Abstract
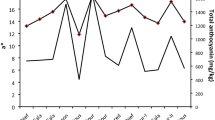
Phenolic compounds of the first and second racking wine lees, including anthocyanins, were qualitatively and quantitatively analyzed by HPLC–DAD–MS. Wine lees from both rackings displayed similar chromatographic profiles. Therefore, it was impossible to differentiate the qualitative results regarding phenolic compounds. On the other hand, those from the second racking presented, on average, concentration of polyphenols twice as high. While the ones from the first racking displayed ca. 1600 mg phenolic compounds and 400 mg anthocyanins per kg of dry matter, those from the second racking have shown ca. 3300 mg phenolic compounds and 700 mg anthocyanins per kg of dry matter. These outcomes indicate that, although the wine lees from the first racking can be employed as a resource for phenolic compounds recovery, those from the second racking are more appropriate for this purpose.




Similar content being viewed by others
References
Acevedo De la Cruz A et al (2012) Anthocyanin identification and composition of wild Vitis spp. accessions by using LC–MS and LC–NMR. Anal Chim Acta 732:145–152. https://doi.org/10.1016/j.aca.2011.11.060
Alonso ÁM, Guillén DA, Barroso CG, Puertas B, García A (2002) Determination of antioxidant activity of wine byproducts and its correlation with polyphenolic content. J Agric Food Chem 50:5832–5836. https://doi.org/10.1021/jf025683b
Barcia MT, Pertuzatti PB, Gómez-Alonso S, Godoy HT, Hermosín-Gutiérrez I (2014) Phenolic composition of grape and winemaking by-products of Brazilian hybrid cultivars BRS Violeta and BRS Lorena. Food Chem 159:95–105. https://doi.org/10.1016/j.foodchem.2014.02.163
Beres C et al (2017) Towards integral utilization of grape pomace from winemaking process: a review. Waste Manag 68:581–594. https://doi.org/10.1016/j.wasman.2017.07.017
Bimpilas A, Tsimogiannis D, Balta-Brouma K, Lymperopoulou T, Oreopoulou V (2015) Evolution of phenolic compounds and metal content of wine during alcoholic fermentation and storage. Food Chem 178:164–171. https://doi.org/10.1016/j.foodchem.2015.01.090
Castillo-Muñoz N, Gómez-Alonso S, García-Romero E, Hermosín-Gutiérrez I (2007) Flavonol profiles of Vitis vinifera red grapes and their single-cultivar wines. J Agric Food Chem 55:992–1002. https://doi.org/10.1021/jf062800k
Delgado de la Torre MP, Priego-Capote F, Luque de Castro MD (2015) Tentative identification of polar and mid-polar compounds in extracts from wine lees by liquid chromatography–tandem mass spectrometry in high-resolution mode. J Mass Spectrom 50:826–837. https://doi.org/10.1002/jms.3592
Devesa-Rey R, Vecino X, Varela-Alende JL, Barral MT, Cruz JM, Moldes AB (2011) Valorization of winery waste vs. the costs of not recycling. Waste Manag 31:2327–2335. https://doi.org/10.1016/j.wasman.2011.06.001
Dimou C, Vlysidis A, Kopsahelis N, Papanikolaou S, Koutinas AA, Kookos IK (2016) Techno-economic evaluation of wine lees refining for the production of value-added products. Biochem Eng J 116:157–165. https://doi.org/10.1016/j.bej.2016.09.004
Fornairon-Bonnefond C, Camarasa C, Moutounet M, Salmon J-M (2001) New trends on yeast autolysis and wine ageing on lees: a bibliographic review. Journal International des Sciences de la Vigne et du Vin 35:57–78. https://doi.org/10.20870/oeno-one.2001.35.2.990
Giacobbo A, Meneguzzi A, Bernardes AM, de Pinho MN (2017) Pressure-driven membrane processes for the recovery of antioxidant compounds from winery effluents. J Clean Prod 155:172–178. https://doi.org/10.1016/j.jclepro.2016.07.033
Ginjom I, D’Arcy B, Caffin N, Gidley M (2011) Phenolic compound profiles in selected Queensland red wines at all stages of the wine-making process. Food Chem 125:823–834. https://doi.org/10.1016/j.foodchem.2010.08.062
Iora SRF, Maciel GM, Zielinski AAF, da Silva MV, Pontes PVdA, Haminiuk CWI, Granato D (2015) Evaluation of the bioactive compounds and the antioxidant capacity of grape pomace. Int J Food Sci Technol 50:62–69. https://doi.org/10.1111/ijfs.12583
Kammerer DR, Kammerer J, Valet R, Carle R (2014) Recovery of polyphenols from the by-products of plant food processing and application as valuable food ingredients. Food Res Int 65:2–12. https://doi.org/10.1016/j.foodres.2014.06.012
Léauté R (1990) Distillation in alambic. Am J Enol Vitic 41:90–103
Lopes-Lutz D, Dettmann J, Nimalaratne C, Schieber A (2010) Characterization and quantification of polyphenols in Amazon grape (Pourouma cecropiifolia Martius). Molecules 15:8543–8552. https://doi.org/10.3390/molecules15118543
Morata A, Gómez-Cordovés MC, Suberviola J, Bartolomé B, Colomo B, Suárez JA (2003) Adsorption of anthocyanins by yeast cell walls during the fermentation of red wines. J Agric Food Chem 51:4084–4088. https://doi.org/10.1021/jf021134u
Núñez V, Monagas M, Gomez-Cordovés MC, Bartolomé B (2004) Vitis vinifera L. cv. Graciano grapes characterized by its anthocyanin profile. Postharvest Biol Technol 31:69–79. https://doi.org/10.1016/S0925-5214(03)00140-6
OIV—Organization Internationale de la Vigne et du Vin. State of the Vitiviniculture World Market—April 2018 (2018) http://www.oiv.int/public/medias/5958/oiv-state-of-the-vitiviniculture-world-market-april-2018.pdf. Accessed 26 Oct 2018
Oliveira M, Duarte E (2016) Integrated approach to winery waste: waste generation and data consolidation. Front Environ Sci Eng 10:168–176. https://doi.org/10.1007/s11783-014-0693-6
Papoušková B et al (2011) Advanced liquid chromatography/mass spectrometry profiling of anthocyanins in relation to set of red wine varieties certified in Czech Republic. J Chromatogr A 1218:7581–7591. https://doi.org/10.1016/j.chroma.2011.07.027
Pérez-Serradilla JA, Luque de Castro MD (2011) Microwave-assisted extraction of phenolic compounds from wine lees and spray-drying of the extract. Food Chem 124:1652–1659. https://doi.org/10.1016/j.foodchem.2010.07.046
Pomar F, Novo M, Masa A (2005) Varietal differences among the anthocyanin profiles of 50 red table grape cultivars studied by high performance liquid chromatography. J Chromatogr A 1094:34–41. https://doi.org/10.1016/j.chroma.2005.07.096
Ramirez-Lopez LM, DeWitt CAM (2014) Analysis of phenolic compounds in commercial dried grape pomace by high-performance liquid chromatography electrospray ionization mass spectrometry. Food Sci Nutr 2:470–477. https://doi.org/10.1002/fsn3.136
Rockenbach II, Rodrigues E, Gonzaga LV, Caliari V, Genovese MI, Gonçalves AEdSS, Fett R (2011) Phenolic compounds content and antioxidant activity in pomace from selected red grapes (Vitis vinifera L. and Vitis labrusca L.) widely produced in Brazil. Food Chem 127:174–179. https://doi.org/10.1016/j.foodchem.2010.12.137
Rodrigues E, Mariutti LRB, Mercadante AZ (2013) Carotenoids and phenolic compounds from Solanum sessiliflorum, an unexploited Amazonian fruit, and their scavenging capacities against reactive oxygen and nitrogen species. J Agric Food Chem 61:3022–3029. https://doi.org/10.1021/jf3054214
Ruggieri L et al (2009) Recovery of organic wastes in the Spanish wine industry. Technical, economic and environmental analyses of the composting process. J Clean Prod 17:830–838. https://doi.org/10.1016/j.jclepro.2008.12.005
Ruiz A et al (2013) Anthocyanin profiles in south Patagonian wild berries by HPLC–DAD–ESI–MS/MS. Food Res Int 51:706–713. https://doi.org/10.1016/j.foodres.2013.01.043
Simirgiotis MJ, Caligari PDS, Schmeda-Hirschmann G (2009) Identification of phenolic compounds from the fruits of the mountain papaya Vasconcellea pubescens A. DC. grown in Chile by liquid chromatography–UV detection–mass spectrometry. Food Chem 115:775–784. https://doi.org/10.1016/j.foodchem.2008.12.071
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic–phosphotungstic acid reagents. Am J Enol Vitic 16:144–158
Stefova M, Ivanova V (2011) Analytical methodology for characterization of grape and wine phenolic bioactives. In: Tokuşoğlu Ö, Hall C III (eds) Fruit and cereal bioactives. CRC Press, Boca Raton, pp 409–427. https://doi.org/10.1201/b10786-25
Vallverdú-Queralt A et al (2015) Identification of phenolic compounds in red wine extract samples and zebrafish embryos by HPLC–ESI–LTQ–Orbitrap–MS. Food Chem 181:146–151. https://doi.org/10.1016/j.foodchem.2015.02.098
You Q, Chen F, Sharp JL, Wang X, You Y, Zhang C (2012) High-performance liquid chromatography–mass spectrometry and evaporative light-scattering detector to compare phenolic profiles of muscadine grapes. J Chromatogr A 1240:96–103. https://doi.org/10.1016/j.chroma.2012.03.086
Acknowledgements
The authors gratefully acknowledge the Brazilian funding agencies (CAPES, CNPq, FAPERGS, and SEBRAE/RS) and the Portuguese funding agency (FCT) for their financial support, as well as the Vinícola Almaúnica (Brazil) by the financial support and for providing the wine lees.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Giacobbo, A., Dias, B.B., Onorevoli, B. et al. Wine lees from the 1st and 2nd rackings: valuable by-products. J Food Sci Technol 56, 1559–1566 (2019). https://doi.org/10.1007/s13197-019-03665-1
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-019-03665-1