Abstract
Eiseniella neapolitana is a semi-aquatic, diploid earthworm that for many years was related to the cosmopolitan species Eiseniella tetraedra and even considered a subspecies of it. Norealidys andaluciana was described in Spain and is usually synonymized with E. neapolitana. We collected 69 specimens from Italy, Spain, and Cyprus and studied five molecular markers (COI, 16S, 28S, 12S, and ND1) and their morphology to solve this taxonomic problem. Phylogenetic analyses reveal the possible existence of two separate genera confounded under the name Eiseniella, but the study of more molecular markers and species of the genus would be necessary to confirm this. Therefore, the synonymy between Eiseniella and Norealidys is maintained. Various genetic analyses, including species delimitation, confirm the separation between E. neapolitana and E. andaluciana (= N. andaluciana) and excluded that E. neapolitana is a subspecies of E. tetraedra. The resemblance in external appearance despite clear genetic differences of the three species could be explained by convergent adaptation to the aquatic habitat. Despite the expected low haplotype diversity based on the 28S gene, we found a surprisingly high variability in the E. andaluciana (= N. andaluciana) population in Spain. However, its stable predicted secondary structure and its high content of G + C reject the presence of a pseudogene.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The genus Eiseniella Michaelsen, 1900 includes two distinct groups of species, all of which belong to the riparian earthworms (Omodeo & Rota, 1991). The first group includes Eiseniella tetraedra (Savigny, 1826), a parthenogenetic and cosmopolitan earthworm with morphological variability, but in general: quadrangular cross-section, less than 95 segments, male pore usually in XIII, tumid porophores, and unique position of the female pores below the line of setae a (Omodeo & Rota, 1991). This species showed a high genetic diversity in the Iberian Peninsula (de Sosa et al., 2017). Perel (1967) and Zicsi (1972) revised the other group. It includes about ten species, and they are also characterized by a quadrangular cross-section, the number of segments mainly ranging from 95 to 160, male pores without porophores, and female pores open dorsally to setae b or behind (Omodeo & Rota, 1991). The distribution of the individual species of this second group is geographically restricted (Omodeo & Rota, 1991). Currently, there are no data on the genetic diversity of these species.
Eiseniella neapolitana (Örley, 1885) belongs to the latter group. It is a semi-aquatic, circum Mediterranean and diploid earthworm with biparental reproduction (Omodeo, 1952). It was long considered a subspecies of E. tetraedra (Michaelsen, 1900; Stephenson, 1924; Bodenheimer, 1937; Cernosvitov, 1938, 1940; Pop, 1952; Mršić, 1991; Pavlicek et al., 2003). But already, Omodeo (1952, 1956), Omodeo and Rota (1991), and more recently Csuzdi and Pavlicek (2005) considered it a valid species.
Qiu and Bouché (1998) described Reynoldsia andaluciana on the basis of a few specimens from the locality Salobreña (Granada, Spain). The new genus, Reynoldsia, was characterized by rudimentary or absent of calciferous glands (Morren’s glands) and typhlosole and by practically nonexistent nephridial vesicles (Qiu & Bouché, 1998). Blakemore (2008) did not accept Reynoldsia due to homonymy with a genus of Diptera and renamed it Norealidys. To date, N. andaluciana is considered a synonym of E. neapolitana in various databases such as DriloBASE (Drilobase Project, 2020), Earthworm species, a searchable database (http://earthworm.uw.hu; Csuzdi, 2012), or FADA database (http://www.fada.biodiversity.be; Balian et al., 2007). However, in Fauna Europaea (Rota & de Jong, 2015), they are treated as separate species.
In recent years, molecular tools are helping with taxonomic problems of earthworms (e.g., Huang et al., 2007; Marchán et al., 2018; de Sosa et al., 2019; Jiménez-Pinadero et al., 2021). Thus, a solution for the systematics of this group of species could arise from the study of molecular markers.
In their large-scale molecular phylogenetic analysis of Lumbricidae, Domínguez et al. (2015) found that the sole member of the genus in the study, E. tetraedra, appeared closely related to Iberian representatives of the genus Eiseniona Omodeo, 1956 (allocated by other authors in Iberoscolex Qiu & Bouché, 1998). The inclusion of new specimens and species of the Eiseniella and supposedly related genera in the phylogenetic analysis could help to prove or reject this hypothesis.
The aim of the present study is (1) to verify whether E. neapolitana is a valid species, distinct from E. tetraedra; (2) to investigate the genetic diversity of E. neapolitana for the first time; (3) to prove whether N. andaluciana is actually a synonym of E. neapolitana; (4) to place the genus Eiseniella in the phylogenetic tree of lumbricids; and (5) to verify whether Norealidys should fall into synonymy with Eiseniella or not.
Material and methods
Earthworm sampling and morphological studies
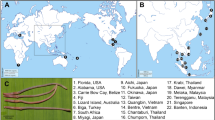
We collected 55 specimens of Eiseniella neapolitana from nine localities in Italy (including the type locality) and one specimen from Cyprus. Also, we gathered thirteen specimens of Norealidys andaluciana from Salobreña (Granada, Spain) (type locality of the species) (Fig. 1, Supplementary Table 1). All individuals were collected by manual sorting in semi-aquatic areas, usually at the edge of bodies of water, washed in distilled water, fixed in 96% ethanol, and stored at − 20 °C in the earthworm collection of the Department of Biodiversity, Ecology and Evolution, Complutense University of Madrid (UCM-LT) with the exception of the specimen from Cyprus, which is housed in the Hungarian Natural History Museum. All newly collected material (except the Cypriot) and specimens of E. tetraedra from de Sosa et al. (2017) were examined morphologically with emphasis on length, weight after fixation, number of segments, position of the clitellum and the tubercula pubertatis, position of male pores, number and position of seminal vesicles and spermatheca, and form and position of the Morren’s glands, typhlosole, and nephridial vesicles. In the case of E. tetraedra, only two specimens found in Naples, Italy, from the de UCM-LT collection (voucher numbers: 30851 and 30854) were examined for the last three characteristics. A portion of the posterior body section was collected and carefully cleaned under a stereomicroscope to remove gut and soil particles. Samples were then stored in ethanol and preserved at − 20 °C until DNA extraction.
Localities sampled. GPS coordinates and number of specimens collected can be found in Supplementary Table 1. Specimens from Italy (3, 4, 5, 6, 7, 8, and 9) and Cyprus (2) belong to E. neapolitana. Specimens from Spain (1) belong to N. andaluciana
DNA extraction, amplification, and sequencing
Total genomic DNA was extracted using the SpeedTools Tissue DNA Kit (Biotools). Four mitochondrial markers, a fragment of cytochrome c oxidase subunit I (COI), 16S rRNA + tRNAs Leu, Ala and Ser (16S), 12S rRNA (12S) and NADH dehydrogenase (ND1), and one nuclear marker (a fragment of 28S rRNA) were amplified. For COI (632 bp), primer sequences, and polymerase chain reactions (PCR) followed (Pop et al., 2003). For 16S-tRNAs (774 bp) and 28S (805 bp), primer sequences and PCR conditions followed (Fernández et al., 2015). For 12S (974 bp) and ND1 (947 bp), primer sequences and PCR conditions followed (Pérez-Losada et al., 2015). Due to the unusual high variability found for the 28S gene (see the “Results” section), four primer pairs were designed to verify the sequences and exclude a possible pseudogene. However, none of the attempts worked.
All PCRs were specific and resolved via 1% agarose gel electrophoresis; they were visualized with GelRed stain (Biotium). All products were purified using ExoSAP-IT reagent (Thermo Fisher Scientific). PCR products were sequenced by Macrogen Spain Inc.
Data analyses
Sequences were aligned in MAFFT v.7 (Katoh & Standley, 2013) using default settings and concatenated with BioEdit v7.0.9 (Hall, 1999). Two phylogenetic trees were constructed. The first contains sequences of the studied populations only (COI-16S + tRNAs + 28S), and the second considers the above populations and other lumbricid species (COI-16S + tRNAs + 28S + 12S + ND1) in order to place them in a wider phylogenetic context. Phylogenetic trees based on the concatenated sequences of COI-16S + tRNAs + 28S (2,211 bp) and COI-16S + tRNAs + 28S + 12S + ND1 (3296 bp) were constructed by the Bayesian Inference (BI) with MrBayes v3.1.2 (Ronquist & Huelsenbeck, 2003) and maximum likelihood (ML) using RaxML v7.03 software (Stamatakis, 2006) both implemented in CIPRES Science Gateway v.3.3 (Miller et al., 2010). The best-fitting substitution model selected by jModelTest2 (Darriba et al., 2012) for all markers in the short dataset was GTR + Γ + I, while for the longer dataset, HKY + Γ + I was chosen for 16S, and GTR + Γ was chosen for 12S. Maximum likelihood analysis with rapid bootstrapping was performed with 1000 replicates. Parameters in MrBayes were set to ten million generations, and 10,000 trees were sampled every 1000th generation, initiating the analysis with a random tree. Two independent analyses were performed, and 20% of the trees were discarded as burn-in. Sequences of each molecular marker for a representative of E. tetraedra (two sequences belonged to Clade I and three to Clade II (de Sosa et al., 2022) were retrieved from Domínguez et al. (2015), Pérez-Losada et al. (2015), and de Sosa et al. (2017) and used as outgroup for the studied populations. In the tree containing other lumbricids, Criodrilus lacuum Hoffmeister, 1845 was used as an outgroup (Supplementary Table 2). Obtained phylogenetic trees were visualized using FigTree v1.3.1 (Morariu et al., 2008).
We calculated haplotype and nucleotide diversity for populations and genes using DNAsp v.6 (Rozas et al., 2017). We also estimated uncorrected pairwise distances for COI and 16S-tRNAs within and between our populations (N. andaluciana/E. neapolitana), as well as with E. tetraedra and two more lumbricids: Dendrobaena byblica Rosa, 1893 and Iberoscolex oliveirae (Rosa, 1894) (Supplementary Table 2). Haplotype networks based on each gene were constructed in PopART 1.7 (Leigh & Bryant, 2015) using the TCS inference method.
We employed two different methods for species delimitation with single locus (COI) analyses. Firstly, the Poisson Tree Processes model (PTP) (Zhang et al., 2013) was used to infer molecular clades based on our inferred molecular phylogeny. Secondly, we used bPTP, which is an updated version of the original maximum likelihood PTP. It adds Bayesian support values to delimited species on the input tree. Both analyses were conducted on the web server for PTP (available at https://species.h-its.org/ptp) using the MrBayes topology as advocated for this method (Zhang et al., 2013; Tang et al., 2014).
The RNAfold web server (Gruber et al., 2008) was used in order to estimate the secondary structure of the 28S rRNA for Spanish and Italian specimens and six other different species: Aporrectodea trapezoides (Dugès, 1828), Allolobophora dubiosa (Örley, 1881), Dendrobaena octaedra (Savigny, 1826), D. byblica, I. oliveirae, and E. tetraedra (Supplementary Table 2). Both MFE (minimum free energy) and centroid secondary structures were estimated. We calculated the G + C content of Spanish specimens by DNAsp v.6 (Rozas et al., 2017). Statistical analyses of morphological data were conducted in Statgraphics Centurion 19 (StatPoint Technologies Inc., USA). We used length, dry weight (after letting it drip on filter paper for 30 s), and number of segments of mature specimens to investigate differences between Spanish (N. andaluciana) and Italian (E. neapolitana) populations and E. tetraedra (de Sosa et al., 2017) through non-parametric analyses (Kruskal–Wallis) followed by Fisher LSD post hoc test.
Results
Phylogenetic analyses
Bayesian and Maximum Likelihood approaches reveal trees with congruent topology for the studied populations (Fig. 2). Sequences are nested in three well-supported clades corresponding to each country studied.
Bayesian inference (BI) of the phylogenetic tree based on concatenated sequences of COI, 16S, and 28S of the studied populations. Posterior probability/bootstrap support values (of maximum likelihood analysis, ML) are shown as black squares when higher than 0.9/0.7 (BI/ML). The scale bar represents 0.08 substitutions per position. Sample numbers correspond to references in the UCM-LT collection and can be found at Supplementary Table 1
Bayesian inference for the studied lumbricid species shows a polytomy for the genus Iberoscolex, E. tetraedra, and the studied populations of N. andaluciana/E. neapolitana (Fig. 3). The Spanish population (N. andaluciana) appears to be more related to the Italian and Cypriot populations (E. neapolitana) than to E. tetraedra, supporting the idea that they may all belong to a different genus, Norealidys, other than Eiseniella.
Bayesian inference (BI) of the phylogenetic tree of lumbricids based on concatenated sequences of COI, 16S, 28S, 12S, and ND1 (long dataset). Posterior probability/bootstrap support values (of maximum likelihood analysis, ML) are shown as black squares when higher than 0.9/0.7 (BI/ML). Gray triangles represent two or more species of a genus-level clade collapsed to facilitate interpretation of the figure. Asterisk represents several singleton species in an unresolved polytomy: Aporrectodea rosea, Aporrectodea georgii, Panoniona leoni, Allolobophora bartolii, Helodrilus patriarchalis, and Proctodrilus antipai. Specimens from Italy and Cyprus belong to E. neapolitana. Specimens from Spain belong to N. andaluciana. Complete names of species are shown in Supplementary Table 2
Genetic diversity and genetic divergence
Within N. andaluciana/E. neapolitana, a total of 17 haplotypes have been identified among 42 sequences for the COI gene, 22 haplotypes within 60 sequences for 16S-tRNAs, and 21 haplotypes within 68 sequences for 28S. Haplotype diversity (H) and nucleotide diversity (π) based on COI including all the specimens within the study are 0.91 and 0.11, respectively. H and π based on 16S-tRNas are 0.90 and 0.04. Genetic diversity parameters based on 28S are 0.58 (H) and 0.01 (π). Values of haplotype and nucleotide diversity for each population are shown in Table 1. A remarkably high haplotype diversity based on 28S has been found for the Spanish population.
Genetic divergence among populations based on COI ranges from 1.57 to 17.81% (Table 2). Values above 9–15% fall into the ambiguous gap between intraspecific and interspecific divergence in earthworms suggested by Chang and James (2011) (which, on the other hand, was based on the slightly higher Kimura-2-parameters distances). Thus, if E. neapolitana from Italy seems to represent by itself a cryptic species complex, according to genetic divergence, the samples from the Spanish population, Cyprus, and Italy seem to belong to three different species. Values obtained for the populations based on 16S are in the range of 0.38 to 10.22% and also show a high variability (Table 2). Genetic distances between the studied populations of N. andaluciana/E. neapolitana and E. tetraedra, D. byblica, and I. oliveirae are also shown in Table 2. COI gene-based distances among E. tetraedra and the studied populations of N. andaluciana/E. neapolitana are higher than the threshold of 9–15% proposed by Chang and James (2011) for interspecific distances. Moreover, distances based on 16S are even higher, reaching about 30%.
The haplotype network for COI shows each population to be highly distant from other neighboring populations, with number of mutational steps from 14 (Italy 4–Italy 6) to 73 (Italy 5–Italy 6) (Fig. 4). Samples from different countries are separated from one another by a high number of mutational steps. Italy 5 is the most heterogeneous population. In contrast, the haplotype network based on 28S shows high homogeneity for Italian haplotypes, whereas Spanish haplotypes show remarkably high differences, as evidenced in their haplotype diversity (Fig. 5). Here as well, populations from different countries appear well separated from one another.
Haplotype network based on COI. Each black circle represents a different haplotype; their size is proportional to the number of samples belonging to that haplotype. Each perpendicular line indicates a mutational step; if there were more than four, it is shown with the number of mutational steps. White circles are hypothetical intermediate haplotypes. Branch length does not contain any information
Haplotype network based on 28S. Each black circle represents a different haplotype; their size is proportional to the number of samples belonging to that haplotype. Each perpendicular line indicates a mutational step, if there were more than four it is shown with the number of mutational steps. White circles are hypothetical intermediate haplotypes. Branch length does not contain any information
To reject the amplification of a pseudogene in the sequences of 28S for the Spanish population (N. andaluciana), we examined the secondary structure of this gene in the studied populations and other Lumbricidae species (Fig. 6). The Italian specimens (E. neapolitana) show the same structure as other species such as A. trapezoides, D. octaedra, and I. albolineatus. However, the Spanish specimens, A. dubiosa and D. byblica, show different forms. The minimum free energy prediction of all sequences was low, ranged from − 397 to − 420 kcal/mol. The G + C content for the Spanish population is 66.50%.
Predicted secondary structures of 28S gene for Spanish and Italian specimens from the studied populations and of six different species: A. trapezoides, A. dubiosa, D. octaedra, D. byblica, I. albolineatus, and E. tetraedra. The structure is colored according to the probability of base pairing: high probability in red, medium probability in green and yellow, and low probability in blue
Species delimitation
The two methods used to delineate species produced the same results (Fig. 7). They recognized seven different species. These analyses perfectly separate E. tetraedra and E. neapolitana, as well as the populations of the latter species (species 1, 2–5, and 6). For E. neapolitana, as many as four different species are delineated in Italy, coinciding with populations in the south (species 2), populations in the north (species 3), and populations in the center of the peninsula (species 4 and 5). Support values are presented in Table 3.
Bayesian inference (BI) of the phylogenetic tree based on sequences of COI of the studied populations and E. tetraedra. Posterior probability is shown as black squares when higher than 0.9. The scale bar represents 0.04 substitutions per position. Sample numbers correspond to references in the UCM-LT collection and can be found at Supplementary Table 1. Results of the PTP model are shown in pink vertical lines. Results of the bPTP model are shown in yellow vertical lines
Morphological studies
Morphological characters of the specimens are shown in Table 4. Only mature individuals were used for the morphological analyses. The Cypriot specimen is not included in the morphological analysis because we only have the postclitellar region of the body. Spermatheca and male funnels are in 10 and 11 and are iridescent in all specimens. The clitellum position of all individuals from the Spanish population is 21–25, while the Italian populations show some variability in this trait. There is also some variability in the position of the tubercula pubertatis in the Italian populations, but in all Spanish specimens, they are 22–23–24. No variability has been found for the male pore, which is always located at segment 15.
The Italian specimens show the Morren’s glands in 10–14 with diverticula in 10, lamellae in 11–13, and little distinct in 14, while none of these structures are observed in the individuals from the Spanish population (except for a very doubtful structure in 11, which should be confirmed histologically) (Fig. 8A; compare with Fig. 8B: E. tetraedra). In the nephridia, the glandular part is broadened both in the Italian and Spanish specimens, and a well-differentiated nephridial vesicle does not appear, but the nephridial tube gradually tapers to open into the nephridial pore (Fig. 8C; compare with Fig. 8D: E. tetraedra). The typhlosole is present in all specimens. It is small and simple at first, and then a slightly pronounced central furrow appears, giving it a certain bilobed appearance (Fig. 8E; compare with Fig. 8F: E. tetraedra).
No statistically significant differences (p > 0.05) are found between the Spanish and Italian populations and E. tetraedra for length and weight. In contrast, a number of segments show statistically significant differences (p = 0.002) between two homogenous groups: Italian and Spanish populations vs. E. tetraedra (Fig. 9).
Discussion
Eiseniella neapolitana was described by Örley (1885) as Allurus neapolitanus and considered by many authors (Michaelsen, 1900; Stephenson, 1924; Bodenheimer, 1937; Cernosvitov, 1938, 1940; Pop, 1952; Mršić, 1991; Pavlicek et al., 2003) as a subspecies of Eiseniella tetraedra, while Omodeo (1952, 1956), Omodeo and Rota (1991), and, more recently, Csuzdi and Pavlicek (2005) claimed it a valid species. The holotype of E. neapolitana was collected by Örley (1885) in Sebeto River (Napoli, Italy). Our specimens from Italy 1 (i.e., locality 3 in Fig. 1) originate from the type locality. However, all Italian specimens of E. neapolitana collected in this study correspond to the morphological characters indicated by Örley (1885) as diagnostic for this species.
The genus Reynoldsia was established by Qiu and Bouché (1998). This name entered into homonymy with the Diptera Reynoldsia Malloch, 1934, so Blakemore (2008) replaced it with the name Norealidys, with the only species N. andaluciana. This genus was discovered in Salobreña, in the south of Spain, on the sea coast, in a sugar cane cultivation, on a sandy soil rich in organic matter, and very moist (Qiu & Bouché, 1998). Our specimens of the Spanish population were collected in the same locality. Currently, the sugar cane fields have disappeared, so the specimens were searched for in the area where this culture used to be found, on a riverbank with sandy and moist soil that was not flooded. As for the morphological details, the position of the clitellum and the tubercula pubertatis of the studied individuals are consistent with those diagnostic for N. andaluciana. The external appearance is very similar to that of the genus Eiseniella. The anatomical traits of N. andaluciana, namely the absence or reduction of the Morren’s glands, typhlosole, and nephridial vesicles, probably indicating an adaptation to a humid environment (Qiu & Bouché, 1998) are all found in the individuals collected by us at the Salobreña site, so we can conclude that they do indeed belong to this species. Qiu and Bouché (1998) described the species as earthworms without skin pigmentation and of small size (between 47 and 72 segments). Our studied individuals were pigmented and had a larger number of segments. This contradiction could be due to the fact that only two individuals were used to describe the species, and they were not sufficient to capture the intraspecific variability of N. andaluciana.
Specimens from Italy (E. neapolitana) and Spain (N. andaluciana) were nested in the same clade in our phylogenetic tree, but with long and well-separated branches. The genetic distances among them, as well as the distances from the other species studied (E. tetraedra, D. byblica, and I. oliveirae), are high (from 16.27 to 21.38%). However, Szederjesi et al. (2018) proved that within Eisenia lucens (Waga, 1857) genetic distances can be higher than 19%. These authors also highlighted that genetic distances are rather clade-specific; therefore, setting a general threshold is not possible. Nevertheless, species delimitation analyses confirmed that E. tetraedra is a separate species from E. neapolitana. On the other hand, they also showed that N. andaluciana is a different species from E. neapolitana, which invalidates the proposal of synonymy between these two species. Moreover, the specimen from Cyprus seems to represent a species distinct from E. neapolitana. However, since it is a single specimen and we only had the posterior section of its body, we cannot describe or name this species until we have examined more material from this population. Finally, these analyses and the haplotype networks demonstrate the status of species complex of E. neapolitana in Italy, which needs to be further investigated.
The phylogenetic tree distinguishes two different clades comprising, on one hand, E. tetraedra and on the other hand all the other populations studied in this work. Based on the depth of the branches, one can assume the existence of two different genera. However, other molecular markers that could resolve the polytomy obtained by our phylogenetic analyses need to be investigated in addition to the other species within the genus Eiseniella. Thus, the most conservative stance is to maintain the synonymy between the genera Eiseniella and Norealidys in the meantime.
The genus Eiseniella appears in the phylogenetic tree in a polytomy with Iberoscolex (= Eiseniona), a genus whose relationships with the other lumbricids and internal phylogeny will be the subject of future studies. Although the general appearance of Eiseniella species is very similar, such as the quadrangular posterior section with dorsal groove and the anterior position of the clitellum, there are differences in the position of clitellum and tubercula pubertatis. We also found significant differences in the number of segments between E. neapolitana and E. andaluciana (= N. andaluciana) with E. tetraedra. Their similar external appearance (small size, anterior clitellum, and posterior dorsal sulcus) could be due to convergence to aquatic or semi-aquatic habitat. Earthworms from the semi-aquatic family Almidae also present quadrangular body at the posterior end and round body at the anterior and an anterior clitellum (Chanabun et al., 2020). Other semi-aquatic species, such as those of the family Criodrilidae, also share some of these morphological characteristics (Blakemore, 2008; Omodeo, 1984). However, the most closely related genus in the phylogenetic tree, Iberoscolex (= Eiseniona), does not present these characters. Therefore, these traits do not appear to have a phylogenetic signal, being shared by species from different families.
E. neapolitana showed well-developed Morren’s glands, which would place it close to E. tetraedra. However, molecular markers placed it together with E. andaluciana (= N. andaluciana), which tends to lack or underdevelop these structures (Qiu & Bouché, 1998). Future studies could resolve these inconsistencies and show whether structures such as the Morren’s glands could correspond to analogous organs and what evolutionary pressures intervene in their development, similar to those indicated by Marchán et al. (2016) for the typhlosole of the Hormogastridae.
As a result of concerted evolution, all copies of rDNA families are generally rapidly homogenized within individuals and species, but interspecific divergence can be high (Hillis & Dixon, 1991). Intraspecific divergence has been found to be high in animals such as corals (Márquez et al., 2003), grasshoppers (Keller et al., 2006), and nematodes (Hugall et al., 1999; Pereira & Baldwin, 2016). At protein-coding loci, pseudogenes can be detected by the presence of stop codons and frame shifts, the absence of substitution bias at third position, or the changes in otherwise invariant amino acid residues (e.g., Arctander, 1995). These indicators cannot be used for nonprotein-coding genes (such as 28S rRNA); however, selection acts to preserve the secondary structure of functional RNA molecules. Current methods for predicting RNA secondary structure are mainly based on the minimum free energy algorithm, which finds the optimal folding state of RNA in vivo using an iterative method to satisfy the minimum energy or other constraints (Zhang et al., 2019). In general, secondary structure stability (low free energies) and pattern of nucleotide substitution in other ribosomal genes such as 5.8S or ITS appeared to be the most powerful approaches to distinguish putative pseudogenes from presumed functional sequences (Razafimandimbison et al., 2004). Thus, the presence of a stable predicted secondary structure (Pereira & Baldwin, 2016) and high G + C content (Zheng et al., 2008) do not support the presence of pseudogenes. Therefore, the high intraspecific divergence for the 28S rRNA gene in the studied Spanish populations may not be explained by the presence of pseudogenes. The mode of reproduction has also been associated with rRNA heterogeneity. Cross-fertilization may increase intraspecific variation due to recombination in the cyclic parthenogenetic Daphnia pulex Leydig, 1860 (Crease & Lynch, 1991) and in polyploid obligate mitotic parthenogens of the nematode genus Meloidogyne (Hugall et al., 1999). Although the studied Spanish population appears to be sexual based on the presence of sperm in the spermatheca, suggesting copulation with another individual, laboratory experiments are required to investigate whether it may also be parthenogenetic. Therefore, its hybrid origin could explain the hypervariability in the 28S rRDNA.
Differences in predicted secondary structure of 28S rRNA between different earthworms’ species may be due to hypervariability rather than pseudogenes or lack of function. They also do not appear to have a phylogenetic signal, as species of the same genus have different structures.
Conclusions
Phylogenetic analyses based on several markers, genetic divergence based on COI, and species delimitation analyses support the validity of E. neapolitana and N. andaluciana as two distinct species. They also invalidate E. neapolitana as a subspecies of E. tetraedra. E. neapolitana also appears as a cryptic complex in the studied populations. According to the phylogenetic analysis, we could hypothesize the existence of two different genera. However, the molecular tree shows the studied species in an unresolved polytomy position, which indicates that the markers used have no power enough to resolve their phylogenetic relationship; the dataset should be increased by adding further molecular markers and the remaining species of the genus Eiseniella seeking to resolve the polytomy recovered in our analyses. Thus, the synonymy between the genera Eiseniella and Norealidys is maintained pending further confirmation. The high variability of the 28S rRNA gene for E. andaluciana (= N. andaluciana) could not be explained by the presence of pseudogenes due to its stable secondary structure and high G+C content.
Availability of data and material
We collected 55 specimens of Eiseniella neapolitana from nine localities in Italy (including the type locality), thirteen specimens of Norealidys andaluciana from Salobreña (Granada, Spain) (type locality of the species), and one specimen from Cyprus (Supplementary Table 1). Voucher specimens were deposited in the earthworm collection of the Department of Biodiversity, Ecology and Evolution, Complutense University of Madrid (UCM-LT). In addition, all nucleotide sequences generated in this study were deposited in the GenBank (Supplementary Table 2).
References
Arctander, P. (1995). Comparison of a mitochondrial gene and a corresponding nuclear pseudogene. Proceedings of the Royal Society of London b: Biological Science, 262, 13–19. https://doi.org/10.1098/rspb.1995.0170
Balian, E. V., Segers, H., Martens, K., & Lévêque, C. (2007). An introduction to the freshwater animal diversity assessment (FADA) project. In Freshwater Animal Diversity Assessment (pp. 3–8). Springer, Dordrecht.
Blakemore, R. J. (2008). Cosmopolitan earthworms – an Eco-Taxonomic Guide to the Species (2nd ed.). VermEcology.
Bodenheimer, F. S. (1937). Prodromus faunae Palestinae. Essai sur les éléments zoogeographiques et historiques du sud-ouest du sous-règne Paléarctique. Mémoires Présentés a L’institut D’egypte, 33, 1–286.
Černosvitov, L. (1938). The Oligochaeta. In Washbourn R, Jones RF (eds): Report of the Percy Sladen Expedition to Lake Huleh; a contribution to the study of fresh waters of Palestine. Annals and Magazine of Natural History, 11, 535–550.
Černosvitov, L. (1940). On some Oligochaeta from Palestine. Annals and Magazine of Natural History, 11, 438–447.
Chanabun, R., Lin, A., Jirapatrasilp, P., & Bantaowong, U. (2020). The semi-aquatic freshwater earthworm genus Glyphidrilus Horst, 1889 from Myanmar (Oligochaeta: Almidae) with description of a new species. Tropical Natural History, 20, 203–218.
Chang, C. H., & James, S. (2011). A critique of earthworm molecular phylogenetics. Pedobiología, 54, S3–S9. https://doi.org/10.1016/j.pedobi.2011.07.015
Crease, T. J., & Lynch, M. (1991). Ribosomal DNA variation in Daphnia pulex. Molecular Biology and Evolution, 8, 620–640.
Csuzdi, C., & Pavlíček, T. (2005). Earthworms from Israel. II. Remarks on the genus Perelia Easton, 1983 with descriptions of a new genus and two new species. Acta Zoologica Academiae Scientiarum Hungaricae, 51, 75–96.
Csuzdi, C. (2012). Earthworm species, a searchable database. Opuscula Zoologica Budapest, 43(1), 97–99.
Darriba, D., Taboada, G. L., Doallo, R., & Posada, D. (2012). jModelTest 2: More models, new heuristics and parallel computing. Nature Methods, 9, 772–772. https://doi.org/10.1038/nmeth.2109
de Sosa, I., Marchán, D. F., Novo, M., Almodovar, A., & Díaz Cosín, D. J. (2017). Bless this phylogeographic mess–Comparative study of Eiseniella tetraedra (Annelida, Oligochaeta) between an Atlantic area and a continental Mediterranean area in Spain. European Journal of Soil Biology, 78, 50–56. https://doi.org/10.1016/j.ejsobi.2016.11.006
de Sosa, I., Díaz Cosín, D. J., Csuzdi, C., Paoletti, M. G., & Marchán, D. F. (2019). Placing Eophila tellinii (Oligochaeta, Lumbricidae) in a molecular phylogenetic context advances the century-old controversy around the problematic genus. European Journal of Soil Biology, 94, 103–114. https://doi.org/10.1016/j.ejsobi.2019.103114
de Sosa, I., Marchán, D. F., Novo, M., Almodóvar, A., & Díaz Cosín, D. J. (2022). Phylogeography of a riparian earthworm shows environmental factors influence genetic structure. Journal of Biogeography. https://doi.org/10.1111/jbi.14518
Domínguez, J., Aira, M., Breinholt, J. W., Stojanovic, M., James, S. W., & Pérez-Losada, M. (2015). Underground evolution: New roots for the old tree of lumbricid earthworms. Molecular Phylogenetics and Evolution, 83, 7–19. https://doi.org/10.1016/j.ympev.2014.10.024
Drilobase Project. (2020). DriloBASE: World Earthworm Database. http://taxo.drilobase.org
Dugès, A. L. (1828). Recherches sur la circulation, la respiration et la reproduction des Annélides abranches. Annales des Sciences naturelles, 15(1), 284–336.
Fernández, R., Novo, M., Marchán, D. F., & Díaz Cosín, D. J. (2015). Diversification patterns in cosmopolitan earthworms: Similar mode but different tempo. Molecular Phylogenetics and Evolution, 94, 701–708. https://doi.org/10.1016/j.ympev.2015.07.017
Gruber, A. R., Lorenz, R., Bernhart, S. H., Neuböck, R., & Hofacker, I. L. (2008). The Vienna RNA Websuite. Nucleic Acids Research, 36, 70–74. https://doi.org/10.1093/nar/gkn188
Hall, T. A. (1999). BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symposium, 41, 95–98.
Hillis, D. M., & Dixon, M. T. (1991). Ribosomal DNA: Molecular evolution and phylogenetic inference. The Quarterly Review of Biology, 66, 411–453.
Huang, J., Xu, Q., Sun, Z. J., Tang, G. L., & Su, Z. Y. (2007). Identifying earthworms through DNA barcodes. Pedobiologia, 51, 301–309. https://doi.org/10.1016/j.pedobi.2007.05.003
Hugall, A., Stanton, J., & Moritz, C. (1999). Reticulate evolution and the origins of ribosomal internal transcribed spacer diversity in apomictic Meloidogyne. Molecular Biology and Evolution, 16, 157–164. https://doi.org/10.1093/oxfordjournals.molbev.a026098
Jiménez Pinadero, S., Marchán, D. F., Novo, M., Trigo, D., Domínguez, J., & Díaz Cosín, D. J. (2021). Sorry atlanticus, you are not my type: Molecular assessment splits Zophoscolex (Lumbricidae: Crassiclitellata) into French and Iberian genera. Zoological Journal of the Linnean Society, zlab011.
Katoh, K., & Standley, D. M. (2013). MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Molecular Biology and Evolution, 30, 772–780. https://doi.org/10.1093/molbev/mst010
Keller, I., Chintauan-Marquier, I. C., Veltsos, P., & Nichols, R. A. (2006). Ribosomal DNA in the grasshopper Podisma pedestris: Escape from concerted evolution. Genetics, 174, 863–874. https://doi.org/10.1534/genetics.106.061341
Leigh, J. W., & Bryant, D. (2015). Popart: Full-feature software for haplotype network construction. Methods in Ecology and Evolution, 6, 1110–1116. https://doi.org/10.1111/2041-210X.12410
Leydig, F. (1860). Naturgeschichte der Daphniden:(Crustacea cladocera). H. Laupp'sche Buchhandlung, Laupp & Siebeck.
Malloch, J. R. (1934). Diptera of Patagonia and South Chile. British Museum (Natural History) Publisher.
Marchán, D. F., Novo, M., Fernández, R., De Sosa, I., Trigo, D., & Díaz Cosín, D. J. (2016). Evaluating evolutionary pressures and phylogenetic signal in earthworms: A case study-the number of typhlosole lamellae in Hormogastridae (Annelida, Oligochaeta). Zoological Journal of the Linnean Society, 17, 4–14. https://doi.org/10.1111/zoj.12410
Marchán, D. F., Fernández, R., de Sosa, I., Sánchez, N., Díaz Cosín, D. J., & Novo, M. (2018). Integrative systematic revision of a Mediterranean earthworm family: Hormogastridae (Annelida, Oligochaeta). Invertebrate Systematics, 32, 652–671. https://doi.org/10.1071/IS17048
Marquez, L. M., MacKenzie, J. B., Takabayashi, M., Smith, C. R., & Chen, C. A. (2003). Difficulties in obtaining microsatellites from acroporid corals. Proceedings of the 9th International Coral Reef Symposium, 1, 139–143.
Michaelsen, W. (1900). Oligochaeta. In: Das Tierreich X. Friedländer & Sohn, Berlin, pp. 575.
Miller, M. A., Pfeiffer, W., & Schwartz, T. (2010). Creating the CIPRES science gateway for inference of large phylogenetic trees. Proceedings of the Gateway Computing Environments Workshop (GCE), 1–8.
Morariu, V. I., Srinivasan, B. V., Raykar, V. C., Duraiswami, R., & Davis, L. S. (2008). Automatic online tuning for fast Gaussian summation. Advances in Neural Lnformation Processing Systems, 21, 1113–1120.
Mršić, N. (1991). Monograph on earthworms (Lumbricidae) of the Balkans II. Monografija o deževnikih (Lumbricidae) Balkana I. Slovenia: Slovenska Akademija Znanosti in Umetnosti, Ljubljana, 456–757.
Omodeo, P. (1952). Oligocheti della Turchia. Annuario Istituto e Museo Di Zoologia Dell’università Di Napoli, 4(2), 1–20.
Omodeo, P. (1956). Contributo alla revisione dei Lumbricidae. Archivio Zoologico Italiano, 41(24), 143.
Omodeo, P. (1984). On aquatic Oligochaeta Lumbricomorpha in Europe. Hydrobiologia, 115, 187–190.
Omodeo, P., & Rota, E. (1991). Earthworms of Turkey II. Italian Journal of Zoology, 58, 171–181.
Örley, L. (1881). A magyarországi Oligochaeták faunája I. Terricolae. Mathematikai és természettudományi Közlemények, 16, 562–611.
Örley, L. (1885). A palaearktikus övben élő Terrikolák-nak revisiója és elterjedése. Magyar Tudományos Akadémia, 15–18.
Pavlícek, T., Csuzdi, C., & Nevo, E. (2003). Species richness and zoogeographic affinities of earthworms in the Levant: The 7th international symposium on earthworm ecology Cardiff Wales 2002. Pedobiologia, 47, 452–457. https://doi.org/10.1078/0031-4056-00212
Pereira, T. J., & Baldwin, J. G. (2016). Contrasting evolutionary patterns of 28S and ITS rRNA genes reveal high intragenomic variation in Cephalenchus (Nematoda): Implications for species delimitation. Molecular Phylogenetics and Evolution, 98, 244–260. https://doi.org/10.1016/j.ympev.2016.02.016
Perel, T. S. (1967). Earthworms of the relict forests of Western Transcaucasia and Talysh region (Russian). Pedobiologia, 7, 93–120.
Pérez-Losada, M., Breinholt, J.W., Aira, M., & Domínguez, J. (2015). An updated multilocus phylogeny of the Lumbricidae (Annelida: Clitellata: Oligochaeta) earthworms. Journal of Phylogenetics and Evolutionary Biology, 3, 140. https://doi.org/10.4172/2329-9002.1000140
Pop, V. (1952). Revizuirea sistematica a genului de Lumbricidae Eiseniella. Studii Şi Cercetări Ştiinţifice, 3(3–4), 1–15.
Pop, A. A., Wink, M., & Pop, V. V. (2003). Use of 18S, 16S rDNA and cytochrome c oxidase sequences in earthworm taxonomy (Oligochaeta, Lumbricidae): The 7th international symposium on earthworm ecology. Cardiff, Wales, 2002. Pedobiologia, 47, 428–433. https://doi.org/10.1078/0031-4056-00208
Qiu, J. P., & Bouché, M. B. (1998). Contribution à la taxonomie des Eiseniellini (Oligochaeta: Lumbricidae). Reynoldsia gen. nov. et deux nouveaux Eiseniella. Institut National de la Recherche Agronomique (ed.). Documents pédozoologiques et intégrologiques, 4, 102–108.
Razafimandimbison, S. G., Kellogg, E. A., & Bremer, B. (2004). Recent origin and phylogenetic utility of divergent ITS putative pseudogenes: A case study from Naucleeae (Rubiaceae). Systematic Biology. https://doi.org/10.1080/1063515049042327
Ronquist, F., & Huelsenbeck, J. P. (2003). MRBAYES 3: Bayesian phylogenetic inference under mixed models. Bioinformatics, 19, 1572–1574. https://doi.org/10.1093/bioinformatics/btg180
Rosa, D. (1893). Viaggio del Dr. E. Festa in Palestina, nel Libano e regioni vicine. – II. Lumbricidi. Bollettino dei Musei di Zoologia ed Anatomia Comparata della R. Università di Torino, 8, 1–14.
Rosa, D. (1894). Allolobophora Ganglbaueri ed A. Oliveirae nuove specie di Lumbricidi europei. Bollettino dei Musei di Zoologia ed Anatomia Comparata della R. Universitá di Torino, 9, 1–3.
Rota, E., & de Jong, Y. (2015). Fauna Europaea: Annelida - Terrestrial Oligochaeta (Enchytraeidae and Megadrili). Aphanoneura and Polychaeta. Biodiversity Data Journal, 3(e5737), 1–48. https://doi.org/10.3897/BDJ.3.e5737
Rozas, J., Ferrer-Mata, A., Sánchez-Del Barrio, J. C., Guirao-Rico, S., Librado, P., Ramos-Onsins, S. E., & Sánchez-Gracia, A. (2017). DnaSP 6: DNA sequence polymorphism analysis of large data sets. Molecular Biology and Evolution, 34, 3299–3302. https://doi.org/10.1093/molbev/msx248
Savigny, J. C. (1826). Analyses des travaux de l’Académie Royale des Sciences pendant l’année 1821, partie physique. Zoologie. Mémoires de l’Academie Royale des Sciences de l’Institut de France, 5, 176–184.
Stamatakis, A. (2006). RAxML-V1-HPC: Maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics, 22, 2688–2690. https://doi.org/10.1093/bioinformatics/btl446
Stephenson, J. (1924). On some Indian Oligochaeta, with a description of two new genera of Ocnerodrilinae. Records of the Indian Museum, 26, 317–365.
Szederjesi, T., Felföldi, T., Krizsik, V., Pop, V. V., & Csuzdi, C. (2018). DNA barcoding reveals deep genetic divergences in Eisenia lucens (Waga, 1857) and supports Eisenia spelaea (Rosa, 1901) as a separate species (Clitellata: Megadrili). European Journal of Soil Biology, 87, 33–39. https://doi.org/10.1016/j.ejsobi.2018.05.001
Tang, C. Q., Humphreys, A. M., Fontaneto, D., & Barraclough, T. G. (2014). Effects of phylogenetic reconstruction method on the robustness of species delimitation using single-locus data. Methods in Ecology and Evolution, 5(10), 1086–1094. https://doi.org/10.1111/2041-210X.12246
Waga, A. (1857). Sprawozdanie z podrozy naturalistov odbytej w r. 1854 do Ojcowa. Bibliotheca Warszawie, 2, 161–227.
Zhang, H., Zhang, C., Li, Z., Li, C., Wei, X., Zhang, B., & Liu, Y. (2019). A new method of RNA secondary structure prediction based on convolutional neural network and dynamic programming. Frontiers in Genetics, 10, 467. https://doi.org/10.3389/fgene.2019.00467
Zhang, J., Kapli, P., Pavlidis, P., & Stamatakis, A. (2013). A general species delimitation method with applications to phylogenetic placements. Bioinformatics, 29(22), 2869–2876. https://doi.org/10.1093/bioinformatics/btt499
Zheng, X., Cai, D., Yao, L., & Teng, Y. (2008). Non-concerted ITS evolution, early origin and phylogenetic utility of ITS pseudogenes in Pyrus. Molecular Phylogenetics and Evolution, 48, 892–903. https://doi.org/10.1016/j.ympev.2008.05.039
Zicsi, A. (1972). Eiseniella koreana eine neue Regenwurm-Art (Oligochaeta: Lumbricidae) aus Korea. Annales Musei Historico-Naturalis Hungarici, 64, 129–132.
Acknowledgements
We would like to thank Nuria Sánchez for field support.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. IS was supported by a Predoctoral Fellowship grant by Universidad Complutense de Madrid, Spain. DF was supported by a Juan de la Cierva Formación grant from Spanish Government (FJCI-2017–32895) and a MOPGA grant from the French Government (mopga-postdoc-3–6111272103), and MN was supported by Ramón y Cajal Fellowship (RYC2018-024654-I) of the Spanish Government. AJ was supported by 2014/15/B/NZ8/00266 grant from the National Science Centre, Poland. This research was funded by projects CGL2013-42908-P and PGC2018-095851-B-C66 from the Spanish Government.
Author information
Authors and Affiliations
Contributions
Irene de Sosa, Daniel F. Marchán, Marta Novo, Ana Almodóvar, and Darío Díaz Cosín contributed to the study conception and design. Material preparation and data collection were performed by Irene de Sosa, Timea Szederjesi, Misel Jelic, Aleksandra Jabłońska, and Raúl Navarro. Analysis were performed by Irene de Sosa. The first draft of the manuscript was written by Irene de Sosa, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Consent for publication
All authors approved the final version of the manuscript for publication.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
de Sosa, I., F. Marchán, D., Novo, M. et al. Guess who? Taxonomic problems in the genus Eiseniella revisited by integrated approach. Org Divers Evol 23, 295–308 (2023). https://doi.org/10.1007/s13127-022-00593-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13127-022-00593-5