stability of the enzyme was examined in the presence of organic solvents (25%, v/v) including methanol, tert-butyl alcohol, toluene, hexane, and cyclohexane. The lipolytic isolate with maximal stability in multiple solvents was identified based on molecular characterization and preserved at − 196 °C for further investigation. Further details are mentioned in the Supplementary Material.
The enzyme assay
The enzyme substrate p-NPP was used to assay the lipase activity according to the method of Hasan-Beikdashti et al. (2012). The substrate solution (1 mM) was prepared and added to the reaction with the incubation of mixture performed at 45 °C for 10 min. The lipase activity was stopped by acetone/ethanol (1:1, v/v) and the release of p-NP was monitored at 420 nm. One lipase unit is defined as the amount of the enzyme required to release 1 µmol p-NP/min mL.
Production optimization of lipase
The optimization of the lipase production was performed in the fermentation medium containing (g/L) yeast extract, (0.3–6.2); peptone, (1.6–18.4); olive oil, (0.5–3.0% v/v), NH4Cl, 5; NaCl, 9; MgSO4·7H2O, 0.5 by means of response surface method (RSM). A central composite design (CCD) was employed including peptone (X 1), yeast extract (X2), and olive oil (X3). The variables were determined at five levels (− α, − 1, 0, + 1, and + α), and the central point with six replications. A total of 20 experiments with different settings were obtained by Design Expert software (version 10.0.7, Stat-Easy Inc. Minneapolis, MN, USA). The significance of experimental data was evaluated by Analysis of Variance (ANOVA) statistical test.
Immobilization and purification of the bacterial lipase
The mesoporous SBA-15@oleic acid was synthesized according to the method reported previously (Imanparast and Faramarzi 2018). Briefly, Pluronics P123 and TEOS were utilized as a structure-directing agent and silica source, respectively. The crystallization of the resulting gel was performed at 130 °C for 24 h followed by calcination to remove surfactant at 550 °C for 6 h. APTES was used for amino-functionalization of SBA-15. SBA-15@oleic acid was prepared by mixing the amine-functionalized support with DMF, alkalinized oleic acid, and trimethylamine for 24 h. The enzyme immobilization was carried out by mixing 1 g of SBA-15@oleic acid with 50 mL of concentrated protein solution in phosphate buffer (50 mM, pH 7.4) at 25 °C for 6 h. The obtained immobilized enzyme was washed three times with phosphate buffer to discard unbound proteins. Thereafter, a linear gradient of 0–1% SDS in Tris buffer (50 mM, pH 8) was applied to elution of the support to separate unwanted bound proteins. The eluted fractions with lipase activity were pooled and lyophilized. Specific activity of the purified lipase was calculated with measuring total activity of the purified enzyme/ mass of the enzyme in mg. The total amount of the enzyme was estimated according to the method of Bradford (1976) by bovine serum albumin (BSA) as a standard. The yield (IY) and efficiency (IE) of lipase immobilization was calculated by the following equations (Tarasi et al. 2018): \({\text{IY}}\left( \% \right)\, = \,\left[ {\left( {{\text{Y}}_{0} \, - \,{\text{Y}}_{1} } \right)/{\text{Y}}_{0} } \right]\, \times \,100\), \({\text{IE }}\left( \% \right)\; = \;\left[ {{\text{E}}_{1} /{\text{E}}_{0} } \right]\, \times \,100\); where E0 and E1 represent the activity of total and immobilized enzyme, respectively. Also, Y0 and Y1 indicate the amounts of total protein and protein that did not bind to SBA-15@oleic, respectively.
SDS-PAGE and zymography
The crude and purified lipases were subjected to SDS-PAGE gel electrophoresis on 12% polyacrylamide and stained with silver nitrate (Laemmli 1970). The molecular weight of proteins was determined with the migration rates of standard proteins. Zymography was also performed to visualize the lipase activity. Triton X-100 (2.5%) was utilized to wash and remove SDS. The washed gel along with an agar plate containing phenol red (0.01%) and 1% olive oil (pH 7.3–7.4) were incubated at 45 °C for 20 min. The emergence of distinct yellow bands against a pink background represented lipase activity on the gel (Singh et al. 2006).
Biochemical characterizations of the purified lipase
Thermal and pH stability and the activity of lipase
Glycine–NaOH (pH 10.0–11.0), Tris–HCl (pH 9.0), phosphate (pH 7.0–8), and citrate (pH 3.0–6.0) buffers were used as general solutions to evaluate the simultaneous effect of pH (3.0–11.0) and temperature (15–65 °C) on the relative enzyme activity. The pH and temperature stability were investigated after 60 min of incubation at 25–65 °C in the mentioned buffers. The percentage of residual lipase activity was determined under standard assay conditions.
Organic solvent stability of the enzyme
The effects of different organic solvents on the lipase activity under the standard assay conditions were determined by mixing the enzyme aliquots in 25, 50, and 80% (v/v) of solvents at 25 °C for 60 min. The residual activity was calculated by measuring the enzyme activity in the presence of organic solvent/ the enzyme activity in the absence of organic solvent × 100.
Effect of metal ions, detergents, and inhibitors
The enzyme stability was evaluated in the presence of metal ions including Mg2+, Al3+, Hg2+, Fe2+, Ca2+, Mn2+, and Cu2+ at the concentrations of 1, 5, and 10 mM. Additionally, the influence of some inhibitors and surfactants on the enzyme stability was estimated after 60 min of incubation at 25 °C. The enzyme inhibitors and surfactants were 2-mercaptoethanol (2-ME) (0.1, 0.25, and 0.5%), ethylenediaminetetraacetic acid (EDTA; 1, 5, and 10 mM), urea (1, 5, and 10 mM), sodium dodecyl sulfate (SDS; 0.1, 0.25, and 0.5%), tween 80% (0.1, 0.25, and 0.5%), triton X-100 (0.1, 0.25, and 0.5%), and docusate sodium salt (AOT; 1, 5, and 10 mM). The mentioned materials were prepared in potassium phosphate buffer (pH 7.0).
Enzymatic improvement of n6/n3 ratio in fenugreek seed oil
Extraction of the crude oil
Fenugreek (Trigonella foenum-graceum) seeds were purchased from a local grocery store in Kerman, Iran. They were cleaned and crushed using an electric grinder. The Soxhlet apparatus was used to extract 100 g of the seed flour. The obtained oil was maintained under nitrogen inside dark bottles at − 20 °C prior to further analysis. The synthesis process was divided into four main stages: (i) the extraction of FFAs from fenugreek seed oil, (ii) PUFA concentration, (iii) enzymatic esterification, and (iv) GC analysis. In all cases, the yield of each step was calculated as follows: \({\text{Yield}}\, = \,\left( {{\text{Recovered mass of the desired compound}};{\text{ g}}/{\text{Initial mass of substrate or raw material}};{\text{ g}}} \right)\, \times \,100.\)
Preparation of FFAs from fenugreek seed oil
Free fatty acids from fenugreek seed oil were obtained by saponification (Wanasundara and Shahidi 1999). The oil sample (50 g) was saponified in 1.2 N KOH/EtOH (172 mL) and water (22 mL) at 62 °C for 1 h under a blanket of nitrogen. The unsaponifiable part was discarded by adding 100 mL distillated water and extracted using hexane. The saponifiable layer was acidified by 3 N HCl and the released free fatty acids were extracted using hexane. FFAs with mass yield of 77.7% (w/w) were recovered by a rotary evaporator and stored at − 60 °C.
Concentration of PUFAs
The PUFAs were concentrated using urea-fatty acid complexation method according a recognized process (Wanasundara and Shahidi 1999). FFAs (10 g) were added to a solution of urea (30 g) in 150 mL of 95% aqueous ethanol, with the mixture heated (60–65 °C) and stirred until a homogeneous solution was obtained. The urea complex was incubated at room temperature for 4 h followed by storage overnight at 4 °C. The formed crystals were separated from the liquid by vacuum filtration. The ethanolic solution consisting of PUFAs was diluted with water, where pH decreased to 4–5. Additionally, hexane was added in equal volume while stirring for 1 h. Then, the hexane layer was eluted with distilled water to discard any residual urea. The mass yield of 45.2% (w/w) was calculated for PUFA concentrations.
Esterification reaction
The acylglycerols were synthesized in a screw-capped crystal flask composed of PUFA concentrates (0.25 g), glycerol (1 g), tert-butanol/n-hexane (3 mL; 40:60, v/v), and 200 mg of the immobilized lipase. The reaction solution was incubated for 4 h in an orbital shaker (150 rpm, 45 °C), with molecular sieves added to remove the water formed during the reaction after 1 h. Finally, the immobilized lipase and the molecular sieves were separated by centrifugation. The obtained FFAs were neutralized by titration using 0.025 mol/L NaOH and subjected to thin-layer chromatography (TLC) and gas chromatography (GC) analyses. The acid value was calculated according to the following equation: \({\text{FFA content }}\left( {{\text{w}}/{\text{w }}\% } \right)\, = \,\left[ {\left( {40\, \times \,{\text{N}}\, \times \,{\text{V}}\, \times \,{\text{MW}}_{{{\text{FFA}}}} } \right)/{\text{m}}} \right]\, \times \,100;\)where V is the used volume in mL of NaOH solution, N represents normality of NaOH, m indicates the mass in g of the product taken for the titration, and MWFFA is the molecular weight of FFA. The molecular weight of 279 g/mol was considered as an average molecular weight of FFAs.
Analysis of fatty acid compositions
The samples were applied for TLC on silica 60 F254 and developed in an acetic acid/chloroform/acetone (1:95:4, v/v/v) mobile phase. The reaction products were visualized by iodine vapor as the reagent. The obtained acylglycerols were converted into fatty acid methyl ester (FAME) according to the Metcalfe method (1966) and analyzed via gas chromatography (GC) equipped with a fused silica capillary column (BPX70, 0.25 mm × 30 m i.d.) and a flame ionization detector (FID). The column temperature was initially set at 180 °C for 9 min and increased at the rate of 20 °C/min to 200 °C, and then maintained for 25 min. The identification of fatty acids from Fenugreek oil was performed based on the comparison of their retention times with the pure FAME standards. The GC analysis of the Fenugreek oil revealed that it was not contained pentadecanoic acid (C15:0), so for quantification of fatty acids pentadecanoic acid methyl ester (10 mg/mL) was applied as internal standard (IS) without interfering with the results and the injection volume was 1 μL. Based on the area of IS peak, the concentration of fatty acids were reported as weight percentages according to the following equation: \({\text{FAME content}}\, = \,\left[ {{\text{The peak area of each methyl ester}}\, \times \,\left( {{\text{The concentration of IS}}/{\text{ The peak area of SI}}} \right)} \right]/{\text{ Mass of the sample}}.\)
Statistical analyses
The experiments of this study were performed in triplicate with the obtained results mentioned as mean. Statistical assessments were conducted by ANOVA and values < 0.05 were considered significant. The statistical analyses were done by SigmaPlot version 14.0.
Results
Solvent-tolerant lipase producing strains
Thirty-five lipase-producing isolates were obtained during a screening program using Rhodamine B-agar plate from soil samples. Among them, 5 isolates with a high activity in the liquid medium were then selected for stability screening in various organic solvents (Tables S1 and S2). An isolate was chosen with the maximal lipase activity and organic solvent stability. The isolate was identified as Streptomyces pratensis MV1 (Gene-Bank accession No. MK447746) based on 16S rRNA gene-sequence analysis following comparison to reference strains by EzTaxon database (Fig. S1). The extracellular lipase production of the strain reached to 1950 and 900 U/L after approximately 5 days in LB and basal salt medium, respectively (Fig. S2). More details have been mentioned in the Supplementary Material.
Optimization of lipase production by statistical model
According to the obtained results of RSM (Tables S3 and S4), a quadric polynomial model was proposed. The coefficients of independent variables were determined as follows: \({\text{R}}_{1} \, = \,6.55\, - \,0.9{\text{ X}}_{1} \, + \,0.42{\text{ X}}_{2} \, + \,0.42{\text{ X}}_{3} \, + \,0.52{\text{ X}}_{1} {\text{X}}_{2} \, - \,1.32 \, \left( {{\text{X}}_{1} } \right)^{2} \, - \,0.98 \, \left( {{\text{X}}_{2} } \right)^{2} \, - \,0.51 \, \left( {{\text{X}}_{3} } \right)^{2}.\)The analysis of variance (ANOVA) and F-test were applied for checking the statistical significance of the equation with X1, X2, X3, X1X2, X12, X22, and X32 considered as the most significant factors on the response. Figure 1 presents the interaction effect of variables through 3D surface and counter plots. The optimized medium composition contained (g/L) yeast extract, 1.5; peptone, 7.0; NaCl, 9; MgSO4.7H2O, 0.5; NH4Cl, 5; and olive oil 2.0% v/v. Under the optimized conditions, the production of lipase was three times higher than that of the basal fermentation medium.
Simultaneous purification and immobilization of S. pratensis lipase
The solvent tolerant lipase from S. pratensis was immobilized and purified by SBA-15@oleic acid nanoparticles. The obtained results of SDS-PAGE and silver staining analysis showed no band at 0–0.5% and 1% of SDS, while one protein band with the molecular weight of about 63 kDa was recognized at 0.75% of SDS (Fig. S3). More results and details have been provided in the Supplementary Material. The obtained results of the purification process of lipase are summarized in Table 1. At the end of the procedure, the purity of the enzyme was estimated to be about 14.7-fold greater than that of the crude extract. The purified lipase with a specific activity of 445.45 ± 8.4 U/mg accounted for about 58.0 ± 0.5 of the total activity. The maximum immobilization efficiency and yield were obtained as 59 ± 2.3% and 72 ± 3.2%, respectively, at pH 7 and 6 h in this research.
Biochemical characterization of the purified enzyme
Optimal pH and temperature of the enzyme stability and activity
The lipase revealed maximum stability at pH 8, and maintained 80% of the activity at a pH of 5.0–9.0 at 25 °C. The lipase kept about 50% of the activity after 1 h incubation at 55 °C and pH 7. The results from our study indicated that the maximum activity occurred at 45 °C and pH 8 with the enzyme maintaining 65% of its maximum activity at pH 9.0. The lipase exhibited pH-sensitivity, with a considerable decline in the enzyme activity detected below pH 8.0 and above 9.0. The data of Fig. 2 also displayed that the enzyme had maximal activity at slightly alkaline pH. The enzyme was active within 15–65 °C and retained about 40% of its total activity at 55 °C and pH 8.0.
The combined effects of pH and temperature on the lipase a stability and b activity by determining the residual activities after 60 min of incubation within a wide range of pH values and temperatures. (a) *p < 0.05 versus 25 °C at pH 11 and pH 3; † p < 0.05 versus 65 °C at pH 8. (b) *p < 0.05 versus 65 °C and 55 °C at pH 8; † p < 0.05 versus pH < 8.0 and > 9.0
Stability of lipase in the presence of organic solvents
The enzyme showed approximately 25, 30, 32, and 45% increases in the activity compared to control in the presence of 25% cyclohexane, hexane, n-hexadecane, and isooctane, respectively (Table 2). Although the stability and activity of lipases in organic solvents such as ethanol, methanol, DMFA, and acetone are rarely detected (Ogino and Ishikawa 2001), in the present study, the results revealed that the enzyme retained 98 and 84% of activity in 25% (v/v) of DMSO and methanol, respectively.
Effectors and inhibitors on lipase stability
The lipase stability was moderately inhibited by Al+2, Cu+2, Fe+2, and Mn+2 at 10 mM and strongly inhibited by Hg+2 at 5–10 mM concentrations. No stabilizing effect was observed in the presence of Mg+2 and Zn+2, but > 100% activity was achieved with some ions such as Fe+2, Ca+2, Mn+2, and Al+3 at 1 mM. Note that after incubation with reducing agents and detergents, the enzyme not only exhibited high stability but also presented > 50% increase in the activity against 2-ME and AOT (Table S5). In this study, it was found that tween 80%, Tris–HCl, and urea stimulated the lipase activity.
Enzymatic improvement of n6/n3 FFAs ratio in fenugreek oil
The esterification reaction was carried out in a substrate molar ratio [1 g glycerol (about 10 mmol): 0.25 g PUFA (about 1 mmol)] = 10:1 and 200 mg of the immobilized lipase with an enzyme/substrate ratio of 2.2%. In general, an increase on the glycerol content will affect the theoretical equilibration values that raise the yield of AGs accordingly. Furthermore, glycerol can act as an effective stabilizer against thermal and solvent deactivation. Nevertheless, the increase of glycerol content will also influence the system polarity, which subsequently affects the system stability and homogeneity. In addition, especially for the enzymes with hydrophilic carriers, glycerol may form a coating surrounding the enzyme, which subsequently leads to enzyme clumping and inactivation (Zhong et al. 2009). In the present study, SBA-15@oleic acid as a hydrophobic support was applied for the enzyme immobilization that can be less affected by high glycerol concentration. This suggests that while glycerol concentration has a significant effect, the type of carrier used to enzyme immobilization may be more important for the glyceride synthesis than glycerol concentration. In accordance to the present study, glyceride synthesis increased with increasing glycerol content and substrate molar ratio of 10:1 and 25:1 were optimal for Candida rugosa lipase and porcine panaceas lipase (Yesiloglu and Kilic 2004).
The obtained results of titration using 0.025 mol/L NaOH that indicated 40% (w/w) of PUFAs were remained as FFAs, so the yield of enzymatic esterification was 60% (w/w). The GC results in Fig. 3a and Table 3 indicate that linoleic acid (C18:2; n-6), α-linolenic acid (C18:3; n-3), oleic acid (C18:1; n-9), palmitic acid (C16:0), and stearic acid (C18:0) were the most fatty acid composition of fenugreek oil. While the n-3 PUFA concentrates obtained by urea complexation method (Fig. 3b) showed a total reduction in saturated (C16:0 and C18:0) and mono-unsaturated (18:1) FFAs contents and remarkable increase in n-3 PUFA (C18:2; n-6 and C18:3; n-3). The percentage of α-linolenic acid (n-3) was enhanced from 30% (76.3 mg) in 250 mg of PUFA concentrate to 45% (67.6 mg) in 150 mg of the final product, while linoleic acid (n-6) decreased from 68% (169 mg) to 52% (78.2 mg) by lipase esterification. These results show that the solvent-tolerant lipase has a high efficiency to n-3 compared to n-6 PUFAs and a reduction in n-6/n-3 ratio is observed. The value of n-6/n-3 ratio as 1.1 in the produced AGs is remarkably distinct from other plant seeds showing a high value of n-6/n-3 ratio (Orsavova et al. 2015).
Discussion
Polyunsaturated fatty acids/ saturated fatty acids (PUFA/SFA) index in vegetable oils is considered as an important parameter for determination of oil nutrition value. The ratio of PUFA/SFA in seed oils with high nutritional value is higher than 1 (Ahmad et al. 2018).
The n-3 and n-6 FFAs are essential PUFAs that cannot be synthesized in the human body and must be obtained through the diet. However, nutritional imbalance in the consumption of n-6/n-3 FFAs ratio can hurt human health and a ratio of 1−5 is recommended (Orsavova et al. 2015). PUFAs are easily dissociated in chemical synthesis, yielding unwanted oxidation polymers and products. Hence, lipase-catalyzed esterification as a feasible alternative could be used for PUFA concentration from vegetable oils for reducing the n-6/n-3 FFA ratio as a valuable alternative in diet. The analysis of FFAs composition of vegetable oils shows that linoleic acid (n-6) within the range of 1.6–79.0% is the most common PUFA in plant oils. On the other hand, ALA (n-3) was detected within the range of 0.1–1.6%, except for flaxseed oil (20–60%) (Orsavova et al. 2015) and fenugreek seed oil (26–40%).
In this study a solvent-tolerant lipase producing strain was isolated from the oil-contaminated regions during primary and secondary screening and identified as Streptomyces pratensis. Under the optimized conditions, lipase production tripled. Ebrahimpour et al. (2008) reported a 4.7-enhancement in lipase production in a similar study. Considering the fact that interfacial activation mechanism is an oriented adsorption of lipases on hydrophobic surfaces and a useful reversible method for simultaneous immobilization and purification of lipases (Bastida et al. 1998), SBA-15@oleic acid nanoparticles were applied as an appropriate functionalized support. The results are comparable with those obtained from few studies on lipase purification by affinity column strategies (Solanki and Gupta 2011; Volpato et al. 2010). The pH stability of the purified enzyme was approximately similar to that of lipases, such as Streptomyces fradiae var. k11 (Zhang et al. 2008) and Streptomyces coelicolor A3 (Côté and Shareck 2008) with at a broad pH range of 4.0–10.0 and 6.0–9.0, respectively.
Lipase-catalyzed reactions are heavily influenced by organic solvent hydrophobicity in the reaction media. Log P as the logarithm of the octanol/water partition coefficient is used as an operative index of the solvent polarity (Lyman et al. 1990). The enzyme stability at 10% polar solvent concentrations have also been reported in few cases such as Serratia marcescens (Zhao et al. 2008), Pseudomonas aeruginosa, Bacillus megaterium, and P. mendocina (Gaur et al. 2008). Water-miscible hydrophilic solvents with log P < 2 such as methanol, acetonitrile, and ethyl acetate are usually incompatible with the activity of a protein; whereas, water-immiscible lipophilic solvents, such as hexane, cyclohexane, isooctane, and n-hexadecan retain the enzyme’s high catalytic activity due to this fact that they do not remove the crucial bound water from the enzyme’s surface leading to its unfolding and reducing stability of the biocatalyst. Also, the activation of lipase by hydrophobic solvents could explain that the molecules of organic solvents interact with the hydrophobic amino acid residues present in the lid that cover the catalytic site of the enzyme, thereby maintaining the enzyme in its open conformation and conducive to catalysis. The other reason for higher stability of the enzyme in the presence of hydrophobic solvents may be resulted from the surface-solvent interaction leading to interfacial activation. However, the solvent-tolerant enzyme showed the activity > 100% in the presence of 25% (v/v) tert-butyl alcohol as a water-miscible hydrophilic solvent. A possible explanation for the lipase stability in the presence of metal inhibitors is that the metals bind to the active site of lipase and change the conformation of the protein for its more proper function (Kumar et al.2016). This phenomenon can be similar to mechanism of the enzyme stability in water-immiscible lipophilic solvents. The enzyme stability in the presence of 2-ME indicated the absence of disulfide linkages in the purified lipase in contrast to other studies on an isolated lipase. Lipases-catalyzed reactions occur at the lipid-water interfacial area and the positive effect of tween 80% as a surfactant could be due to the surfactant’s role in decreasing the surface tension of the liquid and preventing the aggregation of the enzyme and hence increase the enzyme stability (Samaei-Nouroozi et al. 2015). Non-ionic and anionic surfactants improve the stability of surface-active enzymes (Gupta et al. 2004). Urea is a well-known protein-denaturing agent that can affect the enzyme structure by direct interaction with the macromolecule or by an indirect action through effects on the structure and property of the surrounding solvent. However, the enzyme exhibited 109% residual activity in the presence of 10 mM urea. The possible explanation of the urea tolerance of lipase could be due to a conformational change caused by this inhibitor. In accordance to the present study, Pseudomonas aeruginosa mutant, Bacillus licheniformis, and Burkholderia cepacia lipases were reported to be stable with a significant residual activity in the presence of urea (Sonkar and Singh 2020; Chakraborty and Paulraj 2008; Yu et al. 2009). Tris–HCl also forms the complexes with metal ions in solution and can inhibit the enzyme activity in metalloenzymes via chelation. The maintenance > 100% of the activity after incubation with EDTA and Tris–HCl suggests that lipase is not a metalloenzymes (Bose and Keharia 2013). On the other hand, Tris–HCl can protect the enzyme from a variety of denaturing stresses with decreasing the mobility of water molecules and inhibiting autoxidation of the biocatalyst (Ugwu and Apte 2004).
Characterizations of the FA composition of more vegetable oils show that the most abundant PUFA in vegetable oils is linoleic acid (n-6). By contrast, ALA (18:3, n-3) is found to range from 0.1% to 1.6%, so a ratio of n-6/n-3 fatty acids exist too high in most vegetable oils, while a ratio of n-6/n-3 fatty acids of less than 5 is recommended by nutritionists in a healthy diet. This creates a need for functional foods with high content of n-3 PUFAs, especially for vegetarians whose blood contains a high n-6/n-3 ratio. The value of 1.1 for the n-6/n-3 ratio of the produced AGs with S. pratensis MV1 lipase was markedly distinct from seed oils, which normally show a high n-6/n-3 ratio (Orsavova et al. 2015).
Conclusion
The AGs high in n-3 were successfully one-step synthesized via the immobilized lipase with a decline in n-6/n-3 ratio in fenugreek seed oil. The results show that the solvent-tolerant lipase has a high efficiency to n-3 compared to n-6 PUFAs, and it could be considered as a promising candidate in the production of PUFA concentration by a direct esterification procedure. In addition, the purified lipase with stability in organic solvents can be used for the production of fatty acid esters of sugars or sugar alcohols that exhibit high emulsifying, stabilizing, and detergency effects, thus finding applications in the food, cosmetic, detergent, and pharmaceutical industries. Also, it can be applied in inter-esterification and trans-esterification reactions in enantioselective synthesis of drug substances that are a key process in modern chemistry and are particularly important in the field of pharmaceuticals (Kumar et al. 2016).
References
Ahmad BS, Talou T, Saad Z, Hijazi A, Cerny M, Kanaan H, Chokr A, Merah O (2018) Fennel oil and by-products seed characterization and their potential applications. Ind Crops Prod 111:92–98. https://doi.org/10.1016/j.indcrop.2017.10.008
Akoh CC, Sista RV (1995) Enzymatic modification of borage oil: Incorporation of eicosapentaenoic acid. J Food Lipids 4:231–238. https://doi.org/10.1111/j.1745-4522.1995.tb00045.x
Barba FJ, Galanakis CM, Esteve MJ, Frigola A, Vorobiev E (2015) Potential use of pulsed electric technologies and ultrasounds to improve the recovery of high-added value compounds from blackberries. J Food Eng 167:38–44. https://doi.org/10.1016/j.jfoodeng.2015.02.001
Bastida A, Sabuquillo P, Armisen P, Fernández Lafuente R, Huguet J, Guisán JM (1998) A single step purification, immobilization, and hyperactivation of lipases via interfacial adsorption on strongly hydrophobic supports. Biotechnol Bioeng 5:486–493. https://doi.org/10.1002/(SICI)1097-0290(19980605)58:5<486:AID-BIT4>3.0.CO;2-9
Bose A, Keharia H (2013) Production, characterization and applications of organic solvent tolerant lipase by Pseudomonas aeruginosa AAU2. Biocatal Agric Biotechnol 3:255–266. https://doi.org/10.1016/j.bcab.2013.03.009
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 2:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Chakraborty K, Paulraj R (2008) An extracellular alkaline metallolipase from Bacillus lichenformis MTCC 6824: Purification and biochemical characterization. Food Chem 109:727–736. https://doi.org/10.1016/j.foodchem.2008.01.026
Chakraborty K, Raj RP (2009) (2009) Selective enrichment of n−3 polyunsaturated fatty acids with C18–C20 acyl chain length from sardine oil using Pseudomonas fluorescens MTCC 2421 lipase. Food Chem 1:142–150. https://doi.org/10.1016/j.foodchem.2008.09.029
Côté A, Shareck F (2008) Cloning, purification and characterization of two lipases from Streptomyces coelicolor A3 (2). Enzyme Microb Technol 5:381–388. https://doi.org/10.1016/j.enzmictec.2008.01.009
Deng Q, Zinoviadou KG, Galanakis CM, Orlien V, Grimi N, Vorobiev E, Lebovka N, Barba FJ (2015) The effects of conventional and non-conventional processing on glucosinolates and its derived forms, isothiocyanates: extraction, degradation, and applications. Food Eng Rev 7:357–381. https://doi.org/10.1007/s12393-015-9122-2
Ebrahimpour A, Rahman RNZRA, Ch'ng DHE, Basri M, Salleh AB (2008) A modeling study by response surface methodology and artificial neural network on culture parameters optimization for thermostable lipase production from a newly isolated thermophilic Geobacillus sp. strain ARM. BMC Biotechnol 1:96. https://doi.org/10.1186/1472-6750-8-96
El Nasri NA, El Tinay A (2007) Functional properties of fenugreek (Trigonella foenum graecum) protein concentrate. Food Chem 2:582–589. https://doi.org/10.1016/j.foodchem.2006.09.003
El-Sebaiy LA, El-Mahdy AR (1983) Lipid changes during germination of fenugreek seeds (Trigonella foenum-graecum). Food Chem 4:309–319. https://doi.org/10.1016/0308-8146(83)90086-9
Galanakis CM (2012) Recovery of high added-value components from food wastes: conventional, emerging technologies and commercialized applications. Food Sci Technol 26:68–87. https://doi.org/10.1016/j.tifs.2012.03.003
Galanakis CM (2013) Emerging technologies for the production of nutraceuticals from agricultural by-products: a viewpoint of opportunities and challenges. Food Bioprod Process 91:575–579. https://doi.org/10.1016/j.fbp.2013.01.004
Galanakis CM (2018) Phenols recovered from olive mill wastewater as additives in meat products. Trends Food Sci Technol 79:98–105. https://doi.org/10.1016/j.tifs.2018.07.010
Galanakis CM (2020) The Food Systems in the Era of the Coronavirus (COVID-19) Pandemic Crisis. Foods 9:523. https://doi.org/10.3390/foods9040523
Galanakis CM, Tsatalas P, Galanakis IM (2018) Implementation of phenols recovered from olive mill wastewater as UV booster in cosmetics. Ind Crops Prod 111:30–37. https://doi.org/10.1016/j.indcrop.2017.09.058
Gandhi NN (1997) Applications of lipase. J Am Oil Chem Soc 6:621–634. https://doi.org/10.1007/s11746-997-0194-x
Gaur R, Gupta A, Khare S (2008) Purification and characterization of lipase from solvent tolerant Pseudomonas aeruginosa PseA. Process Biochem 10:1040–1046. https://doi.org/10.1016/j.procbio.2008.05.007
Granato D, Barba FJ, Bursać Kovačević D, Lorenzo JM, Cruz AG, Putnik P (2020) Functional foods: Product development, technological trends, efficacy testing, and safety. Annu Rev Food Sci Technol 11:93–118. https://doi.org/10.1146/annurev-food-032519-051708
Guil-Guerrero JL, Lopez-Martinez JC, Rincon-Cervera MA, Campra-Madrid P (2007) One-step extraction and concentration of polyunsaturated fatty acids from fish liver. J Am Oil Chem Soc 84:357–361. https://doi.org/10.1007/s11746-007-1041-9
Gupta R, Gupta N, Rathi P (2004) Bacterial lipases: an overview of production, purification and biochemical properties. Appl Microbiol Biotechnol 6:763–781
Hasan Beikdashti M, Forootanfar H, Safiarian M, Ameri A, Ghahremani M, Khoshayand M, Faramarzi MA (2012) Optimization of culture conditions for production of lipase by a newly isolated bacterium Stenotrophomonas maltophilia. J Taiwan Inst Chem Eng 5:670–677. https://doi.org/10.1016/j.jtice.2012.03.005
Imanparast S, Faramarzi MA (2018) Fabrication of the silica support of SBA-15@oleic acid for one-step purification of lipase. Iranian Pat. Off No. 97451
Imanparast S, Hamedi J, Faramarzi MA (2018) Enzymatic esterification of acylglycerols rich in omega-3 from flaxseed oil by an immobilized solvent-tolerant lipase from Actinomadura sediminis UTMC 2870 isolated from oil-contaminated soil. Food Chem 245:934–942. https://doi.org/10.1016/j.foodchem.2017.11.080
Kovačević DB, Barba FJ, Granato D, Galanakis CM, Herceg Z, Dragović-Uzelac V, Putnik P (2018) Pressurized hot water extraction (PHWE) for the green recovery of bioactive compounds and steviol glycosides from Stevia rebaudiana Bertoni leaves. Food Chem 254:150–157. https://doi.org/10.1016/j.foodchem.2018.01.192
Kumar A, Dhar K, Kanwar SS, Arora PK (2016) Lipase catalysis in organic solvents: advantages and applications. Biol Proced Online 18:2. https://doi.org/10.1186/s12575-016-0033-2.
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 5259:680. https://doi.org/10.1038/227680a0
López Martínez JC, Campra Madrid P, Rincón Cervera MÁ, Guil Guerrero JL (2005) Ecological and simultaneous seed oil extraction/saponification/γ-linolenic acid concentration. Eur J Lipid Sci Technol 3:180–186. https://doi.org/10.1002/ejlt.200401099
Lyman WJ, Reehl WF, Rosenblatt DH (1990) Handbook of chemical property estimation methods: Environmental behavior of organic compounds. J. Am. Chem. Soc. ISBN 0841217610, 9780841217614.
Metcalfe L, Schmitz A, Pelka J (1966) Rapid preparation of fatty acid esters from lipids for gas chromatographic analysis. Anal Chem 3:514–515. https://doi.org/10.1021/ac60235a044
Nicholson T, Khademi H, Moghadasian MH (2013) The role of marine n-3 fatty acids in improving cardiovascular health: a review. Food Funct 4:357–365. https://doi.org/10.1039/c2fo30235g
Ogino H, Ishikawa H (2001) Enzymes which are stable in the presence of organic solvents. J Biosci Bioeng 2:109–116. https://doi.org/10.1263/jbb.91.109
Orsavova J, Misurcova L, Ambrozova J, Vicha R, Mlcek J (2015) Fatty acids composition of vegetable oils and its contribution to dietary energy intake and dependence of cardiovascular mortality on dietary intake of fatty acids. Int J Mol Sci 6:12871–12890. https://doi.org/10.3390/ijms160612871
Samaei-Nouroozi A, Rezaei S, Khoshnevis N, Doosti M, Hajihoseini R, Khoshayand MR, Faramarzi MA (2015) Medium-based optimization of an organic solvent-tolerant extracellular lipase from the isolated halophilic Alkalibacillus salilacus. Extremophiles 19:933–947. https://doi.org/10.1007/s00792-015-0769-7
Sarfarazi M, Jafari SM, Rajabzadeh G, Galanakis CM (2020) Evaluation of microwave-assisted extraction technology for separation of bioactive components of saffron (Crocus sativus L.). Ind Crops Prod 145:111978. https://doi.org/10.1016/j.indcrop.2019.111978
Senanayake SN, Shahidi F (2004) Incorporation of docosahexaenoic acid (DHA) into evening primrose (Oenothera biennis L.) oil via lipase-catalyzed transesterification. Food Chem 4:489–496. https://doi.org/10.1016/S0308-8146(02)00412-0
Singh R, Gupta N, Goswami VK, Gupta R (2006) A simple activity staining protocol for lipases and esterases. Appl Microbiol Biotechnol 6:679–682. https://doi.org/10.1007/s00253-005-0138-z
Solanki K, Gupta M (2011) Simultaneous purification and immobilization of Candida rugosa lipase on superparamagnetic Fe3O4 nanoparticles for catalyzing transesterification reactions. New J Chem 35:2551–2556. https://doi.org/10.1039/C1NJ20277D
Sonkar K, Singh DP (2020) Biochemical characterization and thermodynamic study of lipase from psychrotolerant Pseudomonas punonensis. Biocatal Agric Biotechnol 12:101686. https://doi.org/10.1016/j.bcab.2020.101686
Tarasi R, Alipour M, Gorgannezhad L, Imanparast S, Yousefi-Ahmadipour A, Ramezani A, Ganjali MR, Shafiee A, Faramarzi MA, Khoobi M (2018) Laccase immobilization onto magnetic β-cyclodextrin-modified chitosan: improved enzyme stability and efficient performance for phenolic compounds elimination. Macromol Res 26:755–762. https://doi.org/10.1007/s13233-018-6095-z
Ugwu SO, Apte SP (2004) The effect of buffers on protein conformational stability. Pharm Technol 28:86–109
Umesha SS (2014) Studies on bioactive compounds and nutritional evaluation of n-3 fatty acid rich garden cress (Lepidium sativum L.) seed oil. UOM https://hdl.handle.net/10603/69883.
Volpato G, Filice M, Ayub MA, Guisan JM, Palomo JM (2010) Single-step purification of different lipases from Staphylococcus warneri. J Chromatogr A 4:473–478. https://doi.org/10.1016/j.chroma.2009.11.055
Wanasundara UN, Shahidi F (1999) Concentration of omega 3-polyunsaturated fatty acids of seal blubber oil by urea complexation: optimization of reaction conditions. Food Chem 1:41–49. https://doi.org/10.1016/S0963-9969(03)00052-8
Yesiloglu Y, Kilic I (2004) Lipase-catalyzed esterification of glycerol and oleic acid. J Am Oil Chem Soc 81:281–284. https://doi.org/10.1007/s11746-004-0896-5
Yu L, Xu Y, Yu X (2009) Purification and properties of a highly enantioselective L-menthyl acetate hydrolase from Burkholderia cepacia. J Mol Catal B Enzy 57:27–33
Zahir F, Rizwi SJ, Haq SK, Khan RH (2005) Low dose mercury toxicity and human health. Environ Toxicol Pharmacol 2:351–360. https://doi.org/10.1016/j.etap.2005.03.007
Zhang Y, Meng K, Wang Y, Luo H, Yang P, Shi P, Wu N, Fan Y, Li J, Yao B (2008) A novel proteolysis-resistant lipase from keratinolytic Streptomyces fradiae var. k11. Enzyme Microb Technol 4:346–352. https://doi.org/10.1016/j.enzmictec.2007.10.015
Zhao LL, Xu JH, Zhao J, Pan J, Wang ZL (2008) Biochemical properties and potential applications of an organic solvent-tolerant lipase isolated from Serratia marcescens ECU1010. Process Biochem 6:626–633. https://doi.org/10.1016/j.procbio.2008.01.023
Zhong N, Li L, Xu X, Cheong L, Li B, Hu S, Zhao X (2009) An efficient binary solvent mixture for monoacylglycerol synthesis by enzymatic glycerolysis. J Am Oil Chem Soc 86:783–789. https://doi.org/10.1007/s11746-009-1402-7
Zinoviadou KG, Galanakis CM, Brnčić M, Grimi N, Boussetta N, Mota MJ, Saraiva JA, Patras A, Tiwari B, Barba FJ (2015) Fruit juice sonication: implications on food safety and physicochemical and nutritional properties. Food Res 77:743–752. https://doi.org/10.1016/j.foodres.2015.05.032
Acknowledgements
Research reported in this publication was supported by Elite Researcher Grant Committee under award number 976958 from the National Institutes for Medical Research Development (NIMAD), Tehran, Iran to M.A.F.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Vahidi, M., Imanparast, S., Jahandar, H. et al. An organic solvent-tolerant lipase of Streptomyces pratensis MV1 with the potential application for enzymatic improvement of n6/n3 ratio in polyunsaturated fatty acids from fenugreek seed oil. J Food Sci Technol 58, 2761–2772 (2021). https://doi.org/10.1007/s13197-020-04784-w
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-020-04784-w