Abstract
Pectinases and other carbohydrate-active enzymes are important for the food industry, mainly for juice processing. In addition, the use of peels to produce enzymes can aggregate value to these agro-industrial residues and at the end of the process enhance qualitatively and quantitatively the juice production. In this work, three different extracts produced by Penicillium oxalicum LS09 using agro-industrial residues were optimized and analyzed by mass spectrometry. It was observed an increased production of pectinases in the medium containing orange peel and optimized for production of pectin lyase and pectinesterase (PE). Interestingly, not only pectinases, but also different plant cell wall degrading enzymes (i.e. glucanases, xylanases, arabinases), with a higher ratio (42/73) was identified in the medium optimized for PE. The crude extracts produced by P. oxalicum also reveal the potential for application in the fruit juice industry, showing an increased yield and qualitative characteristics of extracted juices. The presence of other cell wall-degrading enzymes identified by proteomics, reinforce the combination for obtaining clarified and depectinized juice in a single step.

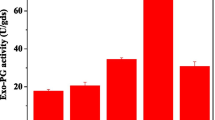
source concentration a, pH b and temperature c upon different pectinolytic extracts produced by Penicillium oxalicum. Fungus was growing in basal medium with 0.5% (White square), 1% (Black circle), 2% (White circle) and 3% (Black square) of respective fruit peels. Data represents mean of three replicates ± SE. Values of p < 0.05 (*) and p < 0.01 (**) were considered to be statistically significant


Similar content being viewed by others
References
Alaña A, Gabilondo A, Hernando F, Moragues MD, Dominguez JB, Llama MJ, Serra JL (1989) Pectin lyase production by a Penicillium italicum strain. Appl Env Microbiol 55:1612–1616
Albersheim P, Killias U (1962) Studies relating to the purification and properties of pectin transeliminase. Arch Biochem Biophys 97:107–115. https://doi.org/10.1016/0003-9861(62)90050-4
Barnby FM, Morpeth FF, Pyle DL (1990) Endopolygalacturonase production from Kluyveromyces marxianus. I. Resolution, purification, and partial characterization of the enzyme. Enzyme Microbial Technol 12:891–897
Byaruagaba-Bazirake GW, Ransburg PV, Kyamuhangire W (2012) Characteristics of enzyme-treated banana juice from three cultivars of tropical and subtropical Africa. African J Food Science Technol 3:277–290
Cantarel BL, Coutinho PM, Rancurel C, Bernard T, Lombard V, Henrissat B (2009) The Carbohydrate-Active enZymes database (CAZy): an expert resource for glycogenomics. Nucleic Acids Res 37:D233–D238. https://doi.org/10.1093/nar/gkn663
Cheng Z, Chen D, Wang Q, Xian L, Lu B, Wei Y, Tang H, Lu Z, Zhu Q, Chen Y, Huang R (2017) Identification of an acidic endo-polygalacturonase from Penicillium oxalicum CZ1028 and its broad use in major tropical and subtropical fruit juices production. J Biosci Bioeng 123:665–672. https://doi.org/10.1016/j.jbiosc.2017.01.013
Dey TB, Banerjee R (2014) Application of decolourized and partially purified polygalacturonase and α-amylase in apple juice clarification. Braz J Microbiol 45:97–104. https://doi.org/10.1590/s1517-83822014000100014
Díaz AB, Alvarado O, Ory I, Caro I, Blandino A (2013) Valorization of grapes pomace and orange peels: Improved production of hydrolytic enzymes for the clarification of orange juice. Food Bioprod Proc 91:580–583. https://doi.org/10.1016/j.fbp.2013.01.007
Diaz-Godinez G, Soriano-Santos J, Augur C, Viniegra-Gonzalez G (2001) Exopectinases produced by Aspergillus niger in solid-state and submerged fermentation: a comparative study. J Ind Microbiol Biotechnol 26:271–275. https://doi.org/10.1038/sj.jim.7000113
Gálvez-López D, Laurens F, Quéméner B, Lahaye M (2011) Variability of cell wall polysaccharides composition and hemicellulose enzymatic profile in an apple progeny. Int J Biol Macromol 49:1104–1109. https://doi.org/10.1016/j.ijbiomac.2011.09.007
Gao Y, Fangel JU, Willats WGT, Vivier MA, Moore JP (2016) Dissecting the polysaccharide-rich grape cell wall matrix using recombinant pectinases during winemaking. Carbohydr Polym 152:510–519. https://doi.org/10.1016/j.carbpol.2016.05.115
Garg G, Singh A, Kaur A, Singh R, Kaur J, Mahajan R (2016) Microbial pectinases: an ecofriendly tool of nature for industries. 3 Biotechnol 6:47. https://doi.org/10.1007/s13205-016-0371-4
Hamdy HS (2005) Purification and characterization of the pectin lyase produced by Rhizopus oryzae grown on orange peels. Ann Microbiol 55:205–211
Hoondal GS, Tiwari RP, Tewari R, Dahiya N, Beg QK (2002) Microbial alkaline pectinases and their industrial applications: a review. Appl Microbiol Biotechnol 59:409–418. https://doi.org/10.1007/s00253-002-1061-1
Houston K, Tucker MR, Chowdhury J, Shirley N, Little A (2016) The plant cell wall: a complex and dynamic structure as revealed by the responses of genes under stress conditions. Front Plant Sci 7:984. https://doi.org/10.3389/fpls.2016.00984
Ismail AS (1996) Utilization of orange peels for the production of multi-enzyme complexes by some fungal strains. Proc Biochem 1:645–650. https://doi.org/10.1016/S0032-9592(96)00012-X
Kashyap DR, Vohra PK, Chopra S, Tewari R (2001) Applications of pectinases in the commercial sector: a review. Biores Technol 77:215–227. https://doi.org/10.1016/S0960-8524(00)00118-8
Kohli P, Gupta R (2015) Alkaline pectinases: a review. Biocat Agric Biotechnol 4:279–285
Mahawar M, Singh A, Jalgaonkar K (2012) Utility of apple pomace as a substrate for various products: a review. Food Bioprod Proc 90:597–605. https://doi.org/10.1080/07388550802368895
Mamma D, Kourtoglou E, Christakopoulos P (2008) Fungal multienzyme production on industrial by-products of the citrus-processing industry. Biores Technol 99:2373–2383. https://doi.org/10.1016/j.biortech.2007.05.018
Mantovani CF, Geimba MP, Brandelli A (2014) Enzymatic clarification of fruit juices by fungal pectin lyase. Food Biotechnol 19:173–181. https://doi.org/10.1080/08905430500316284
Martínez-Trujillo A, Arreguín-Rangel L, García-Rivero M, Aguilar-Osorio G (2011) Use of fruit residues for pectinase production by Aspergillus flavipes FP-500 and Aspergillus terreus FP-370. Lett Appl Microbiol 53:202–209. https://doi.org/10.1111/j.1472-765X.2011.03096.x
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426
Ortega LM, Kikot GE, Rojas NL, López LM, Astoreca AL, Alconada TM (2014) Production, characterization, and identification using proteomic tools of a polygalacturonase from Fusarium graminearum. J Basic Microbiol 54:S170–S177. https://doi.org/10.1002/jobm.201300651
Polizeli MLTM, Damásio ARL, Maller A, Cabral H, Polizeli AM, Rai M (2014) Pectinases produced by microorganisms: properties and applications. In: Polizeli MLRM, Rai M (eds) Fungal enzymes. Taylor & Francis, Abingdon, pp 316–340
Prabasari I, Pettolino F, Liao ML, Bacic A (2011) Pectic polysaccharides from mature orange (Citrus sinensis) fruit albedo cell walls: sequential extraction and chemical characterization. Carbohydr Polym 84:484–494. https://doi.org/10.1016/j.carbpol.2010.12.012
Reyes F, Martinez MJ, Lahoz R, Archer SA (1984) Characterization as glycerol of an inhibitor of pectin lyases from autolysing cultures of Botrytis cinerea. Trans Br Mycol Soc 82(4):689–696. https://doi.org/10.1016/s0007-1536(84)80109-6
Ridley BL, O’Neil MA, Mohnen D (2001) Pectins: structure, biosynthesis, and oligogalacturonide-related signaling. Phytochem 57:929–967
Rui Y, Xiao C, Yi H, Kandemir B, Wang JZ, Puri VM, Anderson CT (2017) Polygalacturonase involved in Expansion3. Functions in seedling development, rosette growth, and stomatal dynamics in Arabidopsis thaliana. Plant Cell 29:2413–2432. https://doi.org/10.1105/tpc.17.00568
Rytioja J, Hildén K, Yuzon J, Hatakka A, de Vries RP, Mäkelä MR (2014) Plant-polysaccharide-degrading enzymes from Basidiomycetes. Microbiol Molec Biol Rev 78:618–649. https://doi.org/10.1128/MMBR.00035-14
Sandri IG, Fontana RC, Barfknecht M, Moura da Silveira M (2001) Clarification of fruit juices by fungal pectinases. LWT Food Sci Technol 44:2217–2222. https://doi.org/10.1016/j.lwt.2011.02.008
Santi L, Beys-da-Silva WO, Berger M, Calzolari D, Guimarães JA, Moresco JJ, Yates JR (2014) Proteomic profile of Cryptococcus neoformans biofilm reveals changes in metabolic processes. J Proteome Res 13:1545–1559. https://doi.org/10.1021/pr401075f
Sharma HP, Patel H, Sugandha S (2017) Enzymatic added extraction and clarification of fruit juices - A review. Crit Rev Food Sci Nutr 57:1215–1227. https://doi.org/10.1080/10408398.2014.977434
Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Provenzano MD et al (1985) Measurement of the principle of protein-dye binding. Anal Biochem 150:76–85
Teixeira JA, Corrêa TL, de Queiroz MV, de Araújo EF (2014) pH-dependent effect of pectinase secretion in Penicillium griseoroseum recombinant strains. J Basic Microbiol 54:133–141. https://doi.org/10.1002/jobm.201200325
Toushik SH, Lee KT, Lee JS, Kim KS (2017) Functional applications of lignocellulolytic enzymes in the fruit and vegetable processing industries. J Food Sci 82:585593. https://doi.org/10.1111/1750-3841.13636
Tu T, Bai YG, Luo HY, Ma R, Wang YR, Shi PJ, Yang P, Meng K, Yao B (2014) A novel bifunctional pectinase from Penicillium oxalicum SX6 with separate pectin methylesterase and polygalacturonase catalytic domains. Appl Microbiol Biotechnol 98:5019–5028. https://doi.org/10.1007/s00253-014-5533-x
Wang D, Yeats TH, Uluisik S, Rose JKC, Seymour GB (2018) Fruit softening: revisiting the role of pectin. Trends Plant Sci 23:302–310. https://doi.org/10.1016/j.tplants.2018.01.006
Wang J, Zhang Y, Qin X, Gao L, Han B, Zhang D, Li J, Huang H, Zhang W (2017) Efficient expression of an acidic endo-polygalacturonase from Aspergillus niger and its application in juice production. J Agric Food Chem 65:2730–2736. https://doi.org/10.1021/acs.jafc.6b05109
Williams BA, Grant LJ, Gidley MJ, Mikkelsen D (2017) Gut fermentation of dietary fibres: physico-chemistry of plant cell walls and implications for health. Int J Molec Sci 18:2203. https://doi.org/10.3390/ijms18102203
Acknowledgments
The authors would like to thank Dr. Maria Inez Sarkis from FIOCRUZ for microorganism identification. This work was supported by grant and fellowship from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
Author information
Authors and Affiliations
Contributions
All authors approved the final article.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Santi, L., Beys-da-Silva, W.O., Berger, M. et al. Penicillium oxalicum secretomic analysis identify plant cell wall degrading enzymes important for fruit juice extraction. J Food Sci Technol 58, 1764–1775 (2021). https://doi.org/10.1007/s13197-020-04688-9
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-020-04688-9