Abstract
Immature embryos of F1 progeny resulting from taxonomically wide hybridizations require tissue culture using complex media to mature, grow and develop into normal and healthy plants. To gain insight as to how progeny of wide-crosses between wild and domestic Triticum species, 12 domestic varieties (Al-Eiz, Babel, Bedhaa, Entisar, Hadbaa, IPA 99, Latifia, Nour, Rabia, Sally, Sham 6, Tamoz 2) were used to determine the genotype response to culture media type. Immature embryos the aforementioned wheat varieties were screened on four media (Murashige and Skoog; MS full strength, ½ strength MS, Gamborg B-5; B-5 full strength, and ½ strength B-5) to determine performance when used as a female in a cross with wild species. The experiment used a completely randomized design with six replications. Traits recorded were final germinations percentage (FGP), shoot length (SL), root length (RL), and root number (RN). Results indicated significant variety x media interaction for all traits studied. Despite the interaction, in a practical sense, all varieties performed adequately on ½ B-5 and varieties Al-Eiz, Entisar, Hadbaa, and Latifia performed well on any of the four media. Tamoz 2 behaved recalcitrantly on all media tested. Data from this study indicated genetic variability among these wheat varieties caused substantial differences in response to each type of media.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bread wheat (Triticum aestivum L.) is the most important crop in the world. It is ranked first world-wide in consumption production, and total planted hectarage followed by rice (Oryza sativa), maize (Zea mays), and barley (Hordeum vulgare) (FAO 2011; Tadesse et al. 2016). Wheat plays an essential role in human food security and international trade because it is a stable for 40% of the global human population. Furthermore, the high nutritional value of wheat can supply roughly 21% of the calories and 20% of the protein for the daily human requirement (Braun et al. 2010; Wu et al. 2022). Wheat has many food uses, including the production of bread, biscuit, confectionary products, ethanol, beer, and protein production. Wheat bran and wheat straw is used as animal feed (Baba and Malik 2020). The human population recently exceeded 8 billion, climate change diseases, and agricultural pests have a substantial impact on wheat demand and production. Continuous cultivation and a lack of genetic diversity in conventional breeding programs led to genetic erosion then narrowing the genetic base of wheat genotypes (Peng et al. 2011). This loss of diversity among wheat genotypes has limited productivity and led to sensitivity to biotic and abiotic stresses exacerbated by climate change (Nevo 2009). Therefore, the incorporation of genetic relatives of wheat, especially the wild relatives, into breeding programs is important to expand domestic wheat’s genetic base (Nevo and Chen; 2010; Solh et al. 2012; Tadesse et al. 2016). However, taxonomically wide hybridizations result in small and shrunken seed, embryo failure and/or endosperm that aborts after 3–5 days of growth; all of which result in very low seed set (Chahal and Gosal 2006; Ogbonnaya et al. 2013). Therefore, embryo rescue is an important method to ameliorate these sexual incompatibility barriers in wheat breeding programs (Houben et al. 2011; Mohammed et al. 2020; Hull et al. 2021). In addition, immature embryo culture is one of most important techniques that is used for producing wheat haploid plants that can be converted to doubled haploids that are 100% homozygous in one generation. Immature embryo culture may also be used to break seed dormancy, test seed viability, callus method, propagate rare plant species, and aid in genetic transformation (Tamas et al. 2004; Aydin et al. 2011; Chauhan and Khurana 2011). Successful embryo rescue depends on the genotype of the plant and the composition of the culture media (Varshney and Altpeter 2002; Tamas et al. 2004). In addition, immature embryos of F1 crosses resulting from these wide-crosses require appropriate nutritional media to complete maturation, germination, and growth into normal plants (Pickering and Johnston 2005; Mohammed et al. 2020).
Characteristics measured in these studies generating wide-cross hybrids include germination percentage, shoot and root length, and root number. Researchers have reported each hybrid requires has a specific medium that may or may not be suitable for other wide-cross hybrids(Tamas et al. 2004; Wu et al. 2008; Gonzalez et al. 2008; Xynias et al. 2014; Ltifi et al. 2019; Mohammed et al. 2020). A number of media types for wheat immature embryo culture have been reported, most frequently Murashige and Skoog (MS), ½ MS, Gamborg B-5 (B-5) media (Singh et al. 2004; Niroula and Bimb 2009; Sourour et al. 2011; Avci and Kutlu, 2020; Patial, et al. 2021). One of the most challenging problems facing plant breeders using interspecific or intergeneric hybridization is germination of embryos compromised by gene imbalances resulting from taxonomically wide-crosses. The question arises, which is the appropriate medium for each F1embryo. To elucidate the appropriate medium for progeny of specific wide-crosses, immature embryos were generated of 12 domestic wheat varieties with the goal of putatively identifying and optimizing germination and growth of immature embryos derived of these varieties crossed with wild species of wheat.
Materials and methods
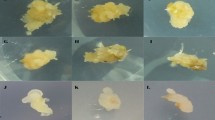
Twelve wheat varieties (Bedhaa, Sham 6, Sally, Tamoz 2, IPA 99, Rabia, Al-Eiz, Latifia, Entisar, Hadbaa, Nour, and Babel) were used to study the effect of two media at two concentrations (MS, ½ MS, B-5, and ½ B-5) on immature embryo culture. These varieties were chosen because they are the 12 most widely grown in Iraq. Seeds of these wheat varieties were received from Seed Technology Center/ Office of Agricultural Research/ Iraqi Ministry of Science and Technology. Field and laboratory experiments were conducted in the Field Crops Station and Plant Tissue Culture Laboratory of Agriculture College/Tikrit University during winter and spring of 2020/2021, respectively. Varieties were planted in the field. Irrigation, fertilization, weed control and other cultural operations were conducted as needed. When embryo length of each variety was 0.5–1 mm, approximately 12–14 days after pollination, spike with its peduncle were harvested suggested by (Tamas et al. 2004; Niroula and Bimb 2009; Devaux 2021). Harvested spikes on their peduncle were placed in a beaker of water and refrigerated until dissection. Excised seed were hand threshed to remove chaff and sterilized for one minute in 70% ethanol. Seed were then surface disinfested with 1% solution of NaClO for 15 min and rinsed three s times with sterile distilled water (Tadesse et al. 2019) in a laminar flow hood (Environmental Air Control, Inc., Hatfield, PA). Disinfested seed were dried on sterile filter paper. Immature embryos were aseptically dissected from each wheat variety. Five embryos from each variety were cultured in a 100 ml baby food jars containing 20 ml of either MS, ½ MS, B-5, or ½ B-5 medium (Sigma-Aldrich Co., M-5519, St. Louis, MO), with no plant growth regulators. Media salts were supplemented with 0.8% agar (Sigma-Aldrich Co., A-1296, St. Louis, MO); 2% and 3% of sucrose (Fisher Scientific, S5-500, Fair Lawn, NJ) for Gamborg B-5 and MS media, respectively. The pH of B-5 and MS media was adjusted to 5.5 and 5.7, respectively, using 0.1 N NaOH (Houben et al. 2011). Jars were wrapped with Parafilm M® PM-996 (Bemis Company Inc., Neenah, WI) labeled and placed in a growth chamber (Percival, Mod. 135LLVL, Controlled Environments. Boone, IA) in a completely randomized arrangement. Chamber conditions were a constant temperature of 20–25 °C with16 h/8 h photoperiod (light/dark) for 10 days (Tadesse et al. 2019). After 10 days, embryos were assessed for normal germination, shoot and root growth. There were six replications of each variety (n = 30). Embryos with roots longer than 2 mm were monitored and counted daily for an additional 10 days. The final germination percentage (FGP) was calculated using the formula of Scott et al. (1984):
Characteristics of embryos included in the final germination percentage included shoot length (SL; cm), root length (RL; cm), root number (RN), and leaf number (LN). These morphological characteristics were assessed on these12 wheat varieties to gain insight to the potential response of rescued wide-cross hybrid embryos to culture media. The experimental design was a completely randomized design (CRD) with six replications (jars) totaling 30 embryos per variety. Data were transformed and statistically analyzed using PROC MEANS and PROC GLM in SAS (Version 9.4, SAS Institute 2011, Cary, NC). Significant mean separation among the 12 wheat varieties, two media types at two concentraions, and variety x media interactions was determined using Fisher’s LSD(α = 0.05).
Results and discussion
Analysis of variance (Table 1) indicated no year effect, but highly significant effects on all traits studied. A significant interaction between varieties x media takes precedence over the main effects. Data from this study showed most mean values could be separated at the P < 0.01 level, for Table 2–5 least significant difference values were reported at P < 0.05. While mean separations do indicate relative ranking among varieties and media formulations, there is a practical aspect of reporting SL, RL, and RN, i.e., the variety x media combination with the greatest shoot length (and ranked greatest) isn’t the only variety x media combination representing embryos that would grow and mature into plants that could survive transplantation. All data with appropriate mean separation are presented in Table 2–5, but the narrative focuses on practical delimiters of length and number, i.e. while more is better, if a single embryo germinates and survives breeding progress can be made.
The significant interaction effect indicates not all wheat varieties responded the same to each of the media formulations. Previous studies have reported significant differences among wheat genotypes, media, and interaction for different characteristics such as FGP, RL, SL and RN (Tamas et al. 2004; Singh et al. 2004; Gonzalez et al. 2008; Wu et al. 2008; Niroula and Bimb 2009; Sourour et al. 2011; Xynias et al. 2014; Ltifi et al. 2019; Avci and Kutlu, 2020 Mohammed et al. 2020; Patial et al. 2021).
Likely the most important characteristic, FGP was measured daily, and finalized on day-10. It is defined as a seed with a radicle equal to or greater than 2 mm. The FGP ranged from 100% (Al-Eiz on both MS formulations) to 0% (Tamoz 2 on full strength MS) (Table 2). While a greater percentage germination would be considered better, it should be noted that all 12 genotypes tested had at least 50% FGP on at least one of the four media formulations tested.
There are media x variety combinations had relatively high values of FGP, such as Latifia on ½ B-5 (90.48%), and Hadbaa x MS, B-5, and ½ B-5 (88.89%). While not the greatest percentage, Bedhaa, Sally, Tamoz 2, IPA 99, Latifia, and Babel varieties had highest value of FGP (83.33, 63.33, 50.0, 66.11, 90.48, and 78.33%, respectively) when cultured on ½ B-5 medium compared to the other three formulations. Sham 6 and Entisar had the greatest FGP when grown on ½ MS medium. Rabia performed best on B-5 medium and Al-Eiz showed the greatest FGP on full and half strength MS. The variety, Nour showed greatest FGP on MS (51.11%) and B-5 (60.22). Hadbaa performed equally on three of the four media formulations (88.89%), except ½ MS medium which gave a lower FGP (77.78%). Wheat varieties used in this study could be used in interspecific or intergeneric hybridization by culturing immature embryos of F1 crosses on appropriate medium of each variety in wheat breeding programs. Similar results have been reported by previous studies. Tamas et al. 2004 found that reducing the concentration of nutrient salts to half in BEG 2 (MS supplemented with vitamins and sucrose), MS-9 (MS supplemented with vitamins, sucrose and 6-benzylaminopurine) and MS media led to increase germination. Cherkaoui et al. (2008) reported that B-5 and ½ MS media were better than MS medium for FGP of durum x maize embryos. Sourour et al. (2011) reported that B-5 medium was significantly better than MS and ½ MS media for immature durum x maize embryo germination. Xynias et al. (2014) found that each wheat cross responded to each type of media independently, and ½ MS and B-5 media were better than MS medium for double haploid embryo germination.
Patial et al. (2021) reported that ½ MS medium followed by modified MS medium gave FGP immature wheat embryos compared to B-5 and modified B-5 media. Similar to this study, they found that genotypes responded individually to each test medium. Mohammed et al. (2020) found the same results working on germination of immature embryos resulting from wide-crosses between domestic (Hordeum vulgare) and wild barley (H. bulbosum). These differing results were attributed to genotype diversity, immature embryos size and components of each medium type, such as macro and microelements, vitamins, sucrose level and pH.
Germination was defined as the production a radicle at least 2 mm long. However, shooting may occur without emergence of a radical (Northam and Callihan, 1994). Shoot growth is necessary for the embryo to maintain growth outside of the culture medium. Mean shoot (SL) length of germinated embryos were evaluated and ranged from 11.27 cm (Hadbaa on ½ strength B-5)to 0 cm (Tamoz 2 onMS)(Table 3). The variety x media type interaction was significant (P < 0.01) for SL. Varieties that were determined to have adequate shoot growth (shoots > 7.0 cm) were Entisar (8.39 cm; on ½ MS,and 8.35 cm; B-5),Hadbaa (11.27 cm on ½ B-5), Latifia and Nour (8.24 and 9.60 cm, respectively, on MS), Rabia (8.62 cm on ½ B-5), and Sally (9.94 cm on B-5). Varieties responding the poorest (SL < 2.0 cm) on these four media formulations were Al-Eiz (1.57 cm on ½ MS), Babel, Bedhaa, and IPA 99 (1.56, 0.43 and 0.17 cm, respectively, on MS). Tamoz 2, based on FGP and SL appears to be recalcitrant on these media. The zero-shoot response of Tamoz 2 (on MS) corresponds to complete radicle failure (Table 2) on the same medium.
When assessing varietal performance based on SL, seven of the 12 varieties had the greatest SL on ½ B-5 medium (Babel, Entisar, Hadbaa, IPA 99, Rabia, Sahm 6 and Tamoz 2), four of 12 on B-5 (Babel, Bedhaa, Entisa, and Sally), two of 12 on ½ MS (Entisar and Latifia)and three of 12 on MS (Al-Eiz, Latifia an Nour). This exceeds 12 because a variety may share maximum growth classification in more than one medium. Based on SL performance of these 12 varieties, MS would seem to be the poorest choice, with four varieties having the shortest shoot length on that medium (Table 3). These SL data fall into trends similar to FGP and confirmthe effects of wheat genotype with types of media.Our results are sustained by previous studies (Burun and Poyrazoglu 2002; Tamas et al. 2004; Russowski et al. 2006; Cherkaoui et al. 2008; Sourour et al. 2011; Mohammedet al. 2020).These authors found similar difficulty in singling out one appropriate medium for different varieties of wheat.
A significant variety x media interaction was present for RL. Root length values ranged from 8.32 cm (Nour on MS) to 0.0 cm (Tamoz 2 on MS; Table 4).
The varieties determined to have RL appropriate for transplanting and sustained growth (RL > 5.0 cm) were Bedhaa (7.48 and 5.54 cm on B-5 and ½ B-5, respectively), Entisar (6.08, 5.64 and 5.02 cm on ½ MS, B-5 and ½ B-5, respectively), Hadbaa (5.52 and 7.08 cm on B-5 and ½B-5, respectively), Latifia (5.24, 5.18, 5.81 cm on MS, ½ MS, and ½ B-5, respectively), Nour 8.32, 6.73, 5.63, 7.00 cm on MS, ½ MS, B-5 and ½ B-5, respectively) and Sally (8.14 cm on B-5). The varieties with the greatest RL were Nour on MS media, Sally, and Bedhaa on B-5 media.
Among the poorest performing (RL < 2.0 cm) were Al-Eiz (1.93 cm on ½ MS), Babel (1.35 cm on MS),Bedhaa, IPA 99 and Tamoz on MS medium (0.7, 0.1 and 0.0 cm, respectively). Consistent with the trend observed with FGP and SL, Tamoz 2 performed poorly with respect to RL on the other three media formulations as well. Tamoz 2 RL measurements were 0.9 cm (on ½ MS), 0.6 cm (on B-5) and 1.69 (on ½ B-5).
When assessing varietal performance by medium based on RL, eight of the 12 varieties had their greatest RL on ½ B-5 media (Al-Eiz, Babel, Hadbaa, IPA 99, Latifia, Rabia, Sahm 6, and Tamoz 2). Of these eight, six had RL > 5.0 cm. Six varieties (Babel, Bedhaa, Entisa, IPA 99, Sally, and Sham 6) performed best on B-5, with five exceeding 5.0 cm in RL. Two of the 12 varieties performed best on ½ MS (Entisar and Latifia). In addition to Entisar and Latifia, Nour had RL > 5.0 cm. On the MS media three varieties (Al-Eiz, Latifia, and Nour) performed best. This exceeds 12 due to a variety having maximum growth in more than a single medium. Indeed, Nour performed exceedingly well on all media and Latifia and Entisar performed well on three of the four media formulations. These RL data fall into a trend similar to SL and FGP.
Based on RL performance of these 12 varieties, ½ B-5 would appear to be the best medium, while MS would seem to be the poorest choice(Table 3). As with FGP and SL, it is difficult to draw a conclusion about the optimal medium type for different varieties. Tamas et al. 2004 (with wheat); Gonzalez et al. 2008 (with T. turgidum); Wu et al. 2008 (with wheat); Niroula and Bimb 2009 (wheat and maize); Mohammed 2018 (with H. vulgare and H. bulbosum); Ltifi et al. 2019 (wheat and maize) found similar results.
Root number is another measure of embryo performance in tissue culture. Mean RN ranged from 5.36 (Entisar on B-5) to 0.0 (Tamoz 2 on MS) on the four media formulations (Table 5). Entisar (5.36 on B-5) gave the highest value, but40 other variety x media combinations produced mean RN of 3.0 or more on the four media formulations. Seven variety x media combinations showed low numbers (< 3.0) of roots produced; they were Babel (2.73 on MS), Bedhaa (2.5 on MS), IPA 99 (0.50 and 2.25 on MS and ½ MS, respectively), and Tamoz 2 (0.0, 2.0, 1.33 on MS, ½ MS and B-5, respectively).
When comparing varietal response to media, all twelve varieties produced > 3 roots on B-5 media, 11 of 12 on ½ B-5 media, 10 of 12 on ½ MS media and eight of 12 on MS media.As with FGP, SL, and RL the data for FN confirmed each wheat variety responded to each medium type individually. Previous studies by Burun and Poyrazoglu 2002 (with barley); Russowski et al. 2006; Cherkaoui et al. 2008 (with Valeriana glechomifolia);Sourour et al. 2011 (with durum wheat x maize crosses); Mohammedet al. 2020 (with barley) found similar difficulty to find a single appropriate medium for different varieties.
Conclusion
Results indicated significant variety x media interaction for all traits studied. Assuming other wheat varieties behave in a manner like these 12, knowing nothing about a wheat variety, the medium formulation ½ B-5 would be the best option for a culture medium. If choosing specific varieties to make wide crosses, Al-Eiz, Entisar, Hadbaa, and Latifia gave FGP from 64 to 100% in every media formulation tested and performed well for SL and RL. Tamoz 2 had the lowest FGP reaching 50% (in ½ B-5) when it did germinate. Tamoz 2’s SL, RL and RN were always among the lowest value of the 48 variety x media combinations.
The data from this study provides insight to the putative performance when using these varieties as females crossed with wild wheat germplasm. However, the significant variety x media interactions indicate broad and absolute statements concerning wide-cross hybrid wheat embryo performance on any specific medium are not possible because of the variability among the males used in this experiment.
References
Avcı S, Kutlu İ (2020) Comparison of orchard-grass and sweet maize for doubled haploid plant production via wide hybridization in bread wheat. Turkish J Agric.-Food Sci. Tech. 8(7):1548–1552
Aydin M, Tosun M, Haliloglu K (2011) Plant regeneration in wheat mature embryo culture. Afric. J. Biotechn. 10(70):15749–15755. https://doi.org/10.5897/AJB11.1495
Baba AY, Malik T (2020) Nutritional values and health effects of wheat. The ABCs of Agriculture. Education Publishing, India, pp 212–219
Braun HJ, Atlin G, Payne T (2010) Multi-location testing as a tool to identify plant response to global climate change. In: Reynolds MP (ed) Climate change and crop production. CABI Publishers, Wallingford, UK, pp 115–138
Burun B, Poyrazoglu EC (2002) Embryo culture in barley (Hordeum vulgare L.). Turk J Biol 26:175–180
Chahal GS, Gosal SS (2006) Tissue culture in crop improvement. Principles and procedures of plant breeding: Biotechnological and conventional approaches. Alpha Sci. Inter. Ltd, Harrow UK, pp 429–456
Chauhan H, Khurana P (2011) Use of doubled haploid technology for developmentof stable drought tolerant bread wheat (Triticum aestivum L.) transgenics. Plant Biotechn. J. 9(3):408–417. https://doi.org/10.1111/j.1467-7652.2010.00561.x
Cherkaoui S, Lamsaouri O, Chlyah A, Chlyah H (2008) Durum wheat× maize crosses for haploid wheat production: influence of parental genotypes and various experimental factors. Plant Breed 119(1):31–36. https://doi.org/10.1046/j.1439-0523.2000.00433.x
Devaux P (2021) Production of wheat doubled haploids through intergeneric hybridization with maize. In: Segui-Simarro JM (ed) Doubled haploid technology. Humana, New York, NY, pp 26–279
FAO.(2011). FAOSTAT. FAO, Rome, Italy. http://faostat.fao.org
Gonzalez JM, Friero E, Jouve N (2008) Influence of genotype and culture medium on callus formation and plant regeneration from immature embryos of Triticum turgidum Desf cultivars. Plant Breed 120(6):513–517. https://doi.org/10.1046/j.1439-0523.2001.00661.x
Houben A, Sanei M, Pickering R (2011) Barley doubled-haploid production by uniparental chromosome elimination. Plant Cell Tiss Organ Cult 104(3):321–327. https://doi.org/10.1007/s11240-010-9856-8
Hull R, Head G, Tzotzos GT (2021) Genetically modified plants. In: Jesy C (ed) Genetically modified plants (Second edit). Academic Press, UK, pp 35–81
SAS Institute. (2011). SAS guide to macro processing. SAS Inst, Cary NC.
Ltifi A, Sahli A, Haouala F (2019) Study of some factors influencing the production of durum wheat haploids by cross wheat× maize. Intern. J. Adv Agric. Sci. Tech. 6(1):17–23
Mohammed AH (2018) Polyploidy hybrids from wide-crosses between Hordeum vulgare and H. bulbosum for Improving Salinity Tolerance Using Embryo Rescue. Mississippi State University, USA, p 1
Mohammed AH, Morrison JI, Baldwin BS (2020) Interspecific crosses between domestic and wild barley and embryo rescue to overcome sexual incompatibilities. Agrosys Geosci Environ. https://doi.org/10.1002/agg2.20130
Nevo E (2009) Ecological genomics of natural plant populations: The Perspective. In: Gustafson J, Langridge P, Somers D (eds) Plant Genomics. Methods in Molecular Biology™. Humana Press, USA, pp 321–344
Nevo E, Chen G (2010) Drought and salt tolerances in wild relatives for wheat and barley improvement. Plant, Cell Environ 33(4):670–685. https://doi.org/10.1111/j.1365-3040.2009.02107.x
Niroula RK, Bimb HP (2009) Overview of wheat x maize system of crosses for dihaploid induction in wheat. World Appl Sci J 7(8):1037–1045
Northham RE, Callihan RH (1994) Interpreting germination results based on differing embryonic emergence criteria. Weed Sci. 42(3):474–481
Ogbonnaya FC, Abdalla O, Mujeeb-Kazi A, Kazi AG, Xu SS, Gosnian N, Lagudah ES (2013) Synthetic hexaploids: Harnessing species of the primary gene pool for wheat improvement. Plant Breed Rev. 37:35–122. https://doi.org/10.1002/9781118497869.ch2
Patial M, Chaudhary HK, Sharma N, Sundaresha S, Kapoor R, Pal D, Kumar J (2021) Effect of different in vitro and in vivo variables on the efficiency of doubled haploid production in Triticum aestivum L. using Imperata cylindrica-mediated chromosome elimination technique. Cereal Res. Comm. 49(1):133–140. https://doi.org/10.1007/s42976-020-00069-2
Peng JH, Sun D, Nevo E (2011) Domestication evolution, genetics and genomics in wheat. Molec Breed 28(3):281–301. https://doi.org/10.1007/s11032-011-9608-4
Pickering R, Johnston PA (2005) Recent progress in barley improvement using wild species of Hordeum. Cytogenet Genome Res 109(1–3):344–349. https://doi.org/10.1159/000082418
Russowski D, Maurmann N, Rech SB, Fett-Neto AG (2006) Role of light and medium composition on growth and valepotriate contents in Valeriana glechomifolia whole plant liquid cultures. Plant Cell Tiss Organ Cul 86:211–218. https://doi.org/10.1007/s11240-006-9109-z
Scott SJ, Jones RA, Williams WA (1984) Review of data analysis methods for seed germination. Crop Sci 24(6):1192–1199. https://doi.org/10.2135/cropsci1984.0011183X002400060043x
Singh S, Sethi GS, Chaudhary HK (2004) Different responsiveness of winter and spring wheat genotypes to maize-mediated production of haploids. Cereal Res. Comm. 32:201–207
Solh, M., Nazari, K., Tadesse, W., and Wellings, C. R. (2012). The growing threat of stripe rust worldwide. In: Borlaug Global Rust Initiative (BGRI) Conference, Beijing, China (pp. 1–4).
Sourour A, Olfa SA, Silva JAT, Hajer SA (2011) Effect of different factors on haploid production through embryo rescue in durum wheat× maize crosses. Intern J Plant Breed 5(2):118–121
Tadesse W, Amri A, Ogbonnaya FC, Sanchez-Garcia M, Sohail Q, Baum B (2016) Wheat. In: Singh M, Upadhyaya HD (eds) Genetic and Genomic Resources for Grain Cereals Improvement. Academic Press, San Diego, CA, pp 81–124
Tadesse W, Sanchez-Garcia M, Tawkaz S, Baum M (2019) Doubled haploid production in wheat. Advances in Breeding Techniques for Cereal Crops. Burleigh Dodds Science Publishing, UK, pp 93–116
Tamas C, Szucs P, Rakszegi M, Tamas L, Bedo Z (2004) Effect of combined changes in culture medium and incubation conditions on the regeneration from immature embryos of elite varieties of winter wheat. Plant Cell Tiss. Organ Cult. 79(1):39–44. https://doi.org/10.1023/B:TICU.0000049447.81409.ed
Varshney A, Altpeter F (2002) Stable transformation and tissue culture response in current European winter wheats (Triticum aestivum L.). Mole Plant Breed 8:295–309
Wu BH, Zheng YL, Liu DC, Zhou YH (2008) Trait correlation of immature embryo culture in bread wheat. Plant Breed 122(1):47–51. https://doi.org/10.1046/j.1439-0523.2003.00778.x
Wu M, Xu Y, Zheng J, Hao Z (2022) North expansion of winter wheat planting area in China under different emissions scenarios. Agriculture 12(6):763. https://doi.org/10.3390/agriculture12060763
Xynias I, Koufalis A, Gouli-Vivdinoudi E, Roupakias D (2014) Factors affecting doubled haploid plant production via maize technique in bread wheat. Acta Biol Cracov Bot 56(2):67–72. https://doi.org/10.2478/abcsb-2014-0022
Funding
This work was funded by Tikrit University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mohammed, A.H., Baldwin, B.S. Investigation of media for wheat (Triticum aestivum L.) immature embryo culture. J. Crop Sci. Biotechnol. 27, 331–337 (2024). https://doi.org/10.1007/s12892-023-00233-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12892-023-00233-0